Introduction
Pelvic lymph node dissection (PLND) at the time of
radical prostatectomy (RP) is currently the most reliable method
for detecting lymph node (LN) metastases in patients with prostate
cancer (1–4). Conventionally, only the obturator LNs
are included in LND for prostate cancer; such a narrow dissection
range may be associated with the low number of node-positive cases.
Indeed, Miyake et al (5)
demonstrated that localized LND in RP was insufficient. Extended
LND (ELND) has been recommended, as it increases the node-positive
rate in localized prostate cancer. However, an increased risk of
complications associated with ELND has also been reported (6,7).
To verify whether the node-positive rate is
increased by widening the dissection range and whether ELND may be
safely performed on a routine basis, we conducted a comparative
study between a group of patients treated with the ELND proposed by
Bader et al (8), where the
obturator foramen LNs were excised along with the internal and
external iliac LNs, and another group treated with conventional
obturator LND alone.
Standard LND for prostate cancer is limited to
obturator LNs, although the internal and external LNs represent the
primary landing zone for prostatic lymphatic drainage. Heidenreich
et al (9) reported that,
considering their data, dissection of the presacral and common
iliac lymphatics does not appear to be necessary, since only 3 of
95 patients (3.1%) had LN metastasis in that region. Heidenreich
et al (9) and Zincke (10) have also demonstrated the high
diagnostic accuracy of pelvic staging LND limited to the internal
and external iliac and the obturator fossa LNs. We referred to LND
limited to the internal and external iliac and obturator fossa LNs
as ‘semi-extended’ LND and compared it to standard LND (obturator
fossa LNs alone).
An anatomically semi-extended PLND and a
conventional limited PLND were performed, in order to assess the
incidence of LN metastasis in cases of clinically localized
prostate cancer and evaluate the cases with metastases to the
LNs.
Patients and methods
Patient stratification by
treatment
We retrospectively queried our database for patients
who underwent open RP between 1988 and 2013 in Yamagata Prefectural
Central Hospital (Yamagata, Japan). Ethics approval was obtained
from the Institutional Review Board of the Yamagata Prefectural
Central Hospital (approval no. 2013-0003). Patients who received
neoadjuvant or immediate adjuvant therapy were included in the
study. A total of 730 consecutive patients underwent RP. The
patients were divided into two groups, the group undergoing
semi-extended PLND, comprising 6 selective fields, namely the
bilateral external iliac, internal iliac and obturator fields, and
the group undergoing standard LND (obturator LNs alone).
Patient stratification by risk
A total of 131 patients who underwent semi-extended
PLND were compared with 599 standard LND patients (Table I). The patients were stratified into
high-risk [prostate-specific antigen (PSA)>20 ng/ml, Gleason
score (GS)≥8], intermediate-risk (PSA 10–20 ng/ml, GS=4+3) and
low-risk (PSA<10 ng/ml, GS≤3+4) subgroups. The patients were
followed up for a median of 40 months (range, 1–261 months).
 | Table I.Number of positive lymph nodes (LNs)
stratified by risk group and extent of LN dissection (LND). |
Table I.
Number of positive lymph nodes (LNs)
stratified by risk group and extent of LN dissection (LND).
|
| LND type |
|---|
|
|
|
|---|
| Risk | Standard | Semi-extended |
|---|
| Low (%) | 0/253 (0.0) | 0/40 (0.0) |
| Intermediate (%) | 1/164 (0.6) | 1/30 (3.3) |
| Higha (%) | 13/182 (7.1) | 12/61
(19.6)b |
Results
Positive LN detection by treatment and
risk group
Following semi-extended LND, positive LNs were
detected in 12/61 (20%) of the high-risk, 1/30 (3%) of the
intermediate-risk and 0/40 (0%) of the low-risk cases. Standard LND
identified positive LNs in 13/182 (7%) of the high-risk, 1/164
(0.6%) of the intermediate-risk and 0/253 (0%) of the low-risk
cases. In the high-risk group, the detection rate of LN metastasis
was significantly higher following extended LND compared with that
following standard LND (Table I,
P<0.01).
In 9 of 13 patients (69%), metastases were
identified in the internal and external iliac regions, despite
negative obturator LNs. The range, mean and median number of
harvested LNs were 0–24, 6.7 and 6, respectively, with standard LND
and 0–34, 10.8 and 9, respectively, with semi-extended LND
(P<0.01). The PSA of patients with LN metastasis ranged between
3.3 and 252.7 ng/ml (mean, 49.7 and median, 38 ng/ml). All the
patients with a single obturator LN metastasis underwent standard
LND.
In the 2 intermediate-risk patients exhibiting LN
metastasis, the serum PSA level (GS) was 6.7 ng/ml (4+3) and 16.1
ng/ml (4+3), respectively.
Complications and outcome
There were no significant differences regarding
intraoperative and postoperative complications or blood loss
between the two groups. There was no lymphocele formation in
patients undergoing either standard or semi-extended LND. The
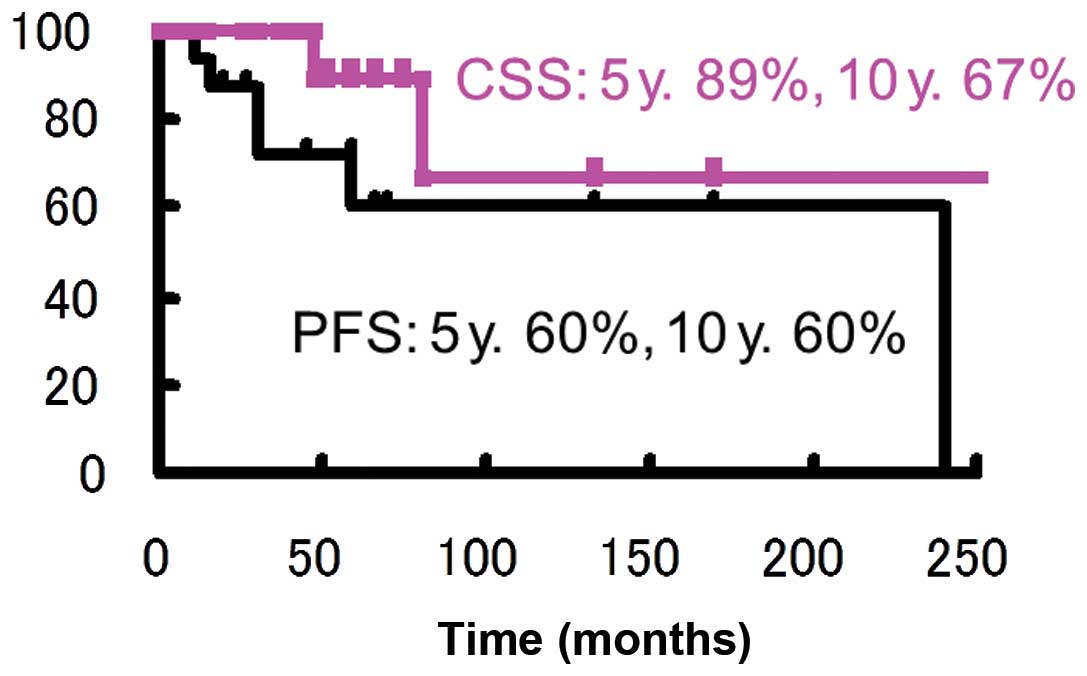
Kaplan-Meier curves for the oncological outcomes of
progression-free survival (PFS) and cause-specific survival (CSS)
are shown in Fig. 1.
Discussion
LND in RP is considered to be of diagnostic value;
however, its curative significance is limited. The detection rate
of LN metastasis may be increased by widening the dissection range,
although the extent of the dissection remains controversial. Clark
et al (11) performed a
prospective comparative study on extended and localized LND and
observed no difference in the node-positive rate; in addition, ELND
was associated with a higher complication rate. Kroepfl et
al (12) included obturator and
internal iliac LNs, as well as the LNs along the hypogastric vein,
in the range of ELND. Heidenreich et al (9) suggested it was unnecessary to dissect
the presacral and common iliac lymphatics, since only 3 of their 95
patients (3.1%) had LN metastasis in that region. Heidenreich et
al (9) and Zincke (10) demonstrated the high diagnostic
accuracy of staging PLND limited to the internal and external iliac
and obturator fossa LNs. An autopsy study by Weingärtner et
al (13) suggested that 20 LNs
must be retrieved for an adequate PLND. In the present study, the
cancer-specific 5-year survival rate of pN1 patients was 89%, which
supports the curative significance of LND.
Retrospective analyses demonstrated that the number
of positive LNs may affect survival. Certain studies have suggested
that RP may be curative for a proportion of patients with
LN-positive prostate cancer. Sterinberg et al (14) followed 64 patients with positive
nodes, of whom 83 and 68% remained free of detectable tumor at 60
and 80 months following RP, respectively, and identified the tumor
burden of the diseased LNs as an independent prognostic factor.
Our data suggest a delay in disease progression for
prostate cancer patients with minimal LN involvement when radical
surgery is performed. The follow-up duration is insufficient to
reach a definitive decision in terms of possible curative effect.
However, depending on the tumor burden or the number of diseased
LNs, radical surgical treatment appears to improve the outcome in a
not precisely defined but significant percentage of patients. Thus,
meticulous pelvic LND to remove all diseased nodes and ensure
accurate staging, combined with RP, is recommended for clinically
organ-confined prostate cancer. The promising survival rate of
patients with minimal nodal disease makes the value of routine
immediate androgen ablation in all patients with positive LNs at
least questionable, particularly when considering the side effects
of hormone therapy.
In conclusion, semi-extended PLND was associated
with a high rate of LN metastasis detection outside the fields of
standard LND in patients with clinically localized prostate cancer.
LND including the internal and external iliac LNs should be
performed in all patients with high risk-prostate cancer. However,
in the low-risk group, PLND may be omitted.
The results of the present study demonstrated that
semi-extended PLND, as performed in our institution, was associated
with greater nodal yields and may result in improved oncological
outcomes in patients with LN-positive prostate cancer. Although a
prospective trial is required to confirm our results, we currently
recommend considering semi-extended LND at the time of RP for
high-risk prostate cancer patients, whereas, in the low-risk group,
PLND may be omitted.
References
|
1
|
Walsh PC, Partin AW and Epstein JI: Cancer
control and quality of life following anatomical radical retropubic
prostatectomy: Results at 10 years. J Urol. 152:1831–1836.
1994.PubMed/NCBI
|
|
2
|
Catalona WJ and Smith DS: Cancer
recurrence and survival rates after anatomic radical retropubic
prostatectomy for prostate cancer: Intermediate-term results. J
Urol. 160:2428–2434. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gervasi LA, Mata J, Easley JD, et al:
Prognostic significance of lymph nodal metastases in prostate
cancer. J Urol. 142:332–336. 1989.PubMed/NCBI
|
|
4
|
Hull GW, Rabbani F, Abbas F, Wheeler TM,
Kattan MW and Scardino PT: Cancer control with radical
prostatectomy alone in 1,000 consecutive patients. J Urol.
167:528–534. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Miyake H, Fujimoto H, Komiyama M and
Fujisawa M: Development of ‘extended radical retropubic
prostatectomy’: A surgical technique for improving margin positive
rates in prostate cancer. Eur J Surg Oncol. 36:281–286. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Withrow DR, DeGroot JM, Siemens DR and
Groome PA: Therapeutic value of lymph node dissection at radical
prostatectomy: A population-based case-cohort study. BJU Int.
108:209–216. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Schumacher MC, Burkhard FC, Thalmann GN,
Fleischmann A and Studer UE: Good outcome for patients with few
lymph node metastases after radical retropubic prostatectomy. Eur
Urol. 54:344–352. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bader P, Burkhard FC, Markwalder R and
Studer UE: Disease progression and survival of patients with
positive lymph nodes after radical prostatectomy. Is there a chance
of cure? J Urol. 169:849–854. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Heidenreich A, Aus G, Bolla M, et al:
European Association of Urology: EAU guidelines on prostate cancer.
Eur Urol. 53:68–80. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zincke H: Extended experience with
surgical treatment of stage D1 adenocarcinoma of prostate.
Significant influences of immediate adjuvant hormonal treatment
(orchiectomy) on outcome. Urology. 33 (Supp 1):S27–S36. 1989.
View Article : Google Scholar
|
|
11
|
Clark T, Parekh DJ, Cookson MS, et al:
Randomized prospective evaluation of extended versus limited lymph
node dissection in patients with clinically localized prostate
cancer. J Urol. 169:145–147; discussion 147–148. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kroepfl D, Loewen H, Roggenbuck U, Musch M
and Klevecka V: Disease progression and survival in patients with
prostate carcinoma and positive lymph nodes after radical
retropubic prostatectomy. BJU Int. 97:985–991. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Weingärtner K, Ramaswamy A, Bittinger A,
Gerharz EW, Vöge D and Riedmiller H: Anatomical basis for pelvic
lymphadenectomy in prostate cancer: Results of an autopsy study and
implications for the clinic. J Urol. 156:1969–1971. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Steinberg GD, Epstein JI, Piantadosi S and
Walsh PC: Management of stage D1 adenocarcinoma of the prostate:
The Johns Hopkins experience 1974 to 1987. J Urol. 144:1425–1432.
1990.PubMed/NCBI
|