Introduction
Esophageal squamous cell carcinoma (SCC) of the
thoracic esophagus is common in Asia (1). In Japan, >85% of esophageal
carcinoma cases are located in the thoracic part of the esophagus
(2), and preoperative chemotherapy
followed by radical esophagectomy with three-field lymphadenectomy
is considered to be the standard treatment (3). In Western countries, adenocarcinoma
(adenoCa) of the esophago-gastric junction (EGJ) is common
(4), and is mainly treated by
preoperative chemoradiotherapy followed by surgery (5,6).
Siewert et al classified EGJ carcinoma into
three types (7), namely type I
(abdominal esophageal carcinoma), type II (true carcinoma of the
cardia), and type III (gastric cancer). A Japanese randomized
controlled study recommended an abdominal approach rather than a
left thoraco-abdominal approach (TAA) for patients with type II and
III EGJ adenoCa (8). However, there
have been few reports regarding the optimal treatment method for
patients with abdominal esophageal cancer (AEC), including SCC
(Siewert type I cancer).
The aim of the present study was to elucidate the
characteristics and outcomes of AEC patients who were treated with
esophagectomy.
Patients and methods
Patients
A total of 210 patients who had undergone
esophagectomy between June 1999 and March 2017 were retrospectively
reviewed. AEC was defined as cancer located in the abdominal
esophagus (from the hiatus to the EGJ), >2 cm above the EGJ.
Among all patients, 20 (9.5%) were diagnosed with AEC. All the
patients underwent preoperative endoscopy, computed tomography and
esophagography to confirm the clinical diagnosis and determine the
surgical approach. The right TAA was adopted for patients with
cancer tissue extending >3 cm above the EGJ, or with lymph node
metastases (LNM) in the neck or the mediastinum.
The clinicopathological characteristics of the
patients, including age, sex, histological type, TNM stage,
nutritional status, use of neoadjuvant chemotherapy (NAC), surgical
approach (right thoraco-abdominal or abdominal), postoperative
complications, pathological findings, use of adjuvant chemotherapy
and postoperative overall survival, were evaluated. The nutritional
status included body mass index (BMI) and serum albumin levels.
Postoperative complications included pneumonia, anastomotic leakage
and surgical site infection (SSI) grade >II according to the
Clavien-Dindo classification (9).
The pathological findings included microscopic residual disease
(R1), number of LNM and location of the LNM.
Three analyses were then
performed
First, the differences in these findings were
compared between SCC and adenoCa. Second, the differences were
compared between patients who developed postoperative early
recurrence (within 12 months) and patients without recurrence, to
evaluate the factors associated with early recurrence. Third, a
survival analysis of all AEC patients in association with the
clinicopathological variables was performed using the Kaplan-Meier
method and log-rank test.
Statistical analysis
The differences between the two groups were assessed
using the Mann-Whitney U test for continuous variables and the
Chi-squared test for categorical variables. The Kaplan-Meier method
and log-rank test were used to compare the cumulative survival
rates. In the survival analysis, patients were classified by the
median value of the continuous variables. Statistical analysis was
performed using JMP 6 software (SAS Institute Inc., Cary, NC,
USA).
Results
Patient characteristics
The clinicopathological characteristics of the 20
AEC patients are summarized in Table
I. The median age of the patients was 73 years and 15 (75.0%)
of the patients were male. A total of 11 patients (55.0%) had SCC,
8 (40.0%) had adenoCa, and 1 (5.0%) had adeno SCC. Only 2 patients
(10.0%) were diagnosed as stage I, and 9 (45.0%) received NAC. NAC
included cisplatin and 5-fluorouracil, with or without docetaxel,
as performed from 2008 onwards for clinical stage II or III
patients. In 9 patients (45.0%) the right TAA was used, while in
the remaining 11 patients the abdominal approach was used.
Thoracoscopy and laparoscopy were applied in 4 and 10 patients,
respectively. Regarding postoperative complications, 2 patients
developed pneumonia, 3 patients had anastomotic leakage, and 5
patients had SSI. As regards the pathological findings, 18 patients
(90.0%) had LNM. Only 2 patients had cervical LNM, whereas 15
(75.0%) patients had abdominal LNM. A total of 8 patients received
adjuvant chemotherapy, and recurrence occurred in 10 patients. Of
those 10 patients, 4 developed early recurrence. The median
survival time was 57 months. A total of 12 patients (60.0%) had
stage III disease, and their median survival time was 33 months.
There was no statistically significant difference in
disease-specific survival among stages (P=0.388).
 | Table I.Characteristics of abdominal
esophageal cancer patients. |
Table I.
Characteristics of abdominal
esophageal cancer patients.
| Characteristics | Values |
|---|
| Age, years
(range) | 73 (52–85) |
| Sex, male (%) | 15 (75.0) |
| Histology (%) |
|
| Squamous
cell carcinoma | 11 (55.0) |
|
Adenocarcinoma | 8 (40.0) |
|
Other | 1 (5.0) |
| Stage
I/II/III/IV | 2/4/12/2 |
| BMI, kg/m2
(range) | 22.0 (18.1–27.2) |
| Albumin, g/dl
(range) | 3.8 (3.0–4.8) |
| NAC (%) | 9 (45.0) |
| Surgical approach
(%) |
|
|
Thoraco-abdominal | 9 (45.0) |
|
Abdominal | 11 (55.0) |
| Operative time, min
(range) | 350 (245–690) |
| Blood loss, ml
(range) | 305 (50–2,160) |
| Complications
(%) |
|
|
Pneumonia | 2 (10.0) |
|
Leakage | 3 (15.0) |
| Surgical
site infection | 5 (25.0) |
| Hospital stay, days
(range) | 21 (11–198) |
| pN0 (%) | 2 (10.0) |
| Number of LNM, median
(range) | 2 (0–21) |
| Location of LNM
(%) |
|
| Cervical
LNM | 2 (10.0) |
|
Mediastinal LNM | 7 (35.0) |
| Abdominal
LNM | 15 (75.0) |
| Microscopic residual
cancer (%) | 4 (20.0) |
| Adjuvant therapy
(%) | 8 (40.0) |
| Recurrence (%) | 10 (50.0) |
|
Distant | 7 (35.0) |
|
Local | 4 (20.0) |
The differences between the SCC and the adenoCa
groups are summarized in Table II.
Patients in the SCC group were older compared with those in the
adenoCa group, although age was not significantly associated with
poor disease-specific survival. The BMI and the serum albumin
levels in the SCC patients were significantly lower compared with
those in adenoCa patients. There were no differences in terms of
surgical approach, outcome and postoperative complications. As
regards pathological findings, 50% of the adenoCa group and 27.3%
of the SCC group had mediastinal LNM. Abdominal LNM was more
prevalent in the adenoCa group compared with the SCC group (100 vs.
54.6%, respectively; P=0.026); however, there were no significant
differences in the recurrence pattern (local or distant) or in the
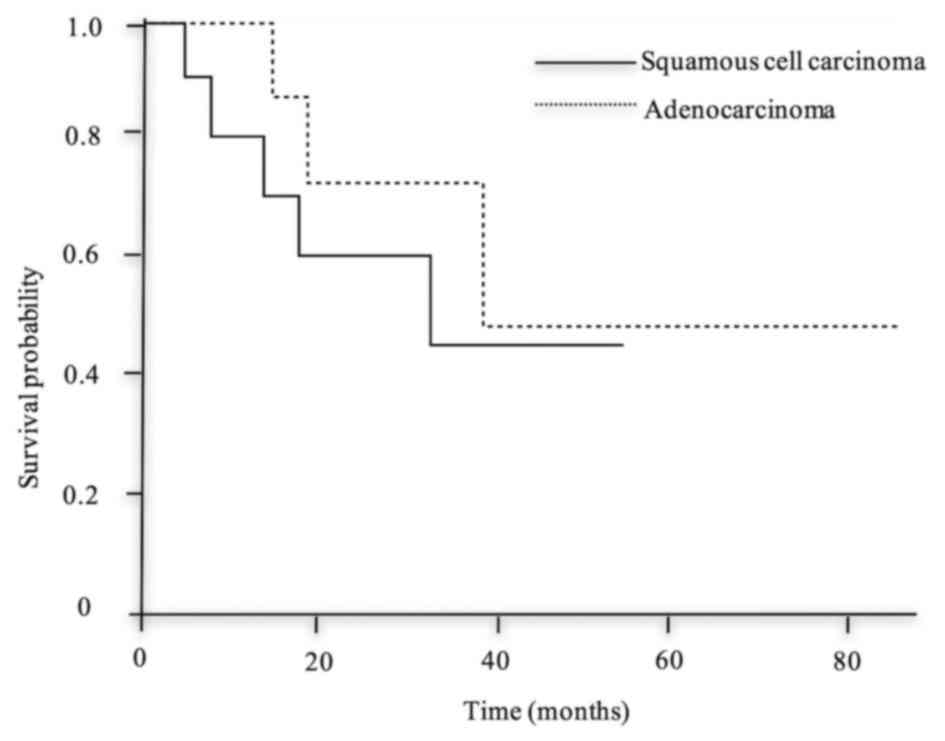
overall survival between the two groups (Fig. 1).
 | Table II.Comparison of the characteristics of
patients with squamous cell carcinoma (SCC) and adenocarcinoma. |
Table II.
Comparison of the characteristics of
patients with squamous cell carcinoma (SCC) and adenocarcinoma.
| Characteristics | SCC (n=11) | Adenocarcinoma
(n=8) | P-value |
|---|
| Sex, male (%) | 9 (81.8) | 6 (75.0) | 0.719 |
| Age, years
(range) | 76 (52–83) | 68 (52–85) | 0.158 |
| Stage
I/II/III/IV | 2/3/5/1 | 0/1/7/0 | 0.266 |
| BMI, kg/m2
(range) | 21.1 (18.5–22.9) | 24.4 (20.6–27.2) | 0.006 |
| Albumin, g/dl
(range) | 3.8 (3.0–4.4) | 4.3 (3.7–4.8) | 0.044 |
| NAC (%) | 5 (45.5) | 4 (50.0) | 0.845 |
| Surgical approach
(%) |
|
| 0.729 |
|
Thoraco-abdominal | 5 (45.5) | 3 (37.5) |
|
|
Abdominal | 6 (54.5) | 5 (62.5) |
|
| Thoracoscopy (%) | 2 | 2 | 1 |
| Laparoscopy (%) | 6 (54.5) | 4 (50.0) | 0.845 |
| Operative time, min
(range) | 355 (245–640) | 313 (245–690) | 0.836 |
| Blood loss, ml
(range) | 300 (50–2160) | 260 (110–580) | 0.620 |
| Complications
(%) |
|
|
|
|
Pneumonia | 2 (18.2) | 0 | 0.485 |
|
Leakage | 3 (27.3) | 0 | 0.107 |
| Surgical
site infection | 3 (27.3) | 2 (25.0) | 1 |
| Hospital stay, days
(range) | 20 (11–198) | 26 (14–35) | 0.710 |
| pN0 (%) | 2 (18.2) | 0 | 0.477 |
| Number of LNM
(range) | 1.0 (0–21) | 2.5 (1–16) | 0.180 |
| Location of LNM
(%) |
|
|
|
| Cervical
LNM | 0 | 1 (12.5) | 0.421 |
|
Mediastinal LNM | 3 (27.3) | 4 (50.0) | 0.377 |
| Abdominal
LNM | 6 (54.6) | 8 (100.0) | 0.026 |
| Microscopic residual
cancer (%) | 3 (27.3) | 1 (12.5) | 0.603 |
| Adjuvant therapy
(%) | 2 (18.2) | 5 (62.5) | 0.048 |
| Recurrence (%) | 5 (45.5) | 5 (62.5) | 0.728 |
|
Distant | 3 (27.3) | 4 (50.0) | 0.377 |
|
Local | 2 (18.2) | 2 (25.0) | 1 |
A comparison between the groups with and without
early recurrence is presented in Table
III. The early recurrence group had a significantly longer
operative time, and a greater prevalence of postoperative SSI and
R1 resection compared with the non-early recurrence group. There
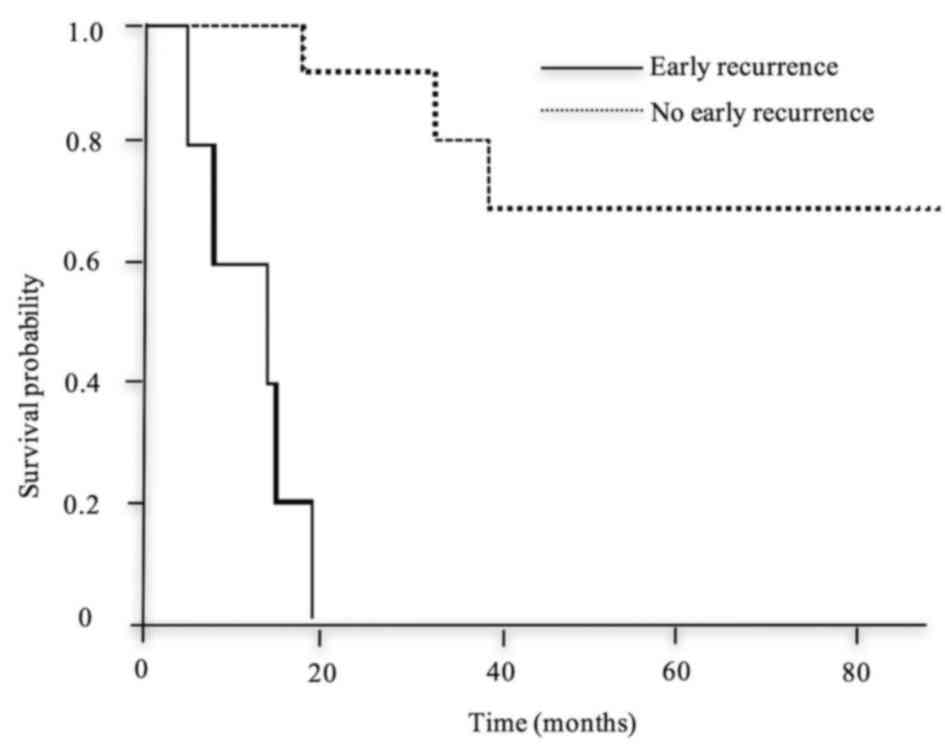
was a significant difference in the overall survival between the
two groups (Fig. 2).
 | Table III.Comparison of the characteristics of
patients with and without early recurrence. |
Table III.
Comparison of the characteristics of
patients with and without early recurrence.
|
Characteristics | Early recurrence
(n=5) | No early recurrence
(n=15) | P-value |
|---|
| Sex, male (%) | 3 (60.0) | 12 (80.0) | 0.371 |
| Age, years
(range) | 72 (52–81) | 73 (52–85) | 0.661 |
| Stage
I/II/III/IV | 0/0/4/1 | 2/4/8/1 | 0.375 |
| BMI,
kg/m2 (range) | 20.3
(19.6–22.6) | 22.2
(18.1–27.2) | 0.861 |
| Albumin, g/dl
(range) | 3.8 (3.0–4.8) | 3.9 (3.3–4.7) | 0.625 |
| NAC (%) | 2 (40.0) | 7 (46.7) | 0.795 |
| Thoraco-abdominal
approach (%) | 4 (80.0) | 5 (33.3) | 0.069 |
| Operative time, min
(range) | 630 (295–690) | 321 (245–641) | 0.032 |
| Blood loss, ml
(range) | 520
(300–2,160) | 240 (50–1,550) | 0.061 |
| Complications
(%) |
|
|
|
|
Pneumonia | 1 (20.0) | 1 (6.7) | 0.447 |
|
Leakage | 2 (40.0) | 1 (6.7) | 0.140 |
|
Surgical site infection | 3 (60.0) | 2 (13.3) | 0.037 |
| Hospital stay, days
(range) | 29 (13–198) | 20 (11–46) | 0.175 |
| Number of LNM
(range) | 7 (1–21) | 2 (0–8) | 0.024 |
| Location of LNM
(%) |
|
|
|
|
Cervical LNM | 1 (20.0) | 1 (6.7) | 0.447 |
|
Mediastinal LNM | 4 (80.0) | 3 (20.0) | 0.031 |
|
Abdominal LNM | 4 (80.0) | 11 (73.3) | 1 |
| Microscopic
residual cancer (%) | 4 (80.0) | 0 (0.0) | 0.001 |
| Adjuvant therapy
(%) | 1 (20.0) | 7 (46.7) | 0.603 |
| Recurrence pattern
(%) |
|
| 0.026 |
|
Local | 1 (20.0) | 2 (13.3) |
|
|
Distant | 4 (80.0) | 3 (20.0) |
|
The results of survival analysis using the
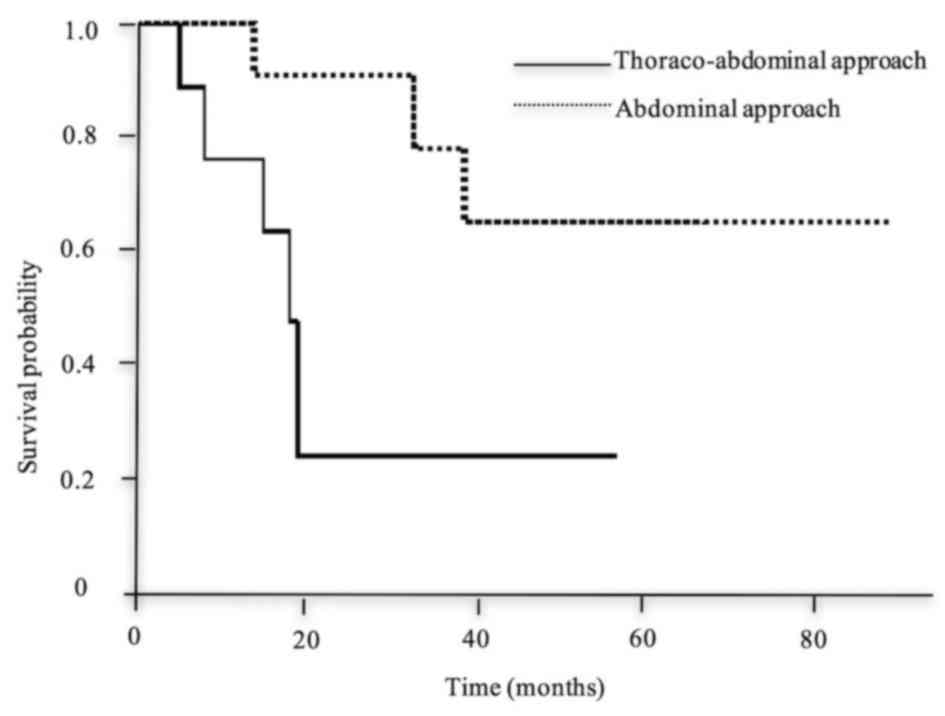
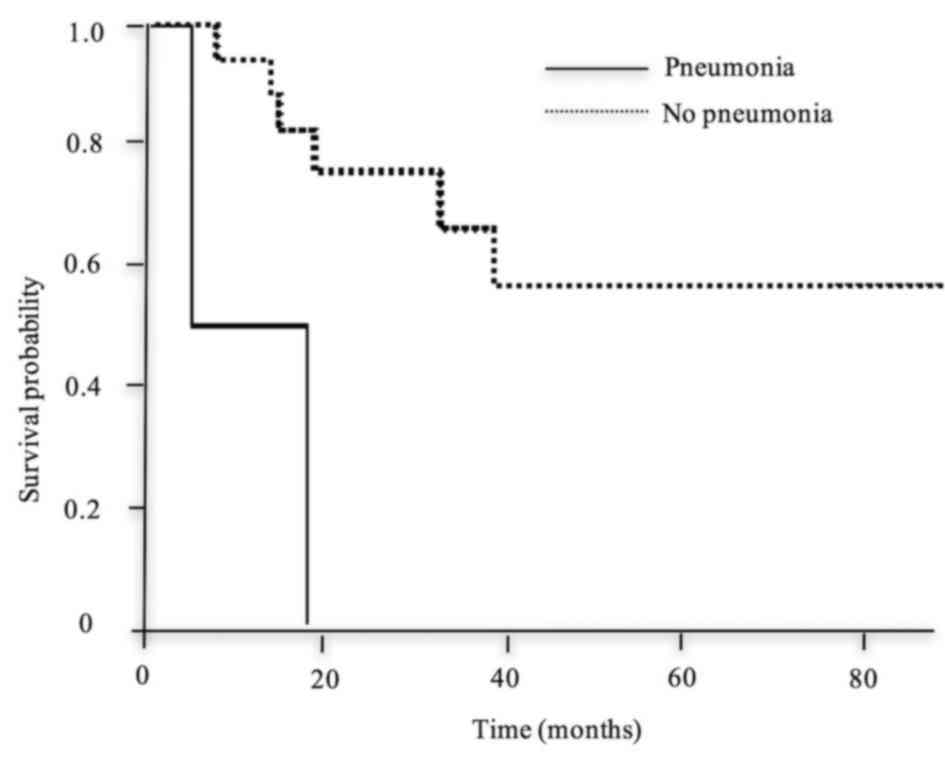
Kaplan-Meier method and log-rank test is shown in Table IV. The survival time of patients who
underwent TAA (Fig. 3), or had
postoperative pneumonia (Fig. 4),
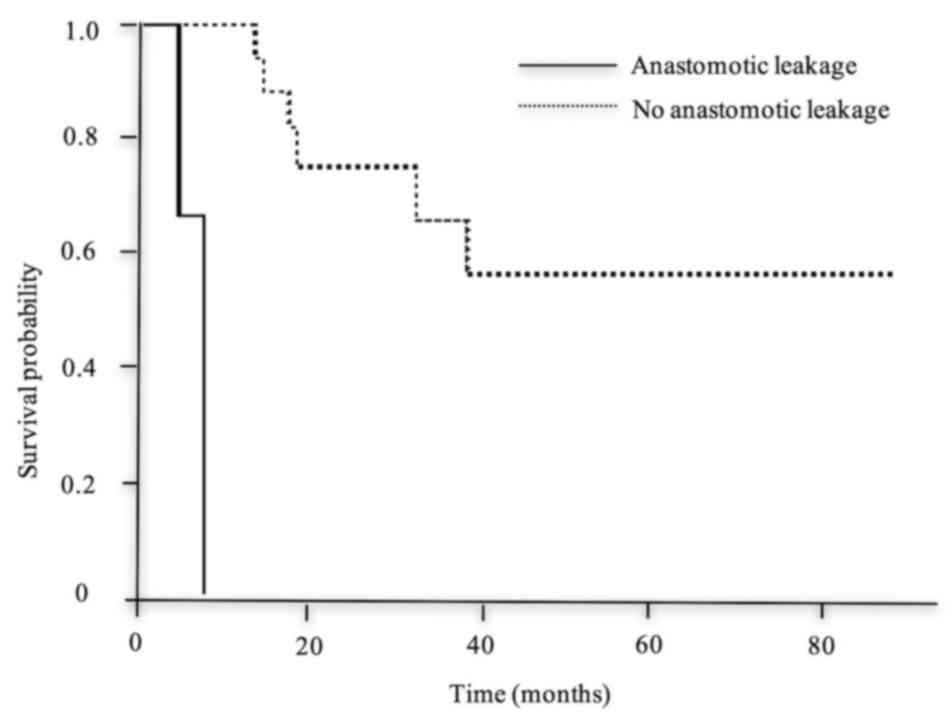
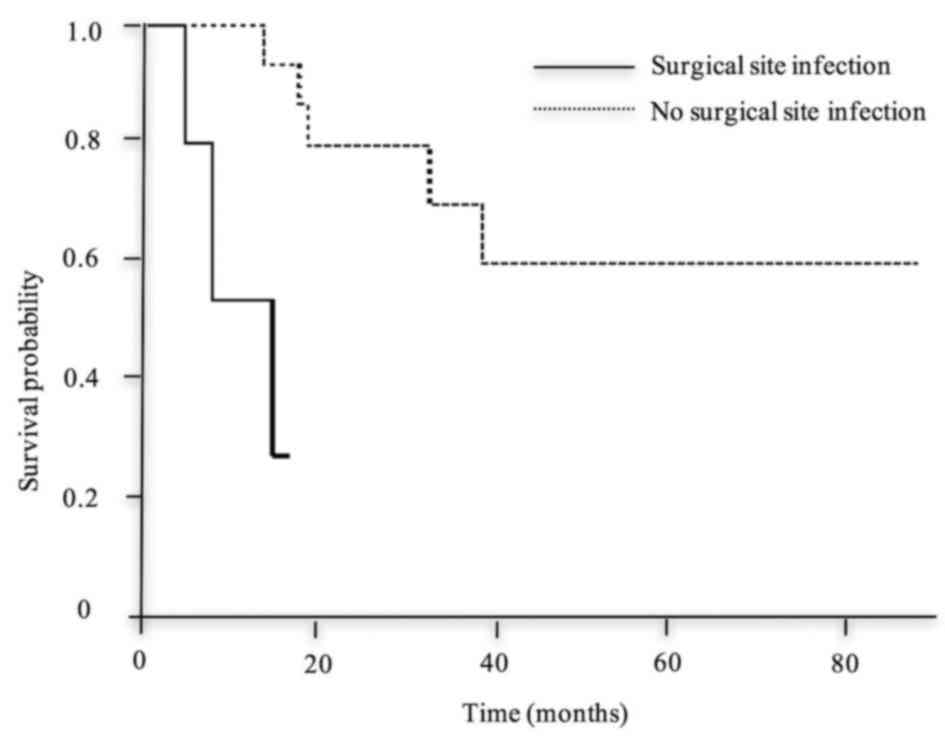
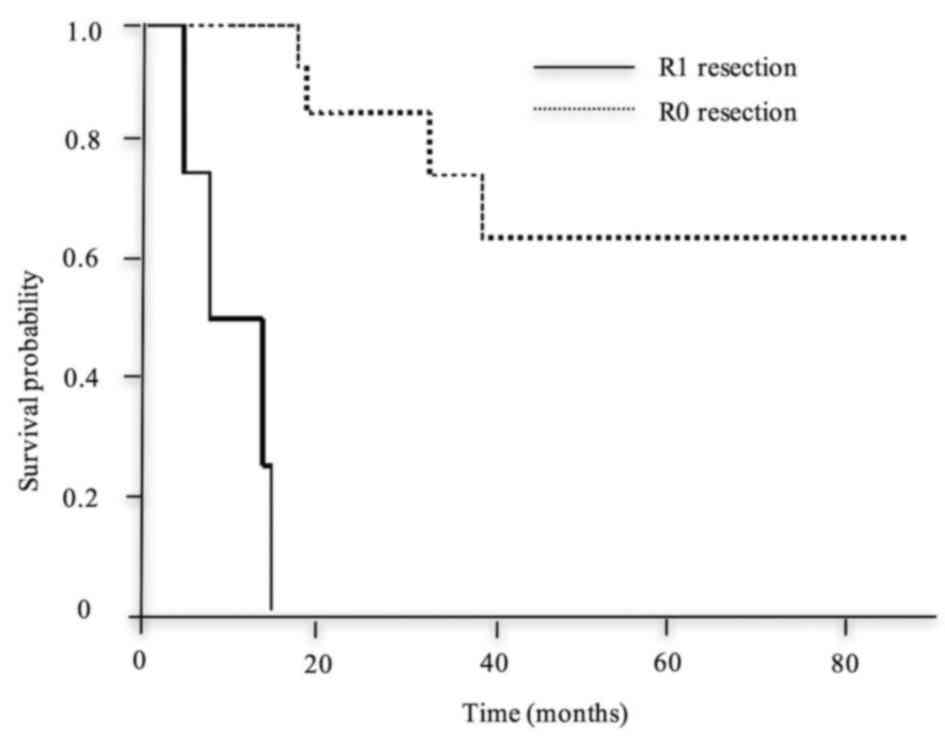
leakage (Fig. 5), SSI (Fig. 6) and R1 (Fig. 7), was significantly shorter compared
with that of patients without these factors.
 | Table IV.Evaluation of variables associated
with disease-specific survival using the Kaplan-Meier method with
the log-rank test. |
Table IV.
Evaluation of variables associated
with disease-specific survival using the Kaplan-Meier method with
the log-rank test.
|
| MST, months |
|
|---|
|
|
|
|
|---|
| Variables | Yes | No | P-value |
|---|
| Sex, male | NR | 33 | 0.199 |
| Age >73
years | 55 | 39 | 0.836 |
| BMI >21.7
kg/m2 | NR | 39 | 0.297 |
| Stage III or
IV | NR | 33 | 0.096 |
| Neo-adjuvant
chemotherapy | NR | 33 | 0.745 |
| Thoraco-abdominal
approach | 18 | NR | 0.028 |
| Operative time
>345 min | 19 | NR | 0.251 |
| Blood loss >310
ml | 19 | NR | 0.615 |
| Pneumonia | 18 | NR | 0.004 |
| Leakage | 8 | NR | <0.001 |
| Surgical site
infection | 15 | NR | 0.001 |
| Microscopic
residual cancer | 14 | NR | <0.001 |
| Number of LNM
>2 | 19 | NR | 0.065 |
| Cervical LNM | NR | 39 | 0.888 |
| Mediastinal
LNM | 19 | NR | 0.083 |
| Abdominal LNM | 39 | 48 | 0.305 |
| Adjuvant
therapy | 57 | 55 | 0.540 |
Discussion
The present study demonstrated that over one-third
of patients with AEC (either SCC or adenoCa) had mediastinal LNM,
and postoperative complications may be associated with poor
prognosis. Conversely, differences in histological type and
surgical approach did not appear to affect postoperative
survival.
Previous studies reported that, in cases with
adenoCa of the EGJ, Siewert type, histological type, tumor size and
number of LNM were associated with prognosis (10). In those studies, prognosis was found
to be better in Siewert type I patients compared with type II or
III patients (11), and in patients
with adenoCa compared with those with SCC (12). In the present study, as over half of
the cohort were SCC patients, it was observed that AEC corresponded
to Siewert type I, with >30% of patients having mediastinal LNM.
However, there was no difference between the SCC and adenoCa groups
regarding the rate of mediastinal LNM or survival. In addition, 80%
of patients who had R1 also experienced early recurrence. For these
reasons, surgery for either SCC or adenoCa, without microscopic
residual cancer (R0 resection), was crucial to the survival of AEC
patients.
NAC or neoadjuvant chemoradiotherapy (NACRT) may
achieve an increase in the R0 resection rate for AEC patients
(13). However, our data do not
support the efficacy of NAC and adjuvant chemotherapy. As NAC was
introduced to treat stage II or III thoracic esophageal SCC
patients in 2009, in accordance with Japanese guidelines, only 9
patients received NAC in the present study. A prospective study
investigating the efficacy of the NAC or NACRT for AEC is required.
The survival of patients who underwent an abdominal approach was
better compared with those who underwent right TAA, possibly
because right TAA was performed for patients with more extensive
cancer, who exhibited mediastinal or cervical LNM or thoracic
esophageal invasion. Furthermore, recent advances in the use of a
thoraco-laparoscopic approach for esophageal cancer may result in
the development of minimally invasive surgery with R0 resection,
which may improve the prognosis of patients compared with a
conventional approach (14).
Our data suggest an association between the
development of postoperative complications and a poor prognosis.
Postoperative pneumonia after thoraco-abdominal esophagectomy has
been reported to be associated with a poor prognosis (15,16).
Markar et al reported an association between severe
anastomotic leakage and a poor prognosis from 2,944 surgically
treated esophageal cancer patients (17). Although it was difficult to directly
accept these data due to the high 90-day mortality rate (7.1%;
209/2,944), these postoperative complications may suppress the
patient's immunological function (18) and result in early recurrence of
cancer.
The limitation of the present study is that it was
retrospectively conducted with a small sample size. A multivariate
analysis was performed using the Cox proportional hazards model,
and found that pneumonia and R1 resection were negative predictive
factors. However, due to small sample size, the hazard ratio and
95% confidence interval values were uncertain. In addition, the EGJ
was difficult to define in some patients with advanced AEC, which
made it more difficult to distinguish between Siewert types I and
II. Further study is required to elucidate the risk of a poor
prognosis for Siewert type I patients with AEC and to establish a
treatment strategy.
The present study demonstrated the significance of
R0 resection and prevention of postoperative complications for
improving the prognosis of AEC patients. Further studies, such as
prospective and multi-center investigations with large cohorts, are
required to confirm short- and long-term outcomes, including the
prognosis of AEC.
The present study was performed in accordance with
the ethical standards laid down in the 1964 Declaration of Helsinki
and its later amendments. Informed consent was obtained from all
individual participants included in this study.
References
|
1
|
Wu M, Zhao JK, Hu XS, Wang PH, Qin Y, Lu
YC, Yang J, Liu AM, Wu DL, Zhang ZF, et al: Association of smoking,
alcohol drinking and dietary factors with esophageal cancer in
high- and low-risk areas of jiangsu province, china. World J
Gastroenterol. 12:1686–1693. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tachimori Y, Ozawa S, Numasaki H,
Fujishiro M, Matsubara H, Oyama T, Shinoda M, Toh Y, Udagawa H, Uno
T, et al: Registration committee for esophageal cancer of the Japan
esophageal society. Comprehensive registry of esophageal cancer in
Japan, 2009. Esophagus. 13:110–137. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ando N, Kato H, Igaki H, Shinoda M, Ozawa
S, Shimizu H, Nakamura T, Yabusaki H, Aoyama N, Kurita A, et al: A
randomized trial comparing postoperative adjuvant chemotherapy with
cisplatin and 5-fluorouracil versus preoperative chemotherapy for
localized advanced squamous cell carcinoma of the thoracic
esophagus (JCOG9907). Ann Surg Oncol. 19:68–74. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
DeMeester SR: Adenocarcinoma of the
esophagus and cardia: A review of the disease and its treatment.
Ann Surg Oncol. 13:12–30. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sjoquist KM, Burmeister BH, Smithers BM,
Zalcberg JR, Simes RJ, Barbour A and Gebski V; Australasian
Gastro-Intestinal Trials Group, : Survival after neoadjuvant
chemotherapy or chemoradiotherapy for resectable oesophageal
carcinoma: An updated meta-analysis. Lancet Oncol. 12:681–692.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
van Hagen P, Hulshof MC, van Lanschot JJ,
Steyerberg EW, van Berge Henegouwen MI, Wijnhoven BP, Richel DJ,
Nieuwenhuijzen GA, Hospers GA, Bonenkamp JJ, et al: Preoperative
chemoradiotherapy for esophageal or junctional cancer. N Engl J
Med. 366:2074–2084. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Siewert JR and Stein HJ: Classification of
adenocarcinoma of the oesophagogastric junction. Br J Surg.
85:1457–1459. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sasako M, Sano T, Yamamoto S, Sairenji M,
Arai K, Kinoshita T, Nashimoto A and Hiratsuka M; Japan Clinical
Oncology Group (JCOG9502), : Left thoracoabdominal approach versus
abdominal-transhiatal approach for gastric cancer of the cardia or
subcarsia: A randomized controlled trial. Lancet Oncol. 7:644–651.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Clavien PA, Barkun J, de Oliveira ML,
Vauthey JN, Dindo D, Schulick RD, de Santibañes E, Pekolj J,
Slankamenac K, Bassi C, et al: The Clavien-Dindo classification of
surgical complications: Five-year experience. Ann Surg.
250:187–196. 2000. View Article : Google Scholar
|
|
10
|
Curtis NJ, Noble F, Bailey IS, Kelly JJ,
Byrne JP and Underwood TJ: The relevance of the Siewert
classification in the era of multimodal therapy for adenocarcinoma
of the gastro-oesophageal junction. J Surg Oncol. 109:202–207.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gertler R, Stein HJ, Langer R, Nettelmann
M, Schuster T, Hoefler H, Siewert JR and Feith M: Long-term outcome
of 2920 patients with cancers of the esophagus and esophagogastric
junction: Evaluation of the new union internationale contre le
cancer/american joint cancer committee staging system. Ann Surg.
253:689–698. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hosokawa Y and Kinoshita T, Konishi M,
Takahashi S, Gotohda N, Kato Y, Daiko H, Nishimura M, Katsumata K,
Sugiyama Y and Kinoshita T: Clinicopathological features and
prognostic factors of adenocarcinoma of the esophagogastric
junction according to Siewert classification: Experiences at a
single institution in Japan. Ann Surg Oncol. 19:677–683. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sjoquist KM, Burmeister BH, Smithers BM,
Zalcberg JR, Simes RJ, Barbour A and Gebski V; Australasian
Gastro-Intestinal Trials Group, : Survival after neoadjuvant
chemotherapy or chemoradiotherapy for resectable oesophageal
carcinoma: An updated meta-analysis. Lancet Oncol. 12:681–692.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kitagawa H, Namikawa T, Munekage M,
Fujisawa K, Munekgae E, Kobayashi M and Hanazaki K: Outcomes of
thoracoscopic esophagectomy in prone position with laparoscopic
gastric mobilization for esophageal cancer. Langenbecks Arch Surg.
401:699–705. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Booka E, Takeuchi H, Nishi T, Matsuda S,
Kaburagi T, Fukuda K, Nakamura R, Takahashi T, Wada N, Kawakubo H,
et al: The impact of postoperative complications on survivals after
esophagectomy for esophageal cancer. Medicine (Baltimore).
94:e13692015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kataoka K, Takeuchi H, Mizusawa J, Igaki
H, Ozawa S, Abe T, Nakamura K, Kato K, Ando N and Kitagawa Y:
Prognostic impact of postoperative morbidity after esophagectomy
for esophageal cancer: Exploratory analysis of JCOG9907. Ann Surg.
265:1152–1157. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Markar S, Gronnier C, Duhamel A, Mabrut
JY, Bail JP, Carrere N, Lefevre JH, Brigand C, Vaillant JC, Adham
M, et al: The impact of severe anastomotic leakage on long-term
survival and cancer recurrence after surgical resection for
esophageal malignancy. Ann Surg. 262:972–980. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Okamura A, Takeuchi H, Matsuda S, Ogura M,
Miyasho T, Nakamura R, Takahashi T, Wada N, Kawakubo H, Saikawa Y
and Kitagawa Y: Factors affecting cytokine change after
esophagectomy for esophageal cancer. Ann Surg Oncol. 22:3130–3135.
2015. View Article : Google Scholar : PubMed/NCBI
|