Introduction
Laparoscopic minimally invasive surgery is widely
used in gynecological operations. Gynecological laparoscopy is a
minimally invasive diagnostic and treatment technology that
integrates modern gynecological surgery and endoscopic diagnosis
and treatment. It has thus become one of the most commonly used
techniques in the field of gynecology. Uterine fibroids are common
benign tumors of the female genital organs and the prevalence has
been reported as high as 20 to 40% (1,2). The
widespread application of pulverizers allows for removal of uterine
fibroids under laparoscopy, which better reflects the superiority
of minimally invasive techniques. However, preoperative diagnosis
of uterine fibroids and uterine sarcoma is very difficult. The
incidence of uterine fibroid sarcoma is approximately 0.03 to 1.00%
(3). Despite the dramatic advances
in imaging technology, diagnostic criteria for preoperative
suspicious leiomyosarcoma are still lacking. The direct use of a
pulverizer under laparoscopic surgery may cause the tumor to spread
in the abdominal cavity, leading to the need for secondary surgery
and affecting the prognosis (4). To
prevent this, after laparoscopic removal of uterine fibroids, the
tumor is placed in a specimen bag, and the modified uterine
fibroids are comminuted in the bag. If laparoscopic hysterectomy is
performed, the isolated uterus is large and difficult to remove
from the vagina at one time. We use a self-made specimen bag to
facilitate smashing of the uterus in the lower pocket of the
vagina. This procedure is described in the present report.
Patients and methods
General information
We identified 82 cases of laparoscopic myomectomy in
our hospital from April 2017 to October 2018. All patients were
diagnosed as intermuscular myoma and subserosal fibroids, according
to preoperative examination. The study protocol was approved by the
Institutional The First Affiliated Hospital of Jiaxing University
(2017-192), and all eligible patients provided written informed
consent prior to treatment. The inclusion criteria were as follows:
i) B type-Ultrasound (transvaginal ultrasound) indicated uterine
fibroids with a single uterine fibroid greater than 4.0 cm in
diameter. ii) For patients with uterine fibroids, preoperative
examination indicated dysplasia in the endometrium, a B
type-ultrasound was performed on the fifth day of menstruation.
Diagnostic curettage or hysteroscopy with diagnostic curettage will
be performed for those whose endometrium was still abnormal.
Patients can only be recruited when there was no endometrial cancer
showed in the postoperative pathology. iii) Patients with rapid
growth of uterine fibroids in short period should receive MRI to
exclude the neoplasm before they can be recruited. The exclusion
criteria included: i) Preoperative examination suggested uterine
fibroids or endometerial canceration. ii) Patients that the
fibroids were found degenerated during the intraoperative stripping
and the consistency was abnormal, swicthed to receivethe
laparatomic surgery. With approval of the ethics committee, 42
cases were randomly selected for the study. The modified uterine
smashing technique was used in the experiment. Patients with other
disease conditions were most common with cardiovascular disease and
metabolic disorders, including 3 patients (7.1%) with hypertension,
2 patients (4.8%) with diabetes, and 1 patient with chronic
bronchitis (2.4%); 1 patient (2.4%) with two or more complications.
Postoperative pathologic examination confirmed 32 cases of uterine
leiomyoma, 9 cases of uterine leiomyoma with degeneration and
abundant cells, 1 case of uterine mucinous myoma. The remaining 40
patients underwent routine surgery without specimen bags. Including
2 patients (5.0%) had hypertension, and 1 patient (2.5%) had
diabetes. Postoperative pathologic examination confirmed 34 cases
of uterine leiomyoma and 6 cases of uterine leiomyoma with
degeneration and abundant cells. There were no significant
differences in the baseline data between the two groups
(P>0.05). There were also no significant differences in the
operation time, blood loss, or average hospital stay between the
two groups (P>0.05; Tables I and II).
We identified 92 patients who underwent laparoscopic
hysterectomy with difficulty in removing the uterus directly from
the vagina from April 2017 to October 2018 in our hospital. The
study protocol was approved by the Institutional The First
Affiliated Hospital of Jiaxing University (2017-192), and all
eligible patients provided written informed consent prior to
treatment. The inclusion criteria were as follows: i) According to
the indications of hysteretomy. ii) If there were indications for
removal of uterus and the preoperative examination showed
abnormalities in the endometrium, a B type-Ultrasound was performed
on the fifth day of menstruation. Diagnostic curettage or
hysteroscopy with diagnostic curettage will be performed for those
whose endometrium was still abnormal. Patients can only be
recruited when there was no endometrial cancer showed in the
postoperative pathology. The exclusion criteria included: The
posibility of malignancy cannot be excluded and the malignancy was
highly suspected, considering the uterus was usually large, a
laparotomic surgery was needed and the patient should be excluded
from the experiment. With approval of the ethics committee, 49
patients were randomly selected for inclusion in the study.
Intraoperative self-made specimen bags were used for the modified
uterine smashing technique. Patients with other disease conditions
were most common with cardiovascular disease and metabolic
disorders, including 5 patients (10.2%) with hypertension, 4
patients (8.2%) with diabetes, and 2 patient with chronic
bronchitis (4.1%); 3 patient (6.1%) with two or more complications.
There were 43 cases of laparoscopic uterine double salpingectomy
and 6 cases of laparoscopic uterine bilateral adnexectomy. In
total, 27 cases of uterine multiple leiomyoma were confirmed by
postoperative pathology, including 4 cases of uterine leiomyoma
with degeneration and abundant cells, 18 cases of myopathy, 3 cases
of endometrial atypical hyperplasia, and 1 case of endometrial
cancer. The patient was a perimenopausal woman, who was diagnosed
with cervical intraepithelial neoplasia III and received cervical
conization. The preoperative examination did not indicate abnormal
endometrium. Because the patient strongly requested to remove the
uterus, so a minimally invasive surgery was performed.
Postoperative pathological accidentally showed endometrial
malignancy. The remaining 43 patients underwent routine surgery
without specimen bags. Including 6 patients (14.0%) with
hypertension, 4 patients (9.3%) with diabetes, and 1 patient with
chronic bronchitis (2.3%); 4 patient (9.3%) with two or more
complications. Postoperative pathologic examination confirmed 31
cases of uterine leiomyoma, including 3 cases of uterine leiomyoma
with degeneration and abundant cells, 10 cases of myopathy, 2 cases
of endometrial atypical hyperplasia. There were no significant
differences in the baseline data between the two groups
(P>0.05). There were also no significant differences in the
uterine weight, operative time, or average length of hospital stay
between the two groups (P>0.05; Tables III and IV).
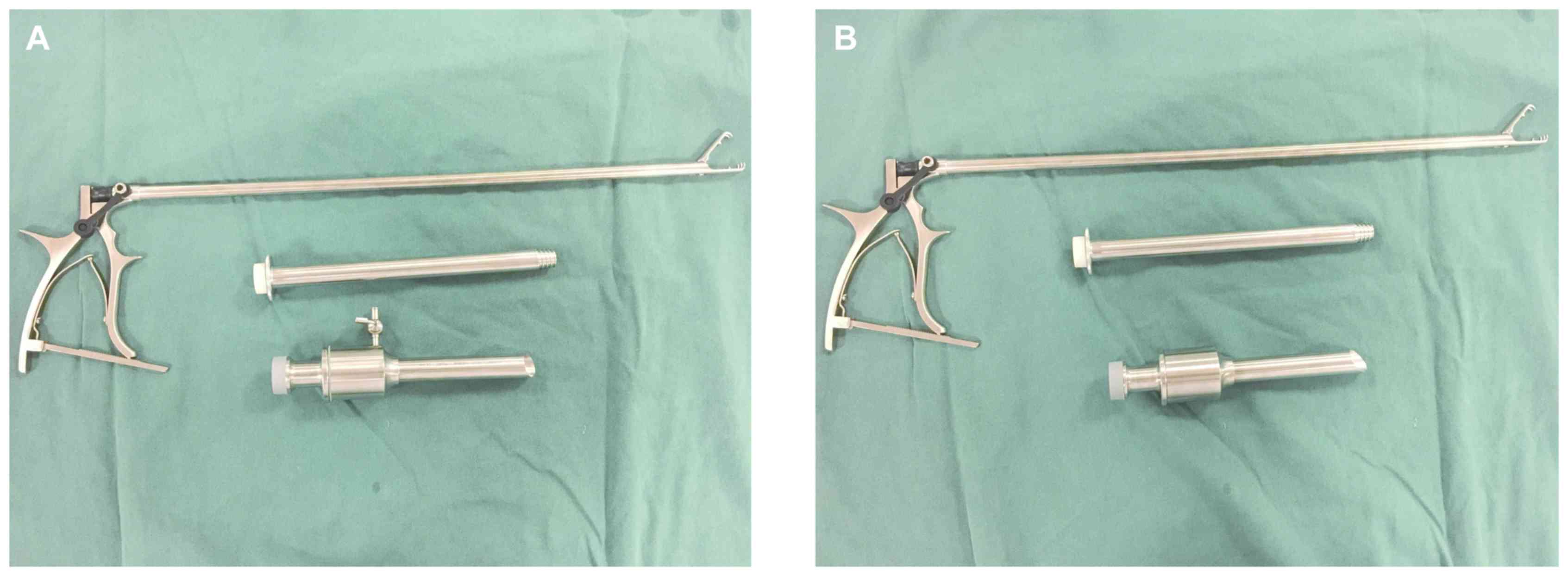
Specimen bag specifications
The width of our hospital's disposable laparoscopic
sheath (disposable suction bag) is 20 cm, and the end with the
white line is selected as the head end of the specimen bag. The
length must be larger than the diameter of the fibroids (generally
approximately 10-15 cm). After determining the length, the bottom
of the specimen bag is knotted or tied with silk thread (Fig. 1).
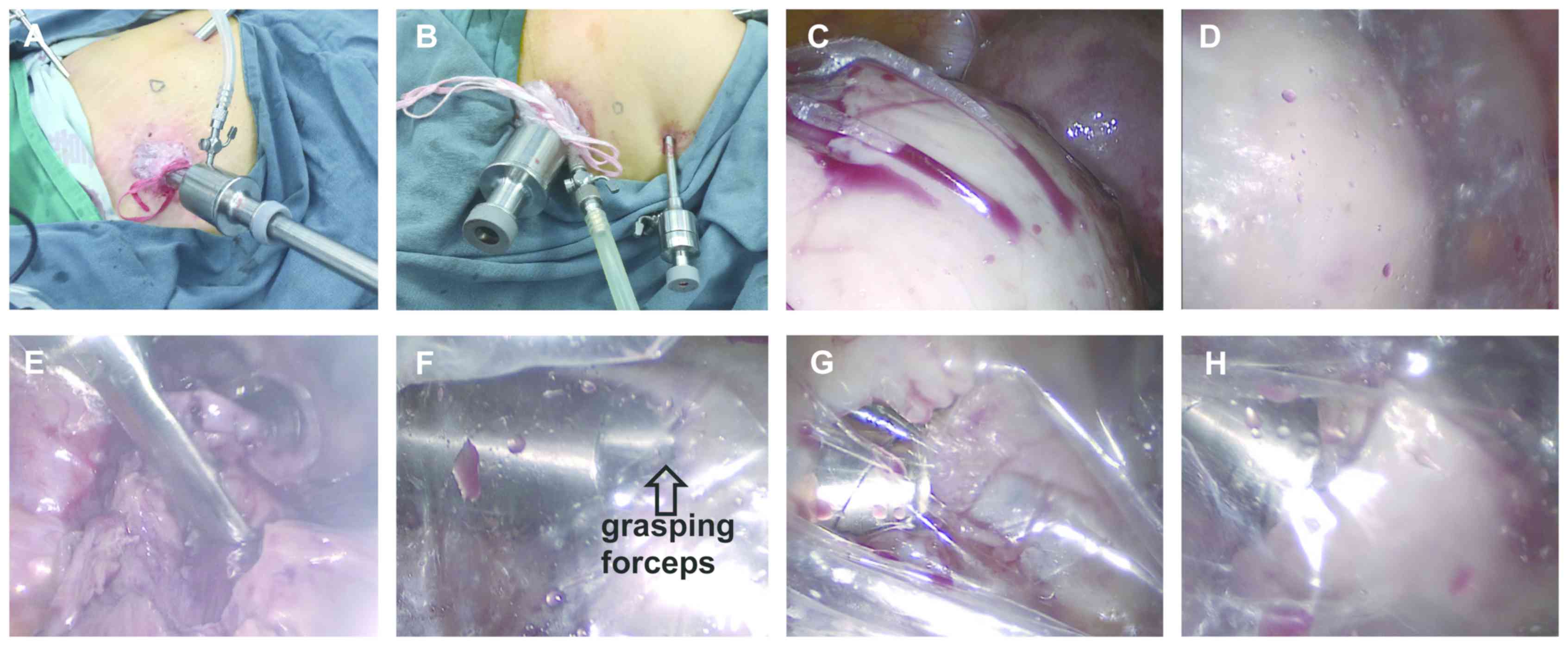
Operation of specimen bag during
uterine myomectomy
After establishment of general anesthesia, the
laparoscopic instrument was appropriately positioned and the
pre-made specimen bag was placed into the abdominal cavity through
a 10-mm trocar. The bag was then placed in the right fossa iliaca
and the mouth of the bag was opened. Laparoscopic myomectomy was
then routinely performed. After the fibroids had been completely
removed, the fibroid specimens were placed directly into the
specimen bag. The surgical wound was routinely sutured, and the
10-mm puncture hole in the left lower abdomen was enlarged to 15
mm. The end of the specimen bag with the white line was then placed
outside the abdominal cavity, and a metal trocar was placed in the
pulverizer. The head end of the specimen bag was fixed and sealed
on the trocar by a white wire, and the inflation port on the trocar
was connected to the pneumoperitoneum tube and opened for inflation
until the laparoscopic direct-view specimen bag bulged and the line
of sight was good (Figs. 2A and
3A). (If the trocar of the
pulverizer has no inflation port, the pneumoperitoneum tube can be
placed into the abdominal cavity together with the trocar). After
the specimen bag was fixed in the same way, the pneumoperitoneum
tube was used to inflate the specimen bag (Figs. 2B and 3B). After successful inflation of the
specimen bag, the fibroids were pulverized within the specimen bag
using the pulverizer under direct vision in the abdominal cavity,
because the laparoscopic mirror is attached to the specimen bag,
the line of sight is slightly blurred, but does not affect the
operation (Fig. 3C-H). During this
process, the specimen can be pulverized to <1.5 cm (i.e., the
size of the incision), and the remaining specimen fragments can be
taken out together with the specimen bag from the abdominal
cavity.
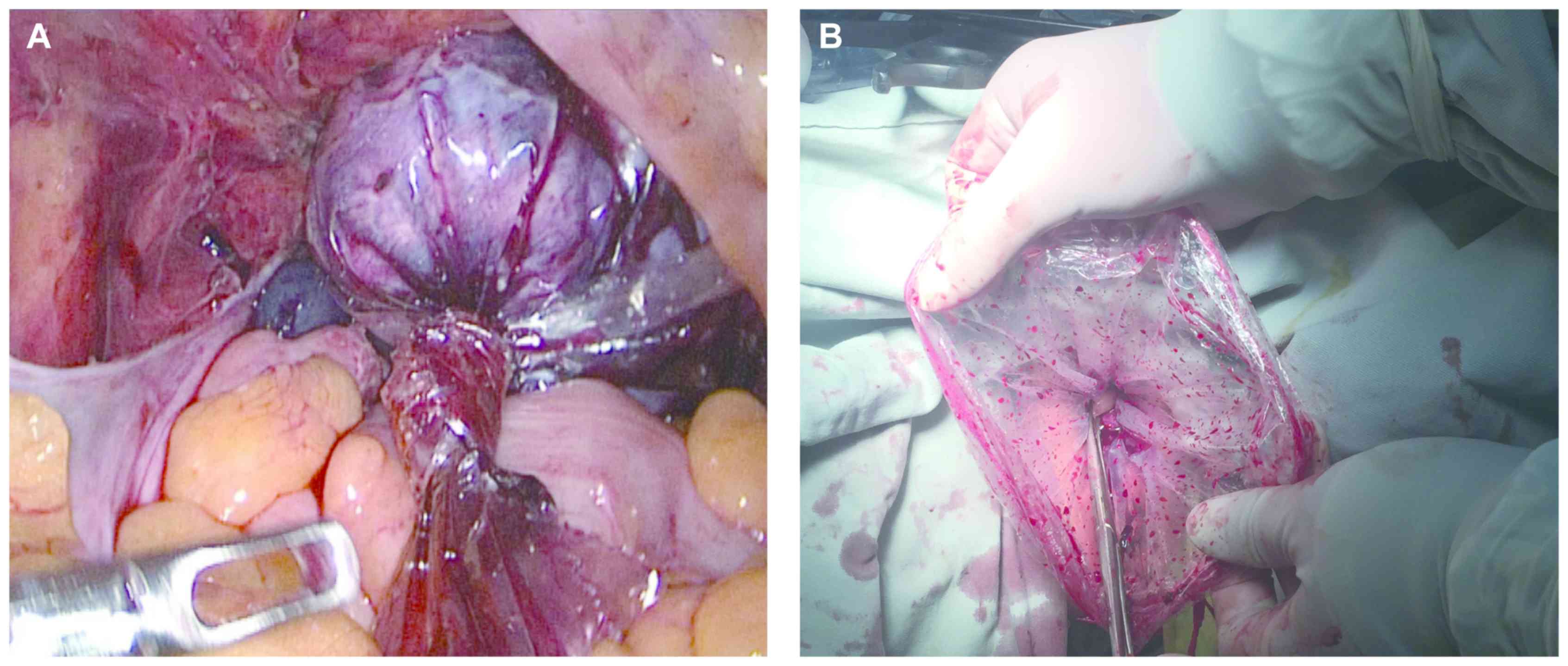
Operation of specimen bag during
hysterectomy
After establishment of general anesthesia, routine
laparoscopic hysterectomy was performed. The vaginal wall was
completely disconnected via laparoscopy, than the self-made
specimen bag which was from the umbilical trocar, was all placed in
the abdominal cavity. All the uterus was placed into the specimen
bag. The end of the specimen bag with the white line was tightened
at the mouth of the uterus outside the uterus cup, and the end of
the bag with the white line was removed from the vagina. After the
mouth of the bag was outside the vaginal opening, the knot was
opened and the uterus was removed. The uterus was then smashed
under gross vision outside the vaginal opening (Fig. 4). The uterus was then taken out
together with the specimen bag. When the specimen was taken in the
vagina, the patient was changed to the supine position, and the
intra-abdominal inflation was stopped.
Statistical analysis
All statistical analyses were performed using SPSS
21.0 software (IBM Corp.). Quantitative data are expressed as the
mean ± standard deviation, and the t-test was used for comparison
between groups. Count data are expressed as a percentage. P<0.05
was considered to indicate a statistically significant
difference.
Results
Results and complications
Using the self-made specimen bag, the fibroids in
the abdominal cavity were crushed and the uterus was smashed and
removed under the vaginal opening. When smashing fibroids, use of
the specimen bag prevents small specimen fragments from splashing
into the abdominal cavity, causing intra-abdominal implantation and
spread. For patients with degenerative uterine fibroids, the bag
also avoids the spread of tumor cells that are invisible to the
naked eye. It is only necessary to smash the specimen to <1.5 cm
during surgery (i.e., the size of the incision), which decreases
the surgical time; finally, the small specimen is removed from the
abdominal cavity together with the specimen bag. No small fibroid
tissue remains in the abdominal cavity. When the uterus is
comminuted in the vagina during total hysterectomy, the protection
of the specimen bag prevents implantation of the tumor including
the endometrium in the abdominal cavity. In the present study, no
damage to the intraoperative specimen bag occurred in any patient,
and the abdominal wall wounds healed after surgery.
Outcomes following the laparoscopic
myomectomy group
In the laparoscopic myomectomy group, the data
analysis showed no significant differences in the preoperative data
between the specimen bag group and the non-standard bag group
(P>0.05). The diameters of the uterine fibroids were not
significantly different between the groups (P>0.05; Table I). There was no significant
difference in the operation time, intraoperative blood loss, or
average hospitalization duration between the two groups (P>0.05;
Table II). These findings indicate
that intraoperative use of the specimen bag is a feasible
procedure. The follow-up ranged from 1 to 23 months (average, 12.6
months), and the follow-up rate was 100%. No postoperative
complications such as wound infection or bleeding occurred.
 | Table IComparison of general basic data
between the two groups in laparoscopic myomectomy. |
Table I
Comparison of general basic data
between the two groups in laparoscopic myomectomy.
| Parameter | Traditional group
(n=40) | Improved group
(n=42) | P-value |
|---|
| Age, years | 42.77±6.25 | 42.19±5.18 | 0.645 |
| BMI,
kg/m2 | 25.57±3.09 | 25.27±3.48 | 0.678 |
| Number of caesarean
section operations in the past | 0.83±0.64 | 0.81±0.71 | 0.917 |
| Number of abdominal
surgeries in the past | 1.08±0.76 | 1.07±0.84 | 0.984 |
| Average diameter of
uterine fibroids, cm | 7.43±2.10 | 7.33±1.54 | 0.822 |
| Number of uterine
fibroids | 1.33±0.57 | 1.33±0.61 | 0.949 |
 | Table IIComparison of surgical data between
the two groups in laparoscopic myomectomy. |
Table II
Comparison of surgical data between
the two groups in laparoscopic myomectomy.
| Parameter | Traditional group
(n=40) | Improved group
(n=42) | P-value |
|---|
| Operation time,
min | 70.10±16.87 | 71.40±17.71 | 0.734 |
| Intraoperative blood
loss, ml | 71.78±29.00 | 68.38±29.94 | 0.603 |
| Average hospital
stays, days | 4.08±0.35 | 4.05±0.22 | 0.669 |
Outcomes following the laparoscopic
hysterectomy group
In the laparoscopic hysterectomy group, the uterus
body was smashed and taken out of the vaginal opening under direct
vision. Five patients had hematuria because of the uterine was so
large that the intravaginal specimen was taken longer during the
operation. Hematuria disappeared by 24 h postoperatively, and the
urethral catheter was removed 3 days postoperatively; no urinary
leakage occurred. The average time for specimen bag placement was
4.42 min, which was negligible. No rupture of the specimen bag
occurred during the operation, no complications occurred after the
operation, and the average length of hospital stay was within the
plan. Postoperative follow-up was performed for 1 to 23 months
(average, 11.3 months). The follow-up rate was 100%. No
complications such as wound infection, hemorrhage, or urinary tract
injury occurred (Table V).
 | Table VSpecific surgical data of the specimen
bag group in laparoscopic hysterectomy. |
Table V
Specific surgical data of the specimen
bag group in laparoscopic hysterectomy.
| Parameter | Surgical data |
|---|
| Average uterus
weight, g | 372.31 (210-710) |
| Uterine weight ≥300
g, n | 32 (65.31%) |
| Place specimen bag
time, min | 4.47 |
| Intraoperative
complications, n | 5 (10.20%) |
| Intraoperative
specimen bag rupture, n | 0 (0%) |
| Transfer to open,
n | 0 (0%) |
| Postoperative
complications, n | 0 (0%) |
| Average hospital
stays, days | 4.76 (4-6) |
Discussion
Traditional open surgery, specimens can be easily
removed from the abdominal cavity. However, in laparoscopic
myomectomy and laparoscopic hysterectomy, the specimen is too large
to be directly removed from the abdominal wall incision or the
vagina. In 1991, Semm reported the first manual crushing device
(5). In 1993, Steiner et al
invented the electric splitter (6).
The US Food and Drug Administration (FDA) reviewed and passed the
use of an electric splitter in myomectomy (K946147) and total
hysterectomy (K993801) in 1995 and 2000, respectively, which
undoubtedly promoted the rapid development of gynecological
laparoscopy. However, many clinical data analyses have shown that
the use of electric shredders can actually increase the
dissemination of uterine fibroids (7,8), while
the dissemination rate of uterine sarcoma after abdominal
dissection is as high as 13 to 57% (7,9,10). Park et al (4) retrospectively analyzed 56 cases of
uterine leiomyosarcoma, 25 of which had involved the use of
electric shredders. They found that use of the electric shredder
was associated with a worse prognosis and shorter survival time.
Morice et al (11) reported
that the recurrence and metastasis rates in the pelvic and
abdominal cavities increased within 3 months after malignant tumor
tissue was divided. At the end of 2014, the FDA issued a statement
on the use of electric breakers in minimally invasive surgery for
uterine fibroids, indicating that the use of shredders in patients
with suspected or clear uterine malignancies should be avoided
(12). So some scholars have
suggested abandoning use of the shredder (13). The literature indicates that the
incidence of uterine fibroid sarcoma is approximately 0.03 to 1.00%
(14). Although imaging techniques
have been greatly improved, the differential diagnosis of uterine
fibroids and uterine sarcoma before surgery is difficult, and
diagnostic criteria for preoperative suspicious leiomyosarcoma are
still lacking. Therefore, diagnosis of uterine malignant tumors is
still often made after pathological diagnosis of uterine fibroids,
which leads to intraoperative tumor dissemination. Even in patients
with benign uterine leiomyoma, peritoneal disseminated leiomyoma
may occur. However, when a pulverizer is applied to the whole
uterus, it may cause endometrial and malignancy dissemination. In
this way, minimally invasive surgery has become extremely
important.
To this end, we considered placing the specimen in a
sealed specimen bag for improved comminution. Although specialized
closed specimen bags are commercially available, but they are
expensive, and increase not only the patient's economic burden but
also the hospital's expenditure on medical resources. We modified
the disposable luminal sheath in the basic configuration of the
laparoscope into a specimen bag. The specimen bag is free to the
patient. For the medical institution, the material of the specimen
bag is a necessary material for the endoscope, even in primary
hospitals. The low price of this material will not significantly
increase the medical expenses. Notably, however, the improved
smashing of the uterus specimen is associated with limited vision,
which makes the operation more difficult and indirectly prolongs
the operation time. If we can improve the material of the specimen
bag and make the visual field high, we can solve this problem.
To resolve the above difficulties and better avoid
tumor spread, the author recommends the following based on clinical
experience (1). When laparoscopic
uterine fibroids are removed, the specimen bag is placed before the
fibroids are removed (2). The
incision on the surface of the uterine fibroids should be large
enough to facilitate removal of the fibroids, and the removal of
fissures during the process of removing the fibroids should be
minimized. This is also beneficial to reduce the spread of tumor
cells during the process of stripping the fibroids (3). After the fibroids have been removed,
the specimens are put directly in the specimen bag that has been
pre-placed in the uterine rectal sulcus to reduce the exposure time
of the fibroids in the abdominal cavity (4). The uterus was sutured before the
specimen was taken. This not only reduces the bleeding of the wound
surface but also slightly reduces the volume of the in vitro
specimen, which is conducive to clear vision when crushing
(5). The specimen bag should be
large enough, and its length should be larger than the diameter of
the specimen by 5 to 10 cm. When the fibroids are smashed, the
specimen bag is large enough to be inflated, and an adequate length
of the specimen bag outside the abdominal cavity is also
guaranteed. When the whole uterus specimen is removed from the
vagina, it is necessary to ensure that the mouth of the specimen
bag is located outside the vaginal opening to fully protect the
vagina and the abdominal cavity (6).
When smashing the fibroids, the specimen bag is preheated with warm
water before being placed in the abdominal cavity to reduce the fog
generated in the intraoperative bag. Some surgeons can master
intraoperative use of a mirror, and sometimes the mirror can even
be attached to the specimen bag to observe the specimen in the bag
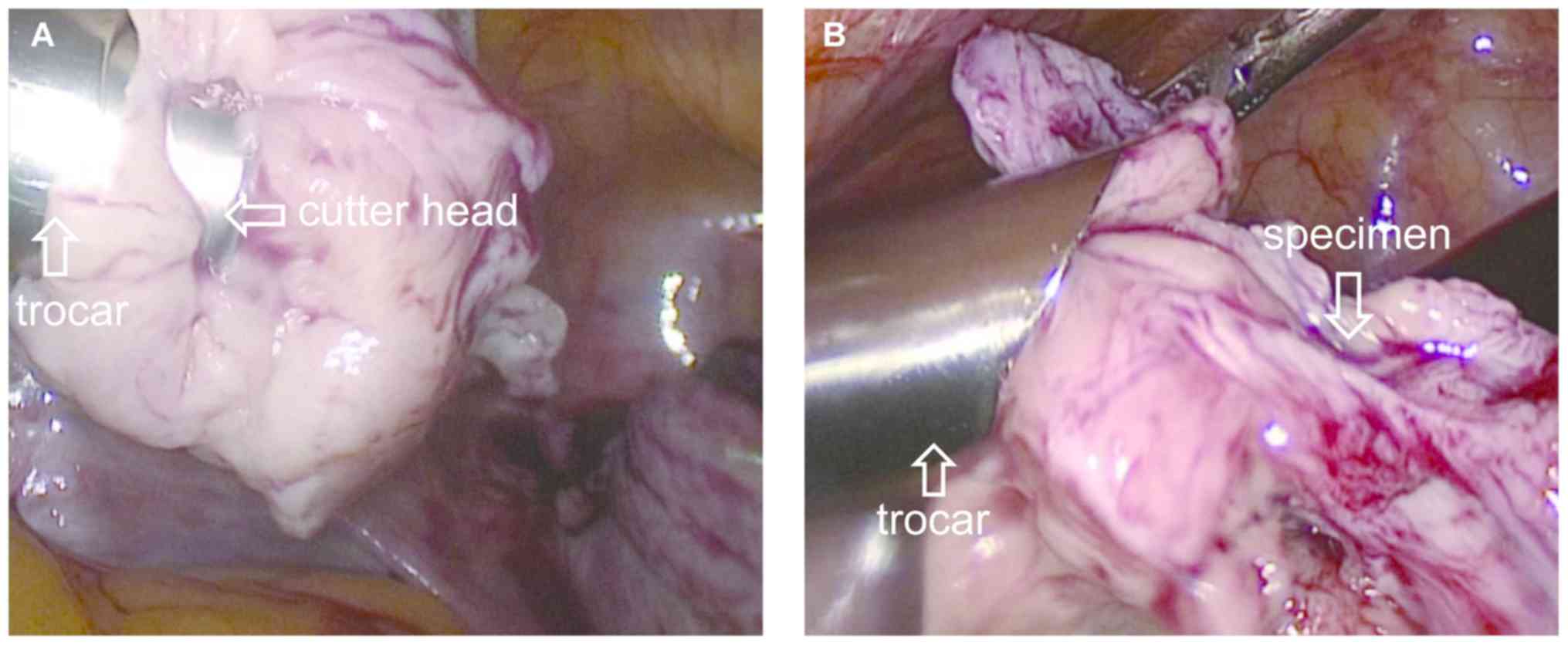
(7). When smashing fibroids, it is
necessary to master the use of a pulverizer. The trocar of the
pulverizer should be placed in the center of the specimen bag, and
the cutter head should be located approximately 5 cm below the
abdominal wall. The cutter head of the pulverizer can be mostly or
even all hiddend in the trocar, which prevents the specimen from
rotating with the cutter head and prevents damage to the
surrounding organs by the cutter head (Fig. 5). When pulverizing the fibroids, the
surgeon should start from the surface; the inside of the fibroids
should not be penetrated, and good vision and surgical safety
should be ensured. Specimens that are pulverized to a diameter of
≤1.5 cm can be removed without being taken out together with the
specimen bag, and the time for pulverizing the specimen is
reduced.
Our hospital has improved the laparoscopic uterine
smashing technique, which has been applied to laparoscopic uterine
fibroid removal in 42 cases, laparoscopic uterine double
salpingectomy in 43 cases, and laparoscopic uterine bilateral
adnexectomy in 6 cases. Among these 91 cases, none were abandoned
due to accidents. During laparoscopic myomectomy, creation and
insertion of the specimen bag took a short time, and use of the
specimen bag reduced the time required for smashing and removal of
the specimen debris; thus, the operation time was not significantly
extended. During laparoscopic hysterectomy, when the specimen is
too large to be removed from the vagina at one time, we have
adopted an improved method that only increases the time to place
the specimen bag (average time of 4.47 min) for the entire
procedure. This increase is negligible in terms of the entire
operation. No signs of tumor dissemination were observed throughout
follow-up in the present study. However, the follow-up time was
limited, and the postoperative pathologic examination confirmed
that the data of malignant tumors was too little. Thus, longer-term
follow-up and additional samples are needed. Additionally, the
specimen bag can be used for the laparoscopic removal of ovarian
tumors. Because the pulverizer is not involved, it does not need to
be inflated and can be directly removed from the abdominal cavity
or vagina. During the removal process, the ovarian tumor can be
aspirated and drained in the bag to capture the specimen. Moreover,
our self-made specimen bags are made from disposable laparoscopic
sheaths. Even in primary hospitals, such sheaths will be used as
long as the endoscopes are used. This product is inexpensive, does
not incur an extra charge, and does not significantly increase the
medical costs. This can be described as a ‘win-win situation.’ The
herein-described self-made specimen bag technology is suitable for
a wide range of applications, especially in primary hospitals.
Acknowledgements
Not applicable.
Funding
The present study was supported by a grant from The
Science and Technology Project of Jiaxing in China (grant no.
2018AD32061; patent no. 2018206067047).
Availability of data and materials
The datasets generated and analyzed during the
current study are available from the corresponding author on
reasonable request.
Authors' contributions
XS and SZ conceived the study and conducted a
critical pre-submission review of the manuscript. XS was
responsible for patients' enrollment, operations practice and the
manuscript writing. LS made substantial contributions to the design
of the study. XS, LS and SZ scanned and identified the relevant
literature according to the inclusion and exclusion criteria. XS,
LS and SZ and modified parts of the manuscript. All the authors
have read and approved the final version of the manuscript for
publication.
Ethics approval and consent to
participate
All patients provided written informed consent prior
to enrolment. The present study protocol was approved by the
institutional The First Affiliated Hospital of Jiaxing University
(2017-192). The procedures followed were in accordance with the
ethical standards of the responsible committee on human
experimentation (institutional or regional) and with the Helsinki
Declaration of 1975, as revised in 2000.
Patient consent for publication
All patients approved the publication of the present
study.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ryan GL, Syrop CH and Van Voorhis BJ:
Role, epidemiology, and natural history of benign uterine mass
lesions. Clin Obstet Gynecol. 48:312–324. 2005.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Wallach EE and Vlahos NF: Uterine myomas:
An overview of development, clinical features, and management.
Obstet Gynecol. 104:393–406. 2004.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Hagemann IS, Hagemann AR, LiVolsi VA,
Montone KT and Chu CS: Risk of occult malignancy in morcellated
hysterectomy: A case series. Int J Gynecol Pathol. 30:476–483.
2011.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Park JY, Park SK, Kim DY, Kim JH, Kim YM,
Kim YT and Nam JH: The impact of tumor morcellation during surgery
on the prognosis of patients with apparently early uterine
leiomyosarcoma. Gynecol Oncol. 122:255–259. 2011.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Semm K: Hysterectomy via laparotomy or
pelviscopy. A new CASH method without colpotomy. Geburtshilfe
Frauenheilkd. 51:996–1003. 1991.(In German). PubMed/NCBI View Article : Google Scholar
|
|
6
|
Steiner RA, Wight E, Tadir Y and Haller U:
Electrical cutting device for laparoscopic removal of tissue from
the abdominal cavity. Obstet Gynecol. 81:471–474. 1993.PubMed/NCBI
|
|
7
|
Tan-Kim J, Hartzell KA, Reinsch CS, O'Day
CH, Kennedy JS, Menefee SA and Harrison TA: Uterine sarcomas and
parasitic myomas after laparoscopic hysterectomy with power
morcellation. Am J Obstet Gynecol. 212:594.e1–10. 2015.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Bogani G, Cliby WA and Aletti GD: Impact
of morcellation on survival outcomes of patients with unexpected
uterine leiomyosarcoma: A systematic review and meta-analysis.
Gynecol Oncol. 137:167–172. 2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Ehdaivand S, Simon RA, Sung CJ, Steinhoff
MM, Lawrence WD and Quddus MR: Incidental gynecologic neoplasms in
morcellated uterine specimens: A case series with follow-up. Hum
Pathol. 45:2311–2317. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Liu FW, Galvan-Turner VB, Pfaendler KS,
Longoria TC and Bristow RE: A critical assessment of morcellation
and its impact on gynecologic surgery and the limitations of the
existing literature. Am J Obstet Gynecol. 212:717–724.
2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Morice P, Rodriguez A, Rey A, Pautier P,
Atallah D, Genestie C, Pomel C, Lhommé C, Haie-Meder C, Duvillard P
and Castaigne D: Prognostic value of initial surgical procedure for
patients with uterine sarcoma: Analysis of 123 patients. Eur J
Gynaecol Oncol. 24:237–240. 2003.PubMed/NCBI
|
|
12
|
Hampton T: Use of morcellation to remove
fibroids scrutinized at FDA hearings. JAMA. 312(588)2014.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Kho KA and Nezhat CH: Evaluating the risks
of electric uterine morcellation. JAMA. 311:905–906.
2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Sala E, Rockall AG, Freeman SJ, Mitchell
DG and Reinhold C: The added role of MR imaging in treatment
stratification of patients with gynecologic malignancies: What the
radiologist needs to know. Radiology. 266:717–740. 2013.PubMed/NCBI View Article : Google Scholar
|