Introduction
Primary central nervous system lymphoma (PCNSL) is a
rare form of non-Hodgkin's lymphoma that is usually confined to the
brain, leptomeninges, spine, cerebrospinal fluid and eyes without
evidence of systemic spread (1).
PCNSL represents approximately 4% of all newly diagnosed central
nervous system (CNS) tumours (2,3), and
most seem to be of late or post-germinal centre B-cell origin
(4,5). Immunodeficiency due to congenital
immunodeficiency syndromes such as ataxia-telangiectasia and
Wiskott-Aldrich syndrome, as well as secondary causes such as
acquired immunodeficiency syndrome and iatrogenic immunosuppression
for transplant procedures, has been implicated in the development
of PCNSL (6).
The prognosis of PCNSL has improved substantially in
recent years, particularly in immunocompetent patients (7). However, treatment of PCSNL may cause
neurotoxicity and compromise health-related quality of life.
Studies have suggested that patients treated with combined
high-dose methotrexate chemotherapy and consolidation whole-brain
radiotherapy (WBRT) in the treatment of PCNSL develop worse
neurotoxicity and cognitive dysfunction than those treated with
chemotherapy alone (8-12).
Thus, consolidation WBRT is often withheld unless necessary,
particularly in elderly patients (13). Conversely, cognition, as measured by
mini-mental state examination (MMSE) scores, frequently improves
following the successful treatment of PCNSL with chemotherapy with
or without immunotherapy (rituximab) and remains stable on
follow-up (10,14-16).
However, currently, no study has described the neurological
prognosis of PCNSL patients with a very poor neurocognitive
function at baseline.
In this study, we reviewed the cases of 3 patients
with neuroimaging- and biopsy-proven PCNSL who had baseline
comatose neurological states at presentation (Table I). All 3 were treated with high-dose
methotrexate-based chemotherapy only without WBRT and achieved
either a partial or complete response to treatment, as assessed
using International PCNSL Collaborative Group criteria (17).
 | Table IClinical characteristics and treatment
outcomes of patients with PCNSL. |
Table I
Clinical characteristics and treatment
outcomes of patients with PCNSL.
| ID | Age at diagnosis
(years) | Gender | Significant
comorbidities | Presenting
symptoms | Histology | Anatomical
location | Treatment
regimen | GCS (at
presentation) | GCS (before
treatment) | Time to
treatment | End of treatment
responseb | GCS (post-
treatment) |
|---|
| 1 | 73 | Male | Hypertension Diabetes
mellitus Hyperlipidaemia Ischaemic heart disease | Lethargy Drowsiness
Behaviour change | DLBCL, non-GCB | Basal ganglia
Thalamus Midbrain Periventricular Corpus callosum | High-dose
methotrexate based (2.5 g/m2) for 4 cycles | E3V1M1a | E2V2M1 | 2 months | Complete
response | E2V2M1 |
| 2 | 42 | Male | Nil | Diplopia Poor visual
acuity | DLBCL, non-GCB | Optic chiasma, optic
nerves and tract Midbrain and pons Hypothalamus | High-dose metho
trexate based (2.5 g/m2) for 5 cycles | E3V4M6 | E1VTM1 | 6 months | Partial response | E2V1M1 |
| 3 | 68 | Male | Hypertension
Hyperlipidaemia Stroke | Unsteady gait Memory
impairment Slow speech | DLBCL, GCB | Periventricular
Corpus callosum Corona radiata | High-dose
methotrexate based (2.5 g/m2) for 5 cycles | E4V4M6 | E1V1M1 | 12 days | Complete
response | E1V1M1 |
Case reports
Case 1
A 73-year-old Chinese man presented with worsening
lethargy and drowsiness, together with behavioural changes for 1
month. He had a medical history of hypertension, diabetes mellitus,
hyperlipidaemia and ischaemic heart disease. During his initial
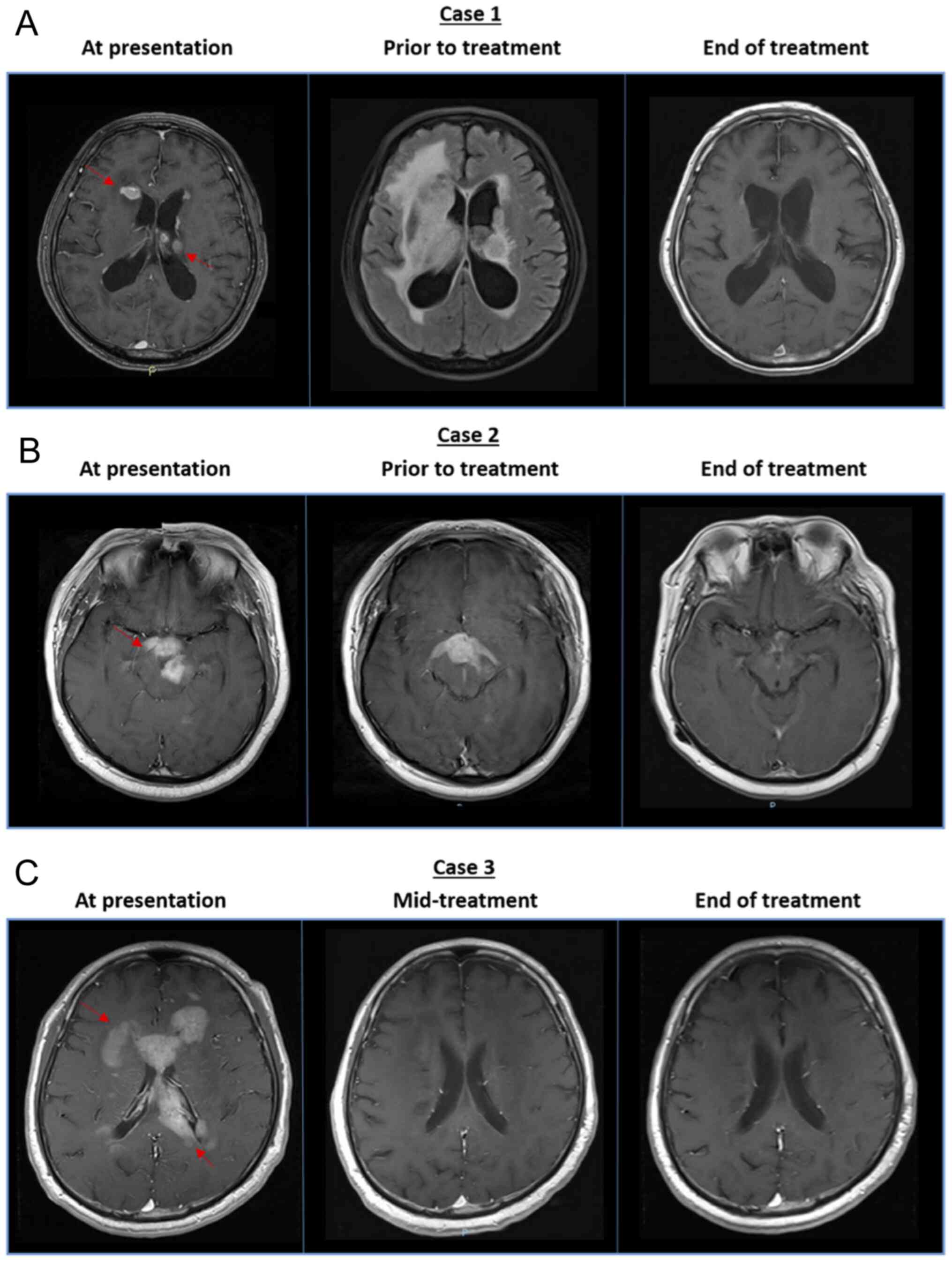
admission, magnetic resonance imaging (MRI) of the brain revealed
multiple foci of abnormal enhancement with low to heterogeneous T2
signals in a periventricular distribution, including the ependymal
margins of both lateral ventricles, infundibular recess of the 3rd
ventricle and pituitary stalk. These findings were highly
suggestive of PCNSL. Despite these findings, the patient declined
further work-up and was discharged against medical advice. He was
subsequently re-admitted a month later for progressive drowsiness
and fever. Neurological examination revealed a severely depressed
level of consciousness with a Glasgow Coma Scale (GCS) of E3V1M1.
Imaging of the brain showed interval disease progression and
obstructive hydrocephalus. The patient subsequently underwent an
external ventricular drain insertion, followed by stereotactic
biopsy of the right caudate region two weeks later.
Histological evaluation confirmed diffuse large
B-cell lymphoma (DLBCL), non-germinal centre B-cell like (non-GCB)
subtype. He was administered dexamethasone, procarbazine,
vincristine and high-dose methotrexate (2.5 g/m2)
(18). At the point of treatment
initiation, the GCS remained poor at E2V2M1. The time from the
initial presentation to time of treatment initiation was 2 months.
Because chemotherapy was complicated by repeated infective
episodes, including pyelonephritis, colitis and pneumonia, only 4
cycles were administered (up to week 8). Brain MRI performed at the
end of treatment showed no residual tumour or interval new tumour,
indicating a complete response (Fig.
1A).
Despite the complete response of the lymphoma to
chemotherapy, the patient's neurocognitive status did not improve
and the GCS remained poor at E2V2M1. The patient eventually died of
relapsed disease 8 months later.
Case 2
A 42-year-old Bruneian man with no significant
medical history initially presented with diplopia and was referred
to the National Cancer Centre, Singapore, for suspected PCNSL on
preliminary brain imaging. Physical examination revealed a GCS of
E3V4M6, cranial nerve III, IV and VI palsy bilaterally, a fixed
left pupil and extremely poor visual acuity with inability to
visualize light bilaterally. Mild left hemiparesis was also
evident. Brain MRI revealed homogeneously enhancing lesions in the
optic chiasma, optic nerves and tract, midbrain and pons, and
hypothalamus.
Stereotactic biopsy of the left suprasellar lesion
confirmed DLBCL, non-GCB type. Unfortunately, the patient became
progressively drowsier because of the interval enlargement of the
known suprasellar mass associated with an increasing mass effect
and worsening hydrocephalus. The patient subsequently underwent
ventriculoperitoneal shunt insertion and tracheostomy, while his
GCS continued to deteriorate to E1VTM1. He was administered
high-dose methotrexate-based chemotherapy for 5 cycles based on the
protocol by Shah et al (rituximab, vincristine, procarbazine
and methotrexate 2.5 g/m2) (19). The time from the initial
presentation to the start of treatment was 6 months. Post-treatment
brain MRI showed a stable hypothalamus lesion and marked
improvement in the lesions in the right temporal lobe, bilateral
basal ganglia and brainstem, indicating an effective partial
response to treatment (Fig.
1B).
Despite the overall response to treatment, the
patient did not achieve significant improvement in cognition or
physical function. His post-treatment GCS remained low at E2V1M1.
Thereafter, he was managed with best supportive care alone at a
hospice.
Case 3
A 68-year-old Chinese man with a significant medical
history of hypertension, hyperlipidaemia and stroke presented with
an unsteady gait with frequent falls, impairment of semantic memory
and slow speech. His physical examination was unremarkable, and he
had no gross neurological deficits. His GCS was E4V4M6. Brain MRI
showed lobulated semi-confluent enhancing lesions at the bilateral
periventricular regions involving the corpus callosum and corona
radiata.
Stereotactic biopsy of the left frontal
periventricular region revealed DLBCL, GCB type. Post operatively,
he was administered levetiracetam and dexamethasone because he had
mild fasciculations of his right thigh and twitching of his left
biceps suggestive of a provoked seizure. He was intubated when his
GCS subsequently deteriorated to E1V1M1, following which a
tracheostomy was performed. Subsequently, he was administered
high-dose methotrexate-based chemotherapy for 5 cycles including
rituximab, vincristine and methotrexate (2.5 g/m2) but
not procarbazine. Post-treatment brain MRI revealed complete
resolution of the lesions along the periventricular region and
corpus callosum, indicating a complete response to treatment
(Fig. 1C).
Similar to cases 1 and 2, despite an effective
tumour response to chemotherapy, he remained in a comatose state
with a GCS of 3 and died 8 months later.
Discussion
We described the cases of 3 patients with extremely
poor neurological statuses before treatment initiation. All 3
patients did not achieve meaningful neurological recovery despite
an effective tumour response to chemotherapy, as evidenced by the
low post-treatment GCS of 5, 4 and 3 for cases 1, 2 and 3,
respectively. These findings contrast those of earlier studies that
reported an improvement in cognitive function following the
successful treatment of PCNSL (10,14-16).
Although these cohorts generally comprise patients with grossly
intact neurological and cognitive function (median MMSE range,
22-23), our study is novel because the 3 patients had extremely
poor neurocognitive function at baseline with a GCS score of 5 or
below before treatment initiation.
The GCS decreased in all 3 patients in the short
time frame between the initial presentation and start of treatment,
ranging from 12 days to 6 months. In case 1, treatment was delayed
because the patient had initially requested for discharge against
medical advice, whereas the delay in case 2 was due to the patient
being referred from an overseas hospital. In case 3, treatment was
promptly commenced. Thus, PCNSL is an aggressive disease with an
unpredictable clinical course. A sharp decrease in the GCS
representing disease progression of PCNSL may indicate a poor
neurological prognosis even if the tumour responds well to
chemotherapy. In such a scenario, best supportive care focusing on
the quality of life may be considered and weighed carefully against
aggressive chemotherapy with curative intent.
Although cases 1 and 3 involved elderly Chinese
patients older than 65 years, case 2 involved a young Bruneian man
aged only 42 years at diagnosis. Despite the differences in both
age and race, all 3 patients had similarly poor neurological
outcomes at the end of chemotherapy treatment. This supports the
hypothesis that a poor neurocognitive status before treatment is a
poor prognostic indicator for the post-treatment neurological
outcome across various demographical factors such and age and
race.
In conclusion, the neurocognitive status of PCNSL
patients can deteriorate quickly, indicating dismal outcomes.
Patients with severe neurocognitive compromise may have a poor
neurological prognosis despite an effective response to treatment.
Further validation studies should be conducted to examine the
neurological prognosis of PCNSL patients with poor neurological
function at baseline who were treated successfully with
chemotherapy, as well as to determine the possible causes of and
prevent poor neurological status in these patients. Our study
suggests that administering early treatment in PCNSL patients and
avoiding unnecessary delays are necessary to achieve optimal
neurocognitive recovery.
Acknowledgements
Not applicable.
Funding
This work was supported by the Singapore Ministry of
Health's National Medical Research Council of Singapore (grant no.
NMRC/FLWSHP/054/2017-00), SHF-Foundation (grant no.
SHF/FG653P/2017) and SingHealth Duke-NUS Academic Medical Centre
and Oncology ACP (grant no. 08-FY2017/P1/14-A28).
Availability of data and materials
Data sharing is not applicable to this article, as
no datasets were generated or analysed during the present
study.
Authors' contributions
RMHL and JYC conceptualized the study and wrote the
manuscript. RMHL acquired, analysed and interpreted the data. JYC
enrolled the study patients, obtained their consent and treated
them. Both authors have confirmed the authenticity of all raw data,
as well as read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Singhealth
Centralised Institutional Review Board (CIRB 2018/3084). Written
informed consent was obtained from all the participants and/or
their legal guardians.
Patient consent for publication
Written informed consent was obtained from the
patients for the publication of this case report and any
accompanying images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Grommes C and DeAngelis LM: Primary CNS
lymphoma. J Clin Oncol. 35:2410–2418. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Villano JL, Koshy M, Shaikh H, Dolecek TA
and McCarthy BJ: Age, gender, and racial differences in incidence
and survival in primary CNS lymphoma. Br J Cancer. 105:1414–1418.
2011.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Hoffman S, Propp JM and McCarthy BJ:
Temporal trends in incidence of primary brain tumors in the United
States, 1985-1999. Neuro Oncol. 8:27–37. 2006.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Montesinos-Rongen M, Brunn A, Bentink S,
Basso K, Lim WK, Klapper W, Schaller C, Reifenberger G, Rubenstein
J, Wiestler OD, et al: Gene expression profiling suggests primary
central nervous system lymphomas to be derived from a late germinal
center B cell. Leukemia. 22:400–405. 2008.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Camilleri-Broët S, Criniè E, Broët P,
Delwail V, Mokhtari K, Moreau A, Kujas M, Raphaël M, Iraqi W,
Sautès-Fridman C, et al: Auniform activated B-cell-like
immunophenotype might explain the poor prognosis of primary central
nervous system lymphomas: Analysis of 83 cases. Blood. 107:190–196.
2006.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Bhagavathi S and Wilson JD: Primary
central nervous system lymphoma. Arch Pathol Lab Med.
132:1830–1834. 2008.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Shiels MS, Pfeiffer RM, Besson C, Clarke
CA, Morton LM, Nogueira L, Pawlish K, Yanik EL, Suneja G and Engels
EA: Trends in primary central nervous system lymphoma incidence and
survival in the U.S. Br J Haematol. 174:417–424. 2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Correa DD, DeAngelis LM, Shi W, Thaler H,
Glass A and Abrey LE: Cognitive functions in survivors of primary
central nervous system lymphoma. Neurology. 62:548–555.
2004.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Batchelor T and Loeffler JS: Primary CNS
lymphoma. J Clin Oncol. 24:1281–1288. 2006.PubMed/NCBI View Article : Google Scholar
|
|
10
|
van der Meulen M, Dirven L, Habets EJJ,
van den Bent MJ, Taphoorn MJB and Bromberg JEC: Cognitive
functioning and health-related quality of life in patients with
newly diagnosed primary CNS lymphoma: A systematic review. Lancet
Oncol. 19:e407–e418. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Doolittle ND, Korfel A, Lubow MA, Schorb
E, Schlegel U, Rogowski S, Fu R, Dósa E, Illerhaus G, Kraemer DF,
et al: Long-term cognitive function, neuroimaging, and quality of
life in primary CNS lymphoma. Neurology. 81:84–92. 2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Thiel E, Korfel A, Martus P, Kanz L,
Griesinger F, Rauch M, Röth A, Hertenstein B, von Toll T,
Hundsberger T, et al: High-dose methotrexate with or without whole
brain radiotherapy for primary CNS lymphoma (G-PCNSL-SG-1): A phase
3, randomised, non-inferiority trial. Lancet Oncol. 11:1036–1047.
2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Sierra del Rio M, Rousseau A, Soussain C,
Ricard D and Hoang-Xuan K: Primary CNS lymphoma in immunocompetent
patients. Oncologist. 14:526–539. 2009.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Fritsch K, Kasenda B, Schorb E, Hau P,
Bloehdorn J, Möhle R, Löw S, Binder M, Atta J, Keller U, et al:
High-dose methotrexate-based immuno-chemotherapy for elderly
primary CNS lymphoma patients (PRIMAIN study). Leukemia.
31:846–852. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Batchelor T, Carson K, O'Neill A, Grossman
SA, Alavi J, New P, Hochberg F and Priet R: Treatment of pimary CNS
lymphoma with methotrexate and deferred radiotherapy: A report of
NABTT 96-07. J Clin Oncol. 21:1044–1049. 2003.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Hoang-Xuan K, Taillandier L, Chinot O,
Soubeyran P, Bogdhan U, Hildebrand J, Frenay M, De Beule N,
Delattre JY and Baron B: European Organization for Research and
Treatment of Cancer Brain Tumor Group. Chemotherapy alone as
initial treatment for primary CNS lymphoma in patients older than
60 years: A multicenter phase II study (26952) of the European
Organization for Research and Treatment of Cancer Brain Tumor
Group. J Clin Oncol. 21:2726–2731. 2003.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Abrey LE, Batchelor TT, Ferreri AJ,
Gospodarowicz M, Pulczynski EJ, Zucca E, Smith JR, Korfel A,
Soussain C, DeAngelis LM, et al: Report of an international
workshop to standardize baseline evaluation and response criteria
for primary CNS lymphoma. J Clin Oncol. 23:5034–5043.
2005.PubMed/NCBI View Article : Google Scholar
|
|
18
|
DeAngelis LM, Seiferheld W, Schold SC,
Fisher B and Schultz CJ: Radiation Therapy Oncology Group Study
93-10. Combination chemotherapy and radiotherapy for primary
central nervous system lymphoma: Radiation Therapy Oncology Group
Study 93-10. J Clin Oncol. 20:4643–4648. 2002.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Shah GD, Yahalom J, Correa DD, Lai RK,
Raizer JJ, Schiff D, LaRocca R, Grant B, DeAngelis LM and Abrey LE:
Combined immunochemotherapy with reduced whole-brain radiotherapy
for newly diagnosed primary CNS lymphoma. J Clin Oncol.
25:4730–4735. 2007.PubMed/NCBI View Article : Google Scholar
|