|
1
|
Howlader N, Noone AM, Krapcho M, Miller D,
Bishop K, Altekruse SF, Kosary CL, Yu M, Ruhl J, Tatalovich Z,
Mariotto A, Lewis DR, Chen HS, Feuer EJ and Cronin KA: SEER Cancer
Statistics Review. pp. 1975–2013. National Cancer Institute;
Bethesda, MD, USA: 2016
|
|
2
|
DeSantis CE, Fedewa SA, Goding Sauer A,
Kramer JL, Smith RA and Jemal A: Breast cancer statistics, 2015:
Convergence of incidence rates between black and white women. CA
Cancer J Clin. 66:31–42. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gampenrieder SP, Rinnerthaler G and Greil
R: Neoadjuvant chemotherapy and targeted therapy in breast cancer:
Past, present, and future. J Oncol. 2013:7320472013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Fisher B, Brown A, Mamounas E, Wieand S,
Robidoux A, Margolese RG, Cruz AB Jr, Fisher ER, Wickerham DL,
Wolmark N, et al: Effect of preoperative chemotherapy on
local-regional disease in women with operable breast cancer:
Findings from national surgical adjuvant breast and bowel project
B-18. J Clin Oncol. 15:2483–2493. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Earl HM, Hiller L, Dunn JA, Blenkinsop C,
Grybowicz L, Vallier AL, Abraham J, Thomas J, Provenzano E,
Hughes-Davies L, et al: ARTemis Investigators: Efficacy of
neoadjuvant bevacizumab added to docetaxel followed by
fluorouracil, epirubicin, and cyclophosphamide, for women with
HER2-negative early breast cancer (ARTemis): An open-label,
randomised, phase 3 trial. Lancet Oncol. 16:656–666. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Huober J, von Minckwitz G, Denkert C,
Tesch H, Weiss E, Zahm DM, Belau A, Khandan F, Hauschild M,
Thomssen C, et al: Effect of neoadjuvant anthracycline-taxane-based
chemotherapy in different biological breast cancer phenotypes:
Overall results from the GeparTrio study. Breast Cancer Res Treat.
124:133–140. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gerber B, Loibl S, Eidtmann H, Rezai M,
Fasching PA, Tesch H, Eggemann H, Schrader I, Kittel K, Hanusch C,
et al: German Breast Group Investigators: Neoadjuvant bevacizumab
and anthracycline-taxane-based chemotherapy in 678 triple-negative
primary breast cancers; results from the geparquinto study (GBG
44). Ann Oncol. 24:2978–2984. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Levasseur N, Clemons M, Hilton J, Addison
C, Robertson S, Ibrahim M and Arnaout A: Neoadjuvant endocrine
therapy and window of opportunity trials: New standards in the
treatment of breast cancer? Minerva Chir. 70:181–193.
2015.PubMed/NCBI
|
|
9
|
Dowsett M: Predictive and prognostic
factors. Breast Cancer Res. 12 Suppl 4:S22010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Shinkaruk S, Bayle M, Laïn G and Déléris
G: Vascular endothelial cell growth factor (VEGF), an emerging
target for cancer chemotherapy. Curr Med Chem Anticancer Agents.
3:95–117. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ferrara N, Hillan KJ, Gerber HP and
Novotny W: Discovery and development of bevacizumab, an anti-VEGF
antibody for treating cancer. Nat Rev Drug Discov. 3:391–400. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Summers J, Cohen MH, Keegan P and Pazdur
R: FDA drug approval summary: Bevacizumab plus interferon for
advanced renal cell carcinoma. Oncologist. 15:104–111. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gil-Gil MJ, Mesia C, Rey M and Bruna J:
Bevacizumab for the treatment of glioblastoma. Clin Med Insights
Oncol. 7:123–135. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cohen MH, Gootenberg J, Keegan P and
Pazdur R: FDA drug approval summary: Bevacizumab (Avastin) plus
Carboplatin and Paclitaxel as first-line treatment of
advanced/metastatic recurrent nonsquamous non-small cell lung
cancer. Oncologist. 12:713–718. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Conti RM, Dusetzina SB, Herbert AC, Berndt
ER, Huskamp HA and Keating NL: The impact of emerging safety and
effectiveness evidence on the use of physician-administered drugs:
The case of bevacizumab for breast cancer. Med Care. 51:622–627.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lenzer J: FDA committee votes to withdraw
bevacizumab for breast cancer. BMJ. doi.org/10.1136/bmj.d4244.
|
|
17
|
Cao L, Yao GY, Liu MF, Chen LJ, Hu XL and
Ye CS: Neoadjuvant bevacizumab plus chemotherapy versus
chemotherapy alone to treat non-metastatic breast cancer: A
meta-analysis of randomised controlled trials. PLoS One.
10:e01454422015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Moher D, Liberati A, Tetzlaff J and Altman
DG: PRISMA Group: Preferred reporting items for systematic reviews
and meta-analyses: The PRISMA statement. BMJ.
doi.org/10.1136/bmj.b2535.
|
|
19
|
Hutton B, Moher D and Cameron C: The
PRISMA extension statement. Ann Intern Med. 163:566–567. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tierney JF, Stewart LA, Ghersi D, Burdett
S and Sydes MR: Practical methods for incorporating summary
time-to-event data into meta-analysis. Trials. 8:162007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
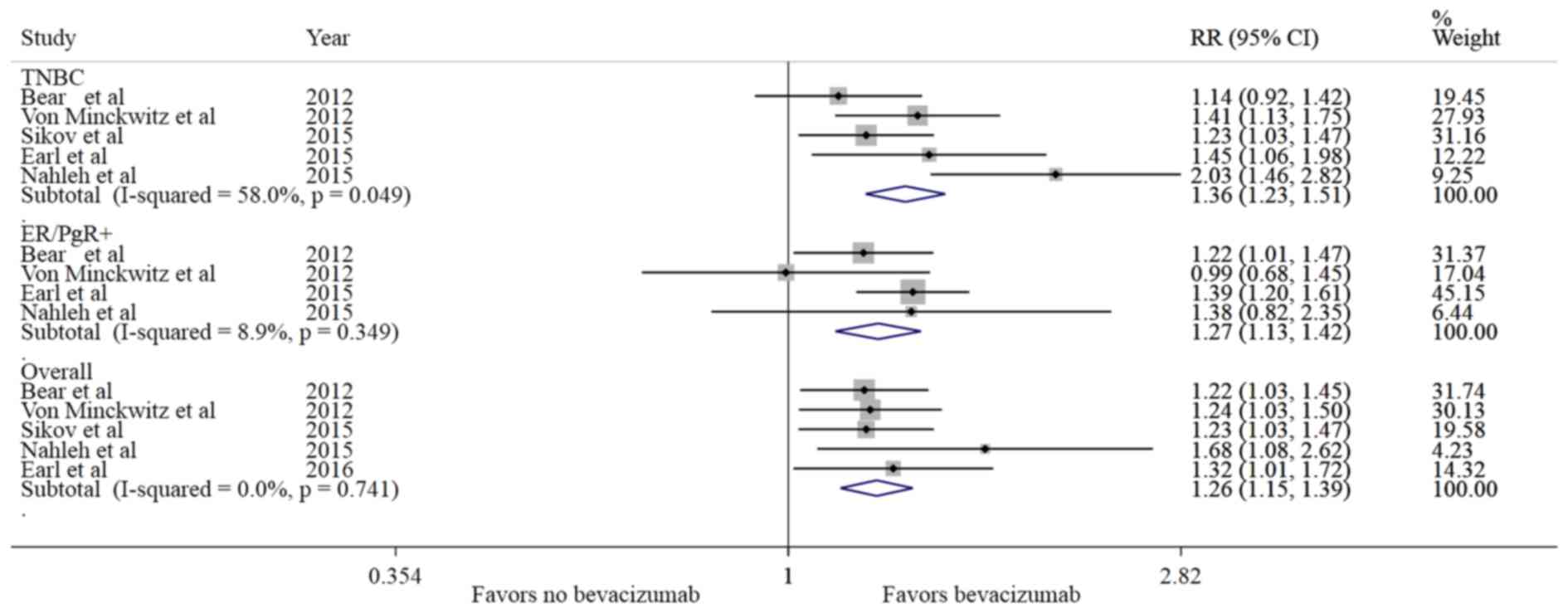
Sikov WM, Berry DA, Perou CM, Singh B,
Cirrincione CT, Tolaney SM, Kuzma CS, Pluard TJ, Somlo G, Port ER,
et al: Impact of the addition of carboplatin and/or bevacizumab to
neoadjuvant once-per-week paclitaxel followed by dose-dense
doxorubicin and cyclophosphamide on pathologic complete response
rates in stage II to III triple-negative breast cancer: CALGB 40603
(Alliance). J Clin Oncol. 33:13–21. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
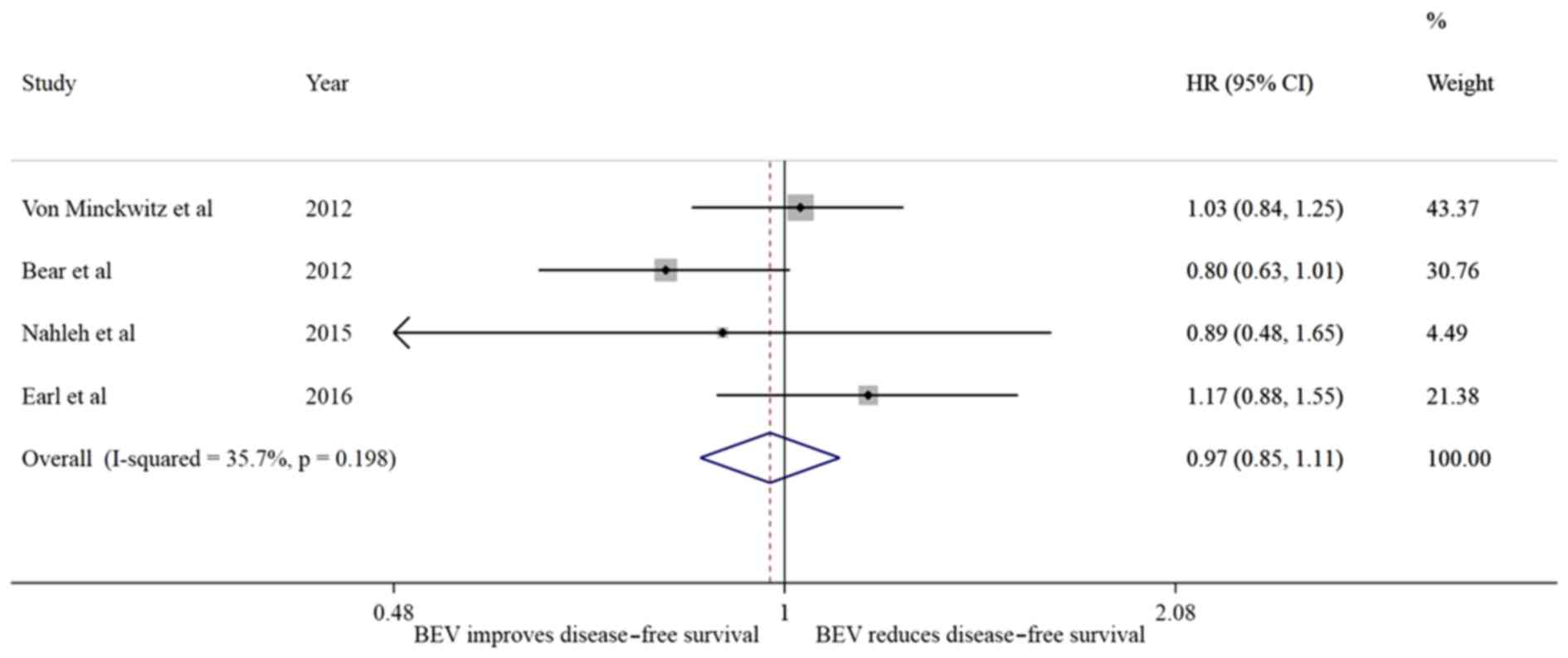
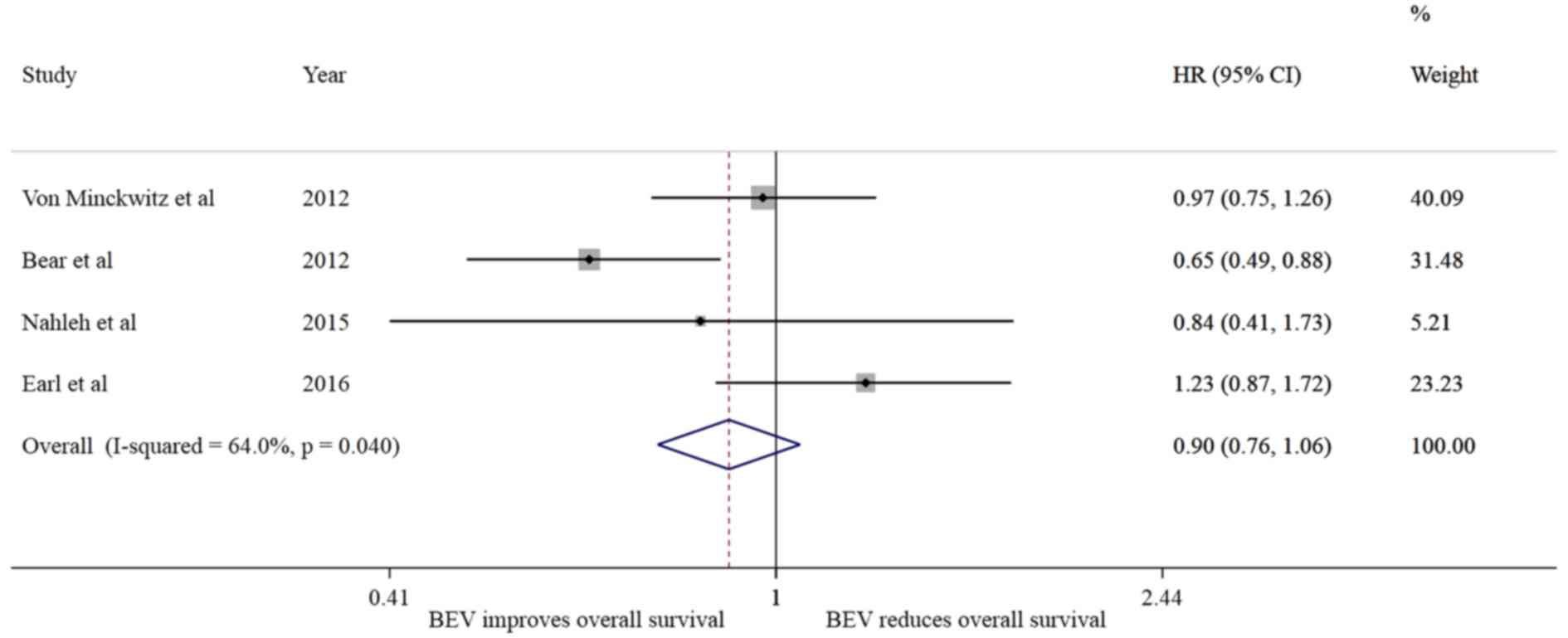
Bear HD, Tang G, Rastogi P, Geyer CE Jr,
Robidoux A, Atkins JN, Baez-Diaz L, Brufsky AM, Mehta RS,
Fehrenbacher L, et al: Bevacizumab added to neoadjuvant
chemotherapy for breast cancer. N Engl J Med. 366:310–320. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
von Minckwitz G, Schneeweiss A, Loibl S,
Salat C, Denkert C, Rezai M, Blohmer JU, Jackisch C, Paepke S,
Gerber B, et al: Neoadjuvant carboplatin in patients with
triple-negative and HER2-positive early breast cancer (GeparSixto;
GBG 66): A randomised phase 2 trial. Lancet Oncol. 15:747–756.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Nahleh ZA, Barlow WE, Hayes DF, Schott AF,
Gralow JR, Sikov WM, Perez EA, Chennuru S, Mirshahidi HR, Corso SW,
et al: SWOG S0800 (NCI CDR0000636131): Addition of bevacizumab to
neoadjuvant nab-paclitaxel with dose-dense doxorubicin and
cyclophosphamide improves pathologic complete response (pCR) rates
in inflammatory or locally advanced breast cancer. Breast Cancer
Res Treat. 158:485–495. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
von Minckwitz G, Eidtmann H, Rezai M,
Fasching PA, Tesch H, Eggemann H, Schrader I, Kittel K, Hanusch C,
Kreienberg R, et al: German Breast Group; Arbeitsgemeinschaft
Gynäkologische Onkologie–Breast Study Groups: Neoadjuvant
chemotherapy and bevacizumab for HER2-negative breast cancer. N
Engl J Med. 366:299–309. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Berry DA and Hudis CA: Neoadjuvant therapy
in breast cancer as a basis for drug approval. JAMA Oncol.
1:875–876. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mancuso MR, Davis R, Norberg SM, O'Brien
S, Sennino B, Nakahara T, Yao VJ, Inai T, Brooks P, Freimark B, et
al: Rapid vascular regrowth in tumors after reversal of VEGF
inhibition. J Clin Invest. 116:2610–2621. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cameron D, Brown J, Dent R, Jackisch C,
Mackey J, Pivot X, Steger GG, Suter TM, Toi M, Parmar M, et al:
Adjuvant bevacizumab-containing therapy in triple-negative breast
cancer (BEATRICE): Primary results of a randomised, phase 3 trial.
Lancet Oncol. 14:933–942. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Miller K, O'Neill AM, Dang CT, et al:
Bevacizumab in the adjuvant treatment of HER2 negative breast
cancer: Final results from Eastern Cooperative Oncology Group
E5103. 2014 ASCO Annual Meeting (abstract 500). J Clin Oncol. 32
Suppl 15:5002014. View Article : Google Scholar
|
|
30
|
Ulivi P, Scarpi E, Passardi A, Marisi G,
Calistri D, Zoli W, Del Re M, Frassineti GL, Tassinari D, Tamberi
S, et al: eNOS polymorphisms as predictors of efficacy of
bevacizumab-based chemotherapy in metastatic colorectal cancer:
Data from a randomized clinical trial. J Transl Med. 13:2582015.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lambrechts D, Claes B, Delmar P, Reumers
J, Mazzone M, Yesilyurt BT, Devlieger R, Verslype C, Tejpar S,
Wildiers H, et al: VEGF pathway genetic variants as biomarkers of
treatment outcome with bevacizumab: An analysis of data from the
AViTA and AVOREN randomised trials. Lancet Oncol. 13:724–733. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Massuti Sureda B, Jantus-Lewintre E,
Gonzalez-Larriba JL, Rodriguez Abreu D, Juan OJ, Domine M,
Provencio Pulla M, de Castro J, Camps C and Rosell R: 37PDSNPS in
angiogenic factors as predictive markers for outcome in patients
(P) with advanced non-squamous NSCLC (NS-NSCLC) treated with
carboplatin, paclitaxel (CP), and bavacizumab (BEV). Final results
of angiomet spanish lung cancer group trial. Ann Oncol. 26 Suppl
1:i102015. View Article : Google Scholar
|
|
33
|
Mattern J, Koomägi R and Volm M:
Association of vascular endothelial growth factor expression with
intratumoral microvessel density and tumour cell proliferation in
human epidermoid lung carcinoma. Br J Cancer. 73:931–934. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sikov WM, Barry WT, Hoadley KA, et al:
Impact of intrinsic subtype by PAM50 and other gene signatures on
pathologic complete response (pCR) rates in triple-negative breast
cancer (TNBC) after neoadjuvant chemotherapy (NACT) plus/-
carboplatin (Cb) or bevacizumab (Bev): CALGB 40603/150709
(Allianc). Cancer Res. 75:S4–S05. 2015. View Article : Google Scholar
|