Introduction
Liver cancer is one of the most common types of
cancer in humans globally (1). It
is often diagnosed at an advanced stage and is resistant to the
majority of treatment options currently available. Therefore, it is
important to further investigate the biology of liver cancer and
identify novel therapeutic strategies for the management of liver
cancer.
Cancer results from a number of genetic and
epigenetic alterations (2), which
together lead to the deregulation of gene expression, protein
interaction networks and cell metabolism. It is now widely accepted
that the initiation, progression and maintenance of a cancer cell
relies on the collaboration of multiple signaling pathways, which
function simultaneously in a non-linear manner. Therefore,
targeting a single gene or pathway may not be sufficient to
eradicate cancer cells. It was previously identified that silencing
a single β-catenin gene inhibited liver cancer cell growth but was
not able to eliminate the liver cancer completely (3). Simultaneous inhibition of two or more
key cancer growth-dependent signaling pathways may lead to the
induced death of cancer cells.
Deregulation of the Wnt/β-catenin and signal
transducer and activator of transcription 3 (STAT3) signaling
pathways has been observed in the majority of cancer types and is
closely associated with the genesis and development of liver
cancer. The multifunctional protein β-catenin is the activation
center of the Wnt signaling pathway (4-6). The
Wnt/β-catenin signaling pathway is able to affect the growth of
liver cancer through regulating the cell cycle, apoptosis,
angiogenesis, telomerase activity and other cell growth signaling
pathways (3,7). For example, pharmacological
inhibition of the Wnt/β-catenin signaling pathway promotes cell
apoptosis in neuroblastoma cell lines (8). The phosphatase and tensin
homolog/phosphoinositide 3-kinase/Akt and Wnt/β-catenin signaling
pathways are involved in the regulation of human colon cancer cell
proliferation (9). The oncogene
STAT3 is at the convergence of numerous tumor-associated tyrosine
kinase signaling pathways; thus, it is important in the regulation
of cancer cell growth and apoptosis. In addition, STAT3 is
upregulated in numerous types of cancer (10,11).
For example, inactivation of STAT3 signaling induced apoptosis in
HCT116 human colon cancer cells (12) and inhibited the proliferation and
metastasis of hepatocellular carcinoma (13). In addition, inhibition of STAT3
activation was reported to suppress the tumorigenicity and growth
of nasopharyngeal carcinoma cells (14).
Regarding the importance of β-catenin and STAT3 in
tumorigenesis, their synergistic role during the initiation,
progression and maintenance of cancer was investigated. The aim of
the present study was to investigate the levels of cell growth and
apoptosis following simultaneous silencing of β-catenin and STAT3
genes by siRNAs in HepG2 liver cancer cells.
Materials and methods
Cells and reagents
HepG2 liver cancer cells were purchased
from the cell bank of the Chinese Academy of Sciences (Shanghai,
China) and cultured in Dulbecco’s minimum Eagle’s medium (DMEM)
with high glucose (Gibco-BRL, Invitrogen Life Technologies,
Carlsbad, CA, USA) supplemented with 10% (vol/vol) fetal calf serum
(FCS; TBD Biotechnology Corporation, Tianjin, China) at 37°C in a
humidified, 5% carbon dioxide atmosphere. Lipofectamine™ 2000 was
purchased from Invitrogen Life Technologies, and an apoptosis kit
was purchased from BD Biosciences (Franklin Lakes, NJ, USA). Small
interfering RNAs (siRNA) against β-catenin or STAT3 were
synthesized by Shanghai Genepharma (Shanghai, China). The siRNA
sequences directed against the genes were as follows: β-catenin
sense, 5′-GGGUUCAGAUGAUAUAAAUTT-3′ and anti-sense,
5′-AUUUAUAUCAUCUGAACCCAG-3′; STAT3 sense,
5′-CAUCUGCCUAGAUCGGCUAdTdT-3′ and anti-sense,
5′-UAGCCGAUCUAGGCAGAUGdTdT-3′. The scrambled siRNA control sequence
was: sense, 5′-UUCUCCGAACGUGUCACGUTT-3′ and anti-sense,
5′-ACGUGACACGUUCGGAGAATT-3′. The mouse monoclonal β-actin (1:200;
cat. no. sc-47778), mouse monoclonal β-catenin (1:200; cat. no.
sc-7963) and rabbit polyclonal STAT3 (1:200; sc-7179) antibodies
were purchased from Santa Cruz Biotechnology, Inc. (Dallas, TX,
USA). The rabbit polyclonal caspase-3 (1:1,000; cat. no. 9662),
rabbit monoclonal cleaved caspase-3 (1:1,000; cat. no. 9664),
rabbit polyclonal poly(ADP-ribose) polymerase (PARP; 1:1,000; cat.
no. 9542) and rabbit monoclonal cleaved PARP (1:1,000; cat. no.
5625) antibodies were purchased from Cell Signaling Technology
(Danvers, MA, USA). The secondary antibodies, horseradish
peroxidase (HRP)-conjugated goat anti-mouse (1:5,000; cat. no.
sc-2005) and goat anti-rabbit (1:5,000; sc-2004), were also
purchased from Santa Cruz Biotechnology, Inc.
Transient transfection
A total of 3×105 cells were plated in
six-well plates in triplicate and grown to 30–50% confluency. For
transfection, 100 nM siRNA in 10 µl Lipofectamine™ 2000 was
administered to each well containing 2 ml DMEM without FCS. At 6 h
after the transfection, the media was replaced with DMEM containing
10% FCS. After 72 h, the cells were harvested and the proteins were
isolated using radioimmunoprecipitation assay buffer (Solarbio
Technology, Beijing, China) supplemented with a protease inhibitor
(phenylmethylsulfonyl fluoride; Solarbio Technology) and
phosphatase inhibitors (Applygen Technologies, Inc., Beijing,
China). In the co-transfection group, 100 nM siRNA against
β-catenin and 100 nM siRNA against STAT3 were transfected into
cells with Lipofectamine™ 2000 according to the manufacturer’s
instructions. All experiments were performed in triplicate and
representative results are presented.
Determination of hepatocellular carcinoma
(HCC) cell growth using an MTT assay
A total of 1.5×104 cells were plated in
96-well plates in triplicate and were grown to 30–50% confluency at
the time of transfection. The siRNA-Lipo-fectamine™ 2000 complex
(50 µl) was administered to each well containing 100
µl DMEM without FCS. The medium was replaced with DMEM
containing 10% (vol/vol) FCS at 6 h of transfection. MTT (20
µl; 5 mg/ml; Solarbio Technology) diluted in
phosphate-buffered saline (PBS; Beyotime Institute of
Biotechnology, Shanghai, China) was added to the medium at 24, 48
and 72 h after the transfection. After an incubation for 4 h, the
medium was removed and the cells remained at the bottom of the
wells. Dimethyl sulfoxide (200 µl; Solarbio Technology) was
added to each well to dissolve the formazan crystals in the cells.
The absorbance was measured using a microplate reader (MK3; Thermo
Labsystems Inc., Beverly, MA, USA) at 540 nm to determine the
quantities of viable cells. All experiments were performed in
triplicate. Data were normalized to their respective controls and
presented as a bar graph.
Detection of apoptosis by flow
cytometry
Cell apoptosis was detected using an annexin
V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) apoptosis
detection kit (BD Biosciences). At 72 h post-transfection, the
cells were collected, centrifuged three times at 1,000 × g for 5
min, and resuspended in 500 µl 1X binding buffer. The cell
number was adjusted to 1×106 cells/ml. The cells (100
µl) were placed in a tube, annexin V-FITC (5 µl) and
PI (5 µl) were added and then the cell suspension was
incubated for 15 min in darkness at room temperature. Staining of
cells was immediately quantified using a flow cytometer (FACSCanto™
II; BD Biosciences). For each determination, a minimum of 50,000
cells were analyzed. Each experiment was performed three times.
Western blot analysis
The cells were lysed in radioimmunoprecipitation
assay buffer supplemented with with a protease inhibitor
(phenylmethylsulfonyl fluoride) and phosphatase inhibitors on ice
for 15 min. The protein concentrations were determined using the
bicinchoninic acid assay (Beyotime Institute of Biotechnology). A
total of 30 µg total proteins were separated using 10%
SDS-PAGE (Beyotime Institute of Biotechnology) and then transferred
onto polyvinylidene difluoride membranes (Millipore, Billerica, MA,
USA). The membranes were blocked with skimmed milk (Beyotime
Institute of Biotechnology) in PBS at room temperature for 1 h, and
then incubated with specific antibodies (1:200 or 1:1,000 dilution)
at 4°C overnight. On the following day, the membranes were washed
three times with PBS, followed by incubation with HRP-conjugated
secondary antibodies (1:5,000 dilution) at 37°C for 1 h. Following
washing with PBS, the bands were developed in a solution of
3,3′-diamino-benzidine (Santa Cruz Biotechnology, Inc.). Once the
bands were visible on the membranes, the color development was
stopped and the membranes were rinsed in distilled water. Images
were captured using a gel imaging system (Champgel 5500; Beijing
Sage Creation Science and Technology Ltd., Beijing, China) and
analyzed using Quantity One 4.5.2 software (Bio-Rad, Hercules, CA,
USA). The quantitative results of gray-scale analysis were
subjected to statistical analysis.
Statistical analysis
Values are expressed as the mean ± standard
deviation, and were compared using the Student’s t-test and
analysis of variance. Statistical analyses were conducted using
GraphPad Prism 5.0 (GraphPad Software, Inc., La Jolla, CA, USA).
P<0.05 was considered to indicate a statistically significant
difference.
Results
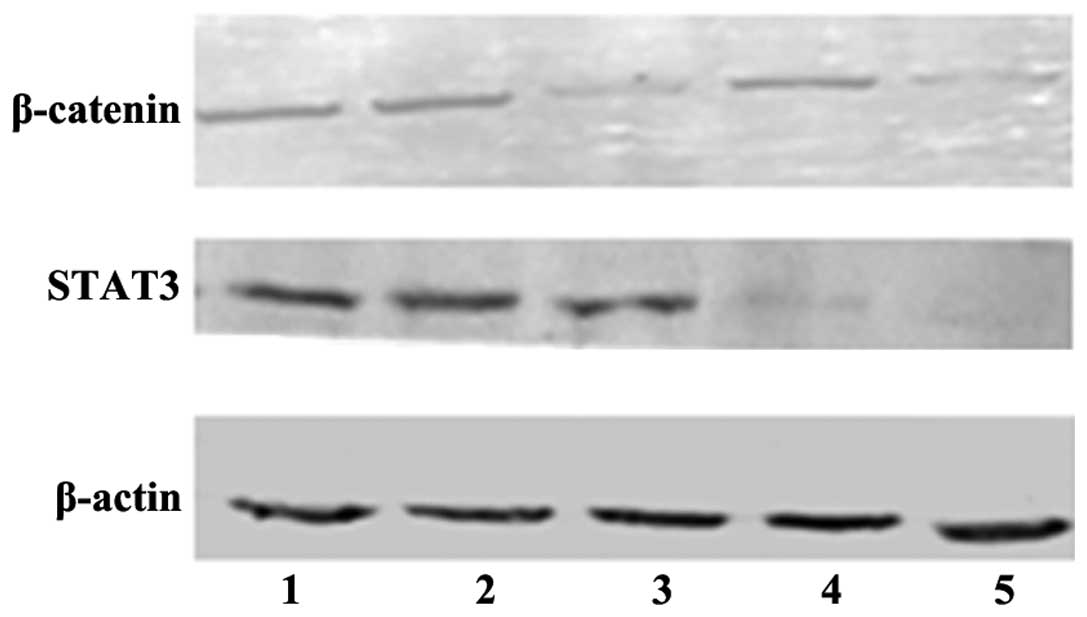
Efficient downregulation of β-catenin and
STAT3 proteins by siRNAs in HepG2 cells
Following siRNA transfection in HepG2
cells for 72 h, the β-catenin and STAT3 protein levels were
determined by western blot analysis. β-catenin was efficiently
decreased in the β-catenin transfection group and in the β-catenin
and STAT3 co-transfection group after 72 h (P<0.05). Similarly,
STAT3 siRNA downregulated STAT3 protein expression in the STAT3
transfection group and in the β-catenin and STAT3 co-transfection
group (P<0.05; Fig. 1).
Accordingly, HepG2 cells transfected with these siRNAs
for 72 h were used for subsequent analysis.
Simultaneous silencing of β-catenin and
STAT3 by siRNAs enhances the loss of HCC cell viability
The cell viability of HepG2 cells was
determined following β-catenin and STAT3 silencing with siRNAs
using an MTT assay. The cell viability in the control group
exhibited no significant difference compared with that in the
negative siRNA control group; however, the transfection group
exhibited a significant decrease in the number of viable cells
(P<0.05). No significant difference was identified in the number
of viable cells between the β-catenin and STAT3 transfection
groups. By contrast, the β-catenin and STAT3 co-transfection group
revealed a more marked decrease in cell viability than the
β-catenin or STAT3 transfection groups (P<0.05; Fig. 2). The cell growth inhibition ratios
were 14.2% at 24 h, 19.6% at 48 h and 27.6% at 72 h (β-catenin
transfection group); 11.4% at 24 h, 17.6% at 48 h and 24.9% at 72 h
(STAT3 transfection group); and 19.6% at 24 h, 27.1% at 48 h and
39.9% at 72 h (β-catenin and STAT3 co-ransfection group). These
results revealed that the growth inhibition rate in the β-catenin
and STAT3 co-transfection group was greater than that in the groups
transfected with β-catenin or STAT3 alone, particularly at 72 h.
These results suggested that simultaneous inhibition of β-catenin
and STAT3 resulted in an enhanced reduction of the cell viability
of liver cancer cells.
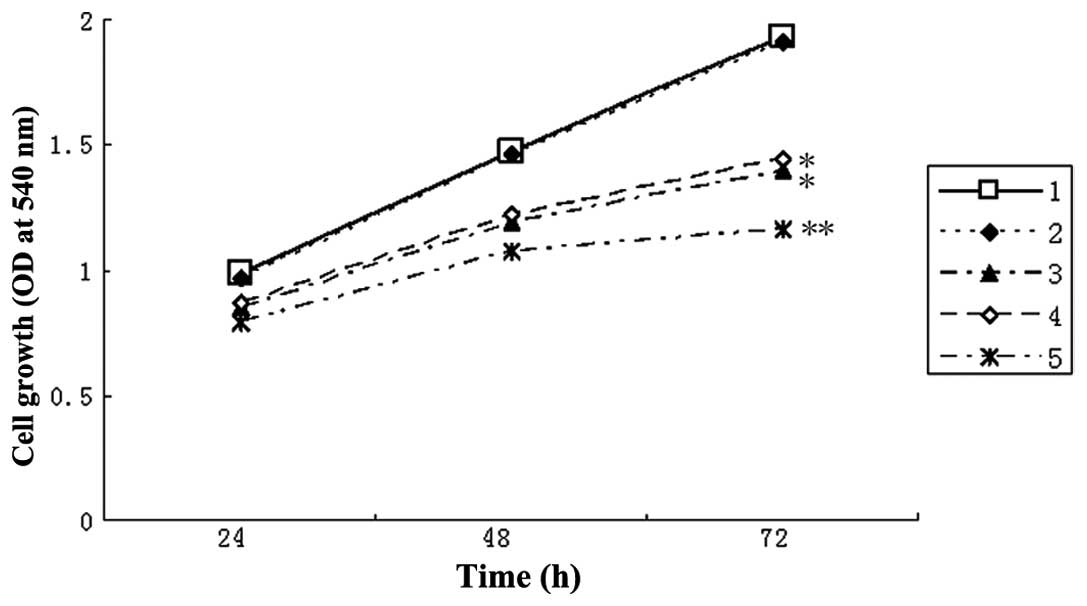 | Figure 2Simultaneous inhibition of β-catenin
and STAT3 results in enhanced reduction of cell viability of
HepG2 cells. HepG2 cells were transfected
with β-catenin siRNA and STAT3 siRNA alone or in combination with
Lipofectamine™ 2000 for the indicated times. The cell viability was
assessed using an MTT assay. 1, Control group; 2, negative siRNA
control group; 3, β-catenin transfection group; 4, STAT3
transfection group; 5, β-catenin and STAT3 co-transfection group.
*P<0.05, compared with the control group;
**P<0.05, compared with the control group and the groups
transfected with β-catenin or STAT3 alone. STAT3, signal transducer
and activator of transcription 3; siRNA, small interfering RNA; OD,
optical density. |
Co-transfection of siRNAs targeting
β-catenin and STAT3 increases cell apoptosis in HCC cells
Cell apoptosis was assessed following siRNA
transfection for 72 h using flow cytometry. Compared with that the
control group, the early apoptotic rates of the three transfection
groups were significantly increased (P<0.05). However, the early
apoptotic rate was not significantly different between the
β-catenin siRNA and STAT3 siRNA transfection groups. In addition,
the early and late apoptotic rates of the β-catenin and STAT3
co-transfection group revealed a more marked increase than those of
the β-catenin or STAT3 transfection group alone (P<0.05;
Fig. 3).
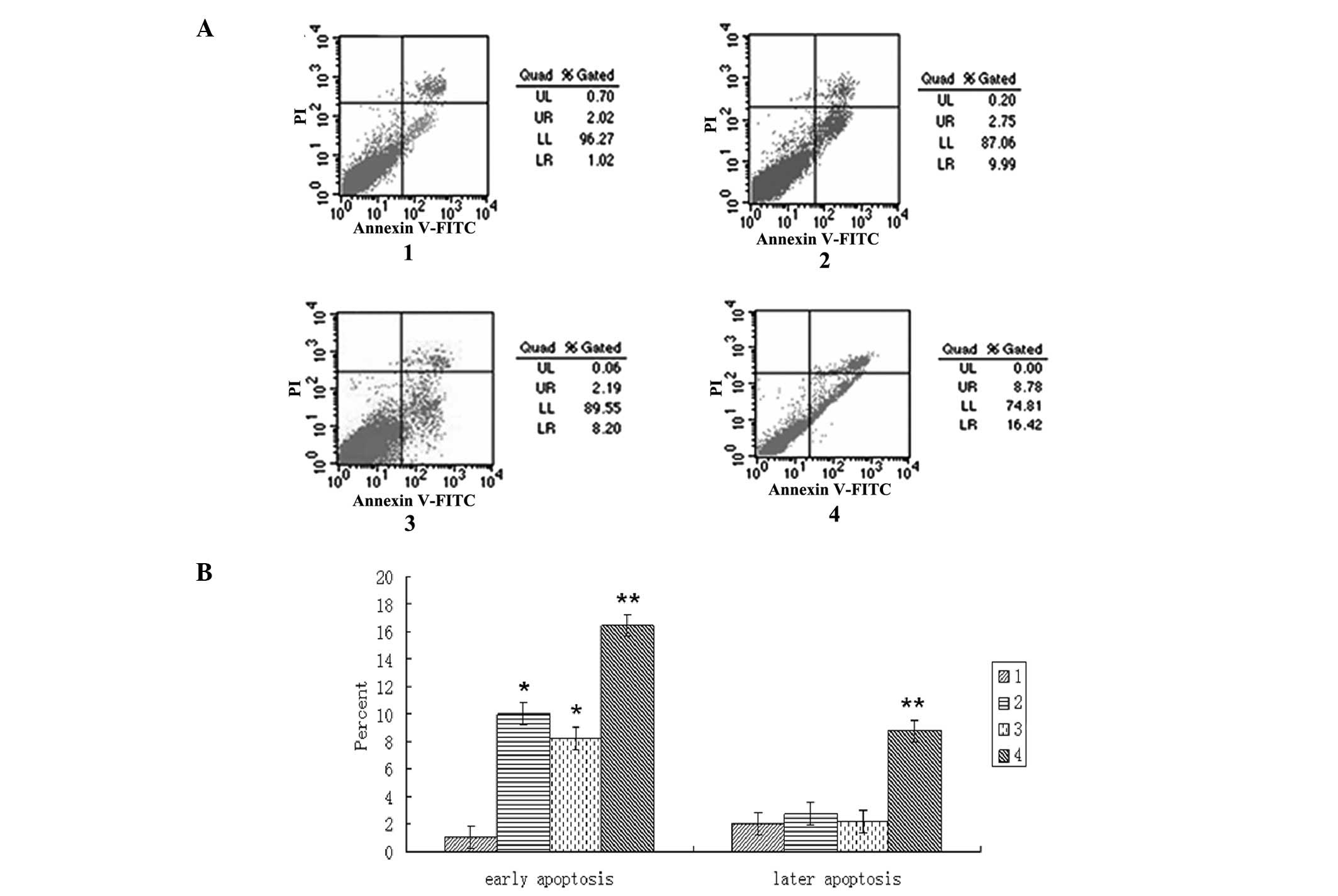 | Figure 3Co-transfection of siRNAs targeting
β-catenin and STAT3 leads to increased apoptosis in
HepG2 cells. HepG2 cells were transfected
with β-catenin siRNA and STAT3 siRNA alone or in combination using
Lipofectamine™ 2000 for 72 h. Cell apoptosis was determined by PI
and annexin V-FITC staining followed by flow cytometric analysis.
(A) Viable cells were negative for PI and annexin V-FITC, apoptotic
cells were positive for annexin V-FITC and negative for PI, whereas
late apoptotic/dead cells were positive for annexin V-FITC and PI
staining. Non-viable cells, which underwent necrosis, were positive
for PI but negative for annexin V-FITC. In the scatter diagram, the
upper left quadrant represents mechanically damaged cells, the
upper right quadrant represents late apoptotic cells, the lower
left quadrant represents viable cells and the lower right quadrant
represents early apoptotic cells. (B) Data were normalized to an
internal control and expressed as the mean ± standard deviation.
The Y-axis indicates the ratio of early and late apoptosis,
respectively. *P<0.05, compared with the control
group; **P<0.05, compared with the control group and
the groups transfected with β-catenin or STAT3 alone. 1, Control
group; 2, β-catenin transfection group; 3, STAT3 transfection
group; 4, β-catenin and STAT3 co-transfection group. STAT3, signal
transducer and activator of transcription 3; siRNA, small
interfering RNA; FITC, fluorescein isothiocyanate; PI, propidium
iodide. |
Simultaneous downregulation of β-catenin
and STAT3 by siRNAs synergistically promotes the cleavage of
caspase-3 and PARP
Finally, the protein expression levels of the cell
apoptosis markers caspase-3, cleaved caspase-3, PARP and cleaved
PARP were determined using western blot analysis following siRNA
transfection for 72 h. The caspase-3 and PARP protein levels
decreased in the three transfection groups, while no significant
difference was observed between the β-catenin and STAT3
transfection groups alone. However, the caspase-3 and PARP protein
levels were more markedly decreased in the β-catenin and STAT3
co-transfection group (P<0.05). By contrast, the cleaved
caspase-3 and cleaved PARP protein levels increased in the three
transfection groups; however, they exhibited no significant
difference between the β-catenin and STAT3 transfection groups
alone. Increased cleavage of caspase-3 and PARP protein was clearly
observed in the β-catenin siRNA and STAT3 siRNA co-transfection
group (P<0.05; Fig. 4).
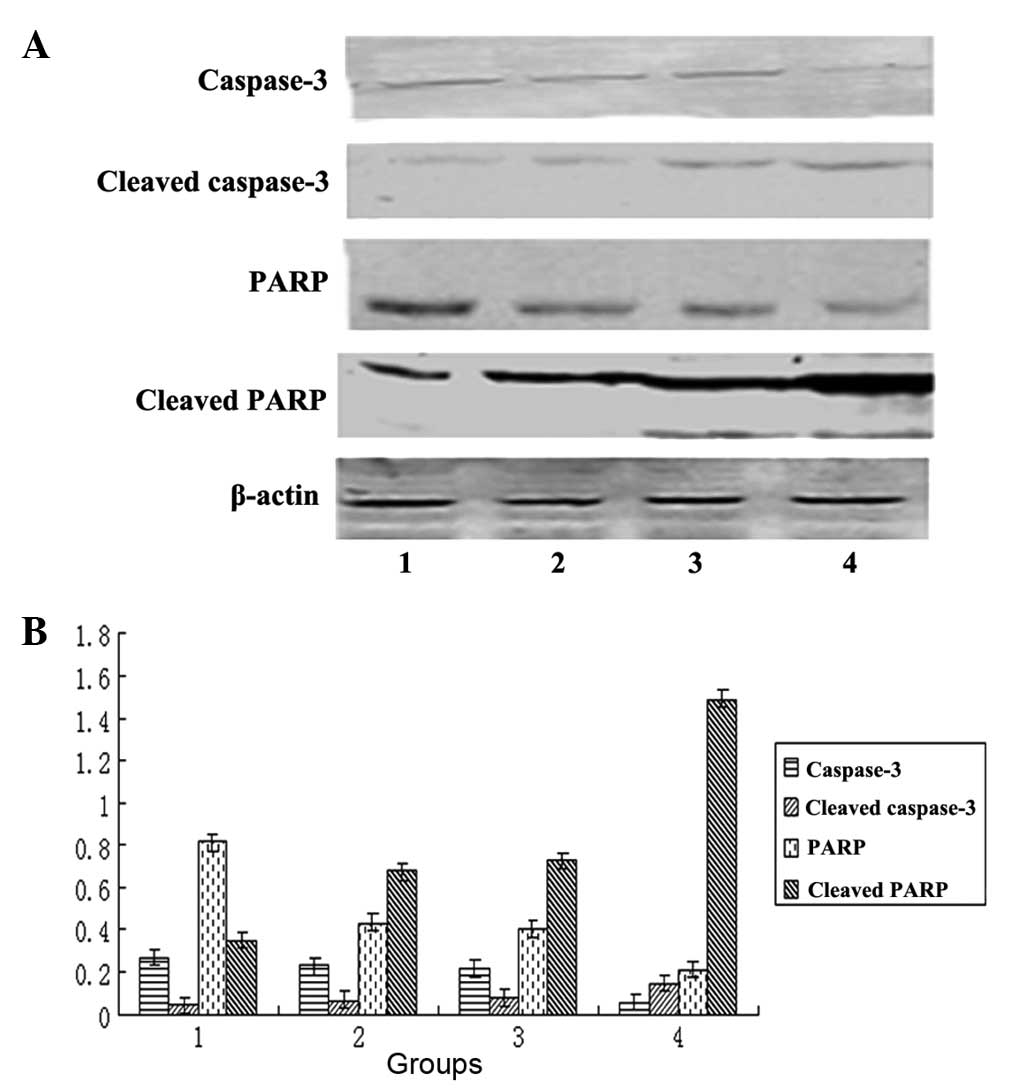 | Figure 4Co-transfection of β-catenin and STAT3
siRNAs synergistically promotes the cleavage of caspase-3 and PARP.
HepG2 cells were transfected with β-catenin siRNA and
STAT3 siRNA alone or in combination using Lipofectamine™ 2000 for
72 h. (A) Immunoblots were performed to determine the protein
levels of caspase-3, cleaved caspase-3, PARP and cleaved PARP, with
β-actin as the loading control. (B) Quantification of A. Data were
normalized to an internal control (β-actin) and expressed as the
mean ± standard deviation. The X-axis indicates the following: 1,
Control group; 2, β-catenin transfection group; 3, STAT3
transfection group; 4, β-catenin and STAT3 co-transfection group.
The Y-axis is the grayscale ratio of the protein of interest to the
β-actin protein. *P<0.05, compared with the control
group; **P<0.05, compared with the control group and
the groups transfected with β-catenin or STAT3 alone. STAT3, signal
transducer and activator of transcription 3; siRNA, small
interfering RNA; PARP, poly(ADP-ribose) polymerase. |
Discussion
It has been well documented that the β-catenin and
STAT3 signaling pathways are important in the evasion of apoptosis
of cancer cells (15–18). The final critical stage of
apoptosis is achieved through caspase activation, in which the role
of caspase-3 is particularly important. PARP, a cell death
substrate, protects the structural integrity of chromosomes and is
involved in DNA repair (19). PARP
was the first identified substrate of caspase-3 during apoptosis
and is the most characteristic proteolytic substrate (20). Following DNA damage, PARP is
activated, and rapidly recognizes and binds to damaged DNA sites to
promote DNA repair (21).
In the present study, β-catenin and STAT3 protein
expression levels were markedly inhibited by siRNA transfection for
72 h, demonstrating that the transfection was effective. Silencing
β-catenin and STAT3 gene expression inhibited HCC cell growth and
promoted apoptosis. In addition, silencing β-catenin and STAT3 gene
expression decreased the caspase-3 and PARP protein levels and
increased the cleavage of caspase-3 and PARP proteins. The decrease
in the expression of PARP would impair DNA damage repair, thereby
promoting cell apoptosis. Similar to the present findings, in human
squamous cell lung carcinomas, overexpression of the protein
inhibitor of activated STAT3 promoted mitochondrial depolarization,
leading to cytochrome C release, caspase-9 and caspase-3
activation, as well as PARP cleavage (22). In glioma cells (23), ovarian cancer cells (24), a urethane-induced lung tumor model
(25), human renal cell carcinoma
and melanoma cell lines (26),
inhibition of STAT3 activity has been revealed to elevate the
levels of cleaved caspase-3 and PARP, which is concordant with the
results of the present study. In addition, β-catenin signaling is
involved in the regulation of cell growth and apoptosis in breast
cancer (27). Accordingly,
inhibition of β-catenin signaling induces apoptosis through
activation of caspase-3, induction of B-cell lymphoma 2
(Bcl-2)-associated X protein (Bax), inhibition of Bcl-2 and
cleavage of PARP in gastric cancer cells (28). In addition, significant
dephosphorylation of glycogen synthase kinase-3β at serine 9
reduced the activation of caspase-3, as well as Bax and PARP
cleavage in human neural progenitor cells (29).
In the present study, the alterations in caspase-3,
cleaved caspase-3, PARP and cleaved PARP protein levels in the
β-catenin and STAT3 co-transfection group were more marked than
those in the β-catenin or STAT3 siRNA alone transfection group. In
addition, the ratios of liver cancer cell growth inhibition and
apoptosis in the β-catenin and STAT3 co-transfection group were
significantly greater than those in the groups transfected with
β-catenin or STAT3 siRNA alone. Similar to the results of the
present study, simultaneous silencing of vascular endothelial
growth factor, telomerase reverse transcriptase and Bclextra large
expression improved the inhibition of cell growth and promotion of
apoptosis in laryngeal squamous carcinoma (30).
The tumorigenesis of liver cancer is the result of
the collaboration of multiple pathways. Inhibiting the functions of
the key genes of several pathways together is expected to achieve
improved results for the purpose of cancer therapy (30). In agreement with this hypothesis,
it was demonstrated that silencing β-catenin and STAT3 genes
together led to enhanced cell apoptosis and loss of cell viability
of HCC cells as compared with silencing of β-catenin and STAT3
genes individually. The present study provided proof-of-concept
that targeting several key components of different pathways
together may be a potential strategy for future development of
anti-cancer drugs.
In conclusion, it was identified that simultaneous
silencing of β-catenin and STAT3 by siRNAs resulted in enhanced
loss of cell viability and induction of apoptosis of liver cancer
cells. The present study provides a basis for further preclinical
and clinical development of anti-cancer therapeutic strategies
targeting several cancer growth-promoting signaling pathways
simultaneously.
Acknowledgments
The present study was supported by the Health
Department of Heilongjiang Province, China.
References
|
1
|
Yamakado K and Kudo M: Treatment
strategies of intermediate-stage hepatocellular carcinomas in Japan
(Barcelona Clinic Liver Cancer stage B). Oncology. 87(Suppl 1):
78–81. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chan LH, Luk ST and Ma S: Turning hepatic
cancer stem cells inside out - a deeper understanding through
multiple perspectives. Mol Cells. Feb 4–2015.Epub ahead of print.
View Article : Google Scholar
|
|
3
|
Wang XH, Sun X, Meng XW, et al: β-catenin
siRNA regulation of apoptosis- and angiogenesis-related gene
expression in hepatocellular carcinoma cells: potential uses for
gene therapy. Oncol Rep. 24:1093–1099. 2010.PubMed/NCBI
|
|
4
|
Zulehner G, Mikula M, Schneller D, et al:
Nuclear beta-catenin induces an early liver progenitor phenotype in
hepatocellular carcinoma and promotes tumor recurrence. Am J
Pathol. 176:472–481. 2010. View Article : Google Scholar :
|
|
5
|
Li W, Tong H, Huang X, et al: High levels
of β-catenin promote IFNγ-induced apoptosis in hepatocellular
carcinoma cells. Oncol Lett. 4:1092–1096. 2012.PubMed/NCBI
|
|
6
|
Guturi KK, Mandal T, Chatterjee A, et al:
Mechanism of β-catenin-mediated transcriptional regulation of
epidermal growth factor receptor expression in glycogen synthase
kinase 3 β-inactivated prostate cancer cells. J Biol Chem.
287:18287–18296. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang XH, Meng XW, Sun X, et al:
Wnt/β-catenin signaling regulates MAPK and Akt1 expression and
growth of hepatocellular carcinoma cells. Neoplasma. 58:239–244.
2011. View Article : Google Scholar
|
|
8
|
Tian XH, Hou WJ, Fang Y, et al: XAV939, a
tankyrase 1 inhibitior, promotes cell apoptosis in neuroblastoma
cell lines by inhibiting Wnt/β-catenin signaling pathway. J Exp
Clin Cancer Res. 32:1002013. View Article : Google Scholar
|
|
9
|
Liu YZ, Wu K, Huang J, et al: The
PTEN/PI3K/Akt and Wnt/β-catenin signaling pathways are involved in
the inhibitory effect of resveratrol on human colon cancer cell
proliferation. Int J Oncol. 45:104–112. 2014.PubMed/NCBI
|
|
10
|
Fletcher S, Turkson J and Gunning PT:
Molecular approaches towards the inhibition of the signal
transducer and activator of transcription 3 (Stat3) protein.
ChemMedChem. 3:1159–1168. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Morikawa T, Baba Y, Yamauchi M, et al:
STAT3 expression, molecular features, inflammation patterns and
prognosis in a database of 724 colorectal cancers. Clin Cancer Res.
17:1452–1462. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Park KW, Kundu J, Chae IG, Kim DH, Yu MH,
Kundu JK and Chun KS: Carnosol induces apoptosis through generation
of ROS and inactivation of STAT3 signaling in human colon cancer
HCT116 cells. Int J Oncol. 44:1309–1315. 2014.PubMed/NCBI
|
|
13
|
Song X, Wang J, Zheng T, et al: LBH589
Inhibits proliferation and metastasis of hepatocellular carcinoma
via inhibition of gankyrin/STAT3/Akt pathway. Mol Cancer.
12:1142013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tsang CM, Cheung YC, Lui VW, et al:
Berberine suppresses tumorigenicity and growth of nasopharyngeal
carcinoma cells by inhibiting STAT3 activation induced by tumor
associated fibroblasts. BMC Cancer. 13:6192013. View Article : Google Scholar
|
|
15
|
Li W, Huang X, Tong H, et al: Comparison
of the regulation of β-catenin signaling by type I, type II and
type III interferons in hepatocellular carcinoma cells. PLoS One.
7:e470402012. View Article : Google Scholar
|
|
16
|
Zhang Y, Du XL, Wang CJ, et al: Reciprocal
activation between PLK1 and Stat3 contributes to survival and
proliferation of esophageal cancer cells. Gastroenterology.
142:521–530.e3. 2012. View Article : Google Scholar
|
|
17
|
Yang F, Jove V, Buettner R, et al:
Sorafenib inhibits endogenous and IL-6/S1P induced JAK2-STAT3
signaling in human neuroblastoma, associated with growth
suppression and apoptosis. Cancer Biol Ther. 13:534–541. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wang XH, Liu BR, Qu B, et al: Silencing
STAT3 may inhibit cell growth through regulating signaling pathway,
telomerase, cell cycle, apoptosis and angiogenesis in
hepatocellular carcinoma: potential uses for gene therapy.
Neoplasma. 58:158–171. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ogiwara H, Ui A, Shiotani B, et al:
Curcumin suppresses multiple DNA damage response pathways and has
potency as a sensitizer to PARP inhibitor. Carcinogenesis.
34:2486–2497. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yuan K, Sun Y, Zhou T, McDonald J and Chen
Y: PARP-1 regulates resistance of pancreatic cancer to TRAIL
therapy. Clin Cancer Res. 19:4750–4759. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Booth L, Cruickshanks N, Ridder T, et al:
PARP and CHK inhibitors interact to cause DNA damage and cell death
in mammary carcinoma cells. Cancer Biol Ther. 14:458–465. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dabir S, Kluge A, McColl K, et al: PIAS3
activates the intrinsic apoptotic pathway in non-small cell lung
cancer cells independent of p53 status. Int J Cancer.
134:1045–1054. 2014. View Article : Google Scholar :
|
|
23
|
Swiatek-Machado K, Mieczkowski J,
Ellert-Miklaszewska A, et al: Novel small molecular inhibitors
disrupt the JAK/STAT3 and FAK signaling pathways and exhibit a
potent antitumor activity in glioma cells. Cancer Biol Ther.
13:657–670. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tierney BJ, McCann GA, Cohn DE, et al:
HO-3867, a STAT3 inhibitor induces apoptosis by inactivation of
STAT3 activity in BRCA1-mutated ovarian cancer cells. Cancer Biol
Ther. 13:766–775. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lee NJ, Choi DY, Song JK, et al:
Deficiency of C-C chemokine receptor 5 suppresses tumor development
via inactivation of NF-κB and inhibition of monocyte
chemoattractant protein-1 in urethane-induced lung tumor model.
Carcinogenesis. 33:2520–2528. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bill MA, Nicholas C, Mace TA, et al:
Structurally modified curcumin analogs inhibit STAT3
phosphorylation and promote apoptosis of human renal cell carcinoma
and melanoma cell lines. PLoS One. 7:e407242012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xue M, Ge Y, Zhang J, et al: Fucoidan
inhibited 4T1 mouse breast cancer cell growth in vivo and in vitro
via downregulation of Wnt/β-catenin signaling. Nutr Cancer.
65:460–468. 2013. View Article : Google Scholar
|
|
28
|
Kundu J, Wahab SM, Kundu JK, et al: Tob1
induces apoptosis and inhibits proliferation, migration and
invasion of gastric cancer cells by activating Smad4 and inhibiting
β-catenin signaling. Int J Oncol. 41:839–848. 2012.PubMed/NCBI
|
|
29
|
Jaeger A, Baake J, Weiss DG and Kriehuber
R: Glycogen synthase kinase-3beta regulates differentiation-induced
apoptosis of human neural progenitor cells. Int J Dev Neurosci.
31:61–68. 2013. View Article : Google Scholar
|
|
30
|
Wang Y, Tao ZZ, Chen SM, et al:
Application of combination of short hairpin RNA segments for
silencing VEGF, TERT and Bcl-xl expression in laryngeal squamous
carcinoma. Cancer Biol Ther. 7:896–901. 2008. View Article : Google Scholar : PubMed/NCBI
|