Introduction
Trigeminal neuralgia (TN) is typically a sudden,
severe, and very brief attack of unilateral pain confined to one or
more segmental areas of the trigeminal nerve (1-4).
TN can be classified into three types: Classic, secondary, and
idiopathic. The classical type, which accounts for 75% of TN cases,
is caused by intracranial vascular compression of the trigeminal
nerve root, typically due to morphological changes in the superior
cerebellar artery relative to the adjacent trigeminal nerve root.
Secondary TN, which accounts for ~15% of TN cases, is caused by
diseases or lesions such as multiple sclerosis and meningioma,
which lead to changes in the trigeminal nerve root entry zone or
compression. Idiopathic TN is diagnosed when no obvious cause of
neuropathy can be found (1).
Symptomatically, classic or idiopathic TN is rarely associated with
somatosensory hypoesthesia. The treatment of classic or idiopathic
TN is established, with carbamazepine as the first-line treatment
(1,5). However, a standard treatment for
secondary TN has not been established, especially in the case of
metastatic carcinoma of the trigeminal nerve roots. Here, we report
a case of TN secondary to brain metastases from lung cancer,
accompanied by bilateral auditory disturbances. Opioids provided
symptomatic relief from facial pain.
Case report
The patient was a 46-year-old man, who was admitted
to The University of Tokyo Hospital (Tokyo, Japan) in July 2022,
with no medical history and was an ex-smoker of two packs per day
from the age of 10 to 38 years (56 pack-years). They underwent left
upper lobectomy and was diagnosed with pStage IIIA, pT1bN2M0
adenocarcinoma of the lung, which displayed an EGFR mutation of
exon 19 (deletion), and was anaplastic lymphoma kinase
immunohistochemistry and fluorescence in situ hybridization
(ALK-IHC/FISH) positive. After post-lobectomy adjuvant
chemotherapy, the patient was treated with four cycles of cisplatin
and vinorelbine. After 5 years of recurrence-free survival,
computed tomography (CT)-guided lung needle biopsy revealed lung
cancer metastasis in the left chest wall. The patient received
afatinib as first-line chemotherapy for recurrent unresectable lung
carcinoma, and alectinib as second-line treatment. The
adenocarcinoma was refractory to alectinib, and brain metastasis
developed, with no clinical symptoms. The patient further received
osimertinib as third-line chemotherapy, cisplatin and pemetrexed as
fourth-line, and docetaxel and ramcirumab as fifth-line treatment.
However, the tumor slowly progressed, with no obvious clinical
symptoms. Contrast-enhanced magnetic resonance imaging (MRI) and
head CT showed multiple brain metastases (i.e., a mass in the left
Sylvian fissure and significant thickening of the left trigeminal
nerve root). Osimertinib re-challenge was used as the sixth-line
treatment, and Gamma Knife was used to manage the brain metastases.
However, a follow-up contrast-enhanced MRI revealed tumor
aggravation in the cerebellar vermis and bilateral cerebellar
hemispheres. Tumor dissemination along the cerebral surface of the
cerebrum, the brainstem, and the cerebellum, as well as along the
right oculomotor nerve roots (III), bilateral trigeminal nerve
roots (V), bilateral facial (VII) and auditory (VIII) nerve roots,
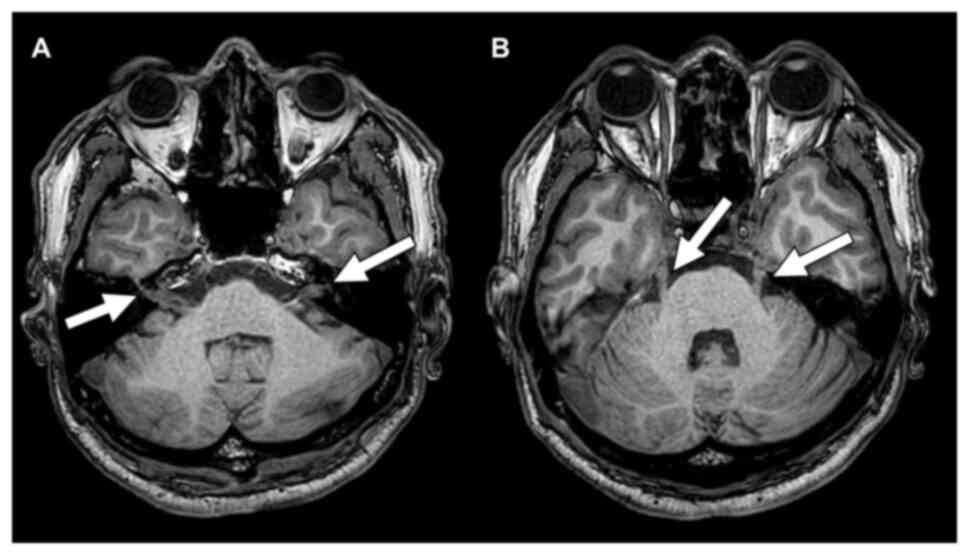
and lower cranial nerve roots (IX-XII) was also found (Fig. 1). Because of disseminated
carcinomatous meningitis, radiation therapy and other
chemotherapies were not administered, and a policy of best
supportive care was adopted 9 years after the surgery. Around the
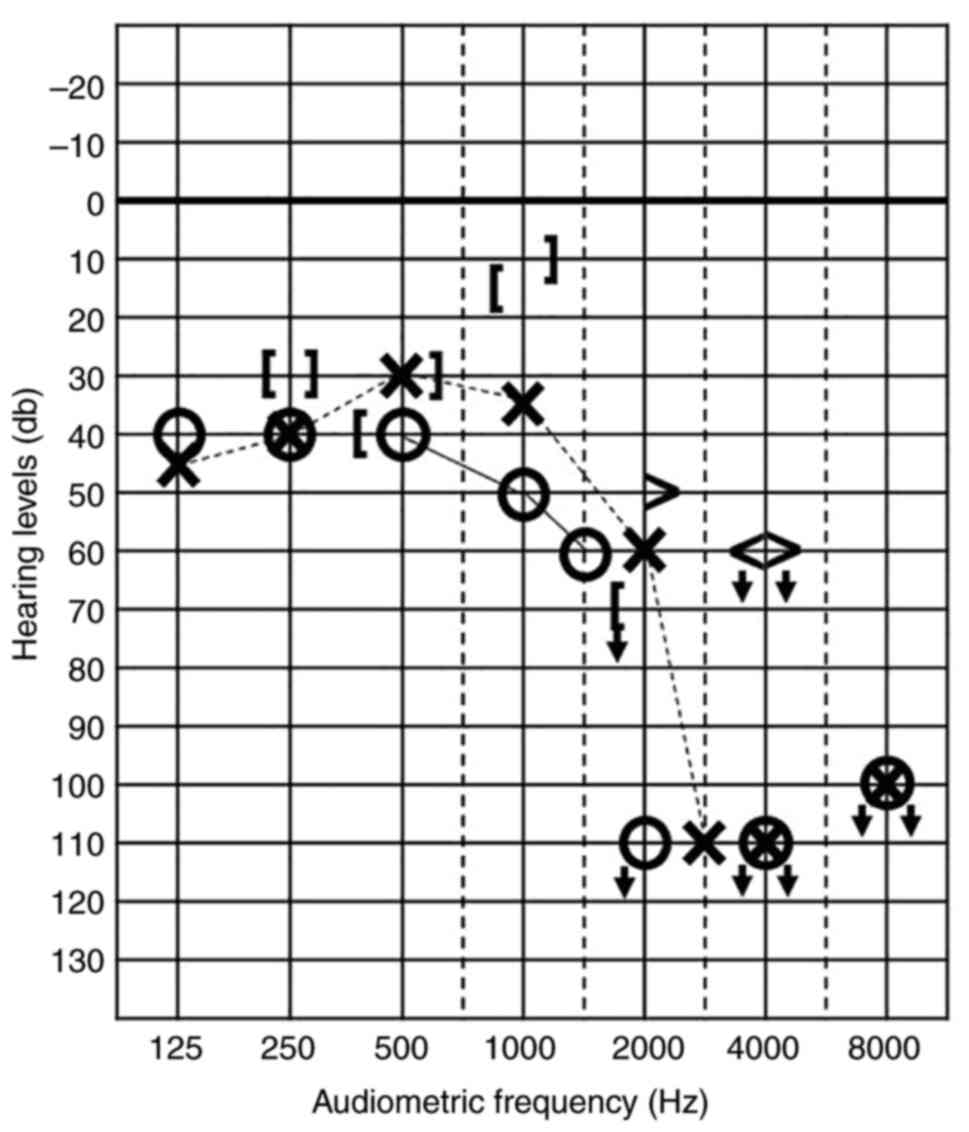
same time, the patient began to experience bilateral auditory
disturbances that were particularly prominent in the high-tone
range (Fig. 2). They
simultaneously developed somatosensory hypoesthesia in the left
facial V1, V2, and V3 regions, explosive pain in the left V1
region, and paresthesia in the tongue. No other neurological
deficits due to cranial nerve root lesions were observed, except
for a slight taste disturbance. Mastication triggered
electroshock-like or stabbing pain in the left facial area, which
made eating difficult. A fentanyl transdermal patch (25 mcg/h) was
initiated following a diagnosis of TN secondary to lung
adenocarcinoma metastasis on the trigeminal nerves. The facial pain
improved rapidly and markedly. Facial pain was not exacerbated
after starting opioids, and the patient was discharged from our
hospital.
Discussion
The most common presentation of TN is the classic
form, with secondary forms occurring less frequently (~15%)
(1). Common causes of secondary TN
include neurological diseases, such as multiple sclerosis and
compression of the trigeminal nerve root by brain tumors (1). Such brain tumors include auditory
schwannomas, meningiomas, epidermoid cysts and pearly tumors
(6). Compression of the trigeminal
nerve by a tumor could result in local demyelination of the
trigeminal nerve root. Similar to the vascular compression of the
root in classic TN, high-frequency discharges in the degenerated
axon can induce persistent pain in the facial region. In the
literature, cases of TN secondary to a metastatic brain tumor, as
in this case, are rare (7). In
contrast to solid brain tumors, the present case did not
demonstrate obvious compression of the trigeminal nerve root by a
brain metastatic tumor, but rather demonstrated significant
thickening of the root. Considering the intracranial cerebrospinal
fluid dissemination in this case, metastatic tumor cells might
infiltrate to the relevant cranial nerve roots. Such thickening was
also observed in the bilateral auditory nerve roots, which were not
obviously compressed, and therefore resulted in bilateral auditory
disturbances.
TN is characterized by sudden, intense, and very
brief attacks of pain confined to the trigeminal region (1). Along with this typical pain
expression, the present case presented with concurrent
somatosensory hypoesthesia in the facial region. Very few cases of
TN itself present with somatosensory loss, which is characterized
by explosive pain (8). While some
types of trigeminal neuropathy secondary to brain tumor compression
and post-herpetic neuralgia occasionally show somatosensory loss,
their pain is continuous, burning, and tingling. Therefore, the
present case, which demonstrated the typical pain characteristics
of TN but was accompanied by somatosensory loss, was considered
atypical. Furthermore, this case was accompanied by slight taste
disturbances and profound bilateral auditory disturbances, both of
which were caused by facial and auditory nerve root thickening and
disseminated brain metastasis. A giant auditory schwannoma can
compress the trigeminal nerve root and secondarily induce TN;
however, in this case the auditory disturbance would be unilateral.
Therefore, the patient's symptoms were considered atypical. If
clinicians encounter such an atypical presentation of TN
accompanied by other cranial nerve impairment(s), imaging studies
such as contrast-enhanced MRI and/or CT should be promptly
considered.
The treatment of TN generally includes
pharmacotherapy in addition to other invasive procedures. Among the
pharmacotherapy options, carbamazepine is the first-line drug of
choice and is usually associated with good pain control. In case of
trigeminal neuropathy, tricyclic antidepressants such as
amitriptyline and gabapentinoids might have additional analgesic
effects (5,9,10).
Opioids are not generally applied for TN despite the fact that even
a small amount of opioids can provide significant symptomatic
relief in the setting of brain metastases from a malignant tumor.
The present case was an atypical example of TN caused by
disseminated brain metastasis. We treated the facial pain with
opioids which rapidly improved as a result. Therefore, this case
suggests that opioids are a possible treatment option for secondary
TN caused by malignancy.
In conclusion, we report a case of secondary TN with
bilateral auditory disturbances secondary to brain metastases from
lung cancer. Pain symptoms were rapidly and markedly improved with
small doses of opioids. Although opioids are not generally used for
TN, this case suggests the importance of considering the use of
opioids as a possible option to treat secondary oncologic TN.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
KK, HA and MS diagnosed and treated the patient. TM
contributed to the acquisition of data. TM, KK and MS interpreted
the data and wrote the manuscript. TE and HA interpreted data and
performed critical reviewing. TM, KK and MS confirm the
authenticity of all the raw data. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
The University of Tokyo Clinical Research Review
Board approved the report of this case (approval no. 3678).
Patient consent for publication
Written consent was obtained for the publication of
this case report. All accompanying images were anonymized.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Cruccu G, Di Stefano G and Truini A:
Trigeminal Neuralgia. N Engl J Med. 383:754–762. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Di Stefano G, Maarbjerg S, Nurmikko T,
Truini A and Cruccu G: Triggering trigem-inal neuralgia.
Cephalalgia. 38:1049–1056. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Cruccu G, Finnerup NB, Jensen TS, Scholz
J, Sindou M, Svensson P, Treede RD, Zakrzewska JM and Nurmikko T:
Trigeminal neuralgia: New classification and diagnostic grading for
practice and research. Neurology. 87:220–228. 2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Maarbjerg S, Gozalov A, Olesen J and
Bendtsen L: Trigeminal neuralgia-a prospective systematic study of
clinical characteristics in 158 patients. Headache. 54:1574–1582.
2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Szok D, Tajti J, Nyári A and Vécsei L:
Therapeutic approaches for peripheral and central neuropathic pain.
Behav Neurol. 2019(8685954)2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Mohammad-Mohammadi A, Recinos PF, Lee JH,
Elson P and Barnett GH: Surgical outcomes of trigeminal neuralgia
in patients with multiple sclerosis. Neurosurgery. 73:941–950;
discussion 950. 2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Viviano M, Donati D and Lorenzini G:
Metastatic carcinoma presenting as neuralgia involving the
trigeminal nerve. J Can Dent Assoc. 77(c32)2012.PubMed/NCBI
|
|
8
|
Luo F, Wang T, Shen Y, Meng L, Lu J and Ji
N: High voltage pulsed radiofrequency for the treatment of
refractory neuralgia of the infraorbital nerve: A prospective
double-blinded randomized controlled study. Pain Physician.
20:271–279. 2017.PubMed/NCBI
|
|
9
|
Bendtsen L, Zakrzewska JM, Abbott J,
Braschinsky M, Di Stefano G, Donnet A, Eide PK, Leal PRL, Maarbjerg
S, May A, et al: European Academy Of Neurology guideline on
trigeminal neuralgia. Eur J Neurol. 26:831–849. 2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Truini A, Barbanti P, Pozzilli C and
Cruccu G: A mechanism-based classification of pain in multiple
sclerosis. J Neurol. 260:351–367. 2013.PubMed/NCBI View Article : Google Scholar
|