Introduction
Lung cancer is a leading cause of mortality
worldwide. Although treatment with epidermal growth factor
receptor-tyrosine kinase inhibitors (EGFR-TKIs) has been reported
to prolong the survival in patients with EGFR mutation-positive
non-small cell lung cancer (NSCLC) (1–3), the
survival period varies widely in individual patients receiving
EGFR-TKI treatment.
Our previous study reported the existence of an
association between the pre-treatment plasma neuron-specific
enolase level and the patient survival in EGFR mutation-positive
NSCLC patients initiated on gefitinib treatment (4). Neuron-specific enolase is a marker of
neuroendocrine tumors, including small cell lung cancer, and
functions as a glycolytic enzyme. Lactate dehydrogenase (LDH), also
an enzyme contributing to anaerobic glycolysis, has been reported
as a prognostic factor in patients with various malignant tumors,
including lung cancer (5–8). The association between the plasma LDH
level and tumor hypoxia is considered as one of the mechanisms
underlying this linkage (6–8). However, the impact of the pre-treatment
plasma LDH level on the survival in patients with EGFR
mutation-positive NSCLC receiving treatment with EGFR-TKIs remains
to be elucidated.
For patients with lung cancer initiated on treatment
with EGFR-TKIs, investigation of the prognostic factors and
identification of patients with a poor prognosis are important for
planning the appropriate follow-up schedule, and may also
contribute to the establishment of novel treatment strategies. The
present study performed a retrospective analysis to investigate the
association between the plasma LDH levels and survival in EGFR
mutation-positive NSCLC patients receiving treatment with
EGFR-TKIs.
Patients and methods
Patient selection and evaluation
The present study retrospectively reviewed the
medical charts of patients with lung cancer receiving treatment at
Toyama University Hospital (Toyama, Japan) between 2007 and 2014.
The inclusion criteria were as follows: i) Cytologically or
histologically confirmed NSCLC; ii) tumor positive for EGFR gene
mutations, with the exception of the T790M mutation; iii) patients
receiving treatment with EGFR-TKIs. Patients with a previous
history of treatment with EGFR-TKIs were excluded from the
analysis.
The following patient data was collected: Age,
gender, number of treatment regimens received previously, disease
stage, tumor EGFR mutation status, patient performance status (PS),
smoking history and presence/absence of distant metastasis. Exon 19
deletions and the exon 21 point mutation of L858R in the
EGFR gene were classified as common mutations, while any
other mutations were classified as uncommon mutations. The disease
stage was classified as stage III/IV or postoperative recurrence.
Presence/absence of metastatic lesions was determined by imaging
examinations, including computed tomography, magnetic resonance
imaging and/or scintigraphy, regardless of the cytological or
histological findings. The present study was performed with the
approval of the Ethics Committee of the University of Toyama
(approval no. 26-158).
Statistical analysis
Survival curves were drawn according to the
Kaplan-Meier method. Progression-free survival (PFS) was calculated
from the date on which the EGFR-TKI administration was initiated
until the date on which disease progression was first documented or
the date of mortality due to any cause, and censored at the last
visit at which no disease progression was observed. The overall
survival (OS) was calculated from the date on which the EGFR-TKI
administration was initiated until the date of mortality due to any
cause, and censored at the last visit at which the patient remained
alive. The association between the survival and the patient
clinical characteristics was analyzed by the log-rank test. With
regards to the analysis regarding the LDH level, the patients were
dichotomized into two groups, according to the median LDH level.
The independent association between each variable and the survival
were analyzed using a Cox proportional hazards model. The variables
with a P-value <0.2 by the log-rank test were entered as
independent variables into the Cox proportional hazards model.
Results
Patient characteristics
Table I lists the
patient characteristics. A total of 65 patients fulfilled the
criteria and were included in the analysis. Of these, 41 patients
who were receiving treatment with gefitinib were also enrolled into
our previous study (4). Female
patients were slightly dominant, accounting for 53.8% of the
population. The median (range) age of the patients was 68 (49–87)
years, and 52.3% were <70-years-old. The majority of the
patients were treated with gefitinib and 28.5% of patients were
treated with erlotinib. The majority of patients (90.8%) exhibited
tumors harboring common EGFR gene mutations. The plasma LDH
levels ranged between 118 and 1,680 IU/l (median value, 208 IU/l).
Disease stage, PS, smoking history, liver metastases and pleural
effusion were imbalanced between the two groups.
 | Table I.Patient characteristics. |
Table I.
Patient characteristics.
| Characteristic | All patients
(n=65) | LDH low (n=35) | LDH high (n=30) |
|---|
| Gender, n (%) |
|
|
|
| Male | 30 (46.2) | 19 (54.3) | 11 (36.7) |
|
Female | 35 (53.8) | 16 (45.7) | 19 (63.3) |
| Age, n (%) |
|
|
|
|
<70 | 34 (52.3) | 18 (51.4) | 16 (53.3) |
| ≥70 | 31 (47.7) | 17 (48.6) | 14 (46.7) |
| EGFR-TKI, n (%) |
|
|
|
|
Gefitinib | 53 (81.5) | 28 (80.0) | 25 (83.3) |
|
Erlotinib | 12 (28.5) | 7 (20.0) | 5 (16.7) |
| Prior regimen, n
(%) |
|
|
|
| 0 | 49 (75.4) | 25 (71.4) | 24 (80.0) |
| ≥1 | 16 (24.7) | 10 (28.6) | 6 (20.0) |
| Stage, n (%) |
|
|
|
|
III/IV | 50 (76.9) | 23 (65.7) | 27 (90.0) |
|
Recurrence | 15 (23.1) | 12 (34.3) | 3 (10.0) |
| EGFR status, n
(%) |
|
|
|
|
Common | 59 (90.8) | 31 (88.6) | 28 (93.3) |
|
Uncommon | 6 (9.2) | 4 (11.4) | 2 (6.7) |
| PS, n (%) |
|
|
|
| 0–1 | 45 (69.2) | 27 (77.1) | 18 (60.0) |
| ≥2 | 20 (30.8) | 8 (22.9) | 12 (40.0) |
| Smoking history, n
(%) |
|
|
|
| Yes | 31 (47.7) | 20 (57.1) | 11 (36.7) |
| No | 34 (52.3) | 15 (42.9) | 19 (63.3) |
| Brain metastases, n
(%) |
|
|
|
| Yes | 19 (29.2) | 10 (28.6) | 9 (30.0) |
| No | 46 (70.8) | 25 (71.4) | 21 (70.0) |
| Liver metastasis, n
(%) |
|
|
|
| Yes | 14 (21.5) | 4 (11.4) | 10 (33.3) |
| No | 51 (78.5) | 31 (88.6) | 20 (66.7) |
| Lung metastasis, n
(%) |
|
|
|
| Yes | 25 (38.5) | 12 (34.3) | 13 (43.3) |
| No | 40 (61.5) | 23 (65.7) | 17 (56.7) |
| Adrenal gland
metastasis, n (%) |
|
|
|
| Yes | 5 (7.7) | 2 (5.7) | 3 (10.0) |
| No | 61 (92.3) | 33 (94.3) | 27 (90.0) |
| Bone metastasis, n
(%) |
|
|
|
| Yes | 31 (47.7) | 15 (42.9) | 16 (53.3) |
| No | 34 (52.3) | 20 (57.1) | 14 (46.7) |
| Pleural effusion, n
(%) |
|
|
|
| Yes | 30 (46.2) | 10 (28.6) | 20 (66.7) |
| No | 35 (53.8) | 25 (71.4) | 10 (33.3) |
Survival
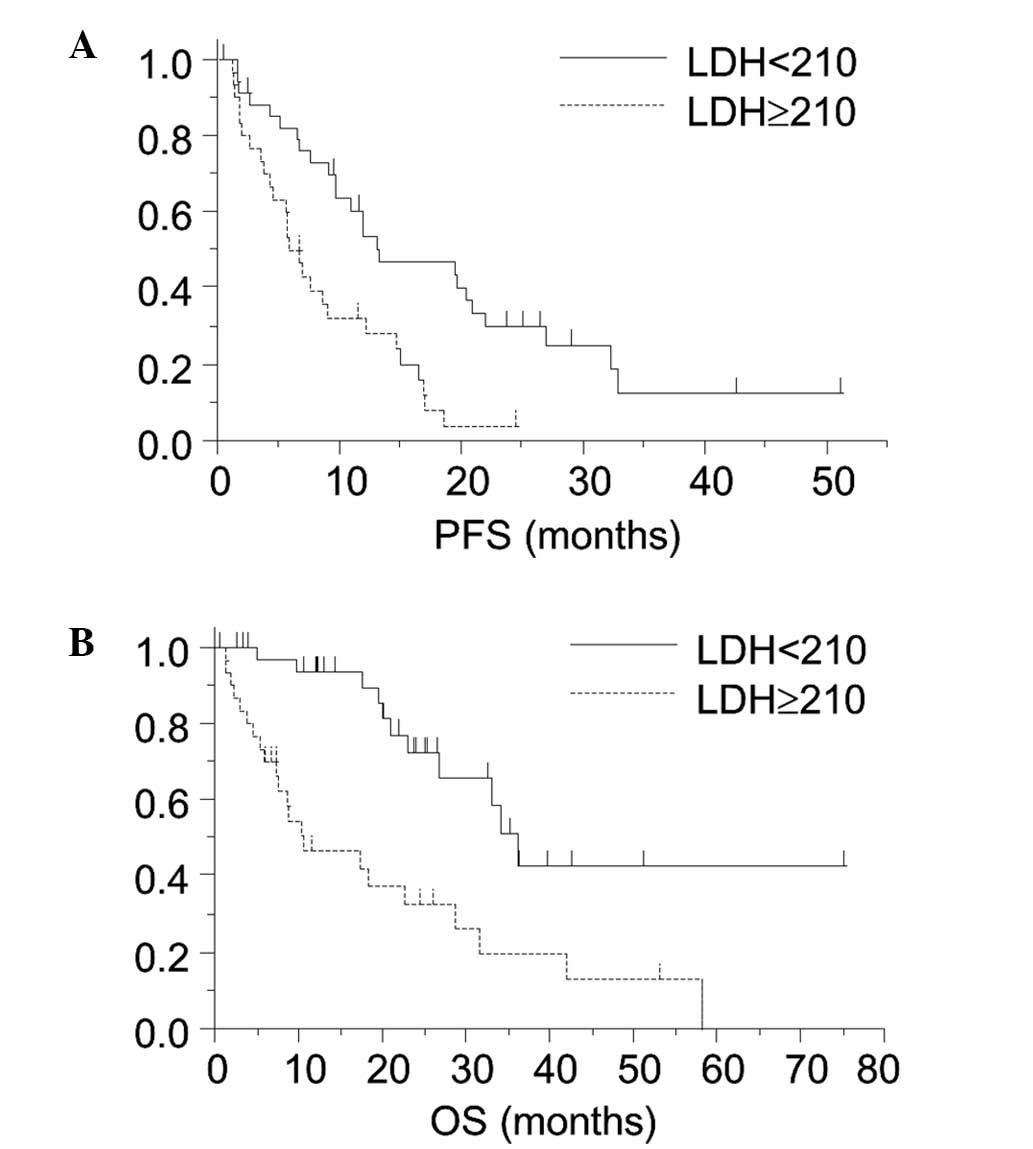
Fig. 1 reveals the
Kaplan-Meier survival curves for the PFS and OS in the patient
groups divided by the plasma LDH level of 210 IU/l. Significant
differences in the PFS (6.2 vs. 13.2 months; P<0.01) and OS
(10.5 vs. 36.1 months; P<0.01) were observed between the
high-LDH and low-LDH groups.
The log-rank test revealed that the P-values for the
disease stage (P=0.02), EGFR mutation status (P=0.17), PS (P=0.01),
presence/absence of brain metastasis (P<0.01), presence/absence
of liver metastasis (P=0.10), presence/absence of pleural effusion
(P=0.12) and the plasma LDH level (P<0.01) were determined to be
<0.2, and these variables were entered into the Cox proportional
hazards model as independent variables. The P-value for gender
(P=0.37), age (P=0.25), type of EGFR-TKI (P=0.60), number of
previous treatment regimens (P=0.91), smoking history (P=0.43),
presence/absence of metastasis to the lung (P=0.83), bone (P=0.90)
and adrenal gland (P=0.95) were >0.2. Table II shows the results of the analysis
performed using the Cox proportional hazards model to assess the
association between each variable and the PFS. The analysis
identified that the EGFR status (P=0.03), presence/absence of brain
metastasis (P<0.01) and the plasma LDH level (P=0.05) were
associated with the PFS.
 | Table II.Analysis using a Cox proportional
hazards regression model for assessing the association between the
plasma lactate dehydrogenase level and progression-free
survival. |
Table II.
Analysis using a Cox proportional
hazards regression model for assessing the association between the
plasma lactate dehydrogenase level and progression-free
survival.
| Variable | HR (95% CI) | P-value |
|---|
| Stage |
| 0.59 |
|
III/IV | 1.28 (0.53–3.31) |
|
|
Recurrence | 1 |
| EGFR status |
| 0.03 |
|
Common | 0.30 (0.11–0.90) |
|
|
Uncommon | 1 |
| PS |
| 0.28 |
| 0–1 | 0.66 (0.32–1.40) |
|
| ≥2 | 1 |
| Brain metastasis |
| <0.01 |
| Yes | 3.39 (1.56–7.37) |
|
| No | 1 |
|
| Liver metastasis |
| 0.47 |
| Yes | 1.32 (0.61–2.70) |
|
| No | 1 |
|
| Pleural effusion |
| 0.19 |
| Yes | 1.74 (0.76–3.92) |
|
| No | 1 |
|
| LDH |
| 0.05 |
|
<210 | 0.49 (0.24–0.99) |
|
| ≥210 | 1 |
With regards to the OS, the P-values for the disease
stage (P=0.07), EGFR mutation status (P=0.07), PS (P<0.01),
presence/absence of pleural effusion (P=0.01) and plasma LDH level
(P<0.01) were <0.2, and these were entered into the Cox
proportional hazards model as independent variables. The P-value
for gender (P=0.29), age (P=0.94), type of EGFR-TKI (P=0.80),
number of previous treatment regimens (P=0.46), smoking history
(P=0.62), and the presence/absence of metastasis to the brain
(P=0.34), liver (P=0.36), lung (P=0.61), bone (P=0.66) and adrenal
gland (P=0.93) were >0.2. Table
III shows the results of the analysis performed using the Cox
proportional hazards model to assess the association between each
variable and the OS. The analysis identified that the EGFR status
(P<0.01), PS (P=0.03) and plasma LDH level (P<0.01) were
associated with the OS.
 | Table III.Analysis using a Cox proportional
hazards regression model for assessing the association between the
plasma lactate dehydrogenase level and the overall survival. |
Table III.
Analysis using a Cox proportional
hazards regression model for assessing the association between the
plasma lactate dehydrogenase level and the overall survival.
| Variable | HR (95% CI) | P-value |
|---|
| Stage |
| 0.42 |
|
III/IV | 1.52 (0.56–4.80) |
|
|
Recurrence | 1 |
|
| EGFR status |
| <0.01 |
|
Common | 0.14
(0.04–0.49) |
|
|
Uncommon | 1 |
|
| PS |
| 0.03 |
|
0–1 | 0.36
(0.14–0.89) |
|
| ≥2 | 1 |
|
| Pleural
effusion |
| 0.14 |
|
Yes | 1.97
(0.80–5.11) |
|
| No | 1 |
|
| LDH |
| <0.01 |
|
<210 | 0.27
(0.11–0.61) |
|
|
≥210 | 1 |
Post-progression treatment
In the higher LDH group, 27 patients experienced
disease progression, 4 (14.8%) patients were treated with EGFR-TKI
re-administration, 5 (18.5%) were treated with platinum doublet
chemotherapy and 5 (18.5%) were treated with single cytotoxic
agent. In the lower LDH group, 25 patients experienced disease
progression, 3 (12.0%) were treated with EGFR-TKI
re-administration, 5 (20.0%) were treated with platinum doublet
chemotherapy and 4 (16.0%) were treated with a single cytotoxic
agent.
Discussion
The present study revealed the existence of a
significant association between the pre-treatment plasma LDH level
and survival in patients with EGFR mutation-positive NSCLC
receiving treatment with EGFR-TKIs. Elevated plasma LDH has been
reported to be associated with a poor prognosis in patients with
several types of malignancies, including lung cancer, receiving
various treatments (5–8). Based on these previous reports, it was
considered that the plasma LDH level may serve as a prognostic
factor in patients with EGFR mutation-positive NSCLC.
Several mechanisms underlying the association
between the serum LDH level and the patient survival have been
proposed. LDH is a glycolytic enzyme that contributes to anaerobic
production of adenosine triphosphate, and the increased production
of LDH can be a direct marker of severe tumor hypoxia. LDH-5, one
of the isoenzymes of LDH, is induced by hypoxia-inducible factor-1α
in the hypoxic environment of the tumor; therefore, high plasma LDH
may reflect an upregulated hypoxia inducible factor-1α-molecular
cascade (7,8). Findings lending support to the
above-mentioned mechanisms were reported by Koukourakis et
al (7) who demonstrated that the
tumor LDH-5 expression was associated with that of
hypoxia-inducible factor and vascular endothelial growth factor. In
addition, LDH activity results in acidification of the
extracellular space through the production of lactic acid, which
may be associated with an increased invasive ability and angiogenic
capability of tumors (6–8).
It is possible that the association between plasma
LDH level and survival demonstrated in the present study partly
resulted from the activity of VEGF. The additional efficacy of
bevacizumab on erlotinib in patients with NSCLC harboring
EGFR gene mutation (9)
suggests that VEGF activity influences patient survival or efficacy
of EGFR-TKI, although the association between VEGF expression and
the patient survival treated with EGFR-TKI remains to be
elucidated.
The present study was retrospective in nature and
the sample size was small. Although multivariate analyses were
performed, it may be difficult to completely exclude the possible
existence of confounding factors affecting patient survival.
Furthermore, it was not confirmed whether the plasma LDH level may
be a predictive factor due to the small sample size and
retrospective design of this study.
In conclusion, the results of the present study
indicated the existence of an association between the pretreatment
plasma LDH level and survival in EGFR mutation-positive NSCLC
patients receiving treatment with EGFR-TKIs, as previous studies
had suggested the existence of a population with a poor prognosis
even among these patients. Evaluation of the pretreatment plasma
LDH level may contribute to follow-up planning in patients
receiving treatment with EGFR-TKIs, and the establishment of a
treatment strategy for the population with a poor prognosis is
expected.
References
|
1
|
Mok TS, Wu YL, Thongprasert S, Yang CH,
Chu DT, Saijo N, Sunpaweravong P, Han B, Margono B, Ichinose Y, et
al: Gefitinib or carboplatin-paclitaxel in pulmonary
adenocarcinoma. N Engl J Med. 361:947–957. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Maemondo M, Inoue A, Kobayashi K, Sugawara
S, Oizumi S, Isobe H, Gemma A, Harada M, Yoshizawa H, Kinoshita I,
et al: Gefitinib or chemotherapy for non-small-cell lung cancer
with mutated EGFR. N Engl J Med. 362:2380–2388. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mitsudomi T, Morita S, Yatabe Y, Negoro S,
Okamoto I, Tsurutani J, Seto T, Satouchi M, Tada H, Hirashima T, et
al: Gefitinib versus cisplatin plus docetaxel in patients with
non-small-cell lung cancer harbouring mutations of the epidermal
growth factor receptor (WJTOG3405): An open label, randomised phase
3 trial. Lancet Oncol. 11:121–128. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Inomata M, Hayashi R, Yamamoto A, Tokui K,
Taka C, Okazawa S, Kambara K, Suzuki K, Ichikawa T, Yamada T, et
al: Plasma neuron-specific enolase level as a prognostic marker in
patients with non-small cell lung cancer receiving gefitinib. Mol
Clin Oncol. 3:802–806. 2015.PubMed/NCBI
|
|
5
|
Albain KS, Crowley JJ, LeBlanc M and
Livingston RB: Survival determinants in extensive-stage
non-small-cell lung cancer: The Southwest Oncology Group
experience. J Clin Oncol. 9:1618–1626. 1991.PubMed/NCBI
|
|
6
|
Ulas A, Turkoz FP, Silay K, Tokluoglu S,
Avci N, Oksuzoglu B and Alkis N: A laboratory prognostic index
model for patients with advanced non-small cell lung cancer. PLoS
One. 9:e1144712014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Koukourakis MI, Giatromanolaki A, Sivridis
E, Bougioukas G, Didilis V, Gatter KC and Harris AL: Tumour
Angiogenesis Research Group: Lactate dehydrogenase-5 (LDH-5)
overexpression in non-small-cell lung cancer tissues is linked to
tumour hypoxia, angiogenic factor production and poor prognosis. Br
J Cancer. 89:877–885. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Danner BC, Didilis VN, Wiemeyer S,
Stojanovic T, Kitz J, Emmert A, Füzesi L and Schöndube FA:
Long-term survival is linked to serum LDH and partly to tumour
LDH-5 in NSCLC. Anticancer Res. 30:1347–1351. 2010.PubMed/NCBI
|
|
9
|
Seto T, Kato T, Nishio M, Goto K, Atagi S,
Hosomi Y, Yamamoto N, Hida T, Maemondo M, Nakagawa K, et al:
Erlotinib alone or with bevacizumab as first-line therapy in
patients with advanced non-squamous non-small-cell lung cancer
harbouring EGFR mutations (JO25567): An open-label, randomised,
multicentre, phase 2 study. Lancet Oncol. 15:1236–1244. 2014.
View Article : Google Scholar : PubMed/NCBI
|