Introduction
Combined spinal-epidural anesthesia (CSE) is a
popular and well-accepted technique for caesarean section (1). Shivering is one of the most frequent
complications associated with spinal or epidural anesthesia, with
an occurrence rate of 45–85% (2).
Neuraxial techniques may obtund skin afferents and decrease the
vasoconstriction threshold even beyond the level of sensory block,
thereby reducing a patient's ability to maintain their core body
temperature (poikilothermia), which may lead to shivering (3,4). However,
the exact mechanism of this is not clear. Shivering is
uncomfortable for affected patients and may interfere with
monitoring of the electrocardiogram, blood pressure and oxygen
saturation (5). It may also increase
oxygen consumption, lactic acidosis and carbon dioxide production,
as well as causing distress to parturients with a low cardiac
pulmonary reserve and a high metabolism (3,6,7). Thus, prevention and treatment of
shivering is an important aspect of patient care during CSE,
resulting in better postoperative outcomes and a reduced incidence
of post-surgical complications (8).
At present, a number of pharmacological methods are
used to treat shivering (9). Many
medications have been investigated for their ability to control
postoperative shivering, including meperidine and tramadol
(10). However, these medications may
have adverse effects on the baby if administered to parturients
prior to delivery (9,11).
Ketamine, which is a noncompetitive antagonist of
N-methyl-D-aspartic acid (NMDA) receptors, serves a role in
thermoregulation on various levels (12). NMDA receptors modulate the
noradrenergic and serotonergic neurons in the locus coeruleus;
consequently, NMDA receptors in the dorsal horn of the spinal cord
facilitate the transmission of ascending nociceptive stimuli
(12). A previous study reported that
intravenous ketamine is able to inhibit postoperative shivering
(13); however, whether epidural
application has the same effect is yet to be established. The aim
of the present study was to investigate whether 0.5 mg/kg of
epidurally administered ketamine affects the incidence and severity
of shivering and to evaluate the efficacy and safety of epidural
ketamine as a prophylactic agent for the prevention of shivering in
patients undergoing caesarean section during CSE.
Materials and methods
Patients and ethics
The present study was approved by the Ethics
Committee of Gansu Provincial Maternity and Child-Care Hospital
(Lanzhou, China). A prospective, randomized, double-blind study was
performed and prior written informed consent was obtained from all
patients. A previous clinical study reported the incidence of
shivering in caesarean section as 55% (10). The sample size required to detect a
40% reduction was a minimum of 28 patients in each group;
therefore, 30 patients were included in each group in the present
study. Thus, a total of 60 female patients (22 to 41 years of age)
with an American Society of Anesthesiologists (ASA) physical status
of I–II and who were scheduled for elective caesarean section under
CSE were recruited for the current trial. Patients were randomly
divided into two groups (group S and group K; n=30 per group) using
a computer-generated code. Exclusion criteria were as follows:
History of severe cardiovascular complications, preoperative
temperature >38°C or <35°C, any contraindications to regional
anesthesia, ketamine allergy, preeclampsia, diabetes mellitus,
significant peripartum hemorrhage or the existence of an absolute
or relative contraindication for the application of the epidural
technique, including neuromuscular disease, bleeding tendency or
local skin infection in the lumbar region. A previous study
(14) reported that intraoperative
shivering is not associated with intraoperative temperature. The
intraoperative room temperature and the temperature of the infusion
liquid were controlled and, although the intraoperative temperature
was monitored during the present study, it was not included as a
detection index. If the intraoperative anesthetic effect was
insufficient or patients reported pain during surgery, 50–100 µg
fentanyl (Yichang Renfu Pharmaceutical Co., Ltd., Yichang, China;
production batch no. 100404) was administered.
Surgical procedure and treatments
The temperature of the operating room was maintained
at 23–25°C, with a humidity of 55–65%. Lactated Ringer's solution
(Sichuan Kelun Pharmaceutical Co., Ltd., Chengdu, China; production
batch no. B12122104) was preheated to 37°C in a warmed cabinet and
10 ml/kg was administered intravenously over 30 min. All
parturients were placed under standard monitoring.
CSE was performed in the left lateral decubitus
position at the L3-L4 lumbar vertebral interspace, with 10 mg 0.5%
hyperbaric bupivacaine by a needle-through-needle technique
(27G/18G spinal/epidural needle; Tuoren Medical Co., Ltd.,
Xinxiang, China). The spinal needle was removed and the epidural
catheter was inserted 4 cm from the end of the needle into the
epidural space and secured aseptically. All treatments were
administered by an anesthesiologist who was blinded to study group
allocation. In group S, all patients received 3 ml saline by
epidural bolus. In group K, all patients received 3 ml ketamine
(0.5 mg/kg; Fujian Gutian Pharmaceutical Co., Ltd., Gutian, China;
production batch no. 1303032) by epidural bolus. Parturient were
subsequently placed in a supine position with a left lateral tilt
(15°).
Monitoring
Sensory and motor assessments were performed at
1-min intervals using pinprick testing and the modified Bromage
score (15), respectively. Surgery
began when adequate anesthesia to the T6 dermatome was achieved.
The time of ketamine/saline administration and the duration of
surgery (from initial skin incision to the last stitch) were
recorded, as were hemodynamic changes and the amount of irrigation
fluid used during the procedure. Side effects, including
hypotension, amnesia, nausea and hallucinations, were also
recorded. The Apgar scale at 1 and 5 min after birth of baby was
recorded (16). All hypotensive
episodes were treated by crystalloid infusion if necessary, with
intravenous ephedrine (5–15 mg; Northeast Pharmaceutical Group Co.,
Ltd., Shengyang, China; production batch no. 131102–1).
Shivering grading
Shivering was graded with a scale similar to that
validated by Crossley and Mahajan (14). Grades were as follows: 0, no
shivering; 1, piloerection or peripheral vasoconstriction with no
visible shivering; 2, muscular activity in 1 muscle group; 3,
muscular activity in >1 muscle group, but not generalized
shivering; and 4, shivering involving the whole body. When
shivering grade was ≥3, patients were treated with 25 mg
intravenous meperidine (Yichang Renfu Pharmaceutical Co., Ltd.;
production batch no. 110406).
Sedation grading
Hallucination, as a side effect, was defined as a
false sensory experience in which the patient reported that they
saw, heard, smelled, tasted, or felt something that was
nonexistent. The attending anesthesiologist also assessed the
degree of sedation on a 5-point scale as follows: 1, fully awake
and oriented; 2, drowsy; 3, eyes closed but rousable to command; 4,
eyes closed but rousable to mild physical stimulation; and 5, eyes
closed but unarousable to mild physical stimulation (17). All data were recorded pre-operatively
and at 10, 30 and 60 min following anesthesia.
Statistical analysis
A power analysis was performed using a power
threshold of 90% and an α-value of 0.05. The Power and Sample Size
statistics program (PS version 3.0.43; Vanderbilt University,
Nashville, TN, USA) was used for power analysis. Data are presented
as the mean ± standard deviation for continuous data and frequency
for non-parametric data. Differences between demographic anesthetic
and surgical characteristics of patients were assessed using an
independent t-test. Fisher's exact test was used to analyze
categorical variables. Differences in heart rate, peripheral
capillary oxygen saturation (SpO2) and mean arterial
pressure (MAP) at different time-points were assessed using
repeated measures analysis of variance with Dunnett's test. SPSS
18.0 (SPSS, Inc., Chicago, IL, USA) was utilized to perform the
tests and P<0.05 was considered to indicate a statistically
significant difference.
Results
Patient characteristics
A total of 60 parturients were enrolled in the
present study and randomized into the two groups (n=30; Fig. 1). No significant differences in
patient characteristics, including age, body mass index,
gestational week and operation duration were observed between
groups (Table I).
 | Table I.Patient characteristics. |
Table I.
Patient characteristics.
| Patient
characteristics | Group S (n=30) | Group K (n=30) |
|---|
| Age (years) | 27.8±6.3 | 28.1±5.7 |
| Body mass index
(kg/m2) | 27.8±3.5 | 27.6±3.3 |
| Gestational age
(weeks) | 39.3±1.0 | 38.9±1.1 |
| Operation duration
(min) | 59.3±13.6 | 60.0±13.7 |
| Heart rate
(bpm) | 94.3±11.5 | 91.9±15.3 |
| SPO2
(%) | 100±0 | 100±0 |
| Apgar score |
|
|
| 5
min | 8.8±0.9 | 8.7±1.2 |
| 10
min | 10±0.0 | 10±0.0 |
No significant differences were observed in the
Apgar scores at 1 and 5 between groups (Table I). Furthermore, no significant
differences in heart rate and SpO2 values between groups
were observed at any time (Table I).
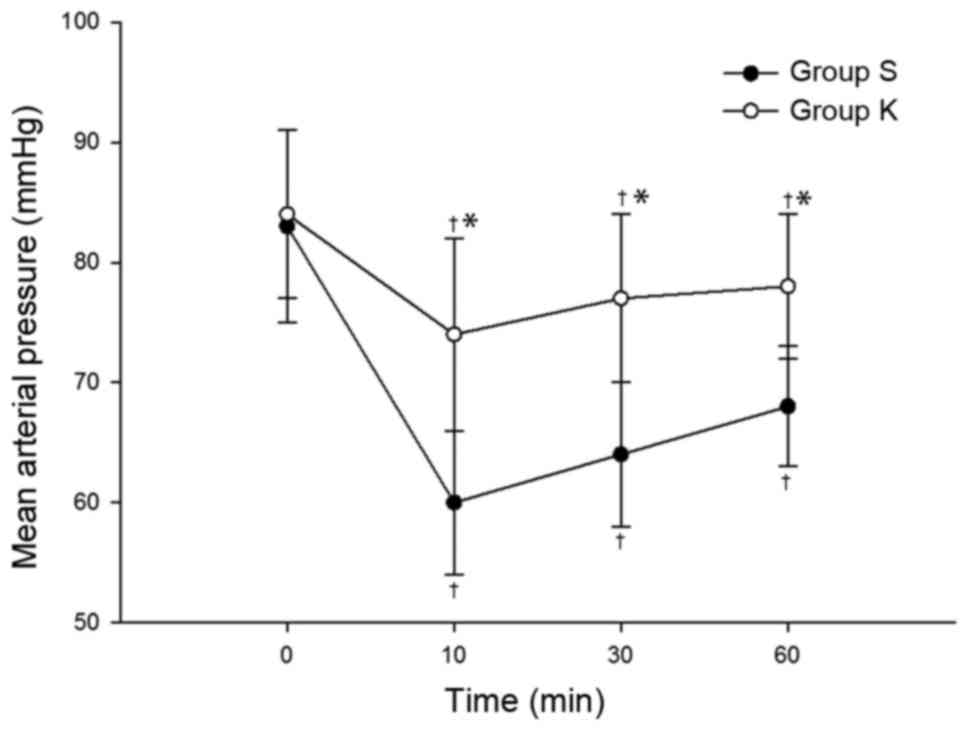
However, MAP in groups S and K were significantly reduced compared
with the baseline at 10, 30 and 60 min (P<0.05; Fig. 2). In addition, MAP was significantly
lower in group S compared with group K at 10, 30 and 60 min
(P<0.05; Fig. 2).
Shivering grade
At 30 min, the incidence and severity of shivering
in group K were significantly lower compared with group S (Table II). At 10 min, 2 patients in group S
were shivering, compared with 1 patient in group K. At 30 min, 10
patients experienced shivering in group S, of whom 2 suffered grade
3 and 2 suffered grade 4 shivering, whereas only 2 patients
experienced shivering in group K, both of grade 1. At 60 min, 3
patients experienced shivering in group S (of whom 2 suffered grade
3 shivering), whereas only 1 patient experienced shivering (grade
2) in group K. Following treatment with intravenous meperidine (25
mg), shivering ceased in all patients.
 | Table II.Shivering grade at different
time-points. |
Table II.
Shivering grade at different
time-points.
|
| Group S (n=30) | Group K (n=30) |
|---|
|
|
|
|
|---|
| Shivering
grade | Pre-operation | 10 min | 30 min | 60 min | Pre-operation | 10 min | 30 min | 60 min |
|---|
| 0 | 30 | 28 | 20 | 27 | 30 | 29 |
28a | 29 |
| 1 | 0 | 0 | 5 | 0 | 0 | 0 | 2 | 0 |
| 2 | 0 | 2 | 1 | 1 | 0 | 1 | 0 | 1 |
| 3 | 0 | 0 | 2 | 2 | 0 | 0 | 0 | 0 |
| 4 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 |
Sedation grade
Sedation was one of the side effects of ketamine. In
group K, 15 patients exhibited grade 2 sedation and 2 patients
exhibited grade 3 sedation at the 10 min time-point. At 30 min, 5
patients experienced grade 2 sedation and at 60 min, 2 patients
experienced grade 2 sedation. No patients experienced sedation at
grade 4 or 5 (Table III). In group
S, patients did not use any sedative drugs, and were all in a
natural awakened state.
 | Table III.Sedation grade in group K (n=30). |
Table III.
Sedation grade in group K (n=30).
| Sedation grade | Pre-operation | 10 min | 30 min | 60 min |
|---|
| 1 | 30 | 13 | 25 | 28 |
| 2 | 0 | 15 | 5 | 2 |
| 3 | 0 | 2 | 0 | 0 |
| 4 | 0 | 0 | 0 | 0 |
| 5 | 0 | 0 | 0 | 0 |
Incidence of side effects
The frequency of nausea and hypotension (systolic
pressure, <90 mmHg) was significantly higher in group S compared
with group K, as was the number of patients who received ephedrine
(P<0.05; Table IV). None of the
common side effects of ketamine, including nystagmus,
hallucinations and amnesia, were observed (Table IV).
 | Table IV.Incidence of side effects. |
Table IV.
Incidence of side effects.
| Symptom | Group S (n=30) | Group K (n=30) |
|---|
| Nausea | 12 |
2a |
| Hypotension | 10 |
3a |
| Nystagmus | 0 | 0 |
| Hallucinations | 0 | 0 |
| Amnesia | 0 | 0 |
| Ephedrine | 6 |
2a |
Discussion
Shivering is a common and uncomfortable side effect
associated with neuraxial anesthesia. Neuraxial techniques may
block the activity of the sympathetic nervous system and reduce a
patient's ability to regulate body temperature (18). Additionally, it may. inhibit
thermoregulatory control centrally and lead to internal
redistribution of heat from the core to the periphery (19,20). In
addition, the release of amniotic fluid from the mother's body may
lead to heat loss (21).
Collectively, all of these factors may increase the incidence of
shivering in parturients.
Shivering during and following caesarean section
increases oxygen consumption and the burden on the heart and lungs,
as well as reducing hepatorenal blood flow, influencing uterine
contractions and extending the anesthesia recovery time (2,11,22). Therefore, intraoperative and
postoperative prevention of shivering in patients receiving
epidural anesthesia is of important clinical significance (23). Although shivering is a protective
mechanism when the body is subjected to low temperatures, there is
no definite linear relationship that exists between body
temperature and the occurrence of shivering during CSE; shivering
may also be observed in normothermic patients under spinal
anesthesia (24), suggesting that
mechanisms other than heat loss and subsequent decrease in core
temperature contribute to the development of shivering. These
mechanisms may include uninhibited spinal reflexes, postoperative
pain, decreased sympathetic activity, pyrogen release, adrenal
suppression and respiratory alkalosis (25). Shivering may therefore be considered
both thermogenic and non-thermogenic (14,23). As
such, a variety of measures are widely used to prevent
postoperative shivering (22,26,27).
A number of pharmacological methods for inhibiting
shivering have been reported (28,29).
Numerous pharmacological interventions have been proposed for the
treatment of postoperative shivering, however the ideal treatment
has not yet been established (9).
Meperidine has been reported to be one of the most effective drugs
used to treat post-anesthetic shivering (30,31). In
the present study, 25 mg meperidine was intravenously administered
if the severity of shivering was above grade 2. The side effects of
intravenous meperidine, including nausea, vomiting, pruritus,
hypotension, bronchospasm, bradycardia and respiratory
insufficiency, have been reported to be dose-related (32) and may limit the application of this
agent, particularly in parturients.
Ketamine was first described in the literature in
1965 (33). Although it was
originally noted for its anesthetic effects, ketamine has numerous
pharmacological properties, including the capacity to block amine
uptake in the descending inhibitory monoaminergic pain pathways,
interactions with muscarinic receptors, local anesthetic action and
the ability to act as a kappa opioid agonist (34). Previous studies have reported that
ketamine administered via continuous intravenous infusion enhances
the analgesic effect of morphine even after the effect of ketamine
had ceased in obstetric patients, while also being safe for both
mother and baby and not affecting breastfeeding (35,36).
Intravenous administration of ketamine has been
reported to effectively prevent and treat shivering in patients
undergoing cesarean section. However, there has been little
research conducted to establish whether ketamine administered
epidurally may produce the same effect. The present study was
designed to determine the effectiveness of epidural ketamine for
the prevention of shivering in patients undergoing caesarean
delivery with CSE. The study was designed to standardize the
possible confounding factors while reflecting normal practice in
our institution. To facilitate this, the temperature of the
operating room was maintained at 22–24°C and the intravenous and
irrigation solutions were also maintained at 37°C during
surgery.
Prophylactic ketamine was administered epidurally
with the aim of reducing postoperative shivering. Following spinal
anesthesia, the incidence and severity of shivering in group K was
significantly reduced compared with group S. Small fluctuations in
MAP were observed, with MAP remaining more stable in group S
compared with group K. Ketamine causes sympathetic stimulation and
vasoconstriction (37) and may
ameliorate the drop in blood pressure caused by spinal anesthesia.
The incidence of nausea in group K was lower compared with group S,
possibly due to fluctuations in MAP, as hypotension may cause brain
stem hypoperfusion, thus triggering nausea and vomiting (38).
Ketamine may decrease the core-to-peripheral
redistribution of heat by direct central sympathetic stimulation
and inhibition of norepinephrine uptake into the postganglionic
sympathetic nerve endings (12).
However, the exact mechanism of ketamine remains unclear. Low-dose
ketamine, as previously defined in the literature (37), was used in the present study as this
dose has previously been used effectively to treat post-operative
shivering (39). The results of the
present study demonstrated that low-dose ketamine reduced shivering
as well as the incidence of adverse effects. No hallucinations or
delirium, which are well-known side effects of ketamine (37), were observed in any of the patient.
Furthermore, none of the patients in group K had sedation scores
>3 or reported amnesia. However, sedation is actually beneficial
during surgery under spinal anesthesia, as it may reduce anxiety
and improve patient comfort and cooperation (40).
In summary, the results of the present study suggest
that prophylactic epidural administration of low-dose ketamine is
able to reduce the incidence and severity of shivering in patients
undergoing caesarean section with CSE. However, the present study
focused on the occurrence and prevention of intraoperative
shivering and only investigated single-dose ketamine. Future
studies should investigate the potential of ketamine to reduce
postoperative shivering in the future and seek to elucidate the
optimal dose.
Acknowledgements
Not applicable.
Funding
This study was supported by the Natural Science
Foundation of Gansu Province of China (grant no. 145RJZA171).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XX, YLv and YLe conceived and designed the study.
YoZ and YaZ performed the majority of the experimental procedures.
YLv analyzed the data. XX wrote the paper. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Gansu Provincial Maternity and Child-Care Hospital
(Lanzhou, China) and obtained informed consent from all
participants.
Consent for publication
Not applicable.
Competing interests
All authors declare that they have no competing
interests.
References
|
1
|
Salman C, Kayacan N, Ertuğrul F, Bıgat Z
and Karslı B: Combined spinal-epidural anesthesia with epidural
volume extension causes a higher level of block than single-shot
spinal anesthesia. Braz J Anesthesiol. 63:267–272. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Crossley AWA, Mcvey FK and Cartwright DP:
Perioperative shivering. Lancet. 338:10261991. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shakya S, Chaturvedi A and Sah BP:
Prophylactic low dose ketamine and ondansetron for prevention of
shivering during spinal anaesthesia. J Anaesthesiol Clin Pharmacol.
26:465–469. 2010.PubMed/NCBI
|
|
4
|
Sessler DI: Temperature monitoring and
perioperative thermoregulation. Anesthesiology. 109:318–338. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Badawy AA and Mokhtar AM: The role of
ondansetron in prevention of post-spinal shivering (PSS) in
obstetric patients: A double-blind randomized controlled trial.
Egypt J Anaesth. 33:29–33. 2017. View Article : Google Scholar
|
|
6
|
Kose EA, Dal D, Akinci SB, Saricaoglu F
and Aypar U: The efficacy of ketamine for the treatment of
postoperative shivering. Anesth Analg. 106:120–122. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zahra FA, Abudallah HM, Shabana RI,
Abdulmageed WM, Abdulrazik SI and Nassar AM: Intramuscular ketamine
for prevention of postanesthesia shivering in children. Saudi Med
J. 29:1255–1259. 2008.PubMed/NCBI
|
|
8
|
Crowley LJ and Buggy DJ: Shivering and
neuraxial anesthesia. Reg Anesth Pain Med. 33:241–252. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kranke P, Eberhart LH, Roewer N and Tramèr
MR: Pharmacological treatment of postoperative shivering: A
quantitative systematic review of randomized controlled trials.
Anesth Analg. 94:453–460. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
de Figueiredo Locks G: Incidence of
shivering after cesarean section under spinal anesthesia with or
without intrathecal sufentanil: A randomized study. Rev Bras
Anestesiol. 62:676–684. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tsai YC and Chu KS: A comparison of
tramadol, amitriptyline, and meperidine for postepidural anesthetic
shivering in parturients. Anesth Analg. 93:1288–1292. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dal D, Kose A, Honca M, Akinci SB, Basgul
E and Aypar U: Efficacy of prophylactic ketamine in preventing
postoperative shivering. Br J Anaesth. 95:189–192. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kose EA, Honca M, Dal D, Akinci SB and
Aypar U: Prophylactic ketamine to prevent shivering in parturients
undergoing Cesarean delivery during spinal anesthesia. J Clin
Anesth. 25:275–280. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Crossley AWA and Mahajan RP: The intensity
of postoperative shivering is unrelated to axillary temperature.
Anaesthesia. 49:205–207. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang W, Li Y, Sun A, Yu H, Dong J and Xu
H: Determination of the median effective dose (ED50) of bupivacaine
and ropivacaine unilateral spinal anesthesia: Prospective, double
blinded, randomized dose-response trial. Anaesthesist. 66:936–943.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Finster M and Wood M: The Apgar score has
survived the test of time. Anesthesiology. 102:855–857. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wilson E, David A, MacKenzie N and Grant
IS: Sedation during spinal anaesthesia: Comparison of propofol and
midazolam. Br J Anaesth. 64:48–52. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Roy JD, Girard M and Drolet P: Intrathecal
meperidine decreases shivering during cesarean delivery under
spinal anesthesia. Anesth Analg. 98:230–234. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sessler DI: Complications and treatment of
mild hypothermia. Anesthesiology. 95:531–543. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rajek A, Greif R and Sessler DI: Effects
of epidural anesthesia on thermal sensation. Reg Anesth Pain Med.
26:527–531. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Giuliano KK and Hendricks J: Inadvertent
perioperative hypothermia: Current nursing knowledge. AORN J.
105:453–463. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhou P, Ge S, Wang Y, Xiong W, Wang T and
Xue Z: Novamin infusion: A new method to cure postoperative
shivering with hypothermia. J Surg Res. 188:69–76. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Webb PJ, James FM III and Wheeler AS:
Shivering during epidural analgesia in women in labor.
Anesthesiology. 55:706–707. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kranke P, Eberhart LH, Roewer N and Tramèr
MR: Single-dose parenteral pharmacological interventions for the
prevention of postoperative shivering: A quantitative systematic
review of randomized controlled trials. Anesth Analg. 99:718–727.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Horn EP, Sessler DI, Standl T, Schroeder
F, Bartz HJ, Beyer JC and Esch Schulte am J: Non-thermoregulatory
shivering in patients recovering from isoflurane or desflurane
anesthesia. Anesthesiology. 89:878–886. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kim G, Kim MH, Lee SM, Choi SJ, Shin YH
and Jeong HJ: Effect of pre-warmed intravenous fluids on
perioperative hypothermia and shivering after ambulatory surgery
under monitored anesthesia care. J Anesth. 28:880–885. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Golembiewski J: Pharmacological management
of perioperative shivering. J Perianesth Nurs. 30:357–359. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gozdemir M, Usta B, Demircioglu RI, Muslu
B, Sert H and Karatas OF: Magnesium sulfate infusion prevents
shivering during transurethral prostatectomy with spinal
anesthesia: A randomized, double-blinded, controlled study. J Clin
Anesth. 22:184–189. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Shukla U, Malhotra K and Prabhakar T: A
comparative study of the effect of clonidine and tramadol on
post-spinal anaesthesia shivering. Indian J Anaesth. 55:242–246.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Pawar MS, Suri N, Kaul N, Lad S and Khan
RM: Hydrocortisone reduces postoperative shivering following day
care knee arthroscopy. Can J Anaesth. 58:924–928. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Haque MF, Rashid MH, Rahaman MS and Islam
MR: Comparison between tramadol hydrochloride & nalbuphine
hydrochloride in the treatment of per-operative shivering after
spinal anaesthesia. Mymensingh Med J. 20:201–205. 2011.PubMed/NCBI
|
|
32
|
Tauzin-Fin P, Maurette P, Vincon G,
Hecquet D, Houdek MC and Bonnet F: Clinical and pharmacokinetic
aspects of the combination of meperidine and prilocaine for spinal
anaesthesia. Can J Anaesth. 39:655–660. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Domino EF, Chodoff P and Corssen G:
Pharmacologic effects of CI-581, a new dissociative anesthetic, in
man. Clin Pharmacol Ther. 6:279–291. 1965. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Aroni F, Iacovidou N, Dontas I, Pourzitaki
C and Xanthos T: Pharmacological aspects and potential new clinical
applications of ketamine: Reevaluation of an old drug. J Clin
Pharmacol. 49:957–964. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Reza FM, Zahra F, Esmaeel F and Hossein A:
Preemptive analgesic effect of ketamine in patients undergoing
elective cesarean section. Clin J Pain. 26:223–226. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang X, Xie H and Wang G: Improved
postoperative analgesia with coadministration of preoperative
epidural ketamine and midazolam. J Clin Anesth. 18:563–569. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
White PF, Way WL and Trevor AJ:
Ketamine-its pharmacology and therapeutic uses. Anesthesiology.
56:119–136. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Shahriari A, Khooshideh M and Heidari MH:
Prevention of nausea and vomiting in caesarean section under spinal
anaesthesia with midazolam or metoclopramide? J Pak Med Assoc.
59:756–759. 2009.PubMed/NCBI
|
|
39
|
Sharma DR and Thakur JR: Ketamine and
shivering. Anaesthesia. 45:252–253. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Hu P, Harmon D and Frizelle H: Patient
comfort during regional anesthesia. J Clin Anesth. 19:67–74. 2007.
View Article : Google Scholar : PubMed/NCBI
|