Introduction
Uterine sarcoma is a rare mesenchymal tumor that
occurs primarily in the uterine corpus, and endometrial stromal
sarcoma is an even rarer type of tumor that accounts for ~10% of
uterine sarcomas (1). Endometrial
stromal sarcoma is classified into high-grade endometrial stromal
sarcoma (HGESS) and low-grade endometrial stromal sarcoma (LGESS)
according to histological characteristics. The tumor cells of HGESS
cases show marked nuclear atypia and increased mitosis. On the
other hand, nuclear atypia is mild or absent, and mitotic figures
are rarely seen in LGESS cases. LGESS may be difficult to
distinguish from uterine leiomyoma by MRI examination, as they are
both mesenchymal tumors, especially in the case of degenerative
leiomyoma. Therefore, there are reports of LGESS cases diagnosed by
postoperative pathological examination after surgery performed
under a preoperative diagnosis of uterine leiomyomas (2). The histopathological findings of
LGESS are characterized by the proliferation of small tumor cells
with round to oval nuclei, similar to proliferative endometrial
stromal cells, and tumor cells are immunohistochemically positive
for CD10. These findings usually make it easy to histologically
distinguish between LGESS and uterine leiomyoma that are usually
immunohistochemically positive for SMA and negative for CD10.
However, LGESS with smooth muscle differentiation is
morphologically and immunohistochemically similar to leiomyoma;
thus it is difficult to distinguish it from uterine leiomyoma when
only the region showing differentiation is observed. The present
report describes the case of LGESS with smooth muscle
differentiation that was diagnosed as uterine leiomyoma by
preoperative needle biopsy and treated with laparoscopic surgery.
The possibility of preoperative diagnosis of similar cases with
reference to the findings obtained from retrospective
immunohistochemical studies in this case was also discussed.
Case report
The patient was a 41-year-old, gravida 1, para 1
female. She visited a nearby clinic with complaints of ovulation
bleeding and lower abdominal pain, and was referred to Osaka City
University hospital due to the presence of a 31x21 mm uterine body
mass with an internal hyperechoic region, as identified by
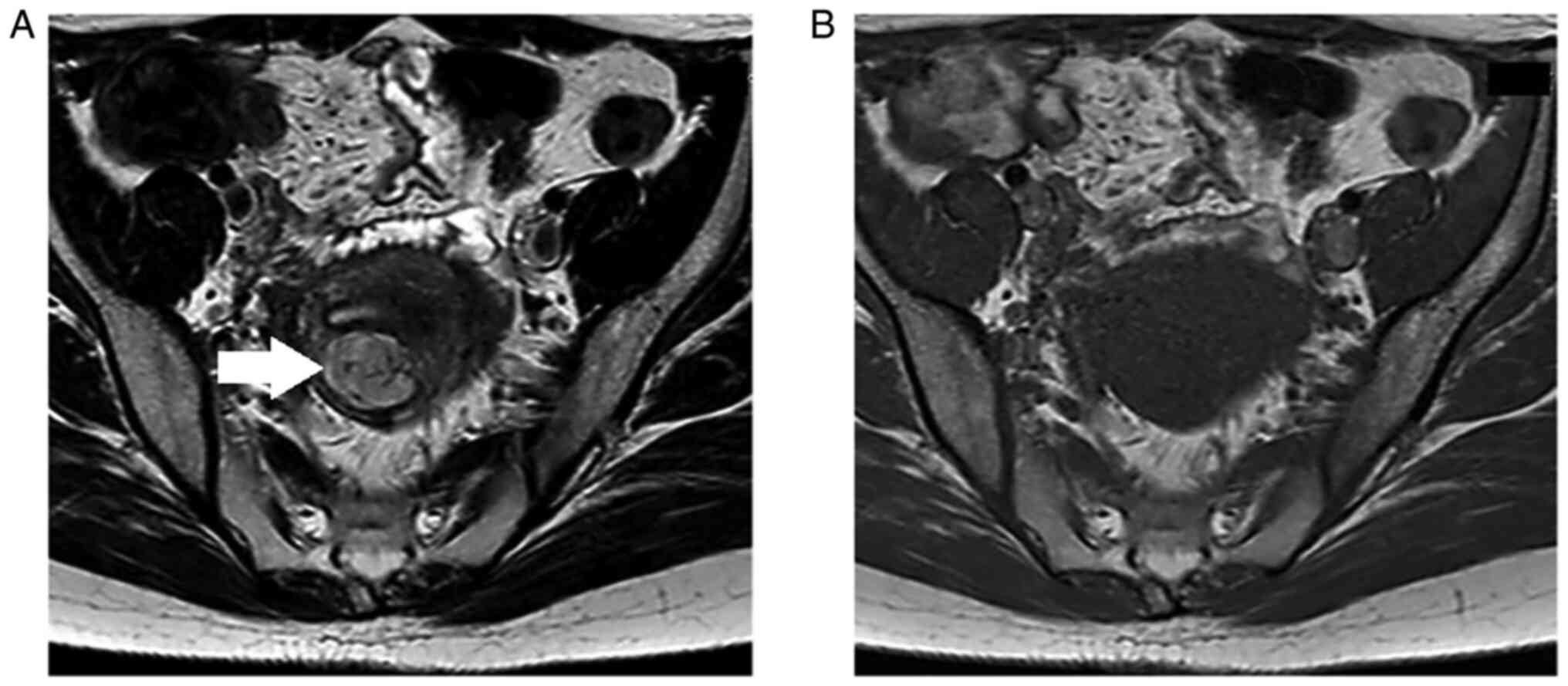
transvaginal ultrasonography. MRI findings showed a diffuse high
signal of a ~30 mm uterine corpus tumor on T2-weighted images, but
a low signal on T1-weighted images (Fig. 1A and B). In addition, since the
diffusion-weighted image showed a high signal, but the apparent
diffusion coefficient did not decrease, the tumor was not
restricted in diffusion and was considered to have few findings
suggestive of malignant disease. The protocol for MRI imaging is as
follows: TR (time to repeat); 4,000 milliseconds, TE (echo time);
85 milliseconds (Sagittal)/100 milliseconds (Axial), receive
bandwidth; 100 Hz, Field of view; 27 cn, Slice thickness; 5 mm,
Matrix number; 512x273, b-value; 0-1,000 second/mm2,
diffusion measurement time; 108. Blood analysis showed no
abnormalities in serum lactate dehydrogenase and CA125 levels.
Based on the aforementioned results, it was considered that there
were few findings supportive of uterine leiomyosarcoma, and the MRI
findings were considered to be different to that of typical uterine
leiomyomas. Therefore, it was decided to perform a histological
examination by transcervical needle biopsy (needle biopsy).
Needle biopsy was performed at the lithotomy
position under transvaginal ultrasound guidance. A puncture
attachment was attached to the transvaginal probe, and a tru-cut
type 17-gauge 250 mm puncture needle was inserted into the tumor to
collect the specimen. The maximum size of the tissue collected by
the biopsy needle was 1x17 mm per tissue, and a total of three
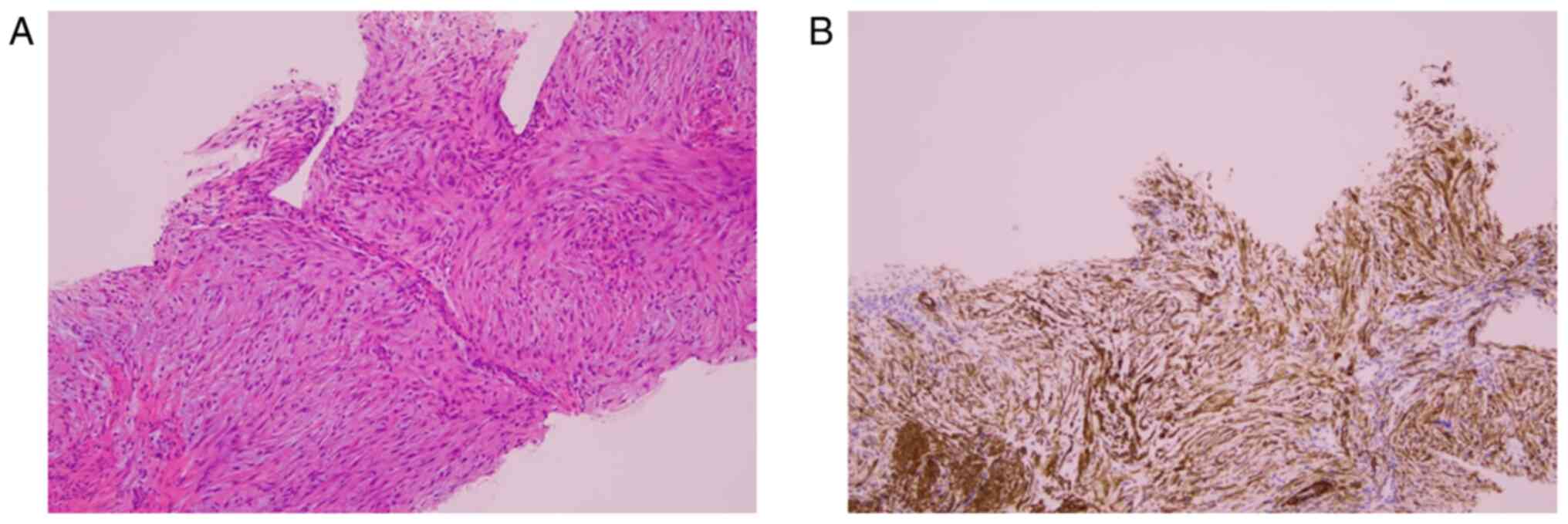
specimen were collected. The findings of the H&E-stained
specimen of the tissue obtained by needle biopsy showed that tumor
cells with spindle nuclei without atypia were arranged in a cord
(Fig. 2A), and the immunostaining
analysis findings were positive for smooth muscle actin (SMA)
(Fig. 2B). The protocols for
H&E staining and immunostaining are as follows: H&E
staining protocol; i) Deparaffinize (dunk for 10 min in xylene, 3
tanks). ii) Remove xylene (dunk for 5 min in 100% ethanol, 3
tanks). iii) Flooding (dunk for 5 min in 95% and 70% ethanol). iv)
Wash with water and distilled water. v) Hematoxylin stain (dunk for
4 min in Hematoxylin stain). vi) Wash with flowing tap water. vii)
Eosin stain (dunk for 2 min in Eosin stain). viii) Dehydration
(dunk for 30 sec in 70 and 95% ethanol, then dunk for 5 min in 100%
ethanol, 3 tanks). ix) Clear (dunk for 10 min in xylene, 3 tanks),
Immunostaining protocol; i) Preheat the antigen retrieval buffer
(100 mM Tris, pH 9.5) to 95˚C. ii) Wash tissues in PBS three times
for 5 min. iii) Incubate the tissues for 20 min with PBS containing
Triton X-100. iv) Wash tissues in PBS three times for 5 min. v)
Incubate tissues with 1% BSA for 30 min. vi) Incubate tissues in
the diluted primary antibody in 1% bovine serum albumin (BSA) in a
humidified chamber for 1 h at room temperature. vii) Decant the
solution and wash the tissues three times in phosphate-buffered
saline (PBS), 5 min each wash. Based on these results, the tumor
was diagnosed as a uterine leiomyoma, and was followed up as she
had no subjective symptoms.
Ultrasonography performed 29 months after the
initial visit showed the appearance of cystic lesions on both sides
of the uterus. The uterine tumor showed a gradual tendency for
growth, and it measured ~60 mm in size at this point. Therefore,
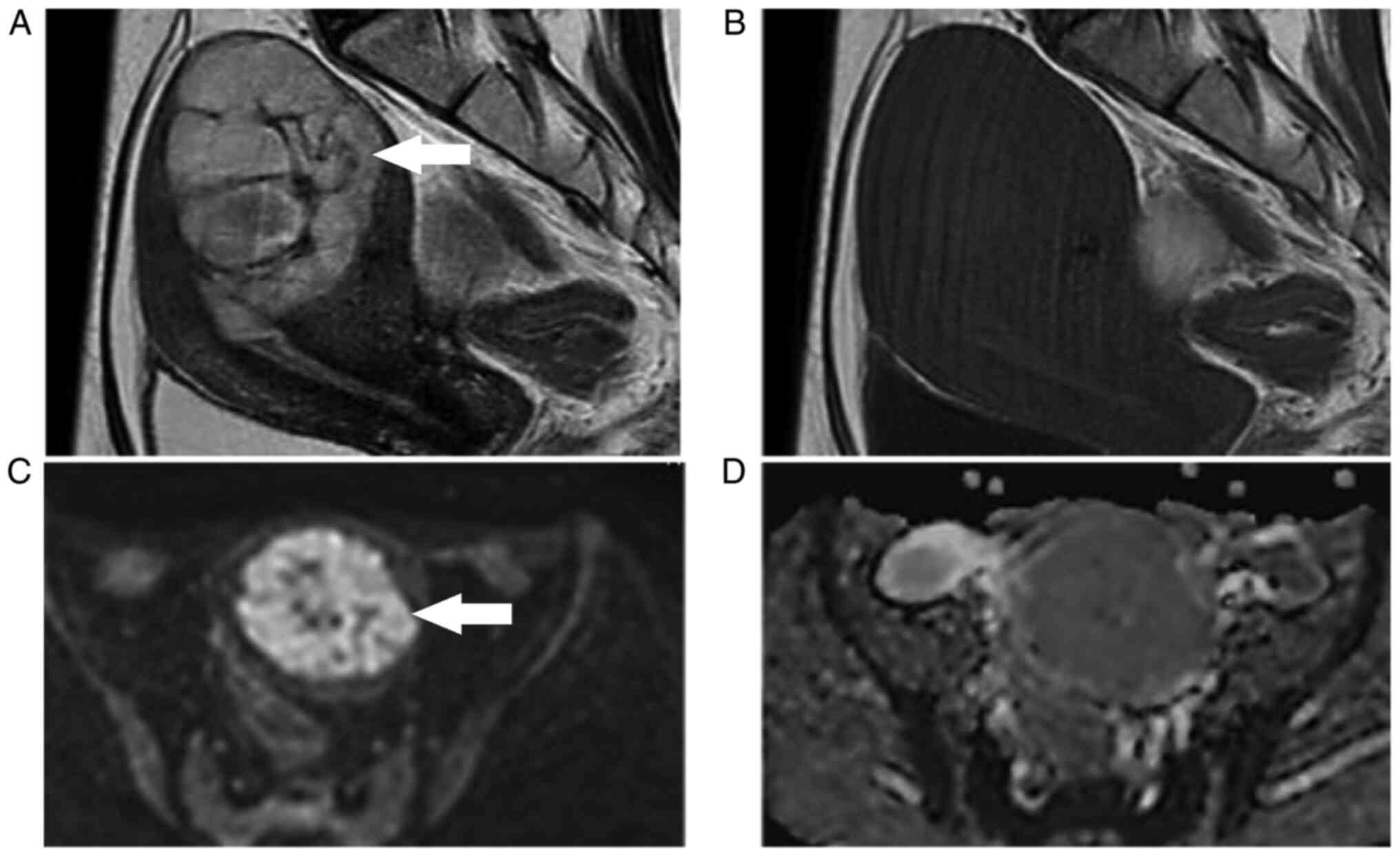
MRI examination was performed again. Cystic lesions of 52x40 mm on
the right dorsal side of the uterus and 36x28 mm on the left dorsal
side of the uterus were observed, both showing an equal signal
intensity on T2-weighted images and a high signal intensity on
T1-weighted images, after performing fat suppression. Bilateral
endometriotic ovarian cysts were suspected from the MRI results.
Conversely, regarding the uterine corpus tumor, although the size
had increased to 68x55 mm, it was considered that the T1-weighted
image showed a low signal intensity and was not subject to
diffusion restriction as before, and there was no change in the
internal properties (Fig.
3A-D).
She was recommended for surgery for the bilateral
ovarian cysts, and she also requested surgery for the uterine tumor
due to her hypermenorrhea. Blood tests were performed on this case
prior to the first needle biopsy, the second needle biopsy and
surgery, but no specific findings were found in the results of
blood analysis including tumor markers except for anemia before
needle biopsy (Table I).
Therefore, it was decided to perform laparoscopic surgery after
histological examination for the uterine tumor by a second needle
biopsy. The second needle biopsy was performed at the lithotomy
position under transabdominal ultrasound guidance. A puncture guide
tube was inserted into the uterine cavity transcervically, and the
tip of the inserted guide tube was guided under transabdominal
ultrasound to the vicinity of the target tumor, and the puncture
needle was continuously inserted into the tumor to collect tissue.
The pathological diagnosis of the tissue collected by needle biopsy
was leiomyoma, as in the first diagnosis (Fig. 4A and B), Based on these results, a laparoscopic
simple total hysterectomy, bilateral ovarian cystectomy and
bilateral salpingectomy were performed. The removed uterus,
bilateral ovarian cysts and bilateral fallopian tubes were placed
in a collection bag and delivered transvaginally. The uterus was
shredded in a bag. Macroscopic findings of the surgical specimen
showed no bleeding inside the uterine corpus tumor and no
remarkable findings in the endometrium. On the microscopic image,
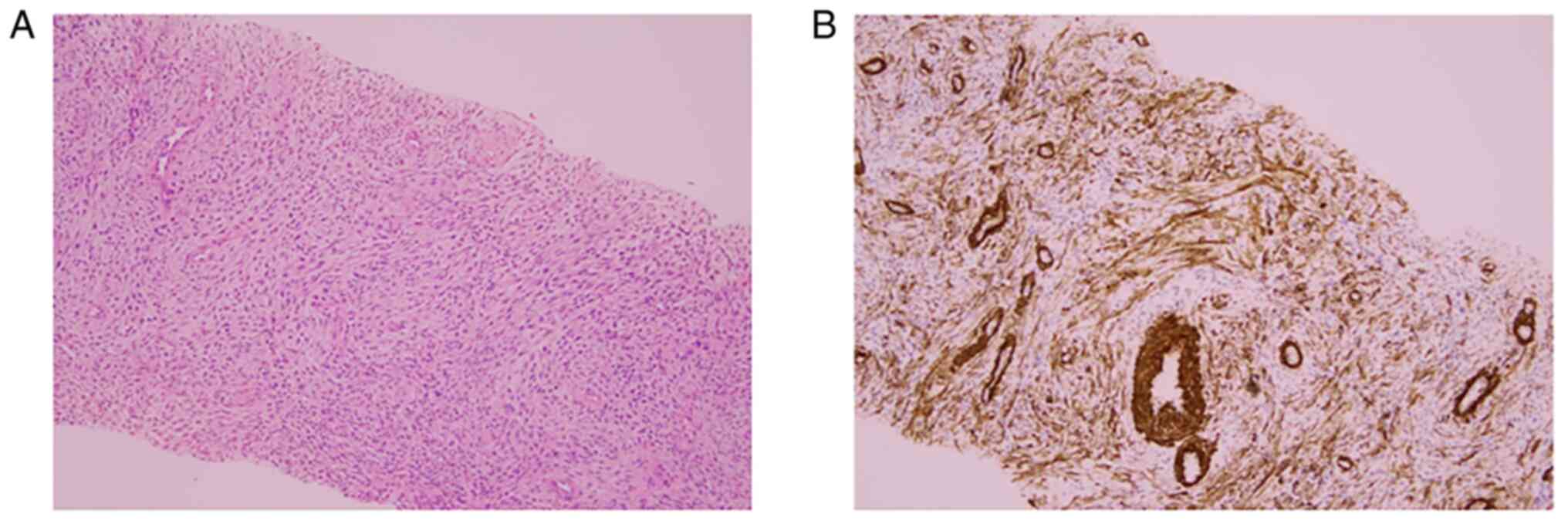
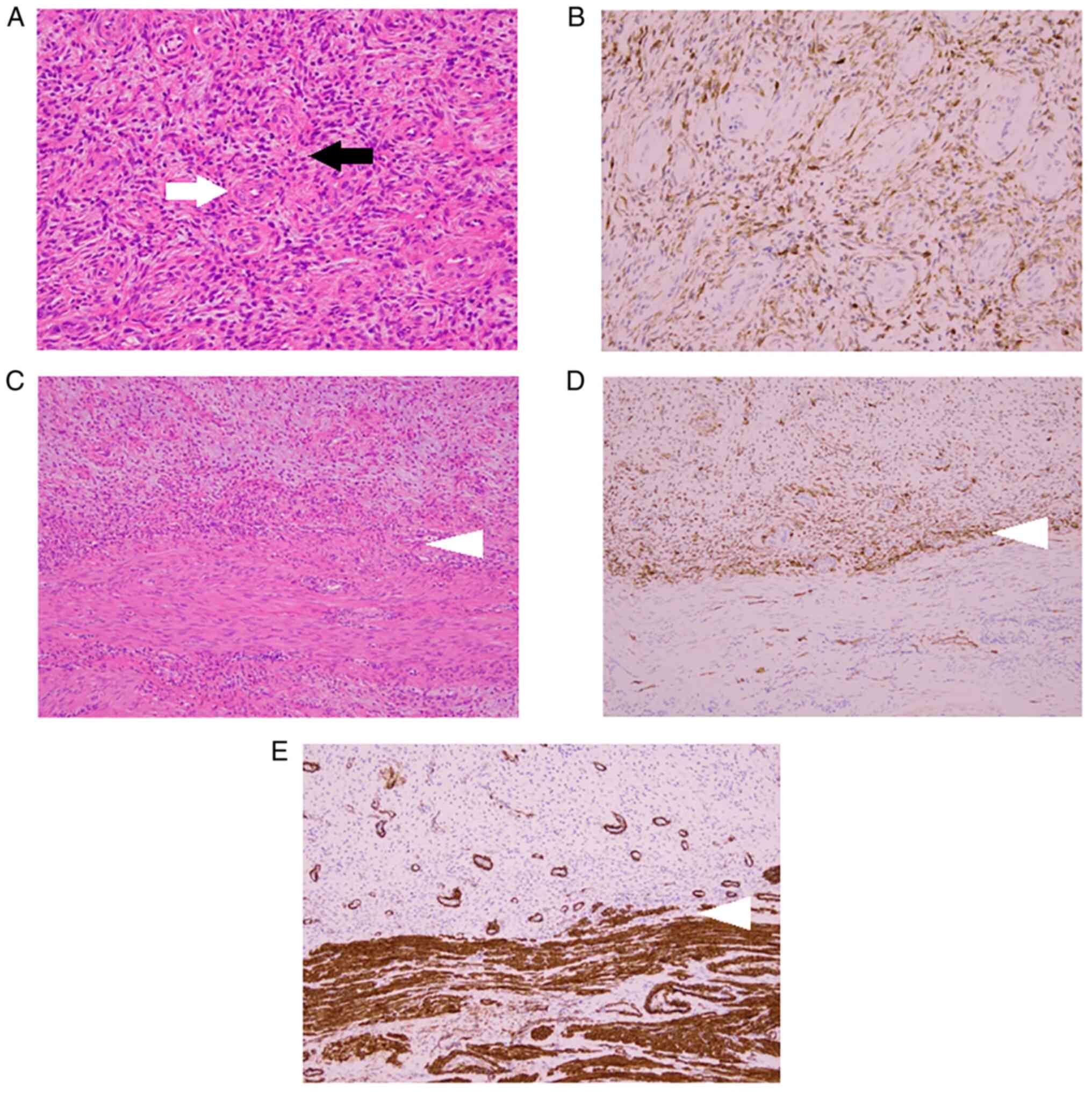
the tumor had infiltrated the surrounding muscle layer. H&E
staining findings showed a region where tumor cells with
spindle-shaped nuclei proliferated and a region where elliptical
and relatively small tumor cells proliferated densely in a swirling
manner around the arterioles, and the former area was considered to
account for ~30% of the specimen. In the latter area,
immunostaining analysis showed extensive positivity for CD10. In
addition, no nuclear atypia or mitotic figures were observed in any
of the regions, so this tumor was diagnosed as LGESS with smooth
muscle differentiation (Fig.
5A-E). Regarding the ovaries, both sides were diagnosed with
benign endometriotic cysts. Although additional treatments, such as
bilateral salpingo-oophorectomy were offered, consent for
additional treatment was not obtained. Therefore, it was decided to
follow up after confirming that there was no neoplastic lesion on
the image via a computed tomography examination. No lesion was
found on the image, and the final diagnosis was LGESS stage IB
(pT1bNxM0). No signs of recurrence were observed 20 months after
the operation.
 | Table IBlood test results. |
Table I
Blood test results.
| A, Results of blood
test performed before the first needle biopsya |
|---|
| Inspection item | Result (normal
range) |
|---|
| White blood
cells | 4,100/µl
(4,300-8,000) |
| Hemoglobin | 10.8 g/dl
(11.3-14.9) |
| Platelets | 26.5/x10,000 µl
(18.0-34.0) |
| Blood urea
nitrogen | 10 mg/dl (8-20) |
| Creatinine | 0.63 mg/dl
(0.40-0.90) |
| Aspartate
amino-transferase | 14 U/l (13-30) |
| Alanine
amino-transferase | 10 U/l (6-27) |
| Total bilirubin | 0.4 mg/dl
(0.2-1.0) |
| Lactate
dehydrogenase | 179 U/l
(124-222) |
| C-reactive
protein | 0.21 mg/dl
(0-0.40) |
| B, Results of blood
test performed before the second needle biopsya |
| Inspection item | Result (normal
range) |
| White blood
cells | 6,300/µl
(4,300-8,000) |
| Hemoglobin | 10.6 g/dl
(11.3-14.9) |
| Platelets | 27.0/x10,000 µl
(18.0-34.0) |
| Blood urea
nitrogen | 11 mg/dl (8-20) |
| Creatinine | 0.67 mg/dl
(0.40-0.90) |
| Aspartate
amino-transferase | 14 U/l (13-30) |
| Alanine
amino-transferase | 11 U/l (6-27) |
| Total bilirubin | 0.3 mg/dl
(0.2-1.0) |
| Lactate
dehydrogenase | 170 U/l
(124-222) |
| C-reactive
protein | 0.05 mg/dl
(0-0.40) |
| C, Preoperative blood
test resultsb |
| Inspection item | Result (normal
range) |
| White blood
cells | 4,400/µl
(4,300-8,000) |
| Hemoglobin | 12.3 g/dl
(11.3-14.9) |
| Platelets | 21.8/x10,000 µl
(18.0-34.0) |
| Blood urea
nitrogen | 12 mg/dl (8-20) |
| Creatinine | 0.60 mg/dl
(0.40-0.90) |
| Aspartate
amino-transferase | 15 U/l (13-30) |
| Alanine
amino-transferase | 11 U/l (6-27) |
| Total bilirubin | 0.4 mg/dl
(0.2-1.0) |
| Lactate
dehydrogenase | 200 U/l
(124-222) |
| C-reactive
protein | 0.04 mg/dl
(0-0.40) |
| Cancer antigen
125 | 22 U/ml (0-35) |
| Carbohydrate antigen
19-9 | 17 U/ml (0-37) |
| Carcinoembryonic
antigen | 2.7 ng/ml
(0-5.0) |
Discussion
LGESS is a rare disease, accounting for <1% cases
of all uterine malignant tumors; however, it is the second most
common malignant mesenchymal tumor of the uterus (3). Amongst the mesenchymal tumors of the
uterus, those derived from the endometrial stromal cells include
LGESS, HGESS, undifferentiated sarcoma (US) and endometrial stromal
nodule (ESN), of which ESN is a benign disease. Histologically,
LGESS is characterized by the proliferation of small round tumor
cells, similar to the proliferative phase of endometrial stromal
cells. Usually, nuclear atypia is absent or mild, and the mitotic
index ranges from zero to several/10 high power field (HPF).
Although LGESS and ESN are similar in cell properties, they are
distinguished by the presence or absence of infiltrative
proliferation in the surroundings. Therefore, strictly speaking, it
is impossible to distinguish between the two tumors without using
an excised uterine specimen containing the tumor. HGESS is also a
mesenchymal tumor consisting of cells similar to the proliferative
endometrial stromal cells, but unlike LGESS, it exhibits a high
degree of nuclear atypia and several necrotic regions. The mitotic
figure is conspicuous and generally shows a mitotic index exceeding
10/10 HPF, which is a distinguishing point from LGESS. US is a
tumor with strong nuclear atypia, which is difficult to determine
as derived from endometrial stromal cells. Characteristically,
LGESS is immunohistochemically positive for CD10, which is a
distinguishing point from other mesenchymal tumors, such as uterine
leiomyoma. However, LGESS can possess several variations, and the
frequency of LGESS with smooth muscle differentiation is 10-30%
(4). LGESS with smooth muscle
differentiation, especially in cases where the leiomyoma-like
region is relatively large, as in this case, when only this region
is observed, proliferation of spindle-shaped tumor cells is
confirmed and SMA is immunohistochemically positive, thus it
becomes difficult to distinguish LGESS from leiomyoma.
A biopsy for mesenchymal tumors of the uterus is not
a commonly performed examination, such as the endometrial biopsy
for endometrial epithelial tumors. With the approval of the
Institutional Review Board and informed consent before examination,
we have performed histological examination for uterine mesenchymal
tumors by needle biopsy in ~700 cases between 1994 and the present
(5). Since malignant tumors are
usually accompanied by nuclear atypia, it can be inferred that the
tumor is a malignant tumor, such as sarcoma, by confirming the
findings of tumor cell proliferation with atypical nuclei in needle
biopsy specimen. Furthermore, since the presence of necrosis is a
characteristic finding of sarcoma, it can be determined that the
tumor is a sarcoma by confirming the coagulative tumor cell
necrosis region with a needle biopsy specimen. Conversely, needle
biopsy is not useful in diagnosing tumors with few nuclear atypia
and without regions of necrosis, and LGESS is one such tumor that
is difficult to diagnose with a needle biopsy. However, needle
biopsies have been performed on 4 previous cases of LGESS, and the
possibility of LGESS from the pathological results in all cases is
considered. The reason why LGESS could be suspected on needle
biopsy specimens is that the growth of small round tumor cells with
few abnormalities was observed, and that the tumor cells showed
positive immunostaining for CD10. These two findings are also
characteristic of ESN, and LGESS and ESN are distinguished by the
presence or absence of infiltrative growth into the surroundings.
Therefore, needle biopsy alone cannot distinguish between the two
tumors, and it does not lead to a definitive diagnosis of LGESS.
However, needle biopsy can at least predict the possibility of
LGESS, and distinguish it from degenerative leiomyoma. The
pathological results of the two needle biopsies of this case were
both judged to be leiomyoma. This was due to the fact that LGESS
with smooth muscle differentiation shows the same H&E staining
and immunostaining findings as leiomyoma depending on the site
where the tissue was collected. The region showing differentiation
in this case was ~30%. Although it was considered a coincidence
that the tissue obtained by needle biopsy was taken from this
region twice, LGESS could not be suspected preoperatively, and
uterine leiomyoma was diagnosed due to these results, thus
laparoscopic surgery was performed.
It is difficult to distinguish between LGESS and
other types of tumors, such as leiomyoma with degeneration or
cellular leiomyoma by MRI examination before surgery. Therefore, it
is not uncommon for LGESS to be identified by postoperative
pathological diagnosis after hysterectomy or tumorectomy for
preoperative diagnosis of uterine leiomyoma, and there are reports
of LGESS being diagnosed during/after laparoscopic surgery
(6-8).
When laparoscopic surgery is performed on LGESS, the risk of
intraperitoneal dissemination should be taken into consideration.
There are reports describing no recurrence after surgery, even when
the tumor or uterus was divided prior to excision, but regarding
the cases in which hysterectomy was performed with diagnosis of
uterine leiomyoma, and finally diagnosed as LGESS, it has been
reported that the recurrence rate is significantly higher in the
divided group in which the tumor was excised separately for
laparoscopic or vaginal surgery compared with the group in which
the tumor was excised without division (9). If possible, it is desirable to avoid
tumorectomy or division of the uterus for LGESS surgery, and for
that purpose, a method for preoperative diagnosis of LGESS is
required.
In the surgically resected specimen in the present
case, the immunostaining findings showed positive staining for CD10
and negative staining for SMA in the region showing a typical LGESS
section, but both SMA and CD10 were positive in the region where
the LGESS was present with smooth muscle differentiation. Apart
from this case, we have experienced 2 patients of uterine leiomyoma
in which CD10 was partially positive. Therefore, a tumor that was
morphologically suspected to be leiomyoma and positive for CD10
immunostaining cannot always be diagnosed as endometrial stromal
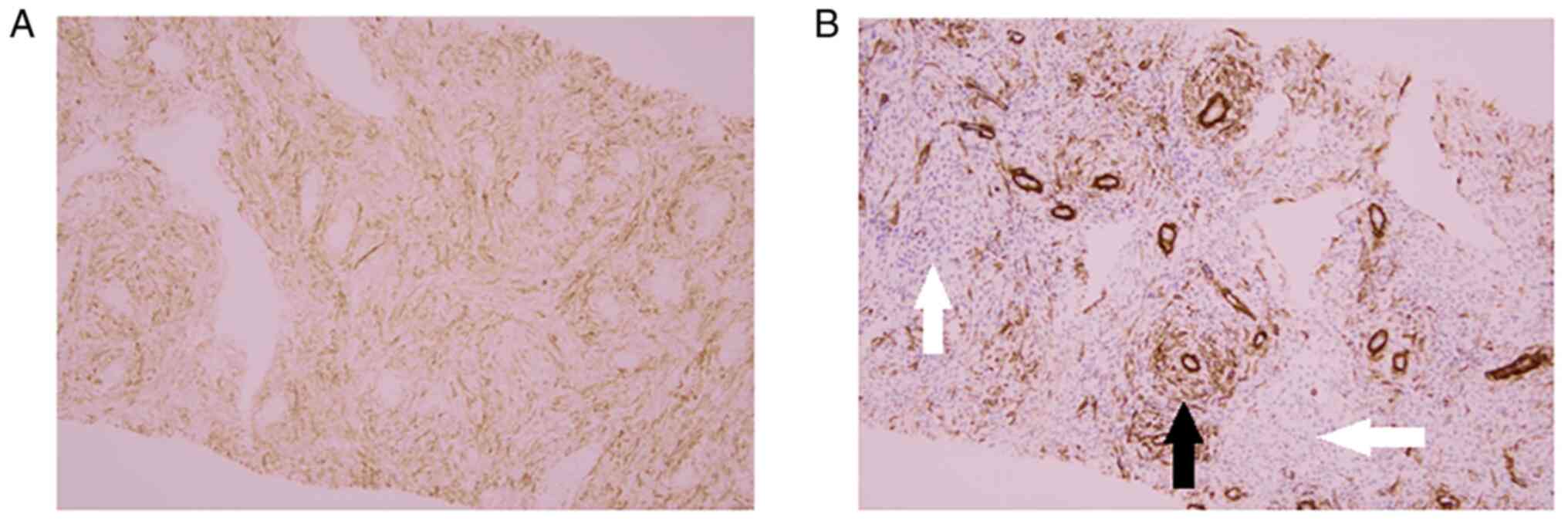
tumor with smooth muscle differentiation. As a retrospective
analysis, CD10 immunostaining on the second needle biopsied
specimen was performed, and it was shown that it too was also CD10
positive (Fig. 6A and B). When this CD10-immunostained section
was observed in detail and compared with SMA-immunostaining of the
same sample, a region where only CD10 was positive was observed.
There are no reports of a uterine leiomyoma in which there is a
region where SMA is negative but CD10 is positive, to the best of
our knowledge. If SMA and CD10 immunostaining is performed on
needle-biopsied specimen reliably collected from uterine tumors,
and a region shows positivity only for CD10, even if the
H&E-stained specimen does not show a typical endometrial
stromal tumor, it can be inferred that the tumor is not
degenerative leiomyoma, but instead an endometrial stromal tumor.
It is a well-known fact that CD10 immunostaining findings are
useful information in the diagnosis of LGESS. In addition, several
other papers have been reported on the differentiation between
endometrial stromal sarcoma and leiomyoma based on immunostaining
findings. Busca et al reported that IFITM1
(interferon-induced transmembrane protein-1) and CD10 had good
sensitivity in differentiating between LGESS and smooth muscle
tumor (10), and Zhu et al
reported that a panel of h-caldesmon, CD10 and CD44v3 was most
useful in differentiating endometrial stromal sarcoma from cellular
leiomyoma (11). Furthermore, Zhao
et al reported that the combination of IFITM1, CD10, SMA,
and h-caldesmon was useful in distinguishing between endometrial
stromal tumor and cellular leiomyoma (12). However, all of these reports are
evaluations of surgically resected specimens, and the purpose of
the study is to make final postoperative pathological diagnosis. On
the other hand, although our report is a retrospective study, it is
a report aimed at preoperative diagnosis by immunohistological
evaluation for biopsy specimens. The results of this study may be
applicable to preoperative diagnosis, unlike previous reports. We
believe that needle biopsy procedure and CD10/SMA immunostaining
may establish a preoperative diagnosis for LGESS. This attempt is a
new initiative that has never been reported.
In conclusion, a case of LGESS that was diagnosed as
leiomyoma by needle biopsy prior to surgery by laparoscopic simple
total hysterectomy is described. Since LGESS is relatively more
common in younger individuals, laparoscopic surgery is likely to be
performed if the preoperative diagnosis is leiomyoma, but
laparoscopic tumorectomy may increase the risk of recurrence. It is
suggested that immunohistochemical examination using anti-SMA and
anti-CD10 antibodies on specimen obtained from needle biopsy may
allow for preoperative diagnosis of LGESS including cases in which
smooth muscle differentiation is present.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
TI, MK and TS conceived and designed this case
report. TI and TS wrote the initial draft. KI, MY, TF and TY
collected clinical data. TI, MK and KI analyzed the data from the
pathological images. TI, MK and TS confirmed the authenticity of
all the raw data. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
Written informed consent was obtained from the
patient prior to surgery and tissue collection.
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of this paper.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Koss LG, Spiro RH and Brunschwig A:
Endometrial stromal sarcoma. Surg Gynecol Obstet. 121:531–537.
1965.PubMed/NCBI
|
|
2
|
Paul PG, Rengaraj V, Das T, Garg R, Thomas
M and Khurd AS: Uterine sarcomas in patients undergoing surgery for
presumed leiomyomas: 10 Years' Experience. J Minim Invasive
Gynecol. 23:384–389. 2016.PubMed/NCBI View Article : Google Scholar
|
|
3
|
WHO Classification of Tumours of Female
Reproductive Organs. 4th edition. 2014.
|
|
4
|
Oliva E, Clement PB, Young RH and Scully
RE: Mixed endometrial stromal and smooth muscle tumors of the
uterus: A clinicopathologic study of 15 cases. Am J Surg Pathol.
22:997–1005. 1998.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kawamura N, Ichimura T, Ito F, Shibata S,
Takahashi K, Tsujimura A, Ishiko O, Haba T, Wakasa K and Ogita S:
Transcervical needle biopsy for the differential diagnosis between
uterine sarcoma and leiomyoma. Cancer. 94:1713–1720.
2002.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Della Badia C and Karini H: Endometrial
stromal sarcoma diagnosed after uterine morcellation in
laparoscopic supracervical hysterectomy. J Minim Invasive Gynecol.
17:791–793. 2010.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Takeda T, Tamada Y, Matoba Y, Saotome K
and Makabe T: Low-grade endometrial stromal sarcoma which relapsed
a month after transcervical resection as fertility-sparing surgery
and diagnosed with laparoscopic hysterectomy. J Gynecol Fertil.
1:1–7. 2016.
|
|
8
|
Zheng Y, Yin Q, Yang X and Dong R:
Fertility-sparing management of low-grade endometrial stromal
sarcoma: Analysis of an institutional series, a population-based
analysis and review of the literature. Ann Transl Med.
8(1358)2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Park JY, Kim DY, Kim JH, Kim YM, Kim YT
and Nam JH: The impact of tumor morcellation during surgery on the
outcomes of patients with apparently early low-grade endometrial
stromal sarcoma of the uterus. Ann Surg Oncol. 18:3453–3461.
2011.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Busca A, Gulavita P, Parra-Herran C and
Islam S: IFITM1 Outperforms CD10 in differentiating low-grade
endometrial stromal sarcomas from smooth muscle neoplasms of the
uterus. Int J Gynecol Pathol. 37:372–378. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Zhu XQ, Shi YF, Cheng XD, Zhao CL and Wu
YZ: Immunohistochemical markers in differential diagnosis of
endometrial stromal sarcoma and cellular leiomyoma. Gynecol Oncol.
92:71–79. 2004.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zhao W, Cui M, Zhang R, Shen X, Xiong X,
Ji X, Tao L, Jia W, Pang L, Sun Z, et al: IFITM1, CD10, SMA, and
h-caldesmon as a helpful combination in differential diagnosis
between endometrial stromal tumor and cellular leiomyoma. BMC
Cancer. 21(1047)2021.PubMed/NCBI View Article : Google Scholar
|