Introduction
Nephrotic syndrome (NS), a collection of signs and
symptoms that may indicate improper kidney function, has many
causes, including primary kidney diseases such as Amyloid A
amyloidosis, minimal-change disease, IgA or membranous nephropathy
(MN) (1). MN is the most common
cause of NS in adults. A previous study estimated that 25% of
patients over 60 years of age with MN experience an associated
cancer, and the overall incidence of cancer in patients with MN was
found to be 7.9% (2). To the best
of our knowledge, few clinical cases of paraneoplastic nephrotic
syndrome (PNS) with lung cancer have been reported, and there is
ongoing dispute about whether immunosuppressive or corticosteroid
treatment combined with primary therapy for PNS is beneficial.
Here, we describe a patient with lung cancer accompanied by NS
treated with radiotherapy.
Case report
An 80-year-old man was admitted to our hospital with
cough and peripheral oedema on December 10th, 2017. His family
history showed no particular disease, and he was physically fit in
the past. Laboratory data at admission are shown in Table I. The following symptoms were
associated with this case: Albuminuria, hyperlipidaemia, oedema,
and hypoalbumina. These clinical findings are consistent with the
diagnosis of NS. To determine the cause of NS, a kidney biopsy was
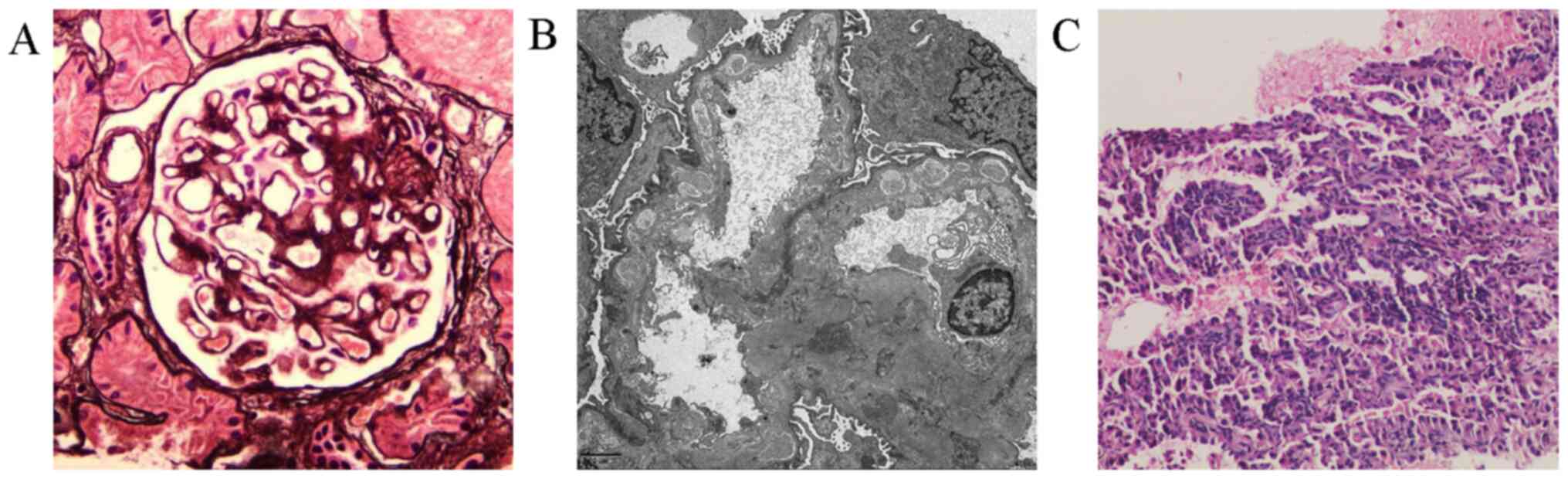
performed. Light microscopy of the renal biopsy in December 2017
revealed mild thickening of the basement membrane and the
absorption of partial immune complexes by periodic acid-methenamine
silver (PASM) staining (Fig. 1A).
Electron microscopy showed electron-dense deposits beneath the
epithelium of the glomerular basement membrane, and partial
electron-dense deposits deposited in the basement membrane were
absorbed (Fig. 1B). MN was
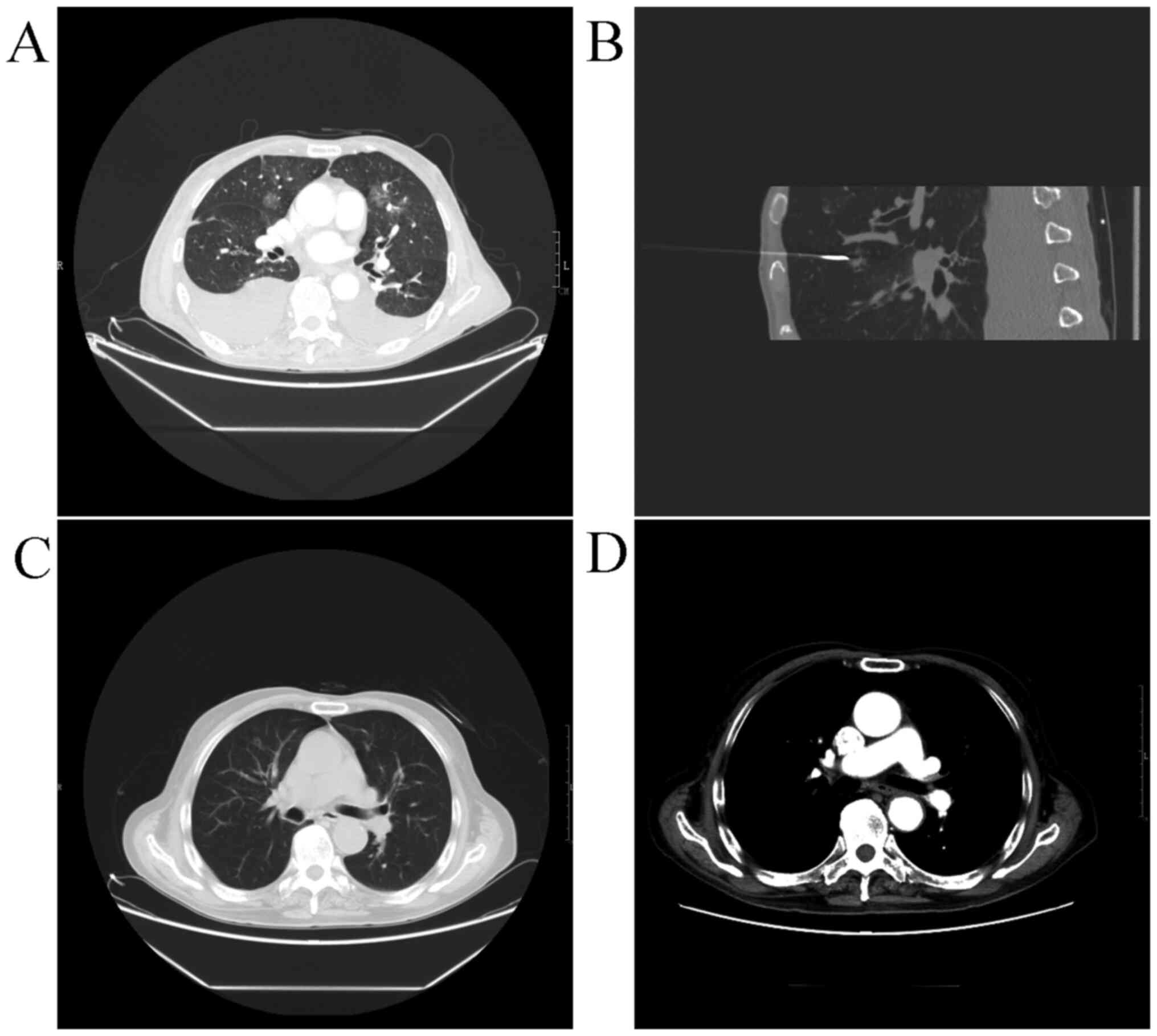
diagnosed on the basis of these findings. Chest computed tomography
(CT) revealed an approximately 16x14 mm nodule in the medial
segment of the right lobe of the lung, with no enlarged lymph nodes
(Fig. 2A). Because the clinical
manifestation was NS, we considered it to be an SNS, and no
glucocorticoids, immunosuppressants, or other drugs to treat the
NS, such as Ras inhibitors, were initiated.
 | Table ILaboratory data at admission. |
Table I
Laboratory data at admission.
| Variable | Value |
|---|
| Haemoglobin,
mg/dl | 9 |
| White blood
cells/m3 | 4920 |
| BUN, mg/dl | 85.82 |
| Creatinine,
mg/dl | 3.33 |
| Total protein,
g/dl | 3.29 |
| Albumin, g/dl | 1.67 |
| 24-h urinary protein
quantity, g/24 h | 6.51 |
| CEA, ng/ml | 2.08 |
| CA19-9, U/ml | 17.92 |
| CA-125, U/ml | 576.4 |
| CYFRA21-1, ng/ml | 4.0 |
| NSE, ng/ml | 19.16 |
After twenty-four days of conservative treatments of
anti-infection, diuretic and supportive therapies, the symptoms of
NS and renal function were not apparently improved. Therefore, the
patient was transferred from the Department of Nephrology to
Oncology. The laboratory data were checked again, with total
protein of 3.56 g/dl, albumin of 1.68 g/dl, creatinine of 3.86
mg/dl, BUN of 53.99 mg/dl, and 24-h urinary protein over 12 g/24 h.
In addition, the serum CA-125 level was remarkably elevated at
726.7 U/ml. The pathology determined from percutaneous cutting
needle biopsy of the lung nodule showed the tumour cells to be
mildly heteromorphic and arranged in the form of adenotubules;
immunohistochemical staining showed positivity for CK, CK7 and
TTF-1, among others (Fig. 1C).
Therefore, the diagnosis was stage I non-small cell lung cancer
(NSCLC), and genetic testing indicated that the patient carried a
wild-type epidermal growth factor receptor gene. We suspected that
NS had developed secondarily to lung cancer. Therefore, we chose
the small wound method for implantation of iodine-125 radioactive
particles into the lung cancer under the guidance of CT (Fig. 2B).
The patients' postoperative course was uneventful.
Three months after the operation, his serum albumin (3.50 g/dl) and
creatinine (1.22 mg/dl) levels improved markedly, the CA-125 level
returned to normal, and the peripheral oedema disappeared. CT
showed a dramatic reduction in the size of the lung nodule
(Fig. 2C), with no enhancement in
the nodule (Fig. 2D). The patient
remains in good shape, with no recurrence of the tumour or
nephrotic syndrome to date.
Discussion
The concept of paraneoplastic glomerulopathy was
introduced in 1922 by Galloway, who noted that PNS can be recovered
with cure of malignant disease outside of the kidney (3). The association between malignant
tumours and nephrotic syndrome was first identified in 1966 by Lee
et al (4). Of their 101
adult patients who presented with NS in a 10-year period, 11 (11%)
were found to have carcinoma (4).
Therefore, physicians should investigate underlying malignant
disease in the elderly, especially in those over 60 years, with
NS.
MN, an antibody-mediated glomerular disease, is the
major cause of NS in adults and is initiated by the in situ
formation of immune deposits between circulating nephritogenic
antibodies and endogenous or exogenous planted antigen (5). The formation of immune deposits on the
outer aspect of the glomerular basement membrane will cause
glomerular damage and proteinuria. MN has three outcomes:
Spontaneous remission has been noted in 20 to 30% of cases and
progressive renal failure in another 20 to 40%; the remaining
patients maintain constant proteinuria even after 5 to 10 years
(6). The factors associated with a
worse prognosis include age, male sex, hypertension, malignancy,
infection, systemic disease and degree of glomerulosclerosis on
biopsy. As few cases of MN caused by NSCLC have been reported since
1922, it is difficult to explain whether the association between
malignant disease and MN in the present patient was a coincidence
or an aetiological association. On rare occasions, remissions and
relapses of glomerular disease have been noted to occur with
removal or relapse of the malignancy (7). In addition, more studies are required
to determine whether patients with MN should receive cancer
screening and whether kidney disease is associated with a specific
type of tumour.
MN has long been controversial. Some patients
experience spontaneous remission after several months or even
years, and drugs such as glucocorticoids, immunosuppressants and
cytotoxic drugs have many side effects (8). Furthermore, it is very difficult to
identify idiopathic MN and SMN by kidney biopsy alone. Indeed, it
is not always the case that SMN can exhibit histopathological clues
distinguishing it from idiopathic MN. In this case, the principal
clinical manifestations were cough, peripheral oedema and renal
dysfunction, with no haemoptysis, expectoration or weight loss.
Although the symptoms of NS, and not lung cancer, were present, we
diagnosed SMN because of the old age, anaemia and nodule in the
lung, among other reasons. Therefore, no corticosteroids or
immunosuppressants for the impaired nephric function of unknown
reasons were applied; additionally, there is a poor effect with the
separate use of glucocorticoids for MN. Glucocorticoids combined
with immunosuppressants can sometimes cure idiopathic MN (9), but the side effects of
immunosuppressants (10) are
frequently highlighted. After nodule puncture, we determined this
case to be MN secondary to lung cancer. The use of
immunosuppressants may reduce body immunity and cause tumour
aggravation.
Secondary NS remission caused by PNS may be achieved
by aggressive tumour therapy, such as traditional surgery combined
with chemotherapy or radiotherapy, independently of corticosteroid
combined with immunosuppressant administration, among other
approaches (11). The reported
prevalence of paraneoplastic syndromes in lung cancer is
approximately 10% (12).
Paraneoplastic syndromes appear to be more common in patients with
small-cell lung cancer than in patients with NSCLC, and the two
types of lung cancer may be associated with different types of
paraneoplastic syndromes (13). For
such tumours, which are particularly difficult to treat, no
standard therapy has been established for the syndrome when it is
associated with cancer, and a radical operation still achieves the
best treatment results. Regardless, only 25% of all patients are
candidates for surgical treatment (14,15).
Martinez-Monge et al (16)
have suggested that CT-guided percutaneous implantation of
radioactive particles is an effective treatment for patients with
medically inoperable stage I NSCLC. In consideration of the
patient, we chose implantation of iodine-125 radioactive particles
into the lung cancer, which is relatively less invasive than other
methods.
PNS related to NSCLC and cured with implantation of
radioactive iodine-125 is rare. Patients with refractory NS caused
by a malignant tumour should pay more attention to the treatment of
the cancer.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analysed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XY, ZF, WC and ZW analyzed and interpreted the
clinical patient data. XY and WC cared for the patient, provided
professional opinions about writing this report. ZF and ZW managed
the whole project, and reviewed and revised the manuscript. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
The patient provided written informed consent for
participation preoperatively.
Patient consent for publication
Written informed consent for the publication of any
associated data was obtained from the patient.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Takane K, Midorikawa Y, Yamazaki S,
Kajiwara T, Yoshida N, Kusumi Y and Takayama T: Gastrointestinal
stromal tumor with nephrotic syndrome as a paraneoplastic syndrome:
A case report. J Med Case Rep. 8(108)2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Zech P, Colon S, Pointet P, Deteix P,
Labeeuw M and Leitienne P: The nephrotic syndrome in adults aged
over 60: Etiology, evaluation and treatment of 76 cases. Clin
Nephrol. 17:232–236. 1982.PubMed/NCBI
|
|
3
|
Galloway J: Remarks on Hodgkin's disease.
Br Med J. 2:1201–1208. 1922.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Lee JC, Yamauchi H and Hopper J Jr: The
association of cancer and the nephrotic syndrome. Ann Intern Med.
64:41–51. 1966.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Vivarelli M, Emma F, Pellé T, Gerken C,
Pedicelli S, Diomedi-Camassei F, Klaus G, Waldegger S, Ronco P and
Debiec H: Genetic homogeneity but IgG subclass-dependent clinical
variability of alloimmune membranous nephropathy with anti-neutral
endopeptidase antibodies. Kidney Int. 87:602–609. 2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Cattran DC, Appel GB, Hebert LA, Hunsicker
LG, Pohl MA, Hoy WE, Maxwell DR and Kunis CL: North America
Nephrotic Syndrome Study Group. Cyclosporine in patients with
steroid-resistant membranous nephropathy: A randomized trial.
Kidney Int. 59:1484–1490. 2001.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Beck LH Jr and Salant DJ: Membranous
nephropathy: Recent travels and new roads ahead. Kidney Int.
77:765–770. 2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Beck LH Jr, Fervenza FC, Beck DM, Bonegio
RG, Malik FA, Erickson SB, Cosio FG, Cattran DC and Salant DJ:
Rituximab-induced depletion of Anti-PLA2R autoantibodies predicts
response in membranous nephropathy. J Am Soc Nephrol. 22:1543–1550.
2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Polanco N, Gutiérrez E, Covarsí A, Ariza
F, Carreño A, Vigil A, Baltar J, Fernández-Fresnedo G, Martín C,
Pons S, et al: Spontaneous remission of nephrotic syndrome in
idiopathic membranous nephropathy. J Am Soc Nephrol. 21:697–704.
2010.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Ye WL, Tang N, Wen YB, Li H, Li MX, Du B
and Li XM: Underlying renal insufficiency: The pivotal risk factor
for Pneumocystis jirovecii pneumonia in immunosuppressed patients
with non-transplant glomerular disease. Int Urol Nephrol.
48:1863–1871. 2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Fujita F, Ishii S, Hirano S, Izumi S,
Takeda Y and Kobayashi N: Response of paraneoplastic nephrotic
syndrome to corticosteroids combined with chemotherapy for advanced
lung cancer: A case report and literature review. Int Canc Conf J.
1:88–92. 2012.
|
|
12
|
Hauber HP: Paraneoplastic syndromes in
lung cancer. Pneumologie. 65:347–358. 2011.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Miret M, Horváth-Puhó E, Déruaz-Luyet A,
Sørensen HT and Ehrenstein V: Potential paraneoplastic syndromes
and selected autoimmune conditions in patients with non-small cell
lung cancer and small cell lung cancer: A population-based cohort
study. PLoS One. 12(e0181564)2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Jensen AD, Münter MW, Bischoff H,
Haselmann R, Timke C, Krempien R, Sterzing F, Nill S, Heeger S,
Hoess A, et al: Treatment of non-small cell lung cancer with
intensity-modulated radiation therapy in combination with
cetuximab: The NEAR protocol. BMC Cancer. 6(12)2006.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Huo X, Huo B, Wang H, Wang L, Cao Q, Zheng
G, Wang J, Chai S, Zhang Z, Yang K, et al: Implantation of computed
tomography-guided Iodine-125 seeds in combination with chemotherapy
for the treatment of stage III non-small cell lung cancer. J
Contemp Brachytherapy. 9:527–534. 2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Martinez-Monge R, Pagola M, Vivas I and
López-Picazo JM: CT-guided permanent brachytherapy for patients
with medically inoperable early-stage non-small cell lung cancer
(NSCLC). Lung Cancer. 61:209–213. 2008.PubMed/NCBI View Article : Google Scholar
|