On the molecular origins of life
There is extensive research and available information in the scientific literature to suggest that the earliest molecule of life is RNA, based on the fact that is it structurally less complex than DNA (1,2). However, there are a number of limitations to this theory as the origin of this RNA has not been established (3). Herein, a new scenario on the onset of molecular life is proposed based on evolutionary biology, biochemistry and genetic entropy (genentropy). This hypothesis on how life began on Earth may complete the puzzle and may provide the basis for the understanding of chemistry, evolution, molecular biology and disease. The story begins almost 13.7 billion years ago with a rather ‘big bang’. After 10-32 sec, energy creates the quark soup and half a second later the 1st proton is created. At the 3-min point, there is the formation of the first helium (He) and lithium (Li) nuclei, and this continues for 380.000 years, during which time atoms are formed. The skies begin to clear, and the first galaxies are formed one billion years after the ‘big bang’. The sun is formed eight billion years later, and Earth is formed out of hot burning intergalactic dust. Planet Earth is a hot rock with no chance of life as is known by humanity. However, trapped energy in the form of heat is released from Earth into the vast space and the planet begins to cool down. Water is condensed and radiation whips the surface of the planet, as there is still no life, no atmosphere and there is only a fraction of the life-giving oxygen that came after cyanobacteria and plants made Earth green. At this stage, there are only elements whirling around in the prehistoric ‘soup’ that is basically an aqueous solution that is exposed to high-energy radiation due to the lack of an atmosphere. These free elements, and as a matter of fact, each and every single element of this early version of Earth, were created long ago in the hearts of exploding stars. The forces of nature that were created with the ‘big bang’ are still ongoing. Thermodynamics and entropy rule and shape the world. Elements bump on each other and eventually, after an unimaginable number of random collisions, pure statistics and crude universal forces, create the first organized molecule of carbons, the building blocks of life as is known.
These universal forces are still ongoing today. These are the forces that push water crystals to form hexagonal shapes. These are the forces that dictate bees to create hexagonal hives, as a trade-off between the optimal 3-dimensional arrangement exploitation and minimal building effort from the bees (4). Benzene rings are the most abundant carbon rings in nature for the exact same reason. Primitive forces shaped the world through the laws of thermodynamics (5). Thus, at this stage of the Earth's chemical history, a critical point is achieved, where the existence of cyclohexanes can be rationalized in the prehistoric soup. There is solid proof in organic cyclization chemistry literature that very conveniently provides the missing link as to how the first cyclohexanes originated (6). Two carbon atoms (methane molecules) established a covalent bond that turned them into an ethane molecule (a simple two carbon atom molecule). Another one then came along, and propane was made, and this went on randomly and for a very long period of time, at which an isoprene unit and other unsaturated hydrocarbon structures could have also been formed. Important though is the question on what is the optimal number of carbon atoms linked together in an aliphatic chain that are able to form a cyclic compound. In order to investigate this, molecular dynamics (MD) simulations were performed using all aliphatic chain lengths from 2 to 12 carbon atoms. Unrestrained MD simulations were carried out using the Molecular Operating Environment (MOE) for 200 ns at 300 K under the MMFF94 forcefield and the Nosé-Poincaré-Andersen (NPA) algorithm (https://www.chemcomp.com/Products.htm). The results verified that the optimal number of carbon atoms in an aliphatic chain that can form a cyclic molecule is six. The only constrain in the MD simulation was that the first and last carbon atoms had to end up in such proximity and geometry as if they were actually covalently bonded. If all carbon-carbon bonds are single bonds, then it is very hard to conclude on the optimal length of carbons for cyclization. It was apparent during the MD simulations that the degrees of freedom for over eight carbon atoms are numerous and the chains turn into knots, thus adopting twisted conformations that would not favor their cyclization. Consequently, the optimal number of carbons was narrowed down between three and seven. At this stage, it is proposed that stereochemistry came into play. Due to sterics, carbon atoms would either adopt a cis or a trans conformation. If they adopt a trans conformation, then they will form long chains, very useful for the hydrophobic parts of phospho- and sphingolipids. However, if they acquire one or two cis bonds, they form a horseshoe that inevitably brings the first and sixth carbon in such a distance and geometry, where cyclization is feasible, and in this manner, cyclohexanes and benzene rings are formed. Subsequently, the copy numbers of cyclohexanes in the simulations were increased following the deterministic path that created the first one, and they began to interact and form decalin rings (Fig. 1A). Another ring was then added, and random new structures were made, finally forming phenananthrene as the most energetically favorable configuration (Fig. 1B) (7). Likewise, a five-membered ring (cyclopentane) was added (Fig. 1D), and the first version of a rather familiar life-as-we-know-it molecule was made: A steroid (Fig. 1C). Steroid hormones are everywhere (8). They control life and all known forms of life, from prokaryotes to mammals, and all life forms respond to steroids. They remember them. There is inherent memory in all life molecules when it comes to steroids.
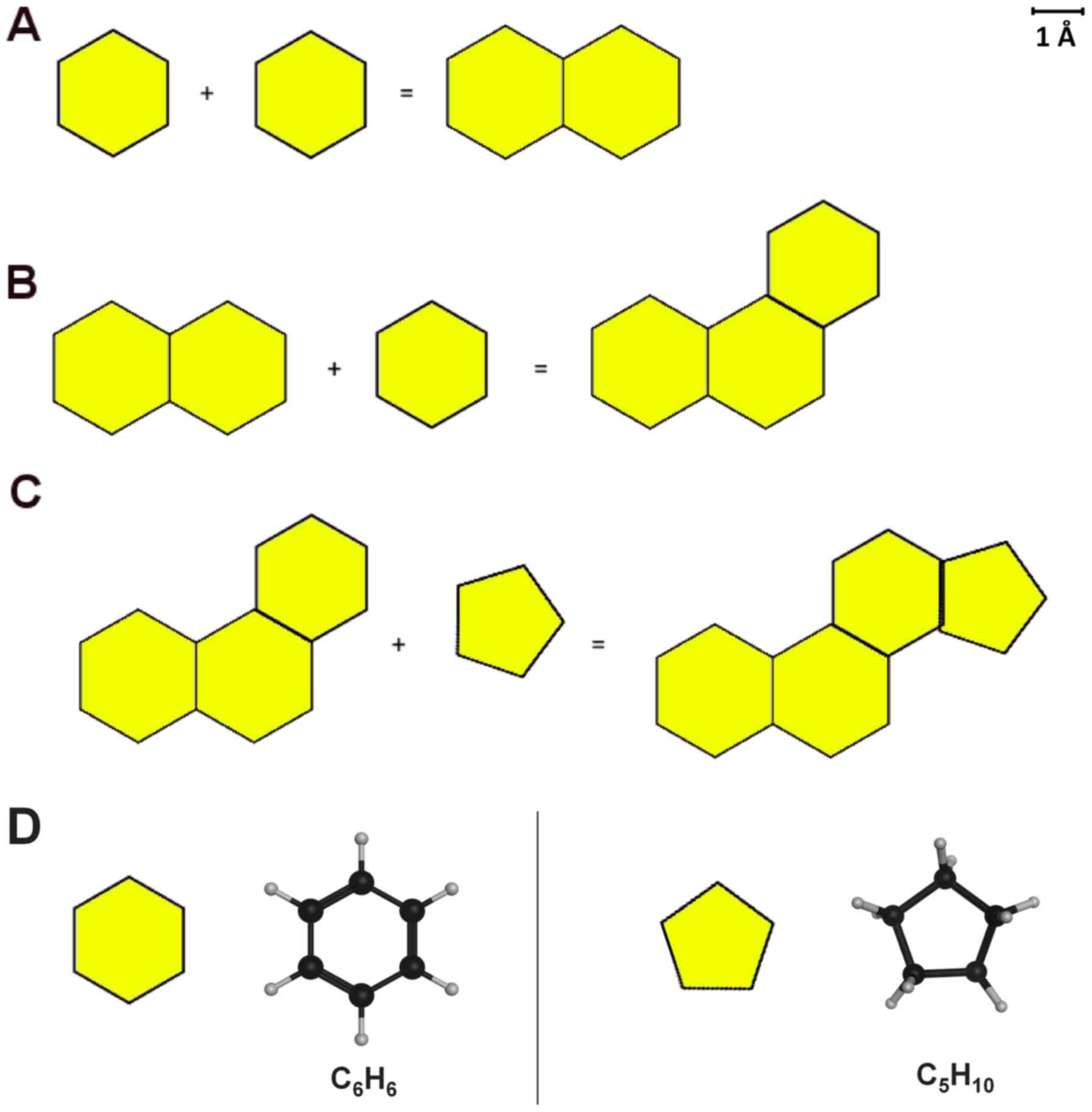 |
Figure 1
A step-by-step representation of the chemical steps for the creation of the first skeleton of the early steroid. (A) The formation of naphthalene rings out of two benzene rings. (B) The formation of phenanthrene rings out of naphthalene and benzene rings. (C) The formation of the first steroid ring. (D) Molecular representation of the depicted chemical structures in yellow.
|
Due to the aromatic nature of carbon rings, eventually, a number of steroids were stacked in an ordered manner (Fig. 2A). This early state of organization was exploited later on by heavier elements, such as phosphorus, quite possibly from nearby erupting stars as they were ending their lives in spectacular supernova explosions. Earth had no atmosphere to provide protection and heavy atom rainfall from outer space is a well-expected phenomenon in pre-atmospheric Earth (9). Phosphorus atoms found their way in close proximity to the stacked steroids, forming an early version of the phosphodiester bond of nucleotide strand backbone. The negative charges of phosphorus atoms were pushing the phosphate groups away from each other. Coupled with bombardment of radiation from outer space, that could have led to the cleavage of the structurally weakest part of the steroid (Fig. 2B). Eventually, the early steroid form was separated into two distinct parts, resembling the structure of RNA (Fig. 2C). Thus, at this point, the universal forces of nature gave birth to the first poly-A and poly-T chains that eventually combined and formed the early versions of today's genetic material.
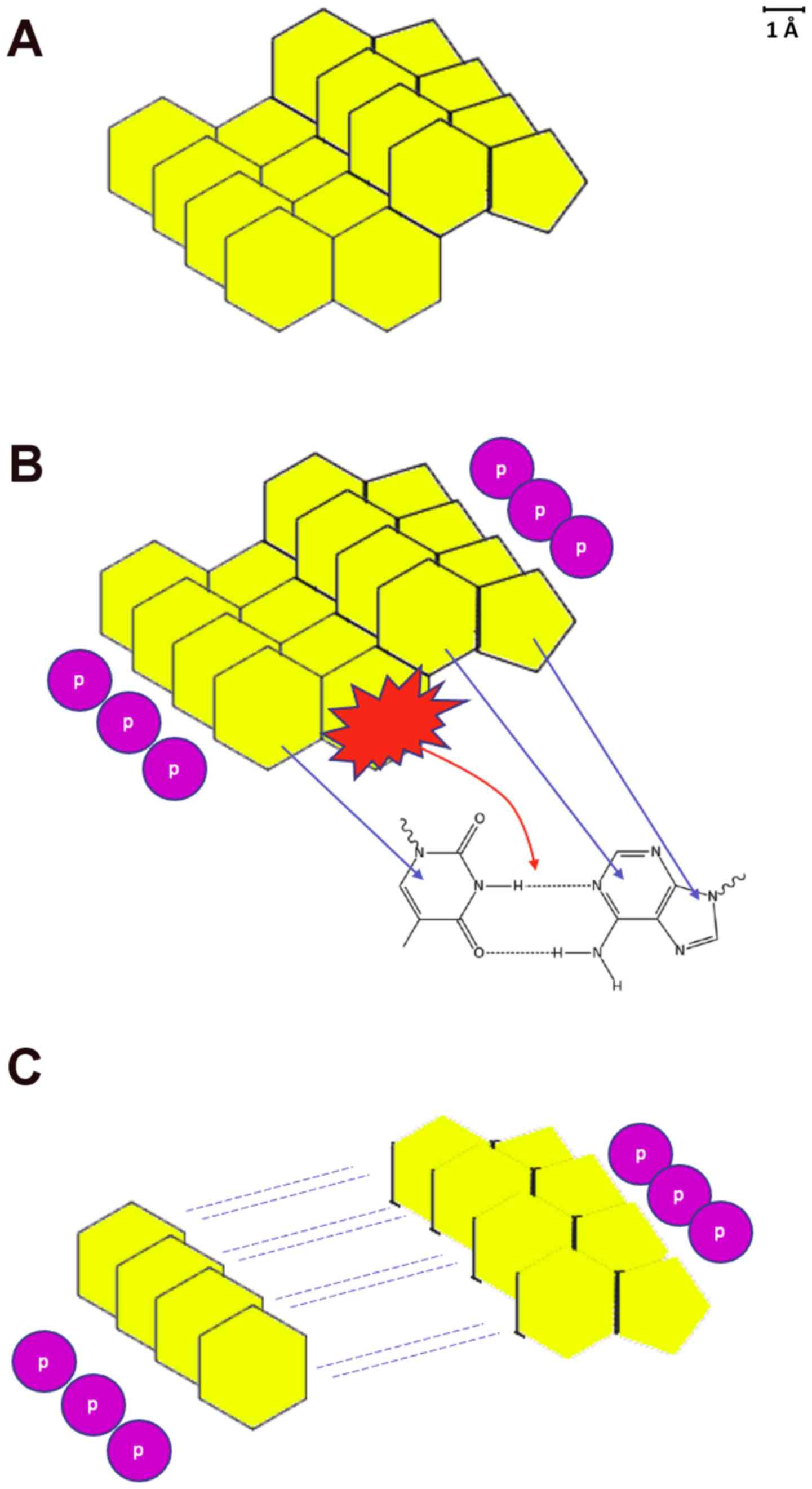 |
Figure 2
The creation of the first RNA molecule out of steroids. (A) The p-stacked due to aromatics steroid molecules. (B) Phosphate groups are established, and the steroids break at their weakest point. The resemblance to A-T DNA base pairing is evident. (C) The first adenosine and thymine early RNA-like molecules are produced.
|
However, it is evident that the molecules that shaped life must have been derived from amino acid catalysis. This suggests that at some point, another path must have produced the building blocks of proteins, as enzymes are necessary for both structural and non-structural functions in a living cell (10). The same applies for cellular membranes, where for instance, terpenoids have been suggested to play a key role in the formation of today's membranes (11). There are also the different paths of genomic evolution, that gave rise to even more complex organisms without marked differences in their genetic code. However, some subtle differences, such as codon usage for specific amino acids could shed light on the early stages of genomic evolution (12). Codon context is another crucial parameter (13). In this direction, a triplet-of-triplet genetic code approach has been suggested for mRNA translation (14). This, coupled with the fact that the physicochemical affinity of nearby residues can influence the resulting protein that the code is translated into, is one of the key elements that may have shaped life as is known (14,15).
The proposed hypothesis on the onset of life on planet Earth is the only scenario that fills the gap of the origin of RNA. The forces of nature that have been at work since the beginning of time are not erroneous and chance is only due to unseen and impossible to calculate permutations of plausible scenarios that occur in parallel. The string theory supports this. All possible scenarios are feasible and only the one that is thermodynamically superior will be captured by our limited senses. The fact of the matter is that, according to Feynman et al (16), any given system has not just one history, but every possible history.
Everyday experience though is hindered by common sense and memory that renders its understanding quite challenging. In an effort to understand and overcome the human bias, a novel approach has been developed: The model-dependent realism. In a few words, if a model is sufficient to describe observation, events and facts, this then tends to be adopted as absolute truth. In the realm of the string theory and the M-theory of everything and all that ever existed, a hypothesis for the path that gave birth to the first early version of the RNA molecule has been described (17). The Newtonian theory due to its inadequate basis to describe the microcosm, gave place to quantum physics and this new hypothesis model of reality is now considered to be the architect of the cosmos and life itself.
Conclusions
Unravelling the mystery of the origins of life is fundamental for the understanding of the laws and architecture of life as is known today. Its quantification in terms of information flow in a physical system can lead to a different perspective on the determination of life and the evolution of cell biology. Information exchange pathways from the macro to the sub-atomic level under a thermodynamics approach have been researched in the past years in an effort to elucidate the energetic costs and entropic forces that eventually determine the accuracy and efficacy of biological processes such as replication or transcription, and under a unified framework, even define the formation of early chemical structures that led to the first RNA molecule and explain all observable universe (18,19). Information exchange on a cell communication level has been also described in a hypothetical basis by Meyl (20), stating that magnetic scalar waves are generated by DNA, with benzene rings being the primary energy source, supporting the significance of this cyclic chemical structure in biological processes. Information is transported with the emission of a magnetic scalar wave that is formed upon the translocation of the bases and the temporary removal of hydrogen bonds during DNA unwinding. In this manner, an information signal is produced that can be received by a suitable system in resonance, and decoding such signals may prove to be invaluable in understanding the genetic and epigenetic mechanisms that govern biology (20). The transition from an ‘RNA world’ to a ‘DNA world’ can be approached once a greater understanding of the early life conditions and chemical world is obtained. An ‘RNA world’ is supported by the fact that protein synthesis cannot be achieved in the absence of RNA; however DNA was eventually selected as the information carrier of living entities (21). The instability of RNA compared to a double-stranded DNA molecule has led to the central dogma of molecular biology as is known, with the information flow directing from DNA to proteins. This selectivity and potential of DNA as an information carrier has been the spawn of epigenetics and deciphering how the first RNA molecule arose; it may be possible to estimate how the first methylation with an epigenetic effect occurred.
The primary perspective herein is to set the ground on the formation of the first RNA molecule through insight on the possible chemical steps for the creation of the first skeleton of the early steroid. Deciphering this process and filling the gap on the ‘RNA world’ hypothesis will help to achieve a better understanding of the chemical world of early Earth and assess the diversity of different planetary systems and their potential in emerging life, as a fundamental motivation. Even though recent findings keep adding to the hypothesis that sugars have been carried by meteorites at early Earth, questions still remain (22). Specifically, these are questions of how were these primitive extraterrestrial organic molecules formed, how did they adapt on the early Earth and how they replicated in the early chemical history. All in all, the proposed path for the creation of RNA is a good Newtonian approximation that is compatible with human senses, observations and logical synthesis. It is obvious that quantum mechanics, physics and chemistry orchestrated at subatomic level the creation of the first molecules that gave rise to the first living cell and, eventually, life.
Acknowledgements
Not applicable.
Funding
DV, GPC and EE would like to acknowledge funding from ‘MilkSafe: A novel pipeline to enrich formula milk using omics technologies’, a research co‑financed by the European Regional Development Fund of the European Union and Greek national funds through the Operational Program Competitiveness, Entrepreneurship and Innovation, under the call RESEARCH - CREATE - INNOVATE (project code: T2EDK-02222).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
DV, GPC and EE contributed to conceptualization, writing, drafting, revising, editing, and reviewing of the manuscript. All authors confirm the authenticity of all the raw data. All authors have read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
GPC is the Editor in Chief of the journal, and DV and EE are Editors of the journal. However, they had no personal involvement in the reviewing process, or any influence in terms of adjudicating on the final decision, for this article.
References
|
1
|
Bernhardt HS: The RNA world hypothesis: The worst theory of the early evolution of life (except for all the others)(a). Biol Direct. 7:23. 2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Lilley DMJ and Sutherland J: The chemical origins of life and its early evolution: An introduction. Philos Trans R Soc Lond B Biol Sci. 366:2853–2856. 2011.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Le Vay K and Mutschler H: The difficult case of an RNA-only origin of life. Emerg Top Life Sci. 3:469–475. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Peterson I: The honeycomb conjecture: Proving mathematically that honeybee constructors are on the right track. Sci News. 156:60–61. 1999.
|
|
5
|
Follmann H and Brownson C: Darwin's warm little pond revisited: From molecules to the origin of life. Naturwissenschaften. 96:1265–1292. 2009.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Mandal DK: Stereochemical Concepts. In: Pericyclic Chemistry. Elsevier, New York, NY, pp41-61, 2018.
|
|
7
|
Poater J, Duran M and Solà M: Aromaticity Determines the Relative Stability of Kinked vs. Straight Topologies in Polycyclic Aromatic Hydrocarbons. Front Chem. 6(561)2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Cole TJ, Short KL and Hooper SB: The science of steroids. Semin Fetal Neonatal Med. 24:170–175. 2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Kasting JF: Earth's early atmosphere. Science. 259:920–926. 1993.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Parsons I, Lee MR and Smith JV: Biochemical evolution II: Origin of life in tubular microstructures on weathered feldspar surfaces. Proc Natl Acad Sci USA. 95:15173–15176. 1998.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Nakatani Y, Ribeiro N, Streiff S, Gotoh M, Pozzi G, Désaubry L and Milon A: Search for the most ‘primitive’ membranes and their reinforcers: A review of the polyprenyl phosphates theory. Orig Life Evol Biosph. 44:197–208. 2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Inouye M, Takino R, Ishida Y and Inouye K: Evolution of the genetic code; Evidence from serine codon use disparity in Escherichia coli. Proc Natl Acad Sci USA. 117:28572–28575. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Chevance FFV and Hughes KT: Case for the genetic code as a triplet of triplets. Proc Natl Acad Sci USA. 114:4745–4750. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Mukai T, Lajoie MJ, Englert M and Söll D: Rewriting the Genetic Code. Annu Rev Microbiol. 71:557–577. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Koonin EV and Novozhilov AS: Origin and evolution of the genetic code: The universal enigma. IUBMB Life. 61:99–111. 2009.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Feynman RP, Leighton RB and Sands ML: The Feynman lectures on physics. Addison-Wesley Pub Co., Reading MA, 1963.
|
|
17
|
Vlachakis D and Champeris Tsaniras S: An introduction to M-theory and its application in biology. J Mol Biochem. 1:65–67. 2012.
|
|
18
|
Parrondo JMR, Horowitz JM and Sagawa T: Thermodynamics of information. Nat Phys. 11:131–139. 2015.
|
|
19
|
Saltalamacchia A, Casalino L, Borišek J, Batista VS, Rivalta I and Magistrato A: Decrypting the Information Exchange Pathways across the Spliceosome Machinery. J Am Chem Soc. 142:8403–8411. 2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Skoruppa E, Nomidis SK, Marko JF and Carlon E: Bend-induced twist waves and the structure of nucleosomal DNA. Phys Rev Lett. 121(088101)2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Lazcano A, Guerrero R, Margulis L and Oró J: The evolutionary transition from RNA to DNA in early cells. J Mol Evol. 27:283–290. 1988.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Furukawa Y, Chikaraishi Y, Ohkouchi N, Ogawa NO, Glavin DP, Dworkin JP, Abe C and Nakamura T: Extraterrestrial ribose and other sugars in primitive meteorites. Proc Natl Acad Sci USA. 116:24440–24445. 2019.PubMed/NCBI View Article : Google Scholar
|
















