Introduction
Gliomas are among the most common brain malignant
tumors and account for ~80% of cancers in the central nervous
system. Over previous decades, the 5-year survival rate of high WHO
grade gliomas, including glioblastoma, was only 2% with a median
survival rate of 1 year (1,2).
Furthermore, since malignant glioma is resistant to radiotherapy,
chemotherapy and adjuvant therapies, its prognosis has not improved
for a long time (3). Thus, the
development of effective therapeutic strategies for the treatment
of glioblastoma are required.
MicroRNAs (miRNAs), endogenous non-coding small RNAs
of 19–25 nucleotides in length, regulate gene expression through
binding to the 3′ untranslated region (UTR) of target mRNAs, which
eventually results in either mRNA degradation or translational
repression. Thus, miRNAs are important in endogenous RNA
interference (4). It is well
established that miRNAs have comprehensive biological functions and
are involved in various processes, including cell survival,
proliferation, differentiation, apoptosis and migration (5). Previously, accumulating evidence has
revealed that miRNAs are crucial in brain physiology and
tumorigenesis (6), making them
promising candidates for the therapy of malignant gliomas. In
addition, over half of the miRNAs are located in cancer-related
genomic regions and the deregulation of certain miRNAs have been
found to be closely associated with the development and progression
of certain types of cancer, by increasing the expression of certain
oncogenes or downregulating the expression of certain tumor
suppressors (7,8).
The aberrant expression of microRNA-203 (miR-203)
has been found to be decreased in various types of cancer,
including melanoma, colon cancer, prostate cancer, hepatocellular
carcinoma, esophageal squamous cell carcinoma, glioma and laryngeal
cancer (9–14). Furthermore, miR-203 has been
suggested to act as a tumor-suppressive microRNA in several types
of cancer by directly targeting certain oncogenes (15–17).
However, the detailed role of miR-203 in glioblastoma has never
been studied.
In the present study, we revealed that the high WHO
grade glioma tissues demonstrated a notably decreased miR-203 level
compared with low WHO grade glioma tissues as well as normal brain
tissues, and a decreasing tendency with increasing WHO grades.
Furthermore, the ectopic overexpression of miR-203 inhibited the
cell proliferation and invasion of U251 glioblastoma cells,
partially at least through inhibiting the protein expression of its
target, phospholipase D2 (PLD2).
Materials and methods
Reagents and materials
Fetal bovine serum (FBS), TRIzol, TaqMan Reverse
Transcription kit, TaqMan miRNA assay kit, Power SYBR-Green kit,
Lipofectamine 2000, miR-203 mimic and enhanced chemiluminescence
reagent were purchased from Thermo Fisher Scientific (Waltham, MA,
USA). Dulbecco’s modified Eagle’s medium (DMEM) was purchased from
Gibco-BRL (Grand Island, NY, USA). The QuikChange site-directed
mutagenesis kit was purchased from Stratagene (La Jolla, CA, USA).
MTT was obtained from Sigma (St. Louis, MO, USA). The pmirGLO
Dual-Luciferase miRNA target expression vector and the
Dual-Luciferase assay kit were purchased from Promega (Madison, WI,
USA). Mouse anti-PLD2 monoclonal antibody, mouse anti-GAPDH
monoclonal antibody and rabbit anti-mouse secondary antibody were
purchased from Abcam (Cambridge, UK). The transwell chamber was
obtained from Corning Inc. (Corning, NY, USA). Matrigel was
obtained from BD Biosciences (Franklin Lakes, NJ, USA). The
PcDNA3.1(+)-PLD2 plasmid was purchased from SuperBio (Changsha,
Hunan, China).
Tissue specimen collection
All protocols in the present study were approved by
the Ethics Committee of Central South University (Changsha, Hunan,
China) and informed consent was obtained from each patient
involved. Thirty-one glioma tissues of different grades (6 cases of
WHO I, 7 cases of WHO II, 9 cases of WHO III and 9 cases of WHO IV)
and 10 normal brain tissues were collected from the Department of
Neurosurgery of The First Xiangya Hospital of Central South
University (Changsha, Hunan, China). Patients with no history of
other tumors were diagnosed as having malignant gliomas and were
untreated. Following surgical removal, all tissues were immediately
frozen in liquid nitrogen and stored at −80°C until use.
Cell culture
The human glioblastoma cell line U251 was purchased
from ATCC (Manassas, VA, USA). U251 cells were cultured at 37°C in
5% CO2 in DMEM containing 10% FBS, 100 U/ml of
penicillin and 100 μg/ml of streptomycin.
Real-time reverse transcription
polymerase chain reaction (RT-PCR) assay
TRIzol reagent was used to extract total RNA, which
was then reverse transcribed into cDNA using a TaqMan Reverse
Transcription kit. A TaqMan miRNA assay kit was used to examine the
expression of mature miR-203 according to the manufacturer’s
instructions. U6 small nuclear RNA was used as an internal control.
The mRNA expression level of PLD2 was determined by a Power
SYBR-Green kit. GAPDH was used as an internal control. The
experiments were performed in triplicate. For miRNA all primer
sequences were as follows: stem-loop RT primer for miR-203: 5′-GTCG
TATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATAC GACCTAGTG-3′; miR-203,
forward 5′-GTGCAGGGTCC GAGGT-3′ and reverse
5′-GCCGCGTGAAATGTTTAGG-3′; U6, forward 5′-CTCGCTTCGGCAGCACA-3′ and
reverse 5′-AACGCTTCACGAATTTGCGT-3′. For the mRNA assay, primer
sequences used were: PLD2, forward 5′-ACTCACG GCGACTTTTCCTG-3′ and
reverse 5′-AACGGCAAATC GAGCCAGAG-3′; GAPDH, forward 5′-GGAGCGAGA
TCCCTCCAAAAT-3′ and reverse 5′-GGCTGTTG TCATACTTCTCATGG-3′.
Transfection
U251 cells (1×105) were harvested,
resuspended and seeded in a 6-well plate and cultured at 37°C in 5%
CO2 for 24 h. Lipofectamine 2000 was used to transfect
U251 cells with the miR-203 mimic, a control miRNA mimic or the
pcDNA3.1(+)-PLD2 plasmid at a final concentration of 200 nM,
according to the manufacturer’s instructions.
Western blot analysis
Tissues or cells were solubilized in cold RIPA lysis
buffer. Proteins (20 μg per lane) were separated with 12% SDS-PAGE
and transferred onto the nitrocellulose membranes, which were then
inhibited in 5% non-fat dried milk in phosphate-buffered saline
with Tween-20 (PBST) for 3 h. Following that, the membranes were
incubated at 4°C overnight with mouse anti-PLD2 monoclonal
antibodies (1:500) or mouse anti-GAPDH monoclonal antibodies
(1:500). After being washed with PBST for 5 min twice, the
membranes were incubated with rabbit anti-mouse secondary
antibodies (1:20,000) for 1 h at room temperature. Enhanced
chemiluminescence reagent was used to detect the signals on the
membranes (Media Cybernetics, Rockville, MD, USA). Image-Pro plus
software 6.0 was then applied to scan for the relative value of
protein expression, which was presented as the density ratio versus
GAPDH.
Luciferase reporter assay
To determine whether or not PLD2 was the direct
target of miR-203, a luciferase reporter assay was performed. The
3′-UTR of PLD2 with a potential target sequence of miR-203 was
cloned into the pmirGLO Dual-Luciferase miRNA target expression
vector. The mutant 3′-UTRs of PLD2 were constructed using a
site-directed mutagenesis kit, bearing a substitution of three
nucleotides (TTT to CCC) in the miR-203 target sequence. The
luciferase reporter plasmids containing wild-type 3′-UTRs or mutant
3′-UTRs of PLD2 were cotransfected with the miR-203 or control
miRNA mimic into U251 cells using Lipofectamine 2000 according to
the manufacturer’s instructions. Following transfection for 72 h,
the luciferase activity in each group was measured using the
Dual-Luciferase Assay kit, in accordance with the manufacturer’s
instructions. The experiments were performed in triplicate.
Cell proliferation assay
U251 cells (1×105) were plated in a
96-well plate and incubated at 37°C in 5% CO2 for 12,
24, 36 or 48 h. An MTT assay was performed to examine cell
proliferation rate. MTT (50 μl; 5 mg/ml) was added and then
incubated at 37°C in 5% CO2 for 4 h. The supernatant was
removed and 200 μl of dimethyl sulfoxide was added. The absorbance
was detected at 570 nm with a microplate reader (Bio-Rad, Hercules,
CA, USA). Each assay was performed in triplicate wells and repeated
three times.
Cell invasion assay
U251 cells were washed in cold PBS and harvested and
resuspended in serum-free DMEM medium. For the invasion assay,
50,000 cells were added into the upper chamber which was pre-coated
with Matrigel. Serum-free DMEM medium was added to the upper
chamber, while DMEM was added to the lower chamber containing 10%
FBS as the chemoattractant. Following 6 h of incubation at 37°C in
5% CO2, cells were fixed with 3.7% formaldehyde and then
stained with crystal violet staining solution. A cotton swab was
used to remove the cells that did not filter through the membrane.
Five fields of the lower surface of the membrane were randomly
selected under the microscope and the cells on it were counted.
Statistical analysis
SPSS 17.0 software (SPSS Inc., Chicago, IL, USA) was
used for data analysis. Data are presented as the mean ± standard
deviation. Statistical analysis was performed using one-way ANOVA
or the Student’s t-test. P<0.05 was considered to indicate a
statistically significant difference.
Results
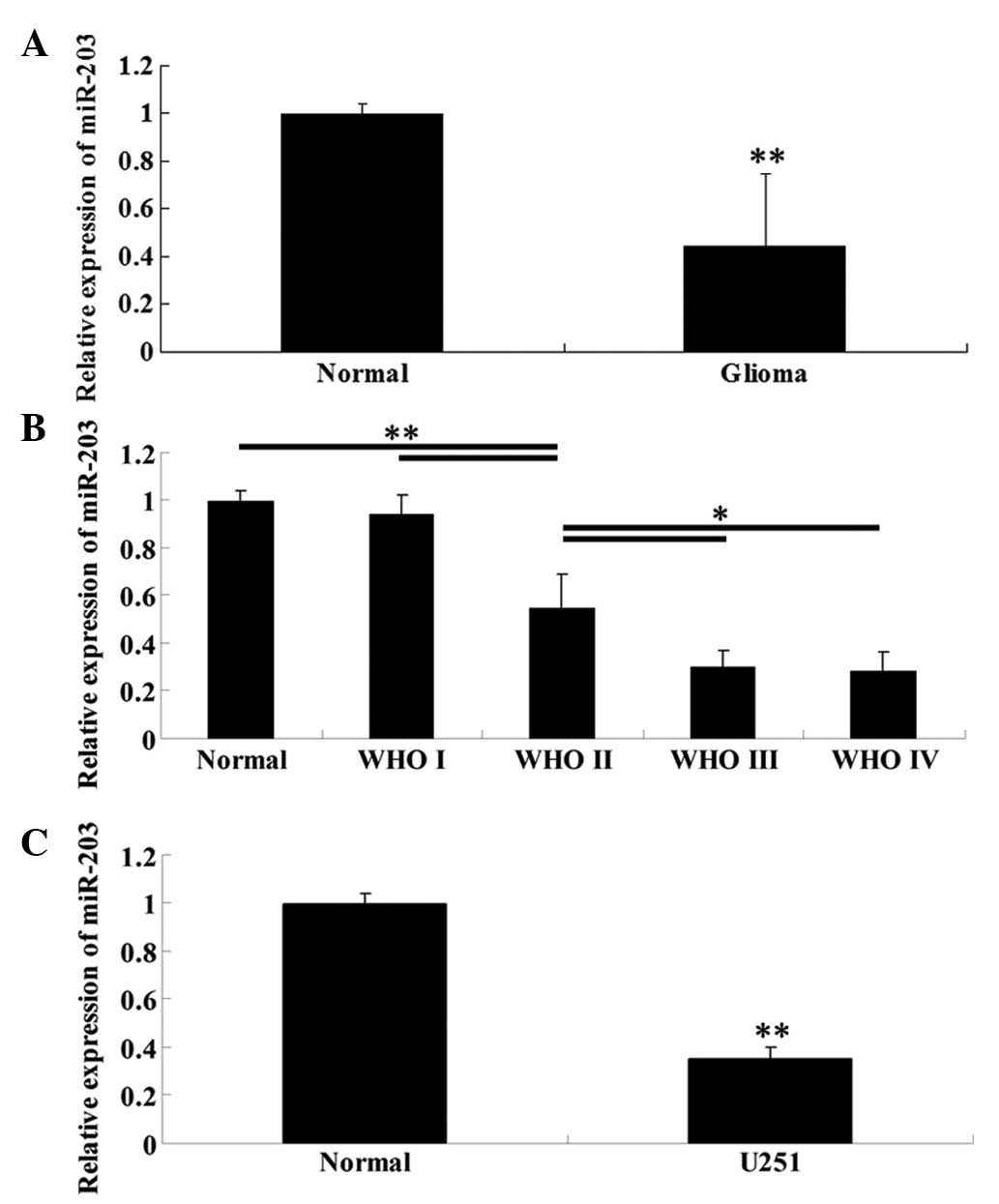
Expression of miR-203 is significantly
reduced in high WHO grade glioma tissues and glioblastoma U251
cells
To determine the expression levels of miR-203 in
glioma tissues of different WHO grades, normal brain tissues, as
well as glioblastoma U251 cells, qRT-PCR was performed. As
demonstrated in Fig. 1A, the
expression of miR-203 in glioma tissues was significantly lower
than that in normal brain tissues. Furthermore, with increasing WHO
grades, the miR-203 expression was gradually downregulated
(Fig. 1B), indicating that its
expression was negatively correlated with the malignancy of glioma.
Furthermore, consistent with the data above, U251 cells also
demonstrated a significantly decreased expression level of miR-203
than normal brain tissues (Fig.
1C).
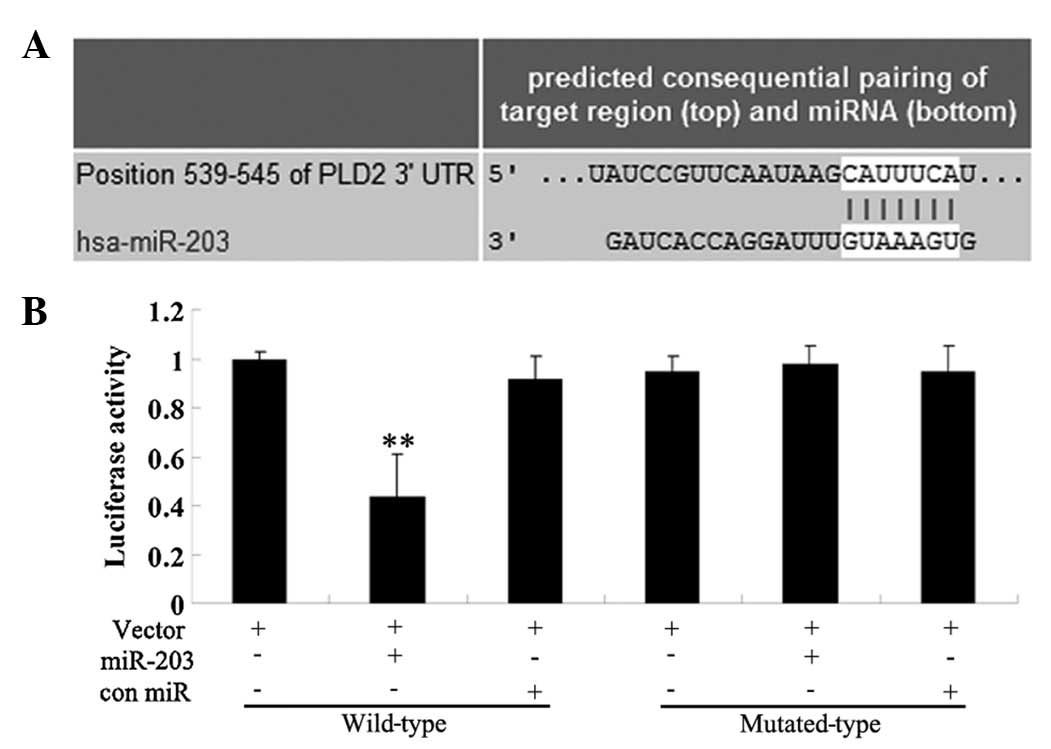
Identification of PLD2 as a direct target
of miR-203
A bioinformatical analysis was performed to predict
the potential targets of miR-203 and the results from all nine
prediction softwares predicted that PLD2 was its target, including
DIANAmT, miRanda, miRDB, miRWalk, RNAhybrid, PICTAR5, PITA, RNA22
and Targetscan (Fig. 2A). To
confirm the predictive results, we performed a luciferase reporter
assay. The results demonstrated that the luciferase activity was
significantly lower in miR-203 and the wild type 3′-UTR of PLD2
cotransfected cells, when compared with that in control cells
without transfection with miR-203. However, the luciferase activity
in cells cotransfected with miR-203 and the mutated 3′-UTR of PLD2
demonstrated no difference with the control cells (Fig. 2B). These findings demonstrated that
the 3′-UTR of PLD2 was the direct target of miR-203.
Expression levels of PLD2 are increased
in high WHO grade glioma tissues and U251 cells
The protein expression levels of PLD2 in glioma
tissues of different WHO grades and normal brain tissues were
examined. As demonstrated in Fig.
3A, the PLD2 protein expression in glioma tissues of high WHO
grades was significantly higher than that in normal brain tissues
and low WHO grade glioma tissues. Notably, with ascending WHO
grades, the PLD2 protein expression demonstrated an increasing
tendency. Furthermore, its protein level was also notably higher in
U251 cells compared with that in normal brain tissues (Fig. 3B).
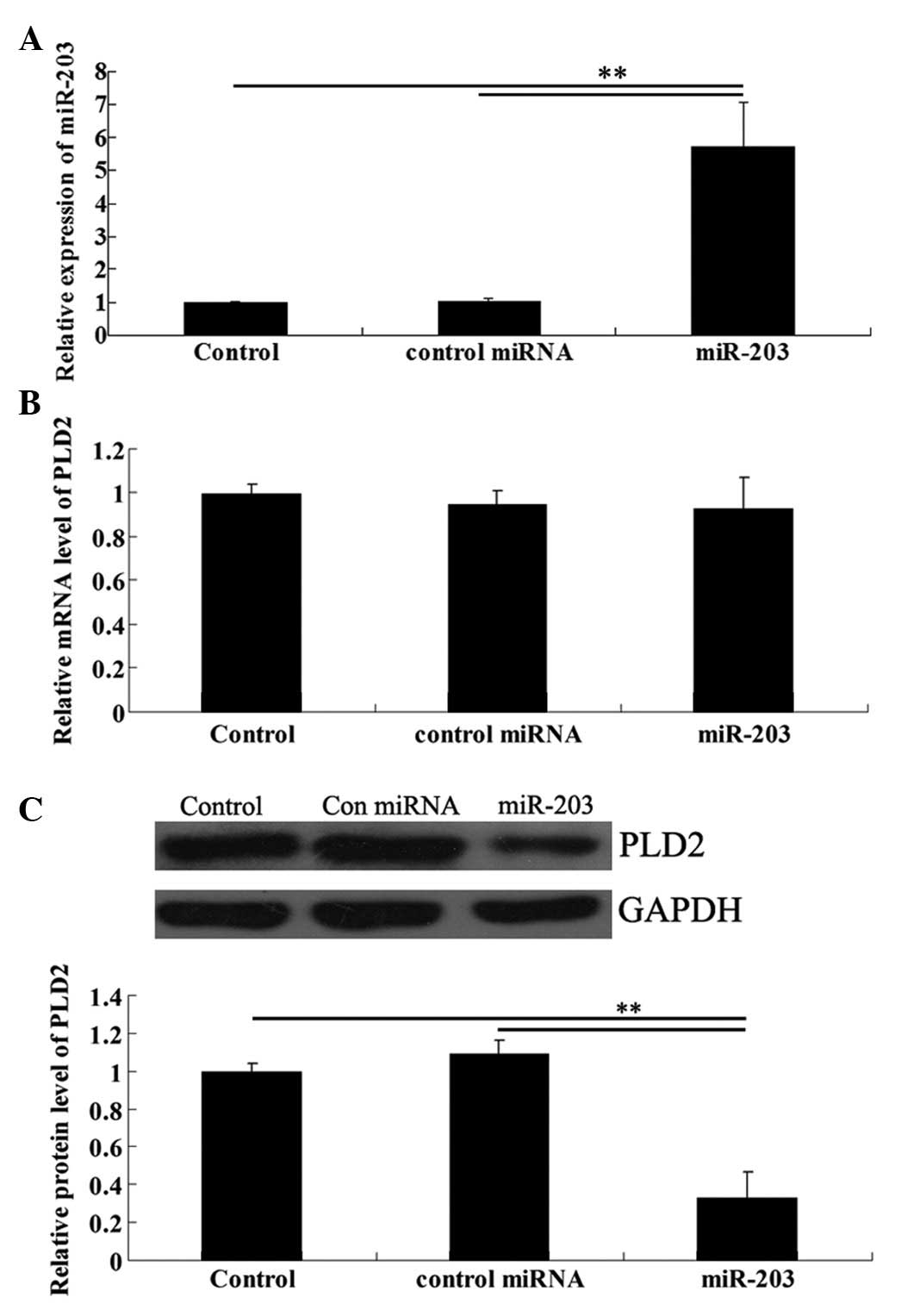
miR-203 overexpression downregulates the
protein level although not the mRNA level of PLD2 in U251
cells
We transfected U251 cells with the miR-203 mimic.
Real-time RT-PCR data confirmed that the expression level of
miR-203 was significantly upregulated following transfection
(Fig. 4A). To further investigate
the effects of miR-203 upregulation on the protein expression of
PLD2 in U251 cells, real-time RT-PCR and western blot analysis were
performed, respectively. As shown in Fig. 4B, although the mRNA level of PLD2
demonstrated no difference, the PLD2 protein level was
significantly reduced following transfection of U251 cells with
miR-203 (Fig. 4B).
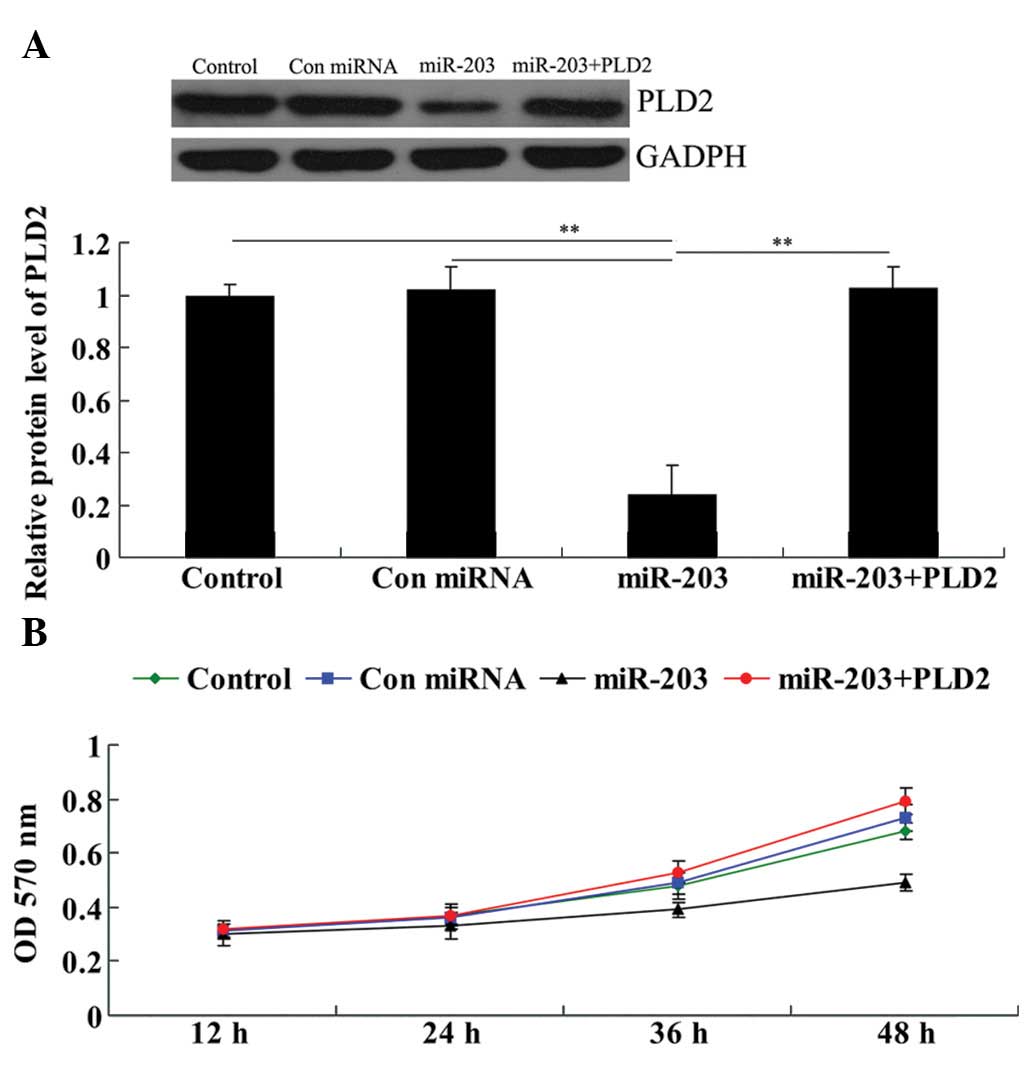
Overexpression of PLD2 significantly
attenuates the inhibitory effect of miR-203 on U251 cell
proliferation
Prior to investigating the roles of miR-203 and PLD2
in gliomas in vitro, we contransfected U251 cells with
miR-203 mimics and the PLD2 plasmid and determined whether PLD2
transfection could rescue the suppressive effect of miR-203 on PLD2
protein expression. As shown in Fig.
5A, the protein level of PLD2 in the miR-203 + PLD2 group was
much higher than that in the miR-203 group and demonstrated no
difference with the control group. These results confirmed that
PLD2 transfection rescued the suppressive effect of miR-203 on PLD2
protein expression in U251 cells.
We performed a cell proliferation assay to examine
the effects of miR-203 and PLD2 on U251 cell proliferation. As
demonstrated in Fig. 5B, in
miR-203-overexpressed U251 cells, the cell proliferation rate was
markedly downregulated. However, the miR-203 + PLD2 group
demonstrated no difference with the control group as well as the
control miRNA group, indicating that the overexpression of PLD2
abrogated the inhibitory effect of miR-203 on U251 cell
proliferation.
Overexpression of PLD2 significantly
attenuates the inhibitory effect of miR-203 on U251 cell
invasion
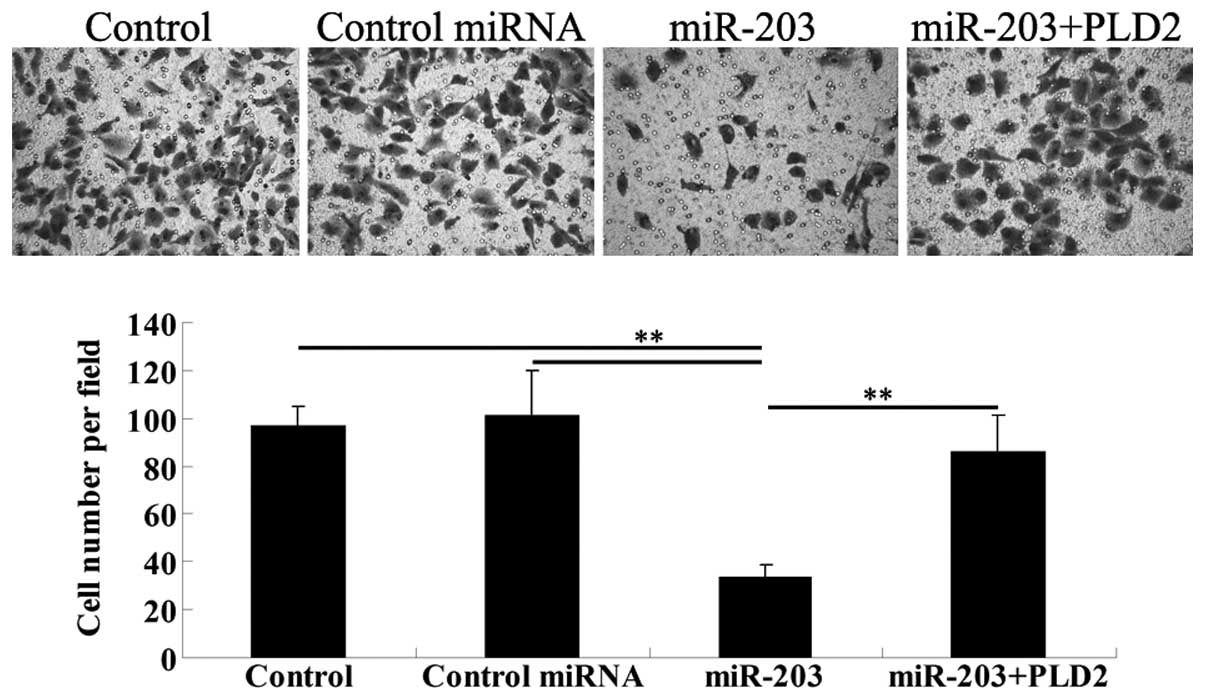
To verify our hypothesis that miR-203 inhibits the
invasion of U251 cells through a PLD2-dependent mechanism, a cell
invasion assay was performed. The results demonstrated that miR-203
significantly downregulated the cell invasion of U251 cells.
However, the overexpression of PLD2 rescued the invasion capacity
of U251 cells transfected with the miR-203 mimic (Fig. 6).
Discussion
In the present study, we revealed that high WHO
grade glioma tissues demonstrated a notably decreased miR-203 level
compared with low WHO grade glioma tissues and normal brain
tissues, as well as a decreasing tendency with increasing WHO
grades. We also demonstrated that ectopic overexpression of PLD2
inhibited the cell proliferation and invasion of glioblastoma U251
cells, at least by suppressing the PLD2 protein expression.
It is well established that the aberrant expression
of multiple miRNAs is associated with the development and
progression of gliomas, including miR-145, miR-383 and miR-34a
(18–20). Previously, He et al reported
that the expression of miR-203 was downregulated in gliomas
compared with normal brain tissues and decreased with increasing
tumor WHO grades (14), which was
consistent with our data. Furthermore, the authors also
demonstrated that miR-203 was a potential prognostic indicator for
the survival rate of patients with glioma (14). Accordingly, those as well as our
findings suggest that miR-203 is important in gliomas. However, no
previous study has revealed the exact role of miR-203 in high WHO
grade glioma cells. Thus, we performed the transfection of
glioblastoma U251 cells with the miR-203 mimic and demonstrated for
the first time, to the best of our knowledge, that miR-203
overexpression inhibited U251 cell proliferation and invasion.
These data confirm the regulatory role of miR-203 in glioblastoma
cells.
Thus far, several targets of miR-203 have been
identified, including interleukin-8, Hakai, LASP1 and Survivin
(17,21–23).
However, no target of miR-203 has been identified in gliomas. The
present study suggests that the effect of miR-203 on U251 cells is
possibly via suppressing the expression of its novel identified
target, PLD2. This gene could encode a protein which catalyzes the
hydrolysis of phosphatidylcholine to phosphatidic acid and choline
and may participate in transcriptional regulation, cell cycle
control and regulated secretion (24). Furthermore, it is crucial in cell
membrane lipid reorganization and acts as a key cell signaling
protein in leukocyte chemotaxis and phagocytosis (25).
PLD2 has been suggested to be important in several
cancer cells, including renal cancer, breast cancer, lung
adenocarcinoma, colorectal carcinoma, ovarian cancer and glioma
(26–31). For instance, a previous study
reported that the PLD2 selective inhibitor ML298 inhibited U87-MG
glioblastoma cell invasion (31).
In the present study, we demonstrated that the overexpression of
PLD2 abrogated the inhibitory effects of PLD2 on cell proliferation
and invasion in glioblastoma U251 cells. According to these
findings and ours, we suggest that PLD2 may be oncogenic in
glioblastoma. Furthermore, the exact regulatory mechanisms by which
PLD2 is involved in the development and progression of several
types of cancer have gradually been identified. For instance, Choi
et al demonstrated that PLD2 upregulation enhanced the
activity of ERK and p38 MAPK signaling pathways and further
activated STAT3, eventually increasing the expression of
anti-apoptotic Bcl-2 in human cervical cancer HeLa cells (32). In addition, COX-2 is involved in
the growth and progression of gliomas and PLD2 was reported to
enhance CoCl(2)-induced COX-2 expression via reactive oxygen
species and p38 MAPK in astroglioma cells (33). Further studies need to focus on the
molecular mechanisms underlying the role of PLD2 in gliomas.
In conclusion, the present study for the first time,
to the best of our knowledge, has demonstrated an inhibitory role
of miR-203 in glioblastoma U251 cells via targeting PLD2 and
suggests that miR-203 is a potential candidate for the treatment of
glioblastoma.
References
|
1
|
Surawicz TS, Davis F, Freels S, Laws ER Jr
and Menck HR: Brain tumor survival: results from the National
Cancer Data Base. J Neurooncol. 40:151–160. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bondy ML, Scheurer ME, Malmer B, et al:
Brain tumor epidemiology: consensus from the Brain Tumor
Epidemiology Consortium. Cancer. 113:1953–1968. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chargari C, Moncharmont C, Lévy A, et al:
Cancer stem cells, cornerstone of radioresistance and perspectives
for radiosensitization: glioblastoma as an example. Bull Cancer.
99:1153–1160. 2012.(In French).
|
|
4
|
Schepeler T: Emerging roles of microRNAs
in the Wnt signaling network. Crit Rev Oncog. 18:357–371. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yates LA, Norbury CJ and Gilbert RJ: The
long and short of microRNA. Cell. 153:516–519. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nikaki A, Piperi C and Papavassiliou AG:
Role of microRNAs in gliomagenesis: targeting miRNAs in
glioblastoma multiforme therapy. Expert Opin Investig Drugs.
21:1475–1488. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Profumo V and Gandellini P: MicroRNAs:
cobblestones on the road to cancer metastasis. Crit Rev Oncog.
18:341–355. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Farazi TA, Hoell JI, Morozov P and Tuschl
T: MicroRNAs in human cancer. Adv Exp Med Biol. 774:1–20. 2013.
View Article : Google Scholar
|
|
9
|
Noguchi S, Mori T, Otsuka Y, et al:
Anti-oncogenic microRNA-203 induces senescence by targeting E2F3
protein in human melanoma cells. J Biol Chem. 287:11769–11777.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Furuta M, Kozaki KI, Tanaka S, Arii S,
Imoto I and Inazawa J: miR-124 and miR-203 are epigenetically
silenced tumor-suppressive microRNAs in hepatocellular carcinoma.
Carcinogenesis. 31:766–776. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Schetter AJ, Leung SY, Sohn JJ, et al:
MicroRNA expression profiles associated with prognosis and
therapeutic outcome in colon adenocarcinoma. JAMA. 299:425–436.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Boll K, Reiche K, Kasack K, et al:
MiR-130a, miR-203 and miR-205 jointly repress key oncogenic
pathways and are downregulated in prostate carcinoma. Oncogene.
32:277–285. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yuan Y, Zeng ZY, Liu XH, et al:
MicroRNA-203 inhibits cell proliferation by repressing DeltaNp63
expression in human esophageal squamous cell carcinoma. BMC Cancer.
11:572011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
He J, Deng Y, Yang G and Xie W:
MicroRNA-203 down-regulation is associated with unfavorable
prognosis in human glioma. J Surg Oncol. 108:121–125. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bian K, Fan J, Zhang X, et al:
MicroRNA-203 leads to G1 phase cell cycle arrest in laryngeal
carcinoma cells by directly targeting survivin. FEBS Lett.
586:804–809. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang C, Zheng X, Shen C and Shi Y:
MicroRNA-203 suppresses cell proliferation and migration by
targeting BIRC5 and LASP1 in human triple-negative breast cancer
cells. J Exp Clin Cancer Res. 31:582012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wei W, Wanjun L, Hui S, Dongyue C, Xinjun
Y and Jisheng Z: miR-203 inhibits proliferation of HCC cells by
targeting survivin. Cell Biochem Funct. 31:82–85. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rani SB, Rathod SS, Karthik S, Kaur N,
Muzumdar D and Shiras AS: MiR-145 functions as a tumor-suppressive
RNA by targeting Sox9 and adducin 3 in human glioma cells. Neuro
Oncol. 15:1302–1316. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
He Z, Cen D, Luo X, et al: Downregulation
of miR-383 promotes glioma cell invasion by targeting insulin-like
growth factor 1 receptor. Med Oncol. 30:5572013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gao H, Zhao H and Xiang W: Expression
level of human miR-34a correlates with glioma grade and prognosis.
J Neurooncol. 113:221–228. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wei T, Xu N, Meisgen F, Ståhle M, Sonkoly
E and Pivarcsi A: Interleukin-8 is regulated by miR-203 at the
posttranscriptional level in primary human keratinocytes. Eur J
Dermatol. Apr 19–2013.(Epub ahead of print).
|
|
22
|
Abella V, Valladares M, Rodriguez T, et
al: miR-203 regulates cell proliferation through its influence on
Hakai expression. PLoS One. 7:e525682012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Takeshita N, Mori M, Kano M, et al:
miR-203 inhibits the migration and invasion of esophageal squamous
cell carcinoma by regulating LASP1. Int J Oncol. 41:1653–1661.
2012.PubMed/NCBI
|
|
24
|
Szumiło M and Rahden-Staroń I:
Phospholipase D in mammalian cells: structure, properties,
physiological and pathological role. Postepy Hig Med Dosw (Online).
60:421–430. 2006.(In Polish).
|
|
25
|
Gomez-Cambronero J: Biochemical and
cellular implications of a dual lipase-GEF function of
phospholipase D2 (PLD2). J Leukoc Biol. 92:461–467. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhao Y, Ehara H, Akao Y, et al: Increased
activity and intranuclear expression of phospholipase D2 in human
renal cancer. Biochem Biophys Res Commun. 278:140–143. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Eisen SF and Brown HA: Selective estrogen
receptor (ER) modulators differentially regulate phospholipase D
catalytic activity in ER-negative breast cancer cells. Mol
Pharmacol. 62:911–920. 2002. View Article : Google Scholar
|
|
28
|
Meacci E, Nuti F, Catarzi S, et al:
Activation of phospholipase D by bradykinin and sphingosine
1-phosphate in A549 human lung adenocarcinoma cells via different
GTP-binding proteins and protein kinase C delta signaling pathways.
Biochemistry. 42:284–292. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Saito M, Iwadate M, Higashimoto M, Ono K,
Takebayashi Y and Takenoshita S: Expression of phospholipase D2 in
human colorectal carcinoma. Oncol Rep. 18:1329–1334.
2007.PubMed/NCBI
|
|
30
|
Snider AJ, Zhang Z, Xie Y and Meier KE:
Epidermal growth factor increases lysophosphatidic acid production
in human ovarian cancer cells: roles for phospholipase D2 and
receptor transactivation. Am J Physiol Cell Physiol. 298:C163–C170.
2010. View Article : Google Scholar
|
|
31
|
O’Reilly MC, Scott SA, Brown KA, et al:
Development of dual PLD1/2 and PLD2 selective inhibitors from a
common 1,3,8-Triazaspiro[4.5]decane Core: discovery of Ml298 and
Ml299 that decrease invasive migration in U87-MG glioblastoma
cells. J Med Chem. 56:2695–2699. 2013.PubMed/NCBI
|
|
32
|
Choi HJ and Han JS: Overexpression of
phospholipase D enhances Bcl-2 expression by activating STAT3
through independent activation of ERK and p38MAPK in HeLa cells.
Biochim Biophys Acta. 1823:1082–1091. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ahn BH, Park MH, Lee YH, Kwon TK and Min
do S: Up-regulation of cyclooxygenase-2 by cobalt chloride-induced
hypoxia is mediated by phospholipase D isozymes in human
astroglioma cells. Biochim Biophys Acta. 1773:1721–1731. 2007.
View Article : Google Scholar : PubMed/NCBI
|