Introduction
Hot flushes, night sweats, pain during sexual
intercourse, hair loss, forgetfulness, depression and difficulties
in initiating and/or maintaining sleep are common problems in
menopausal women, for which there are several different treatment
approaches that been found to be efficient and may be used
according to the patient's individual preferences.
Breast cancer patients are more likely to suffer
from these problems, as chemotherapeutic treatments may induce
temporary or permanent ovarian failure in premenopausal patients.
Furthermore, antihormonal treatment with gonadotropin-releasing
hormone (GnRH) analogues, tamoxifen, aromatase inhibitors and
aromatase inactivators enhances these complaints, occasionally to
such an extent that the patient discontinues the medications. The
Arimidex, Tamoxifen, Alone or in Combination (ATAC) trial reported
discontinuation rates of 14.3% for tamoxifen and 11.1% for
anastrozole (1). The options for
relieving the patients' complaints are limited, as it is generally
accepted that they should not receive oestrogen and related
substances in order to avoid disease recurrence or progression.
Various substances and methods of managing these
conditions have been suggested to alleviate the side effects
(2,3),
originating from conventional medicine as well as from
complementary and alternative medicine. The approaches of
conventional medicine include gabapentin, fluoxetine, venlafaxine
and clonidine (4,5). However, the adverse effects often limit
the general use of these drugs, whereas weight loss, behavioural
therapy and exercise appear to be a promising alternative way of
reducing hot flushes (6,7).
As none of the aforementioned approaches has
convincingly exhibited sufficient efficacy, continued research on
the topic is mandatory. Complementary and alternative medical
practices suggest administration of vitamin E, soy, black cohosh
and red clover. The use of phytooestrogens, however, is not
generally accepted, due to possible stimulation of breast cancer
cells. Another approach is acupuncture, which has been shown to
improve symptoms in several trials, possibly via a calcitonin
gene-related peptide (8–11). However, the fact that acupuncture
leads to an elevation of oestradiol levels and a decrease in the
levels of follicle-stimulating hormone (FSH) and luteinizing
hormone (LH) raises concerns regarding its safety in breast cancer
patients (12). A recent
meta-analysis concluded that soy food intake may be associated with
prolonged survival, particularly in ER−,
ER+/PR+ and postmenopausal patients (13). These finding and additional research,
which demonstrated that oestrogen may induce apoptosis in oestrogen
deprivation-resistant breast cancer, clearly suggest that the
present concepts of antihormonal treatment must be reconsidered
(14). Long-term studies assessing
the benefits and risks of methods from complementary medicine are
currently lacking. Such studies may be of value, as it has been
shown that hot flushes are a strong predictor of
breast-cancer-specific outcome, stronger than age, hormone receptor
status, or even differences in cancer stage at diagnosis (15).
It was recently demonstrated that pollen mixtures
and extracts, as well as bee pollen, may alleviate menopausal
symptoms (16–19). This effect was found to be independent
of hormonal effects (20–22). Pollen was even found to exert
antihormonal effects (23).
A clinical trial was initiated in order to
investigate whether bee pollen is efficient in alleviating
menopausal symptoms in patients under treatment with tamoxifen and
aromatase inhibitors/inactivators.
Patients and methods
Patient recruitment
We conducted a randomised controlled study comparing
a mixture of pollen and honey or pure honey for the alleviation of
menopausal symptoms. The criteria for inclusion were as follows:
Age >18 years, successful surgery without residual tumour, life
expectancy of >12 weeks, initiation of antihormonal treatment at
least 3 months prior to the study, severe menopausal complaints and
the ability to read and write in German. The criteria for exclusion
were distant metastases, pregnancy or no contraception, allergy to
pollen and/or honey, serious concomitant diseases, psychiatric
disease, or conventional concomitant treatment for menopausal
complaints.
In October 2012, we published a contact request in
the local newspapers for breast cancer patients currently under
treatment with tamoxifen and aromatase inhibitors/inactivators with
severe menopausal complaints. We also contacted patients in our
outpatient department who fulfilled the inclusion criteria to
inquire whether they were willing to participate in the study.
Patient randomisation
Following written informed consent, the patients
were randomised to receive either a mixture of pollen and honey
(pollen group) or pure honey (honey group). As there are no
available studies on the effect of honey on menopausal symptoms in
humans, the treatment with honey was considered to be the placebo.
The patients were instructed to ingest a tablespoonful of either
substance for the next 14 days and then return for the second
assessment and a blood sample. After a free interval of at least
another 14 days, crossover to the other study arm was initiated and
the participants received the other alternative for a further 14
days. After that period, the patients were invited for reassessment
(crossover design).
Stratification by treatment
Due to potential differences in the effects,
depending on whether the patients had received tamoxifen or
aromatase inhibitors/inactivators, we stratified for these drugs,
including at least 15 patients on either treatment. The time period
of 14 days was selected in order to be able to assess the efficacy
of either intervention and to avoid exposing patients to a possible
risk due to unforeseeable interactions.
The pollen and honey used in the study were obtained
from Mehler Honig Gmbh (Neichen, Germany). The pollen analysis
performed at the Institute for Honey Analyses (Quality Services
International GmbH, Bremen, Germany) revealed that the honey was
sunflower honey.
Assessment of menopausal complaints. The menopausal
complaints were assessed using the Menopause Rating Scale (MRS), as
introduced by Schneider et al (24,25) and
Heinemann et al (26). This
instrument comprises three subscales that measure psychological,
somato-vegetative and urogenital complaints. We also assessed
demographic data and collected blood samples for measuring
triglycerides, high-density lipoprotein (HDL), low-density
lipoprotein (LDL) and oestradiol. The background for the analyses
of blood lipids is the fact that hormonal treatments may modify
blood lipid levels, affecting cardiovascular disease risks, which
appear to be higher in breast cancer patients (27,28).
The study protocol was approved by the Ethics
Committee of the Justus Liebig University of Giessen, Germany
(application no. 103/12).
Results
Patient characteristics
A total of 46 patients were recruited for this
study. The patient characteristics are summarized in Table I. A total of 31 patients (67.4%;
31/46) completed both phases of the study, whereas 15 patients did
not complete the second part of the study. Of the patients treated
with honey, 5 discontinued the study after having completed the
pollen part and, of the patients treated with pollen, 10
discontinued after having completed the pure honey part, the main
reason being distaste for the pollen. On a 10-point taste scale,
from 1 (very good) to 10 (very bad), honey received an average
score of 2.3, whereas the pollen preparation received an average
score of 6.0. There were some minor adverse effects, which are
summarised in Table II.
 | Table I.Characteristics of breast cancer
patients (n=46). |
Table I.
Characteristics of breast cancer
patients (n=46).
| Characteristics | Values |
|---|
| Age at study
initiation (years) |
|
| Mean
(median; SD) | 60.8 (62.3;
11.2) |
| Age at diagnosis
(years) |
|
| Mean
(median; SD) | 57.4 (58.3;
10.8) |
| Time since last
check-upa (months) |
|
| Mean
(median; SD) | 6.0 (5.1; 3.4) |
| Duration of hormone
therapy (months) |
|
| Mean
(median; SD) | 32.5 (25.5;
28.3) |
| Body mass index |
|
| Mean
(median; SD) | 26.9 (26.5; 4.5) |
| Karnofsky performance
status scale |
|
| Mean
(median; SD) | 91.7 (90.0; 8.2) |
| Earlier treatment,
no. (%) |
|
|
Surgery | 46 (100.0) |
|
Radiotherapy | 39 (84.8) |
|
Chemotherapy | 14 (30.4) |
| Antihormonal
treatment, no. (%) |
|
|
Tamoxifen | 24 (52.1) |
|
Anastrozole/Letrozole | 17 (37.0) |
|
Exemestan | 4 (8.7) |
|
Fulvestrant | 1 (2.2) |
 | Table II.Incidence of minor complaints during
the study. |
Table II.
Incidence of minor complaints during
the study.
| Complaints,
no. | Honey (n=41) | Pollen (n=36) |
|---|
| Exhaustion | 4 | 2 |
| Increased
aggressiveness | 2 | 2 |
| Hot flush on
ingestion | 2 | 1 |
| Prickling of lips
and mouth | – | 3 |
| Oedema of lower
extremities | 2 | – |
| Thin hair | 1 | 1 |
| Mouth odour | 1 | 1 |
| Upper abdominal
pain | 1 | 1 |
| Arthralgia | 1 | 1 |
| Flatulence | 1 | 1 |
| Disgust when
ingesting the product | 1 | – |
| Nausea | 1 | – |
| Vomiting | 1 | – |
| Heartburn | 1 | – |
| Malaise | 1 | – |
| Headache | 1 | – |
| Red eyes | 1 | – |
| Stronger hot
flushes | 1 | – |
| Dizziness | – | 1 |
| Itching of face and
extremities | – | 1 |
Other treatments
All the patients were assessed on their familiarity
with other treatment options for hot flushes and related symptoms.
A total of 9 patients had previous experience with such treatments
and, on average, they had already tried >2. However, the
majority of the patients (37/46; 80.4%) had never used any such
methods. Overall, the efficacy of the previously tested methods was
rated as poor; however, valid analyses were not possible owing to
the low number of patients who had attempted such treatments
(Table III).
 | Table III.Previous experience with treatments
against the side effects of antihormonal treatment (n=9). |
Table III.
Previous experience with treatments
against the side effects of antihormonal treatment (n=9).
| Methods | Patients with
experience of method, no. (%) | Average rating of
method's efficacya |
|---|
| Acupuncture | 3 (6.5) | 3.7 |
| Homeopathy | 7 (15.2) | 4.0 |
| Vitamin E | 3 (6.5) | 2.3 |
| Agnus
castus | 0 (0.0) | – |
| Soy | 2 (4.3) | 4.5 |
| Omega-6 fatty
acids | 2 (4.3) | 4.0 |
| Red clover | 0 (0.0) | – |
| Traditional Chinese
medicine | 1 (2.2) | 2.0 |
| Relaxation
techniques | 3 (6.5) | 2.5 |
| Clonidin | 0 (0.0) | – |
| Venlafaxin | 1 (2.2) | 3 |
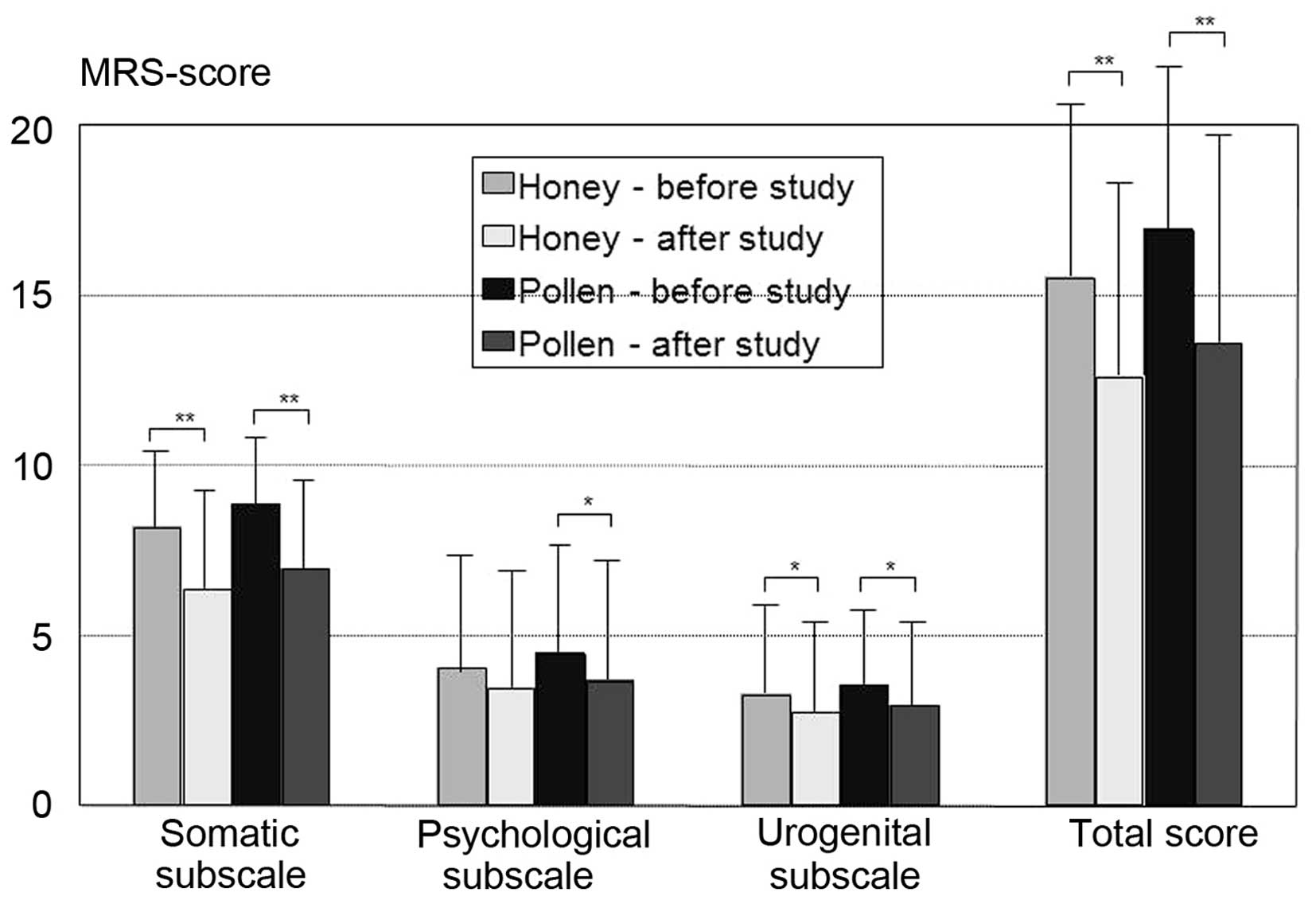
Comparison of honey and pollen
groups
The comparison of all the patients receiving pollen
or honey revealed an improvement in the postmenopausal complaints
in both groups in a pre-post analysis using MRS and its three
subscales, with no significant differences between the two groups
(Fig. 1). Improvements were reported
by 68.3% (28/41) of the patients receiving honey, compared with
70.9% (22/31) of those receiving pollen (the difference was not
significant). If improvements occurred, they were observed 3–4 days
after the initiation of the study, regardless of whether honey or
pollen was administered or whether the patients received tamoxifen
or an aromatase inhibitor/inactivator as antihormonal medication.
Patients taking tamoxifen exhibited a trend towards higher rates of
improvement for pollen (86.7 vs. 58.8%; P=0.087).
A pre-post analysis did not reveal any significant
differences in the serum levels of cholesterol (total, HDL, or LDL
cholesterol), triglycerides or oestradiol.
Patients receiving honey reported a significant
improvement in the symptoms associated with the antihormonal
medication. In patients taking aromatase inhibitors/inactivators,
honey significantly improved symptoms according to the MRS,
compared with patients treated with tamoxifen (FLevene
test = 5.75; P=0.022). The improvements were particularly
associated with the urogenital subscale (FLevene test =
5.36; P=0.027) and there was also a trend in the somatic subscale
(FLevene test = 3.85; P=0.058). However, we also
detected a trend towards an increase in oestradiol levels by honey
in patients receiving aromatase inhibitors (FLevene test
=3.79; P=0.060). Pollen did not exert a significantly different
effect on the two types of antihormonal medication.
Effect of oophorectomy on the
results
Finally, we investigated whether the presence of the
ovaries exerted any effect on the results and observed that
patients who had not undergone oophorectomy benefited more from
honey or pollen treatment (present vs. absent ovaries, 73.1 vs.
33.3%, respectively; P=0.063). However, this result did not reach
the level of significance, as the number of patients who had
undergone oophorectomy was very limited.
Discussion
In this study, we were able to demonstrate that both
honey and the pollen-honey mixture improved menopausal complaints
in breast cancer patients. Over two-thirds of the patients who
completed the study reported an improvement in their symptoms.
Honey was found to be very effective in patients receiving
aromatase inhibitors; however, the increase in oestradiol levels
may raise some concerns regarding the safety of honey in this
setting. Such concerns are not the case with the honey-pollen
mixture. It appears that ovaries may play an important role
regarding the efficacy of honey as well as that of pollen. It was
also demonstrated that neither honey nor pollen exerted a
significant effect on serum cholesterol and lipid levels.
Previous studies on the effects of pollen on
symptoms in menopausal women formed the basis of this trial
(16–19). The mixture of pollen and honey was
used as i) it has been demonstrated that a mixture of honey and
pollen increases the efficacy of pollen (29) and ii) honey improves the taste of
pollen. We demonstrated that the pollen-honey mixture improved
menopausal symptoms even in breast cancer patients undergoing
antihormonal treatment. Certain patients also reported additional
favourable effects, including reduced hair loss (1 case, honey
group), improvement in bowel movement (1 case, honey group; and 1
case, pollen group) and normalised blood pressure (1 case, honey
group).
Of note, honey, which was originally used as placebo
and which contains only minimal amounts of pollen, produced effects
similar to those of pollen. The extent of the effect observed in
this study clearly exceeds that of a placebo effect, which is
believed to be ~25% (30). This may
be related to the findings of animal studies, according to which
Tualang honey prevented uterine atrophy and increased bone density
in ovariectomised rats and was thus considered a possible
alternative to hormone replacement therapy (31). However, the positive findings for
Tualang honey were achieved only with significantly higher doses
(1.0 and 2.0 g/kg body weight) (31).
Unfortunately, the increase in the serum levels of
oestradiol with honey treatment raises some concerns regarding the
safety of honey in patients receiving aromatase
inhibitors/inactivators. This finding is of clinical relevance as
honey is considered to be a healthy food, which is more frequently
consumed by people of higher income and older age (≥60 years),
particularly women (32). Thus, we
may hypothesize that several breast cancer patients on aromatase
inhibitors/inactivators consume honey without being aware of this
potential risk.
The finding that the presence of ovaries may play an
important role in treatment success is supported by an animal
study, which demonstrated that the addition of bee pollen to the
diet reduced the release of insulin-like growth factor-1 (IGF-1),
progesterone and oestradiol from the ovaries (33). This information was not available when
our study was initiated. However, there may also be direct
oestrogen-like effects, as other experiments demonstrated that bee
pollen decreased bone loss, even when the experimental rats were
ovariectomised (34).
A limitation of our study was the high drop-out
rate, particularly in the honey-pollen mixture group. However, this
rate was not as high as that in a comparable study on
rhinoconjunctivitis (35). The
unusual taste of pure pollen, which may be ameliorated only to some
extent by the addition of honey, may limit its potential uses.
Another problem is that there have been no chemical analyses of
honey and pollen. However, it is well known that honey and,
particularly, pollen, contain flavonoids, which are known to reduce
the risk of breast cancer (36,37).
A significant finding of our study was the absence
of any effect of pollen on serum cholesterol and triglycerides,
which was demonstrated in an earlier study by Georgiev et al
(17). This may be due to the fact
that the majority of the patients included in this study had normal
blodd lipid levels at the beginning of the study. In accordance
with earlier studies, no such effect was expected in the honey
group (38,39).
In conclusion, this study provided evidence that
honey and bee pollen improve menopausal symptoms in breast cancer
patients receiving antihormonal treatment. As we observed an
increase in the serum levels of oestradiol with honey treatment in
patients receiving aromatase inhibitors/inactivators, and due to
evidence regarding the effect of honey and bee pollen on ovarian
function and the direct effects of these products, honey and bee
pollen should be used with caution in cancer patients. Whether this
caution is justified remains to be established. As previously
mentioned, the scepticism regarding soy products also does not
appear to be justified, according to Chi et al (13). Honey and bee pollen may be offered to
women who have failed to respond to other reasonable alternatives
to cope with postmenopausal symptoms (e.g., acupuncture) and who
would otherwise discontinue treatment. However, the fact that
flavonoids, which are found in both honey and pollen, have been
found to prevent breast cancer, supports the use of both products
in women with menopausal problems without a history of breast
cancer. The use of honey and pollen for menopausal complaints in
healthy women and patients with breast cancer should be addressed
in future trials.
Acknowledgements
We would like to thank the Dr Abel foundation for
supporting this trial and all the patients for their
participation.
Abbreviations:
|
GnRH
|
gonadotropin-releasing hormone
|
|
FSH
|
follicle-stimulating hormone
|
|
LH
|
luteinizing hormone
|
|
MRS
|
Menopause Rating Scale
|
|
HDL
|
high-density lipoprotein
|
|
LDL
|
low-density lipoprotein
|
|
IGF
|
insulin-like growth factor
|
References
|
1
|
Howell A, Cuzick J, Baum M, Buzdar A,
Dowsett M, Forbes JF, Hoctin-Boes G, Houghton J, Locker GY and
Tobias JS: ATAC Trialists' Group: Results of the ATAC (Arimidex,
Tamoxifen, Alone or in Combination) trial after completion of 5
years' adjuvant treatment for breast cancer. Lancet. 365:60–62.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Loibl S, Lintermans A, Dieudonné AS and
Neven P: Management of menopausal symptoms in breast cancer
patients. Maturitas. 68:148–154. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kligman L and Younus J: Management of hot
flashes in women with breast cancer. Curr Oncol. 17:81–86.
2010.PubMed/NCBI
|
|
4
|
Lavigne JE, Heckler C, Mathews JL, Palesh
O, Kirshner JJ, Lord R, Jacobs A, Amos E, Morrow GR and Mustian K:
A randomized, controlled, double-blinded clinical trial of
gabapentin 300 versus 900 mg versus placebo for anxiety symptoms in
breast cancer survivors. Breast Cancer Res Treat. 136:479–486.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Boekhout AH, Vincent AD, Dalesio OB, van
den Bosch J, Foekema-Töns JH, Adriaansz S, Sprangers S, Nuijen B,
Beijnen JH and Schellens JH: Management of hot flashes in patients
who have breast cancer with venlafaxine and clonidine: A
randomized, double-blind, placebo-controlled trial. J Clin Oncol.
29:3862–3868. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Duijts SF, van Beurden M, Oldenburg HS, et
al: Efficacy of cognitive behavioral therapy and physical exercise
in alleviating treatment-induced menopausal symptoms in patients
with breast cancer: Results of a randomized, controlled,
multicenter trial. J Clin Oncol. 30:4124–4133. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Caan BJ, Emond JA, Su HI, Patterson RE,
Flatt SW, Gold EB, Newman VA, Rock CL, Thomson CA and Pierce JP:
Effect of postdiagnosis weight change on hot flash status among
early-stage breast cancer survivors. J Clin Oncol. 30:1492–1497.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Frisk J, Källström AC, Wall N, Fredrikson
M and Hammar M: Acupuncture improves health-related quality-of-life
(HRQoL) and sleep in women with breast cancer and hot flushes.
Support Care Cancer. 20:715–724. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
de Valois BA, Young TE, Robinson N,
McCourt C and Maher EJ: Using traditional acupuncture for breast
cancer-related hot flashes and night sweats. J Altern Complement
Med. 16:1047–1057. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Walker EM, Rodriguez AI, Kohn B, Ball RM,
Pegg J, Pocock JR, Nunez R, Peterson E, Jakary S and Levine RA:
Acupuncture versus venlafaxine for the management of vasomotor
symptoms in patients with hormone receptor-positive breast cancer:
A randomized controlled trial. J Clin Oncol. 28:634–640. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Spetz Holm AC, Frisk J and Hammar ML:
Acupuncture as treatment of hot flashes and the possible role of
calcitonin gene-related peptide. Evid Based Complement Alternat
Med. 2012:5793212012.PubMed/NCBI
|
|
12
|
Shen X, Du Y, Yan L, Xia Y, Yan H, Han G,
Guo Y and Shi X: Acupuncture for treatment of climacteric syndrome
- a report of 35 cases. J Tradit Chin Med. 25:3–6. 2005.PubMed/NCBI
|
|
13
|
Chi F, Wu R, Zeng YC, Xing R, Liu Y and Xu
ZG: Post-diagnosis soy food intake and breast cancer survival: A
meta-analysis of cohort studies. Asian Pac J Cancer Prev.
14:2407–2412. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ariazi EA, Cunliffe HE, Lewis-Wambi JS, et
al: Estrogen induces apoptosis in estrogen deprivation-resistant
breast cancer through stress responses as identified by global gene
expression across time. In: Proc Natl Acad Sci USA. 108. pp.
18879–18886. 2011; View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mortimer JE, Flatt SW, Parker BA, Gold EB,
Wasserman L, Natarajan L and Pierce JP: WHEL Study Group:
Tamoxifen, hot flashes and recurrence in breast cancer. Breast
Cancer Res Treat. 108:421–426. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Winther K, Rein E and Hedman C: Femal, a
herbal remedy made from pollen extracts, reduces hot flushes and
improves quality of life in menopausal women: A randomized,
placebo-controlled, parallel study. Climacteric. 8:162–170. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Georgiev DB, Metka M, Huber JC, Goudev AR
and Manassiev N: Effects of an herbal medication containing bee
products on menopausal symptoms and cardiovascular risk markers:
Results of a pilot open-uncontrolled trial. MedGenMed.
6:462004.
|
|
18
|
Szanto E, Gruber D, Sator M, Knogler W and
Huber JC: Plazebokontrollierte Untersuchung von Melbrosia zur
Behandlung klimakterischer Beschwerden. Wien Med Wochenschr.
144:130–133. 1994.(In German). PubMed/NCBI
|
|
19
|
Elia D and Mares P: Assessment of the
tolerance and effectiveness of a food supplement Sérélys (Femal)
for menopausal women. Genesis. 135:12–15. 2008.
|
|
20
|
James LF, Foote W, Nye W and Hartley WJ:
Effects of feeding Oxytropis and Astragalus pollen to mice and
Astragalus seeds to rats. Am J Vet Res. 39:711–712. 1978.PubMed/NCBI
|
|
21
|
Einer-Jensen N, Zhao J, Andersen KP and
Kristoffersen K: Cimicifuga and Melbrosia lack oestrogenic effects
in mice and rats. Maturitas. 25:149–153. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hellström AC and Muntzing J: The pollen
extract Femal - a nonestrogenic alternative to hormone therapy in
women with menopausal symptoms. Menopause. 19:825–829. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sarić A, Balog T, Sobocanec S, et al:
Antioxidant effects of flavonoid from Croatian Cystus incanus L.
rich bee pollen. Food Chem Toxicol. 47:547–554. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Schneider HP, Heinemann LA, Rosemeier HP,
Potthoff P and Behre HM: The Menopause Rating Scale (MRS):
Reliability of scores of menopausal complaints. Climacteric.
3:59–64. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Schneider HP, Heinemann LA, Rosemeier HP,
Potthoff P and Behre HM: The Menopause Rating Scale (MRS):
Comparison with Kupperman index and quality-of-life scale SF-36.
Climacteric. 3:50–58. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Heinemann LA, Potthoff P and Schneider HP:
International versions of the Menopause Rating Scale (MRS). Health
Qual Life Outcomes. 1:282003. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chi F, Wu R, Zeng Y, Xing R, Liu Y and Xu
Z: Effects of toremifene versus tamoxifen on breast cancer
patients: A meta-analysis. Breast Cancer. 20:111–122. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
de Sousa-e-Silva EP, Conde DM, Costa-Paiva
L, Martinez EZ, Pinto-Neto AM and Sousa-E-Silva EP: Cardiovascular
risk in middle-aged breast cancer survivors: A comparison between
two risk models. Rev Bras Ginecol Obstet. 36:157–162. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Küpeli Akkol E, Orhan DD, Gürbüz I and
Yesilada E: In vivo activity assessment of a ‘honey-bee pollen mix’
formulation. Pharm Biol. 48:253–259. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sloan JA, Loprinzi CL, Novotny PJ, Barton
DL, Lavasseur BI and Windschitl H: Methodologic lessons learned
from hot flash studies. J Clin Oncol. 19:4280–4290. 2001.PubMed/NCBI
|
|
31
|
Zaid SS, Sulaiman SA, Sirajudeen KN and
Othman NH: The effects of Tualang honey on female reproductive
organs, tibia bone and hormonal profile in ovariectomised rats -
animal model for menopause. BMC Complement Altern Med. 10:822010.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Pocol CB: Modelling the honey consumption
behaviour in Romania by using socio-demographic determinants. Afr J
Agric Res. 6:4069–4080. 2011.
|
|
33
|
Kolesarova A, Bakova Z, Capcarova M, Galik
B, Juracek M, Simko M, Toman R and Sirotkin AV: Consumption of bee
pollen affects rat ovarian functions. J Anim Physiol Anim Nutr
(Berl). 97:1059–1065. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kafadar IH, Güney A, Türk CY, Oner M and
Silici S: Royal jelly and bee pollen decrease bone loss due to
osteoporosis in an oophorectomized rat model. Eklem Hastalik
Cerrahisi. 23:100–105. 2012.PubMed/NCBI
|
|
35
|
Rajan TV, Tennen H, Lindquist RL, Cohen L
and Clive J: Effect of ingestion of honey on symptoms of
rhinoconjunctivitis. Ann Allergy Asthma Immunol. 88:198–203. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Feás X, Vázquez-Tato MP, Estevinho L,
Seijas JA and Iglesias A: Organic bee pollen: Botanical origin,
nutritional value, bioactive compounds, antioxidant activity and
microbiological quality. Molecules. 17:8359–8377. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hui C, Qi X, Qianyong Z, Xiaoli P, Jundong
Z and Mantian M: Flavonoids, flavonoid subclasses and breast cancer
risk: A meta-analysis of epidemiologic studies. PLoS One.
8:e543182013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Münstedt K, Hoffmann S, Hauenschild A,
Bülte M, von Georgi R and Hackethal A: Effect of honey on serum
cholesterol and lipid values. J Med Food. 12:624–628. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yaghoobi N, Al-Waili N, Ghayour-Mobarhan
M, et al: Natural honey and cardiovascular risk factors; effects on
blood glucose, cholesterol, triacylglycerole, CRP, and body weight
compared with sucrose. ScientificWorldJournal. 8:463–469. 2008.
View Article : Google Scholar : PubMed/NCBI
|