Introduction
Wiedemann-Steiner syndrome (WSS) is a genetic
disorder characterized by a wide range of clinical symptoms,
including developmental delay, intellectual disability, distinctive
facial features, and other clinical features (1). Some of the facial features are thick
eyebrows with lateral flare, vertically narrow and downward slanted
palpebral fissures, widely spaced eyes (hypertelorism), long
eyelashes, wide nasal bridge, broad nasal tip, thin vermilion of
the upper lip and thick scalp hair (1,2). In
addition to the aforementioned clinical manifestations, other
features include ophthalmologic anomalies, hand anomalies (like
brachydactyly and clinodactyly), congenital heart defects, as well
as prenatal and postnatal growth restriction (1,2).
WSS is part of a group of disorders known as
chromatinopathies, which are caused by mutations in genes that
encode components of the epigenetic machinery (3). More specifically, WSS has been linked
to heterozygous pathogenic mutations in the lysine
methyltransferase 2A (KMT2A) gene (1,4).
KMT2A encodes the protein lysine methyltransferase 2A, which
is part of the KMT family. This group of proteins is part of the
epigenetic machinery and is crucial for gene expression. More
specifically, the KMT family catalyzes the transfer of methyl
groups from S-adenosylmethionine to lysine residues on histone H3
tails. KMT2A in particular, is responsible for transcriptional
activation through lysine 4 of histone 3 (H3K4) methylation. H3K4
methylation positively regulates the transcription of multiple
genes, including genes involved in hematopoiesis and neuronal
development (5).
The present study reports the case of a 2-year-old
female patient that presented with a variety of clinical features
including hypertelorism, thick eyebrows and epicanthus. Whole exome
sequencing (WES) analysis was performed and the results revealed
the presence of a heterozygous pathogenic mutation in KMT2A,
namely c.517C>T, suggesting a WSS diagnosis. Its de novo
origin was confirmed by DNA analysis of the parents. This is a rare
mutation that has been documented only twice in the ClinVar
database and to the best of our knowledge, there are no reported
cases of this mutation in the scientific literature. Hence, to the
best of our knowledge, the present study describes the first report
of WSS caused by the c.517C>T mutation in KMT2A, that
includes a detailed description of the clinical manifestations in
the patient.
Case report
A 2-year-old girl was referred to Access to Genome,
Clinical Laboratory Genetics for genetic testing due to a variety
of clinical features. More specifically, the girl exhibited
Kabuki-like gestalt with long palpebral fissures, high frontal
hairline, bilateral severe epicanthus, hypertelorism, accentuated
eyebrows with medial flare, a thin upper lip, bulbous nose,
posteriorly rotated ears with anteverted lobuli, hypoplastic
nipples, increased intermamillary distance, hypotonic kyphosis,
mild umbilical hernia, joint laxity, hypotonia, mild dorsal
hypertrichosis, early tooth eruption, thin lower legs with
protruding knees and protruding heels without rocker bottom. She
did not exhibit a delay in gross and fine motor development and
language comprehension; however, she had delayed expressive
language. Moreover, failure to thrive was observed, despite good
feeding behavior. The patient was hyperkinetic, but without
attention deficit. Moreover, an electroencephalogram revealed
normal results, renal, abdominal and heart ultrasound results were
normal, antigliadin antibody tests were negative, hearing test
results were normal, sweat test results were normal, and
hematological, biochemical and endocrinological test results were
also normal. An ophthalmological investigation at 18 months of age
revealed some immaturity of the optic nerve. At 24 months of age,
her weight was 10 kg, between 3rd and 25th percentile. She had a
normal female karyotype (46,XX).
WES analysis was performed on the DNA of the patient
that was isolated from whole blood cells. Exome amplification was
performed using AmpliSeq Exome RDY (Thermo Fisher Scientific,
Inc.). Nucleotide sequencing was performed using the Ion Chef
Instrument in combination with the Ion GeneStudio S5 System (Thermo
Fisher Scientific, Inc.). Subsequently, 4,432 genes associated with
known genetic diseases and syndromes were analyzed. Data evaluation
and interpretation were based on the clinical features of the
patient. The analysis was performed using Alamut Visual and Varsome
Clinical (Saphetor SA) bioinformatic analysis systems. All findings
from the aforementioned analysis were evaluated in accordance with
the international literature and the American College of Medical
Genetics and Genomics (ACMG) guidelines (6). The reference genome was UCSC hg19.
Bioinformatics analysis revealed the presence of a heterozygous
mutation in KMT2A, namely c.517C>T. This is a nonsense
mutation (p.Arg173Ter or R173*) that leads to a premature stop
codon in exon 3.
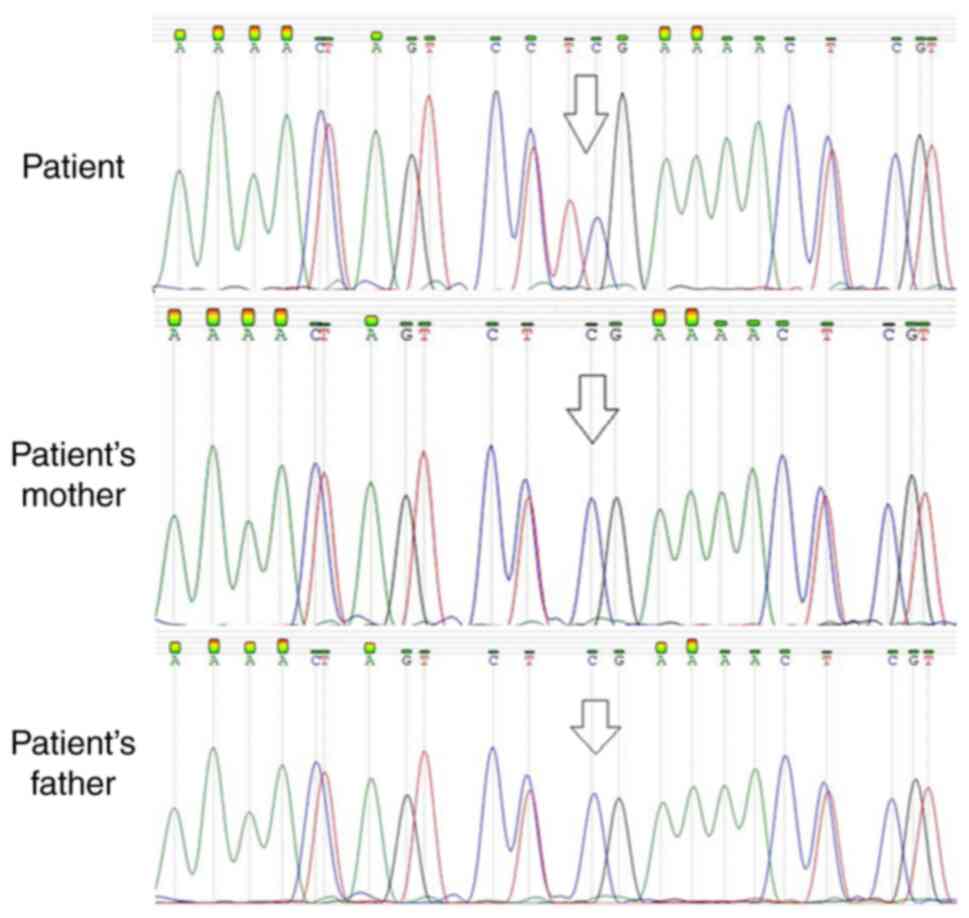
Sanger sequencing was performed in the patient in
order to confirm the presence of the c.517C>T mutation.
Following DNA amplification using PCR, part of the KMT2A
gene was sequenced and was compared to the control sequence. The
sequences of the primers used were: ACTCAAGTTGAACTCAGTACAAAATGG
(forward primer) and CTTTCTTCTTGATCTTATCTCCAGATTTGGT (reverse
primer). The results confirmed the presence of the rare mutation.
In order to establish whether the mutation was inherited or whether
it occurred de novo, Sanger sequencing of this part of
KMT2A was performed on parental DNA that was isolated from
whole blood cells. The mutation was not present in either of the
patient's parents, indicating that this was a de novo
mutation (Fig. 1).
Discussion
The present study describes the case of a 2-year-old
female patient with a phenotype characterized by hypertelorism,
thick eyebrows, epicanthus, dysplastic ears and others. WES
analysis revealed that she is a carrier of the c.517C>T nonsense
mutation in KMT2A and further analysis of her parents
revealed that this mutation occurred de novo. The clinical
features of the patient combined with the identification of the
KTM2A mutation are supportive of a WSS diagnosis.
The KMT2A protein is a component of the epigenetic
machinery, playing a crucial role in epigenetic transcriptional
activation (4). KMT2A is an
evolutionary conserved gene that is critical for various functional
processes during embryonic development, spanning from hematopoiesis
to neurogenesis (4). The encoded
protein mediates chromatin modifications associated with epigenetic
transcriptional activation and functions as a positive regulator
for the expression of numerous target genes (2). Included in those genes are genes that
belong to the Hox complex, as well as other genes involved in
embryonic development (2,7). The disruption of KMT2A leads to the
dysfunction of the epigenetic machinery and the transcriptional
activation of genes that are critical for development. This
ultimately leads to the manifestation of clinical symptoms
associated with WSS (7).
The role of KMTA has been demonstrated in murine and
zebrafish animal models. For instance, a previous study on
zebrafish has demonstrated that KMT2A is essential for neural
development in zebrafish embryos (8). Moreover, previous research has
demonstrated that the complete disruption of KMT2A in mouse embryos
is lethal, while heterozygous animals exhibit a variety of
symptoms, including growth delay and skeletal malformations
(9). The phenotypical differences
between the homozygous and the heterozygous animals suggest a
dosage-sensitive regulation by the KMT2A protein (8). Moreover, the essential role of KMT2A
in neurogenesis was demonstrated in a study where impaired neuronal
differentiation in the postnatal mouse brain was observed in
KMT2A knockout mice (5).
Other studies on mice have also demonstrated that there is a high
expression of KMT2A in adult hippocampal neurons and that
KMT2A is vital for synaptic plasticity, cognition, complex
behaviors and long-term memory (10,11).
Mutations in KMT2A have been observed
throughout the gene, which consists of 37 exons. However, a
pathogenic mutation hotspot in exon 27 exists. The majority of the
observed mutations lead to the loss of function of KMT2A
(4). The c.517C>T mutation is a
nonsense mutation, which leads to a premature stop codon in exon 3.
Nonsense mutations in KMT2A are a known pathogenicity
mechanism for the KMT2A gene. More specifically, these
mutations lead to the nonsense mediated decay of the transcript,
which causes haploinsufficiency, ultimately leading to the clinical
features associated with WSS (1,12).
Notably, the c.517C>T mutation has not yet been reported in the
gnomAD database and only two submissions exist in the ClinVar
database. Moreover, to date, to the best of our knowledge, there is
no report of this variant in the scientific literature. According
to the ACMG guidelines, this variant is categorized as ‘Pathogenic’
based on the PVS1, PM2, PP5 and PM6 criteria (6).
The clinical features of WSS overlap with those of
certain other syndromes (1). Due to
the wide range of phenotypic characteristics of WSS, differential
diagnosis based on the observed phenotype alone is difficult
(2). Notably, mutations in
KMT2A have been identified in cases initially diagnosed as
Coffin-Siris syndrome, Cornelia de Lange syndrome, Kabuki syndrome
and Rubinstein-Taybi syndrome (13-17),
which are also chromatinopathies and share similar clinical
manifestations. In instances where patients exhibit characteristics
suggestive of a chromatinopathy, WES serves as an aid in the
differential diagnosis within this group of disorders.
In the case described herein, a de novo KMT2A
mutation was identified in the patient. This mutation is a nonsense
mutation, resulting in a premature stop codon, a known
pathogenicity mechanism for this gene. Additionally, the symptoms
observed in the patient align with the expected consequences of
this mutation. Based on the aforementioned information, it can be
inferred that the detected KMT2A mutation is the underlying
cause of the patient's condition. To the best of our knowledge,
this is the first report of WSS syndrome caused by the c.517C>T
mutation in KMT2A. The present study provides a detailed
description of the phenotypic characteristics of the patient that
is critical for genetic counseling in cases of prenatal or
postnatal detection of this mutation. Moreover, the case
description in the present study may be valuable for evaluating
other patients who exhibit features of a chromatinopathy. Finally,
the present study emphasizes the importance of utilizing WES for
achieving a differential diagnosis when a chromatinopathy is
suspected.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
CK substantially contributed to the design of the
study and prepared the manuscript. EM was in charge of patient
management and project supervision. EM and IP critically revised
the manuscript. IP, ES and CE performed WES and Sanger sequencing.
YG was responsible for the evaluation of the patient and genetic
counseling. EP, EA and AG were responsible for the medical
treatment and assessment of the patient. EM and IP confirm the
authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
Written informed consent was obtained from the
parents and the patient for the inclusion of their data in the
present case report. Any information revealing the patient's
identity was not included. All procedures followed were conducted
according to The Declaration of Helsinki 1975, as revised in
2008.
Patient consent for publication
Written informed consent was obtained from the
patient's parents for publication of the present case report and
any accompanying images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sheppard SE and Quintero-Rivera F:
Wiedemann-Steiner Syndrome. University of Washington, Seattle, WA,
2022.
|
|
2
|
Yu H, Zhang G, Yu S and Wu W:
Wiedemann-Steiner Syndrome: Case report and review of literature.
Children (Basel). 9(1545)2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Fahrner JA and Bjornsson HT: Mendelian
disorders of the epigenetic machinery: Postnatal malleability and
therapeutic prospects. Hum Mol Genet. 28(R2):R254–R264.
2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Castiglioni S, Di Fede E, Bernardelli C,
Lettieri A, Parodi C, Grazioli P, Colombo EA, Ancona S, Milani D,
Ottaviano E, et al: KMT2A: Umbrella gene for multiple diseases.
Genes (Basel). 13(514)2022.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Lim DA, Huang YC, Swigut T, Mirick AL,
Garcia-Verdugo JM, Wysocka J, Ernst P and Alvarez-Buylla A:
Chromatin remodelling factor Mll1 is essential for neurogenesis
from postnatal neural stem cells. Nature. 458:529–533.
2009.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Richards S, Aziz N, Bale S, Bick D, Das S,
Gastier-Foster J, Grody WW, Hegde M, Lyon E, Spector E, et al:
Standards and guidelines for the interpretation of sequence
variants: A joint consensus recommendation of the American College
of Medical Genetics and Genomics and the Association for Molecular
Pathology. Genet Med. 17:405–424. 2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Fontana P, Passaretti FF, Maioli M,
Cantalupo G, Scarano F and Lonardo F: Clinical and molecular
spectrum of Wiedemann-Steiner syndrome, an emerging member of the
chromatinopathy family. World J Med Genet. 9:1–11. 2020.
|
|
8
|
Huang YC, Shih HY, Lin SJ, Chiu CC, Ma TL,
Yeh TH and Cheng YC: The epigenetic factor Kmt2a/Mll1 regulates
neural progenitor proliferation and neuronal and glial
differentiation. Dev Neurobiol. 75:452–462. 2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Yu BD, Hess JL, Horning SE, Brown GA and
Korsmeyer SJ: Altered Hox expression and segmental identity in
Mll-mutant mice. Nature. 378:505–508. 1995.PubMed/NCBI View
Article : Google Scholar
|
|
10
|
Kim SY, Levenson JM, Korsmeyer S, Sweatt
JD and Schumacher A: Developmental regulation of Eed complex
composition governs a switch in global histone modification in
brain. J Biol Chem. 282:9962–9972. 2007.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Jakovcevski M, Ruan H, Shen EY, Dincer A,
Javidfar B, Ma Q, Peter CJ, Cheung I, Mitchell AC, Jiang Y, et al:
Neuronal Kmt2a/Mll1 histone methyltransferase is essential for
prefrontal synaptic plasticity and working memory. J Neurosci.
35:5097–5108. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Jones WD, Dafou D, McEntagart M, Woollard
WJ, Elmslie FV, Holder-Espinasse M, Irving M, Saggar AK, Smithson
S, Trembath RC, et al: De novo mutations in MLL cause
wiedemann-steiner syndrome. Am J Hum Genet. 91:358–364.
2012.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Bramswig NC, Lüdecke HJ, Alanay Y,
Albrecht B, Barthelmie A, Boduroglu K, Braunholz D, Caliebe A,
Chrzanowska KH, Czeschik JC, et al: Exome sequencing unravels
unexpected differential diagnoses in individuals with the tentative
diagnosis of Coffin-Siris and Nicolaides-Baraitser syndromes. Hum
Genet. 134:553–568. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Yuan B, Pehlivan D, Karaca E, Patel N,
Charng WL, Gambin T, Gonzaga-Jauregui C, Sutton VR, Yesil G,
Bozdogan ST, et al: Global transcriptional disturbances underlie
Cornelia de Lange syndrome and related phenotypes. J Clin Invest.
125:636–651. 2015.PubMed/NCBI View
Article : Google Scholar
|
|
15
|
Parenti I, Teresa-Rodrigo ME, Pozojevic J,
Ruiz Gil S, Bader I, Braunholz D, Bramswig NC, Gervasini C, Larizza
L, Pfeiffer L, et al: Mutations in chromatin regulators
functionally link Cornelia de Lange syndrome and clinically
overlapping phenotypes. Hum Genet. 136:307–320. 2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Sobreira N, Brucato M, Zhang L,
Ladd-Acosta C, Ongaco C, Romm J, Doheny KF, Mingroni-Netto RC,
Bertola D, Kim CA, et al: Patients with a Kabuki syndrome phenotype
demonstrate DNA methylation abnormalities. Eur J Hum Genet.
25:1335–1344. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Negri G, Magini P, Milani D, Crippa M,
Biamino E, Piccione M, Sotgiu S, Perrìa C, Vitiello G, Frontali M,
et al: Exploring by whole exome sequencing patients with initial
diagnosis of Rubinstein-Taybi syndrome: The interconnections of
epigenetic machinery disorders. Hum Genet. 138:257–269.
2019.PubMed/NCBI View Article : Google Scholar
|