Introduction
Vascular endothelial growth factor (VEGF) is one of
the most potent and specific regulators in physiological
angiogenesis and is an important stimulator of tumor-associated
neovascularization (1,2). Angiogenesis also is essential for
tumor progression and metastasis. VEGF is expressed in the majority
of solid tumors, including lung cancer, and its overexpression is
associated with a poor prognosis (3,4). It
has been demonstrated that the VEGF pathway can be inhibited by
either targeting VEGF directly or inhibiting VEGF receptors. VEGF
inhibition is correlated with the suppression of lung cancer growth
and angiogenesis (4–7). Therefore, angiogenesis and VEGF are
important targets for the treatment of lung cancer.
Sanguinarine is a benzophenanthridine alkaloid
derived from the root of Sanguinaria canadensis and other
poppy-fumaria species, and has been shown to possess antimicrobial,
antioxidant and anti-inflammatory properties (8). An increasing number of studies have
suggested that sanguinarine at micromolar concentrations exhibits
potential antitumor activities by the inhibition of cancer cell
growth, migration and invasion, as well as the repression of
angiogenesis (9–12). Additional studies have indicated
that sanguinarine reduces VEGF-induced angiogenesis (13–15).
Moreover, the inhibition of angiogenesis by sanguinarine has
recently been validated in melanoma (10) and colorectal cancer (17). However, the exact, intracellular
molecular-targeted protein of sanguinarine on angiogenesis remains
to be elucidated, although VEGF-induced Akt phosphorylation has
previously been described (15).
The aim of this study, was to investigate the direct impacts of
sanguinarine on VEGF secretion, expression and activation.
Materials and methods
Cell lines and reagents
Human microvascular endothelial cells (HMVECs) were
cultured in MCDB-131 medium containing basic fibroblast growth
factor, heparin, fetal calf serum (FCS), penicillin, streptomycin
and amphotericin-B (Sigma-Aldrich, St. Louis, MO, USA) in tissue
culture dishes pre-coated with 1.5% gelatin in phosphate-buffered
saline (PBS). Human A549 lung cancer cells were obtained from the
American Type Culture Collection (Manassas, VA, USA) and routinely
cultured in RPMI-1640 medium containing 10% heat-inactivated FCS,
penicillin and streptomycin. Recombinant human VEGF was purchased
from R&D Systems (Minneapolis, MN, USA). Sanguinarine chloride
was purchased from Sigma-Aldrich, dissolved in dimethyl sulfoxide
(DMSO) and stored at −20°C.
Tube formation assay
HMVECs from passages 5–8 were seeded on
Matrigel-coated 24-well plates to form confluent monolayers.
Following the stimulation, of monolayers in medium containing VEGF
(30 ng/ml) ± sanguinarine for 48 h, HMVECs were fixed with 0.5 ml
of glutaraldehyde/paraformaldehyde mixture (2.5%) and stained with
modified May-Gruenwald solution (0.25%). Tubular structure
formations were visualized with a Zeiss Axiovert 200 microscope and
photomicrographs were documented by a Nikon CoolPix digital camera
(Tokyo, Japan). Recorded images were assessed for the number and
total length of tube formation. Tube formation was defined as
straight cellular extensions joining two cell masses or at branch
points. At least 10 random fields/wells were used for the
assessment of angiogenesis.
Assay of VEGF secretion
The conditioned media were collected from confluent
cultures of either HMVECs or A549 cells exposed to different
concentrations of sangunarine and centrifuged. VEGF concentrations
were then determined using a quantitative ELISA kit, according to
the manufacturer’s instructions (R&D Systems). The amount of
VEGF immunoreactivity was calculated using recombinant human VEGF
standards present on each plate. Optical densities were determined
at 450 nm using a spectrophotometer.
Migration assay
A549 cells were seeded into fibronectin (16
mg/ml)-coated 6-well tissue culture dishes at a concentration of
2.5×105 cells and cultured to almost confluent cell
monolayers, which were then wounded using 200-μl sterile
pipette tips and any cellular debris was carefully removed by
washing with PBS. The wounded monolayers were incubated in medium
containing 30 ng/m±1.0 or 1.5 μM sanguinarine for 24 h and
photographed under a light microscope. The experiments were
repeated in quadruplicate wells at least three times
(magnification, ×100).
Northern blot analysis
Briefly, total RNA extracted from cells was
performed using TRIzol reagent (Invitrogen, Carlsbad, CA, USA).
Thirty micrograms of each RNA sample were subjected to
electrophoresis on 1% denaturing formaldehyde-agarose gels, blotted
onto nylon membrane overnight by capillary transfer, and UV
cross-linked by a UV Stratalinker 1800 (Stratagene, La Jolla, CA,
USA). The blots were pre-hybridized in rapid hybridization buffer
for 4 h and then hybridized overnight at 42°C with a human VEGF
cDNA probe radiolabeled with (α-32P)dCTP in a standard
random-primed reaction to a specific activity of 1–2×109
cpm/μg. As probe, we used an 800-bp DNA fragment, which
hybridizes to the first exon of the human VEGF gene, and an 800-bp
PstI fragment of the mouse β-actin gene. Hybridization was
performed in QuikHyb solution (Stratagene) and was washed
completely. Autoradiography was performed at −80°C for 3 days. The
filters were stripped and re-probed with β-actin to normalize for
RNA loading.
Assay of luciferase reporter
activity
As described in our previous study (18), subconfluent A549 cells in 24-well
tissue culture dishes were transfected with pGL3 (as the negative
control) or pGL3-VEGF reporter plasmids using Lipofectamine™ 2000
transfection reagent (Invitrogen) according to the manufacturer’s
protocol. The total transfected DNA was kept constant by the
addition of a control vector. Cells were incubated in medium
containing various concentrations of VEGF for 24 h, and harvested
for the luciferase assays. To assess transfection efficacy,
pRSV-β-gal plasmid was co-transfected to allow normalization of
luciferase values to β-galactosidase activity in the same
sample.
Western blot analysis
As described in our previous study (18), cells were harvested and lysed on
ice for 20 min with ice-cold lysis buffer [150 mM NaCl, 10 mM
Tris-HCl (pH 7.5), 1% Triton X-100, 1 mM vanadate, 10 μg/ml
leupeptin, 10 μg/ml aprotinin, 1 mM phenylmethylsulfonyl
fluoride, 0.36 mM phenanthroline]. The lysates were centrifuged for
10 min at 4°C, and the supernatant was collected. Total protein
(100 μg) was boiled in Laemmli sample buffer (125 mM
Tris-HCl, pH 6.8, 20% glycerol, 4% SDS, 10% β-mercaptoethanol and
0.00125% bromophenol blue), separated by SDS-PAGE in 10% gel and
transferred onto 0.2 μm nitrocellulose membrane by
electroblotting. The membrane was blocked with 0.3% bovine serum
albumin (BSA) in PBS containing 0.1% Tween-20 and probed with
polyclonal antibody against protein kinase C (PKC) for 2 h at room
temperature at 4°C. Immunoreactive bands were visualized after an
1-h incubation with horse-radish peroxidase-conjugated anti-mouse
antibody, followed by ECL™ Western blot detection system.
Anti-pSer473 Akt, anti-Akt, anti-p38 and anti-VE-cadherin
antibodies used in the study were purchased from Cell Signaling
Technology, Inc. (Beverley, MA, USA). Polyclonal anti-pTyr658
VE-cadherin and anti-p-Tyr731 VE-cadherin were obtained from EMD
Millipore Co. (Billerica, MA, USA). Anti-pTyr182 p38 antibody and
anti-p-Tyr685 VE-cadherin antibody were purchased from Santa Cruz
Biotechnology, Inc. (Santa Cruz, CA, USA) and BD Biosciences (San
Jose, CA, USA), respectively.
Results
Inhibitory effects of sanguinarine on
VEGF-induced angiogenesis and migration
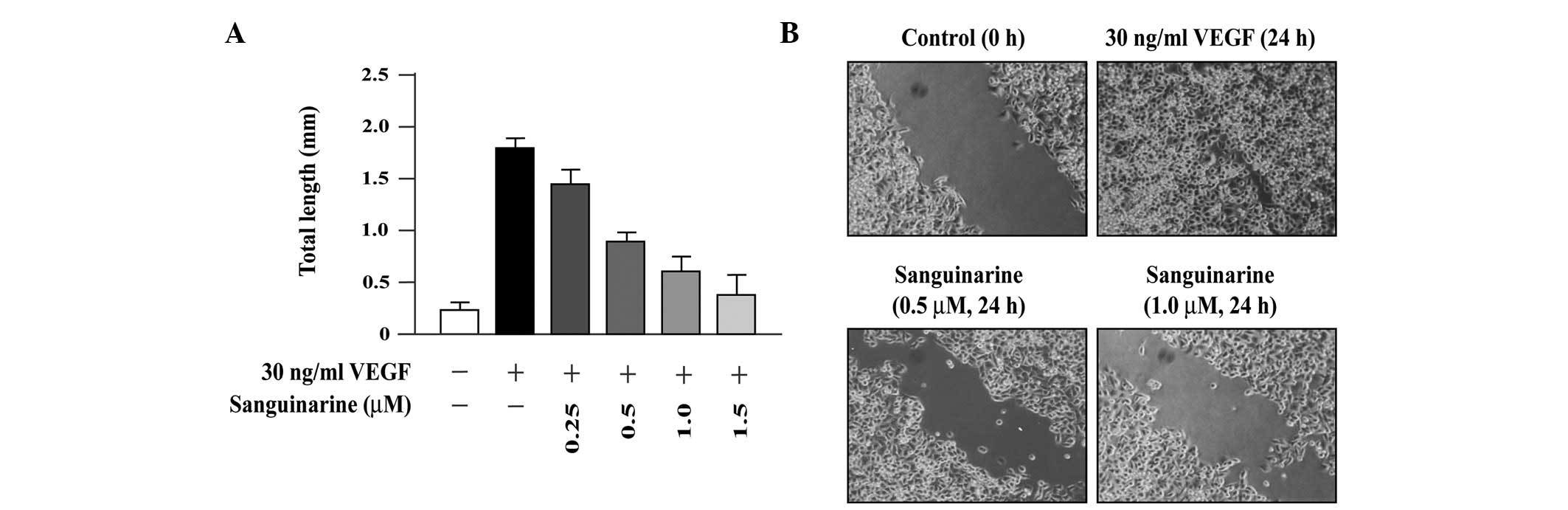
As shown in Fig.
1A, VEGF-induced tubular structure formation of HMVECs was
markedly repressed when HMVECs were cultured in medium supplemented
with sanguinarine at 0.25, 0.5, 1.0 and 1.5 μM for 24 h,
resulting in an 18, 53, 71 and 82% reduction, respectively. These
findings are in agreement with those of previous studies (13–16),
suggesting that sanguinarine is a potent inhibitor of VEGF-mediated
angiogenesis.
To examine whether or not sanguinarine affects the
migration of lung cancer cells, a scratch wound assay was
performed. Confluent monolayers of A549 cells were scratch wounded
with sterile pipette tips and post-incubated for 24 h in medium
containing VEGF ± sanguinarine (0.5 or 1.0 μM; Fig. 1B). In the presence of VEGF, A549
cells rapidly migrated along the edges of the wound and covered the
wound, whereas a significant inhibition of cell flattening and
spreading was observed in the presence of 0.5 or 1.0 μM
sanguinarine. These findings indicate that sanguinarine at
non-toxic concentrations significantly suppressed A549 cell
migration.
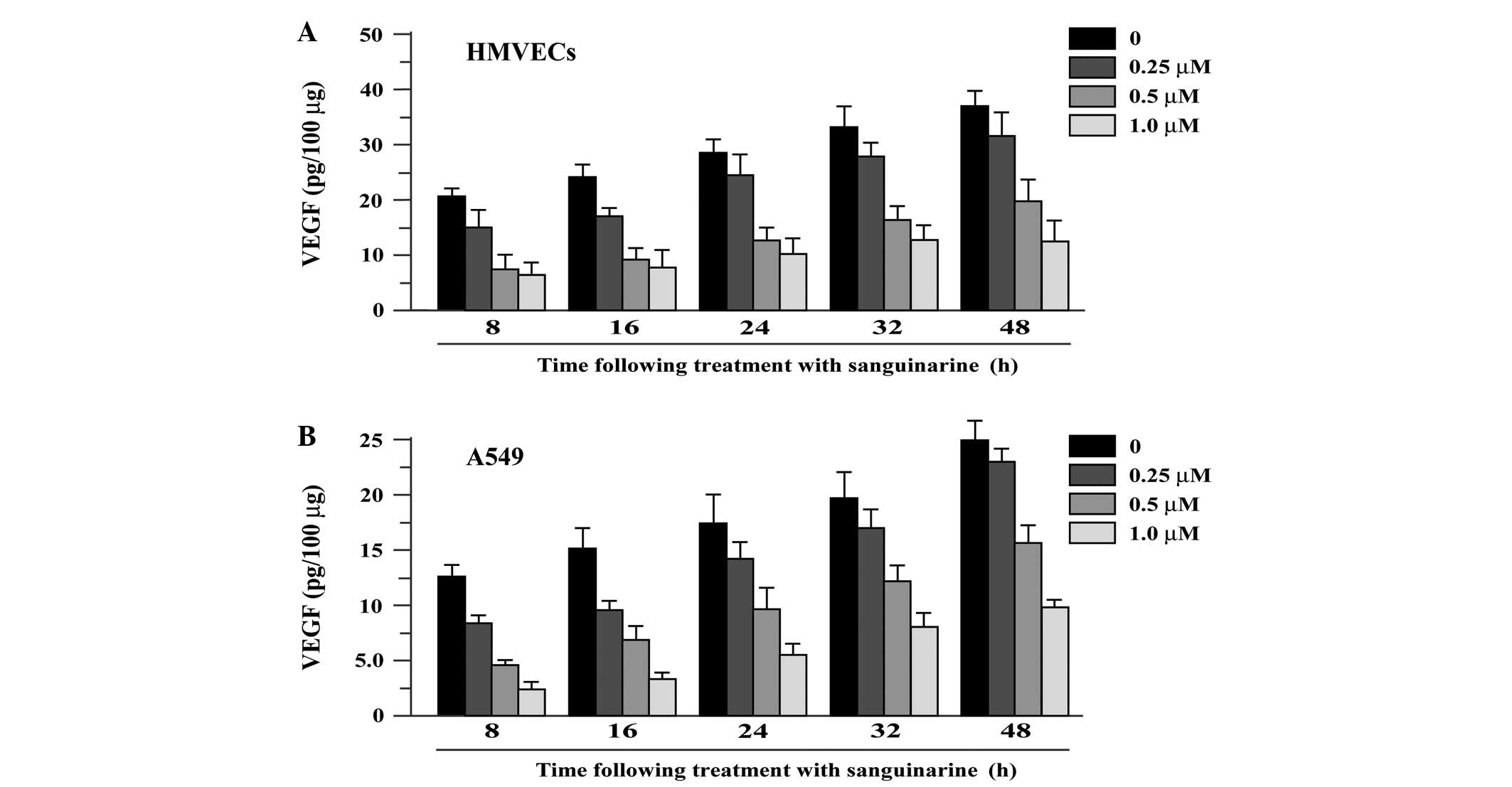
Inhibition of VEGF secretion by
sanguinarine
Compared to untreated cells, sanguinarine caused a
significant dose- and time-dependent inhibition of VEGF secretion
in both HMVEC and A549 cells at all the concentrations examined
(Fig. 2). For example, 0.25, 0.5
and 1 μM sanguinarine resulted in an ∼40, 53 and 80%
reduction of VEGF secretion induced by serum starvation in A549
cells at 24 h (P<0.05), while 1 μM sanguinarine induced
an ∼84, 80, 66, 58 and 60% reduction at 8, 16, 24, 32 and 48 h,
respectively (P<0.05).
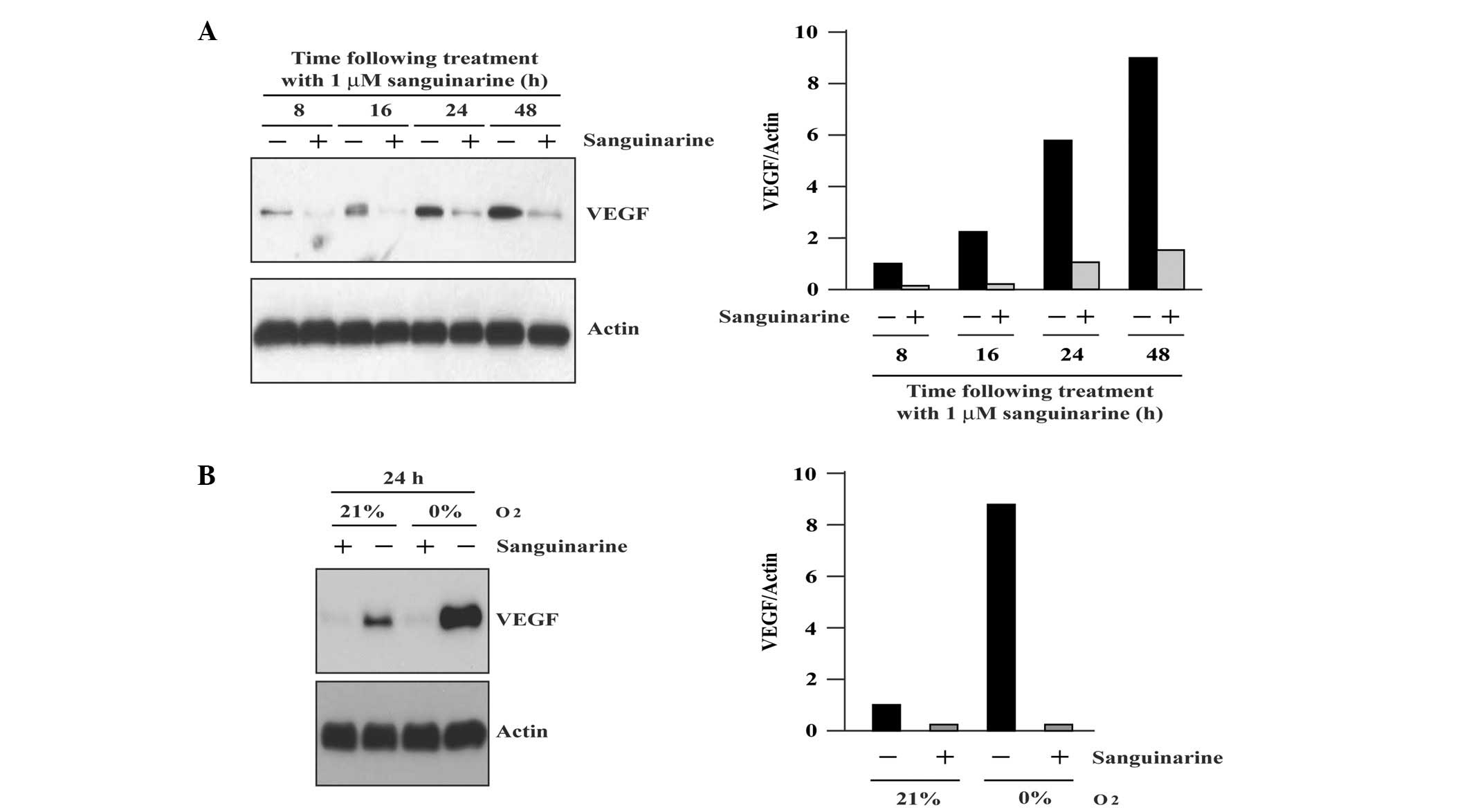
Inhibition of VEGF transcripts by
sanguinarine
Exponentially growing A549 cells were cultured in
serum-free medium with and without 1 μM sanguinarine for 6,
24 or 48 h and harvested for northern blot analysis. As shown in
Fig. 3, a single 3.7-kb VEGF
message in RNA isolated from A549 cells was detectable. Moreover, a
significant time-dependent elevation of VEGF transcripts was
observed in A549 cells cultured in serum-free medium (Fig. 3A). For example, an approximately 6-
and 9-fold increase was obtained by serum starvation at 24 and 48
h, respectively (P<0.05). Moreover, a further increase of VEGF
mRNA was determined when the cells were exposed to 0% O2
(hypoxia) for 24 h (P<0.05) (Fig.
3B). However, the increased expression of VEGF by serum
starvation (P<0.05) and hypoxia (P<0.01) was significantly
repressed in the presence of 1 μM sanguinarine.
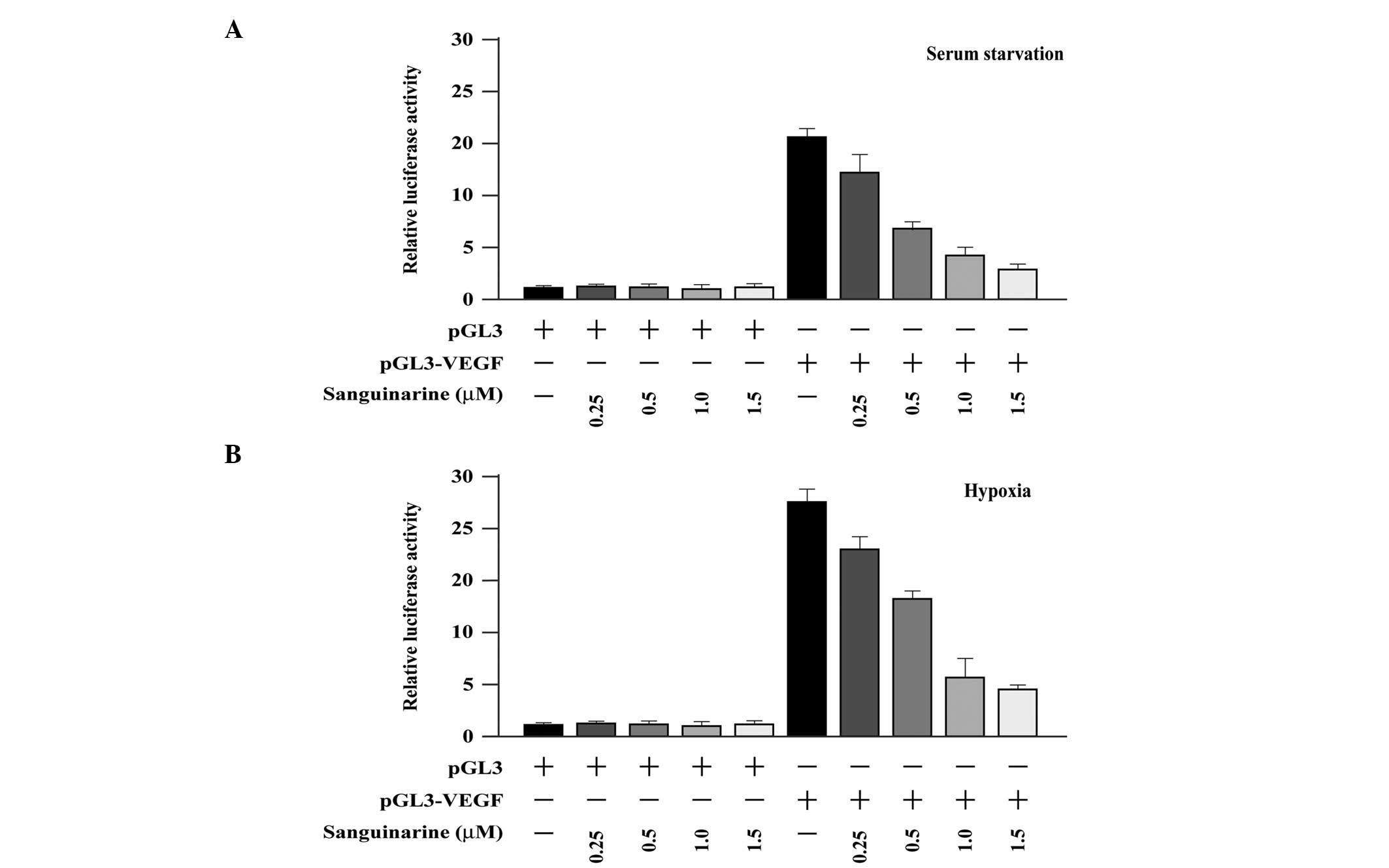
Repression of VEGF promoter activation by
sanguinarine
We also examined the impact of sanguinarine on the
VEGF promoter activity stimulated by serum starvation and hypoxia
in A549 cells transiently transfected with pGL3-VEGF, a VEGF
promoter-luciferase-reporter construct. As expected, a
dose-dependent reduction of pGL3-VEGF activation by serum
starvation (Fig. 4A) or hypoxia
(Fig. 4B) was observed in the
presence of sanguinarine. For example, 1.0 μM sanguinarine
resulted in an ∼65% reduction of VEGF activation by serum
starvation (P<0.05) and in an 82% reduction of VEGF activation
by hypoxia (P<0.05).
Inhibition of VEGF-mediated protein
expression by sanguinarine
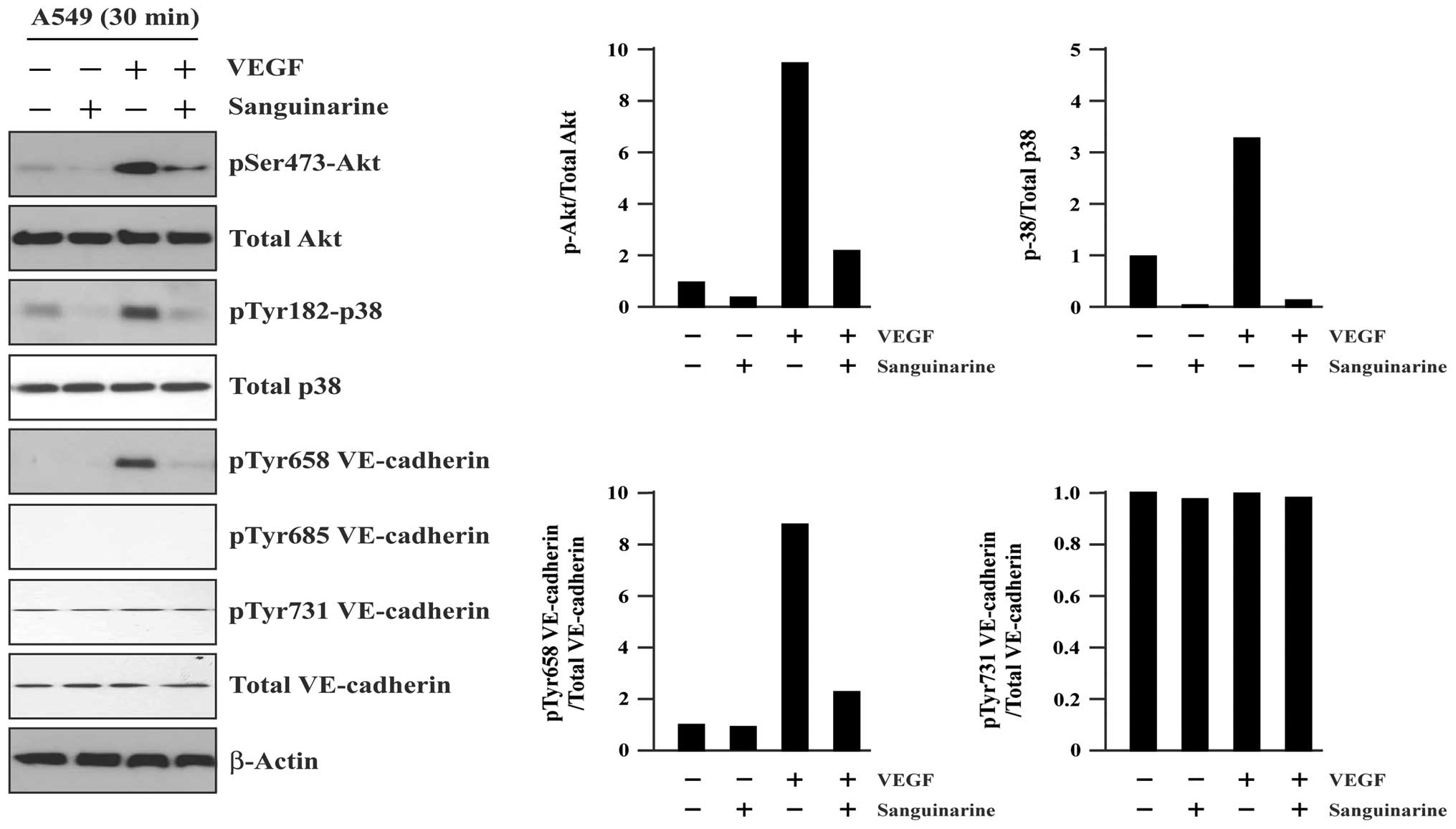
As shown in Fig. 5,
VEGF (20 min exposure) triggered a significant increase of Akt
(Ser473), p38 (Tyr182) and VE-cadherin (Tyr658) phosphorylation,
and did not affect the expression levels of VE-cadherin
phosphorylation at Tyr685 and Tyr731 sites, Akt, p38 and
VE-cadherin protein. However, in the presence of 1.0 μM
sanguinarine, the VEGF-mediated phosphorylation activation was
markedly reduced or completely inhibited. Ssimilar inhibition
findings were also observed in A549 cells treated with 0.5
μM sanguinarine (data not shown).
Discussion
Due to the fact that solid tumors, including lung
cancer, are angiogenesis-dependent, the suppression of endothelial
cells is likely to result in the inhibition of tumor growth,
progression and metastasis. Adhesion molecules, proteases and
cytokines are known to be involved in angiogenesis. Among them, the
best-studied angiogenic growth factor is VEGF (1,2).
VEGF has been demonstrated to bind to the receptor VEGF-R2 and pass
the transcriptional signal to the endothelial cells, while also
binding to VEGF-R1 to induce endothelial cell-cell interactions and
capillary formation. Previous studies demonstrated the effects of
sanguinarine on angiogenesis and metastasis. In 2004, Eun and Koh
(13) first observed that
sanguinarine might inhibit VEGF-induced endothelial cell migration,
sprouting and survival in vitro, as well as suppress blood
vessel formation in vivo in mouse Matrigel plugs and in the
chorioallantoic membrane of the chick embryo model. Subsequently,
in their studies, Basini et al (14–16)
showed that sanguinarine blocked VEGF-induced vessel growth at 300
nM. Findings of additional studies demonstrated that sanguinarine
showed a marked antiangiogenic activity in a mouse model of
melanoma skin cancer (10). Our
findings provide further evidence that sanguinarine inhibits
VEGF-mediated angiogenesis, as observed by the tube formation assay
of HMVECs. In addition, we demonstrated that sanguinarine strongly
inhibited the VEGF-induced migration of human A549 lung cancer
cells. Sanguinarine-reduced adhesion and invasive activities were
also observed in colon cancer cells of NFκB and breast cancer cells
(19,20).
In the present study, we also demonstrated that
sanguinarine strongly inhibits VEGF protein secretion in
conditioned media with serum starvation in either HMVEC or A549
cells. The sanguinarine inhibition of VEGF was also observed at the
level of transcriptional regulation, which was manifested by a
significant decrease in transcription levels and in VEGF promoter
activation by serum starvation and hypoxia. To the best of our
knowledge, these findings indicate for the first time that
sanguinarine affects VEGF secretion and expression, as well as
having a direct impact on VEGF transcription levels and VEGF
promoter activation under various types of stress.
These data also showed that the VEGF-mediated
phosphorylation activation of Akt (Ser473), p38 (Tyr182) and
VE-cadherin (Tyr658), three modulators in the VEGF signaling
pathway, was markedly reduced or blocked by sanguinarine. The
inhibitory effects of Akt phosphorylation by sanguinarine were also
observed by Basini et al (14–16).
Those findings suggested that sanguinarine is able to repress the
activation of VEGF signal transduction, and that the suppression of
Akt (Ser473), p38 (Tyr182) and that VE-cadherin (Tyr658)
phosphorylation activation is critical for the inhibition of
VEGF-mediated angiogenesis by sanguinarine.
These findings provide a mechanistic basis for the
antiangiogenic and anti-invasive activity of sanguinarine in lung
cancer cells, where sanguinatine, a novel VEGF inhibitor, represses
VEGF secretion, expression and transcriptional activation. Current
studies are focused on the potential applications of sanguinarine
alone or in combination with other VEGF inhibiting anticancer drugs
for controlling angiogenesis and metastasis of lung and possibly
other types of cancer. In addition, investigations are underway as
to whether or not other benz[c]phenanthridine alkaloids, such as
chelerythrine, chelidonine, sanguilutine and chelilutine, induce
the an inhibition of VEGF transcriptional activity and expression,
thereby enhancing their antiangiogenic activities.
Acknowledgements
This study was supported by grants
from the NNSFC (nos. 81071906 and 81172127), the Program for
Scientific Innovation Research of College Graduate in Jiangsu
(CX10B-0452); the Outstanding Doctoral Thesis Project of the
Soochow University (233209749), the Medical Scientific Research
Project of Soochow University (HZ200905) and the Program for
Preliminary Scientific Research of Soochow University
(SDY2011A45).
References
|
1
|
Carmeliet P and Jain RK: Molecular
mechanisms and clinical applications of angiogenesis. Nature.
473:298–307. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ebos JM and Kerbel RS: Antiangiogenic
therapy: impact on invasion, disease progression, and metastasis.
Nat Rev Clin Oncol. 8:210–221. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Farhat FS, Tfayli A, Fakhruddin N, et al:
Expression, prognostic and predictive impact of VEGF and bFGF in
non-small cell lung cancer. Crit Rev Oncol Hematol. Apr
9–2012.(Epub ahead of print).
|
|
4
|
Das M and Wakelee H: Targeting VEGF in
lung cancer. Expert Opin Ther Targets. 16:395–406. 2012. View Article : Google Scholar
|
|
5
|
Aggarwal C, Somaiah N and Simon G:
Antiangiogenic agents in the management of non-small cell lung
cancer: where do we stand now and where are we headed? Cancer Biol
Ther. 13:247–263. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Blakely C and Jahan T: Emerging
antiangiogenic therapies for non-small cell lung cancer. Expert Rev
Anticancer Ther. 11:1607–1618. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lind JS and Smit EF: Angiogenesis
inhibitors in the treatment of non-small cell lung cancer. Ther Adv
Med Oncol. 1:95–107. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mackraj I, Govender T and Gathiram P:
Sanguinarine. Cardiovasc Ther. 26:75–83. 2008.
|
|
9
|
Sun M, Liu C, Nadiminty N, Lou W, et al:
Inhibition of Stat3 activation by sanguinarine suppresses prostate
cancer cell growth and invasion. Prostate. 72:82–89. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
De Stefano I, Raspaglio G and Zannoni GF:
Antiproliferative and antiangiogenic effects of the
benzophenanthridine alkaloid sanguinarine in melanoma. Biochem
Pharmacol. 78:1374–1381. 2009.PubMed/NCBI
|
|
11
|
Tsukamoto H, Kondo S, Mukudai Y, et al:
Evaluation of anticancer activities of benzo[c]phenanthridine
alkaloid sanguinarine in oral squamous cell carcinoma cell line.
Anticancer Res. 31:2841–2946. 2011.
|
|
12
|
Jang BC, Park JG and Song DK: Sanguinarine
induces apoptosis in A549 human lung cancer cells primarily via
cellular glutathione depletion. Toxicol In Vitro. 23:281–287. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Eun JP and Koh GY: Suppression of
angiogenesis by the plant alkaloid, sanguinarine. Biochem Biophys
Res Commun. 317:618–624. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Basini G, Bussolati S, Santini SE and
Grasselli F: Sanguinarine inhibits VEGF-induced angiogenesis in a
fibrin gel matrix. Biofactors. 29:11–18. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Basini G, Santini SE, Bussolati S and
Grasselli F: Sanguinarine inhibits VEGF-induced Akt
phosphorylation. Ann N Y Acad Sci. 1095:371–376. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Basini G, Santini SE, Bussolati S and
Grasselli F: The plant alkaloid sanguinarine is a potential
inhibitor of follicular angiogenesis. J Reprod Dev. 53:573–579.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pica F, Balestrieri E, Serafino A, et al:
Antitumor effects of the benzophenanthridine alkaloid sanguinarine
in a rat syngeneic model of colorectal cancer. Anticancer Drugs.
23:32–42. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fan S, Ma YX and Wang C: p300 modulates
the BRCA1 inhibition of estrogen receptor activity. Cancer Res.
62:141–151. 2002.PubMed/NCBI
|
|
19
|
Choi YH, Choi WY, Hong SH, et al:
Anti-invasive activity of sanguinarine through modulation of tight
junctions and matrix metalloproteinase activities in MDA-MB-231
human breast carcinoma cells. Chem Biol Interact. 179:185–191.
2009. View Article : Google Scholar
|
|
20
|
Chang MC, Chan CP, Wang YJ, et al:
Induction of necrosis and apoptosis to KB cancer cells by
sanguinarine is associated with reactive oxygen species production
and mitochondrial membrane depolarization. Toxicol Appl Pharmacol.
218:143–151. 2007. View Article : Google Scholar
|