Technical comment
After reading the paper by Cristian Tomasetti and
Bert Vogelstein (1) and the following
comments, including those from the World Health Organization
International Agency for Cancer Research (Lyon, France) and the
Collegium Ramazzini in Italy (2–10), we
herein present a molecular biology, epidemiological and
mathematical modeling reanalysis that strongly disagrees with their
conclusion, stating that several cancer types may be caused by
random somatic mutations arising during DNA replication of normal
stem cells. Instead of the author's two-group individualized model,
a three-group model is proposed, considerably reducing the fraction
of cancer hypothetically attributable to random mutations.
The hypothesis that the lifetime risk of cancer
differs according to tissue type due to the different total number
of divisions of stem cells in normal tissues, is coherent with our
knowledge on carcinogenesis, since, according to the somatic
mutation theory, cancer is associated with mutations (11,12) and
cell divisions are required for mutations to occur (13).
However, carcinogenesis is a multistep,
multifactorial process, with etiological factors other than the
natural proliferative rate of normal tissue (14). While random mutations, which may occur
in addition to deterministic events during the whole process of
carcinogenesis, are well known, this is more problematic when
applied to cancer initiation.
Cancer may result from gene-environment interactions
mediated by epigenetic mechanisms (15); thus, in addition to innate or acquired
hereditary susceptibility factors, clearly exogenous factors such
as chemicals, radiation and/or microorganisms, play a key role in
carcinogenesis directly, by interfering with DNA, and/or
indirectly, through epigenetic pathways (16–18).
Moreover, it has been estimated that stochastic
mutations, referred to as spontaneous somatic point mutations, may
be in the order of 10−5 to 10−3 per sexual
generation for the entire genome of eukaryotic cells (19), and these mutations are not randomly
distributed within the eukaryotic genome, but mainly in its
non-coding nucleosome-depleted part (20). It is unlikely that random mutations at
this rate may cause cancer initiation and account for cancer
occurrence that is at present significantly more frequent than 1
case out of 1,000. In fact, as random mutations mainly encompass
somatic point mutations rather than cancer-causing (clonogenic)
driver mutations, and cancer results from several driver mutations
(15), random mutation-related cancer
should be much less frequent yet.
We therefore analyzed from the standpoint of
epidemiology the authors' different cancer types in the so called
replicative (R) stochastic group. As depicted in Fig. 1, apart from pancreatic cancer, all R
group cancers have been shown to be associated with an increased
incidence over the last 3 or 4 decades in the USA (21), as well as in Europe (22). We disagree with Tomasetti's response
to our question, that ‘the fact that cancer incidence has increased
over the last 3–4 decades may be explained in part as a consequence
of random replicative mutations, given the longer lifespan of the
population’ and that ‘an increase in screening and awareness
certainly played a role in the increase in the observed incidence
of melanoma, as well as that of many other cancers’. These
arguments are not convincing, since cancer may be initiated at the
fetal stage (23), increased cancer
incidence concerns all age categories, and screening by itself
cannot account for the ‘continuous’ increased incidence of several
cancer types, including those which cannot be screened (24,25). We
therefore concluded that the stochastic interpretation attributed
to the author's R group is not consistent with current
epidemiological and biological data.
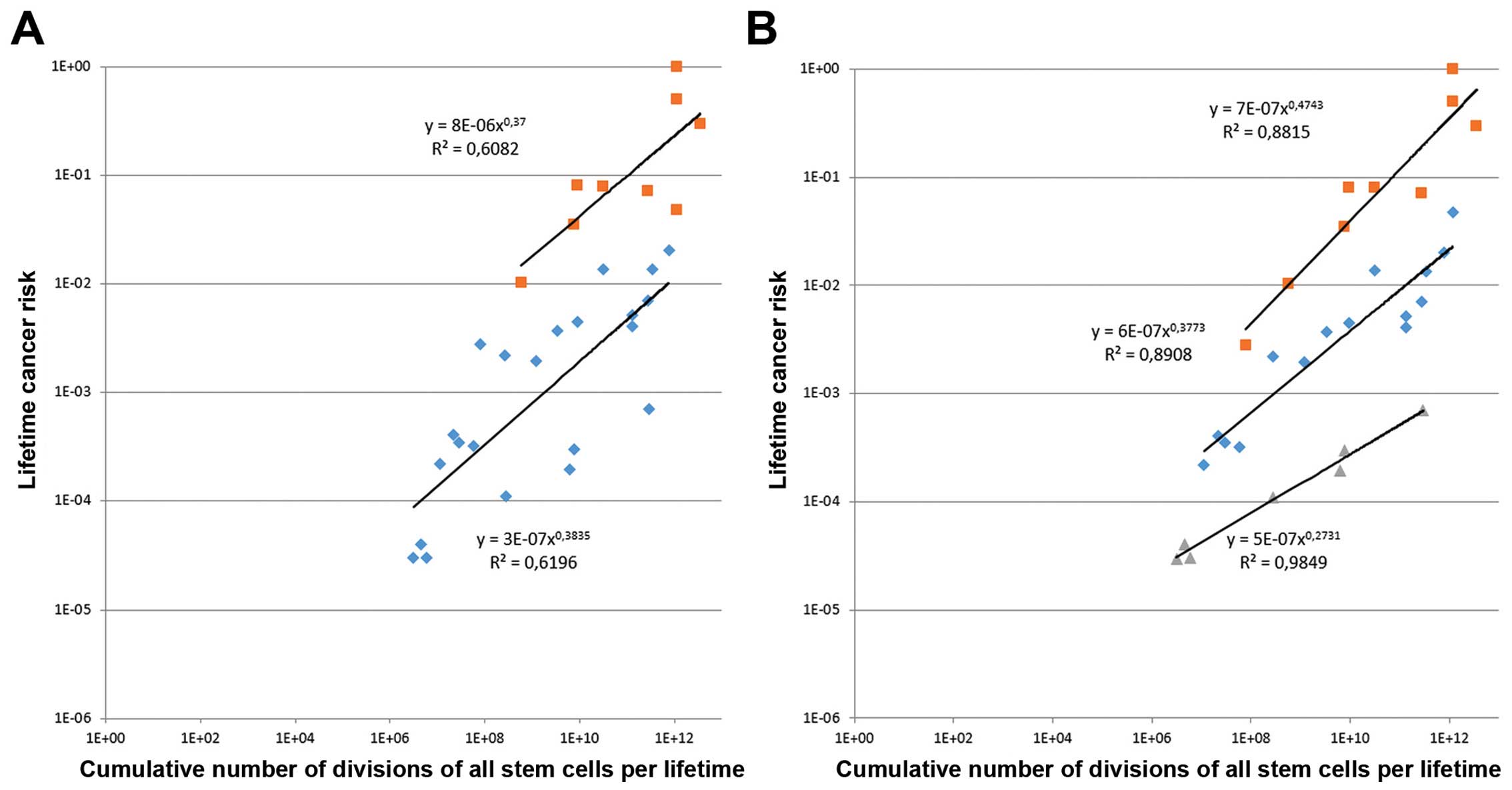
Taking into account the supplementary material of
the article, we investigated the mathematical method that Tomasetti
and Vogelstein used to define their two groups, R and deterministic
(D), the basis of their claim. Considering the association between
the lifetime cancer risk and the total number of stem cell
divisions (Fig. 1A), a strong
correlation at 0.804 was demonstrated statistically by using the
Pearson's Chi-square test. However, applying a linear regression
test, we obtained a coefficient of determination, denoted
R2, of only 0.6463, indicating that a soundly
established linear relation is unlikely. Moreover, we combined
Figs. 1 and 2 of the authors article in a single figure
for the purpose of simplification and better understanding, and
estimated R2 for each of the arbitrarily individualized
R and D groups. Again, we found the same order of R2
(Fig. 2A), confirming that linearity
is not clearly established. We therefore tentatively individualized
a third group from the R and D initial groups. As indicated in
Fig. 2B, we observed a significantly
improved linear regression, with R2 of 0.88, 0.89 and
0.98 for the newly individualized D, intermediate and R groups,
respectively. This suggests that, when restricting the R group to a
significantly smaller number of cancer types, the intermediate
group may in fact include both stochastic and deterministic events,
a hypothesis which appears to be more coherent with what we
previously discussed. Although a very small proportion of cancers
may be caused by spontaneous random mutations, we strongly suggest
that the scientific message of Tomasetti and Vogelstein does not
agree with the currently available biological, epidemiological and
toxicological data. The process of carcinogenesis has not been
sufficiently taken into consideration, so the interpretation of
Tomasetti's and Vogelstein's results may be hazardous, as it
de-emphasizes highly needed primary prevention and limits it to for
a small proportion of cancer types.
Glossary
Abbreviations
Abbreviations:
|
D
|
deterministic
|
|
R
|
replicative
|
|
R2
|
coefficient of determination
|
References
|
1
|
Tomasetti C and Vogelstein B: Cancer
etiology. Variation in cancer risk among tissues can be explained
by the number of stem cell divisions. Science. 347:78–81. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Couzin-Frankel J: Biomedicine. The bad
luck of cancer. Science. 347:122015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ashford NA, Bauman P, Brown HS, Clapp RW,
Finkel AM, Gee D, Hattis DB, Martuzzi M, Sasco AJ and Sass JB:
Cancer risk: Role of environment. Science. 347:7272015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Potter JD and Prentice RL: Cancer risk:
Tumors excluded. Science. 347:7272015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wild C, Brennan P, Plummer M, Bray F,
Straif K and Zavadil J: Cancer risk: Role of chance overstated.
Science. 347:7282015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gotay C, Dummer T and Spinelli J: Cancer
risk: Prevention is crucial. Science. 347:7282015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Song M and Giovannucci EL: Cancer risk:
Many factors contribute. Science. 347:728–729. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
O'Callaghan M: Cancer risk: Accuracy of
literature. Science. 347:7292015. View Article : Google Scholar
|
|
9
|
Tomasetti C and Vogelstein B: Cancer risk:
Role of environment-response. Science. 347:729–731. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Collegium Ramazzini. 17th position
statement: Most types of cancer are cot due to bad luck. http://www.collegiumramazzini.org/download/17_SeventeenthCRStatement(2015).pdfAccessed.
March 10–2015
|
|
11
|
Sjöblom T, Jones S, Wood LD, Parsons DW,
Lin J, Barber TD, Mandelker D, Leary RJ, Ptak J, Silliman N, et al:
The consensus coding sequences of human breast and colorectal
cancers. Science. 314:268–274. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Greenman C, Stephens P, Smith R, Dalgliesh
GL, Hunter C, Bignell G, Davies H, Teague J, Butler A, Stevens C,
et al: Patterns of somatic mutation in human cancer genomes.
Nature. 446:153–158. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Weinstein IB: Mitogenesis is only one
factor in carcinogenesis. Science. 251:387–388. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Skinner MK, Manikkam M and
Guerrero-Bosagna C: Epigenetic transgenerational actions of
environmental factors in disease etiology. Trends Endocrinol Metab.
21:214–222. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Weinberg RA: The Biology of Cancer. (2nd).
Garland Science (New York, NY). 2013.
|
|
16
|
Irigaray P and Belpomme D: Basic
properties and molecular mechanisms of exogenous chemical
carcinogens. Carcinogenesis. 31:135–148. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Elmore SA, Boyle MC, Boyle MH, Cora MC,
Crabbs TA, Cummings CA, Gruebbel MM, Johnson CL, Malarkey DE,
McInnes EF, et al: Proceedings of the 2013 National Toxicology
Program Satellite Symposium. Toxicol Pathol. 42:12–44. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
National Toxicology Program. 13th report
on Carcinogens (2014). http://ntp.niehs.nih.gov/pubhealth/roc/roc13/index.htmlAccessed.
January 12–2015
|
|
19
|
Drake JW, Charlesworth B, Charlesworth D
and Crow JF: Rates of spontaneous mutation. Genetics.
148:1667–1686. 1998.PubMed/NCBI
|
|
20
|
Chen X, Chen Z, Chen H, Su Z, Yang J, Lin
F, Shi S and He X: Nucleosomes suppress spontaneous mutations
base-specifically in eukaryotes. Science. 333:1235–1238. 2012.
View Article : Google Scholar
|
|
21
|
National Cancer Institute, Surveillance,
Epidemiology, and End Results Program. https://seer.cancer.govAccessed. January 12–2015
|
|
22
|
Cancer Research UK. http://www.cancerresearchuk.orgAccessed. January
12–2015
|
|
23
|
Marshall GM, Carter DR, Cheung BB, Liu T,
Mateos MK, Meyerowitz JG and Weiss WA: The prenatal origins of
cancer. Nat Rev Cancer. 14:277–1689. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Belpomme D, Irigaray P, Hardell L, Clapp
R, Montagnier L, Epstein S and Sasco AJ: The multitude and
diversity of environmental carcinogens. Environ Res. 105:414–429.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Belpomme D, Irigaray P, Sasco AJ, Newby
JA, Howard V, Clapp R and Hardell L: The growing incidence of
cancer: Role of lifestyle and screening detection. Int J Oncol.
30:1037–1044. 2007.PubMed/NCBI
|