Introduction
Primary malignant pericardial mesothelioma (PMPM) is
extremely rare, with an incidence of <0.0022% and a poor
prognosis, with a survival from the onset of symptoms of <6
months (1). Unlike pleural and
peritoneal mesothelioma, the association between exposure to
asbestos and PMPM remains controversial (2). Making a definitive diagnosis of PMPM is
extremely difficult. Therefore, the majority of the cases are
diagnosed by autopsy (3). No
specific biomarkers or optimal therapy have been determined thus
far. Little has been reported on the long-term survival of PMPM
cases diagnosed antemortem with an increase in serum mesothelin
levels.
Case report
A 37-year-old female patient complaining of
progressive dyspnea and chest pain was referred to our hospital to
investigate persistent pericardial effusion. The patient had no
past history of exposure to asbestos. The results of the peripheral
blood tests revealed a white blood cell count of 10,800/µl with
72.2% neutrophils, a hemoglobin level of 13.1 g/dl, a platelet
count of 284,000/µl, a C-reactive protein level of 1.2 mg/dl and a
serum brain natriuretic peptide level of 122.9 U/ml, as well as an
increase in serum mesothelin levels (18.5 nmol/l; normal range,
<1.5 nmol/l). The arterial blood gas analysis revealed a pH of
7.43, PaCO2 of 35.5 Torr and PaO2 of 77.0
Torr at room air. A chest radiograph revealed cardiomegaly with
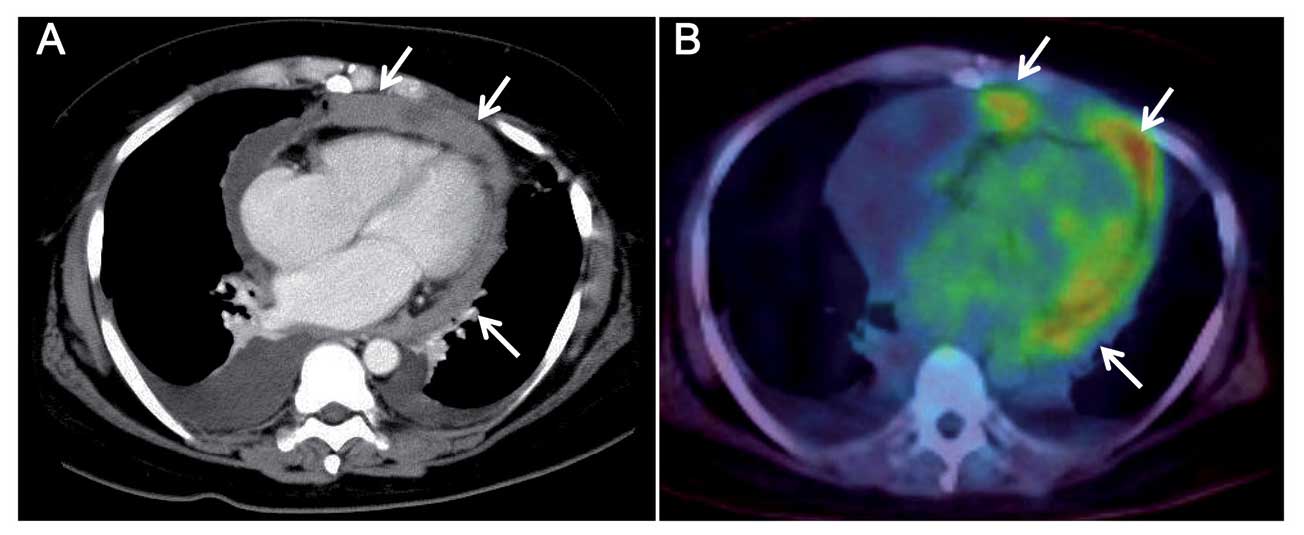
bilateral pleural effusion. Chest computed tomography revealed an
irregular, thickened pericardium with diffuse enhancement, with
loculated large amounts of pericardial and bilateral pleural
effusions (Fig. 1A).
Fluorodeoxyglucose (FDG) positron emission tomography (PET) images
revealed intrapericardial FDG accumulation with a standardized
uptake value of 6.0 (Fig. 1B).
Transthoracic echocardiography showed a thickened pericardium with
pericardial effusion, but no evidence of cardiac tamponade. The
left ventricular function and cardiac valves were normal (Fig. 2).
The results of the pericardial fluid cytology were
class V, with a suspected diagnosis of adenocarcinoma. No fungal,
bacterial, or mycobacterial pathogens were isolated from the
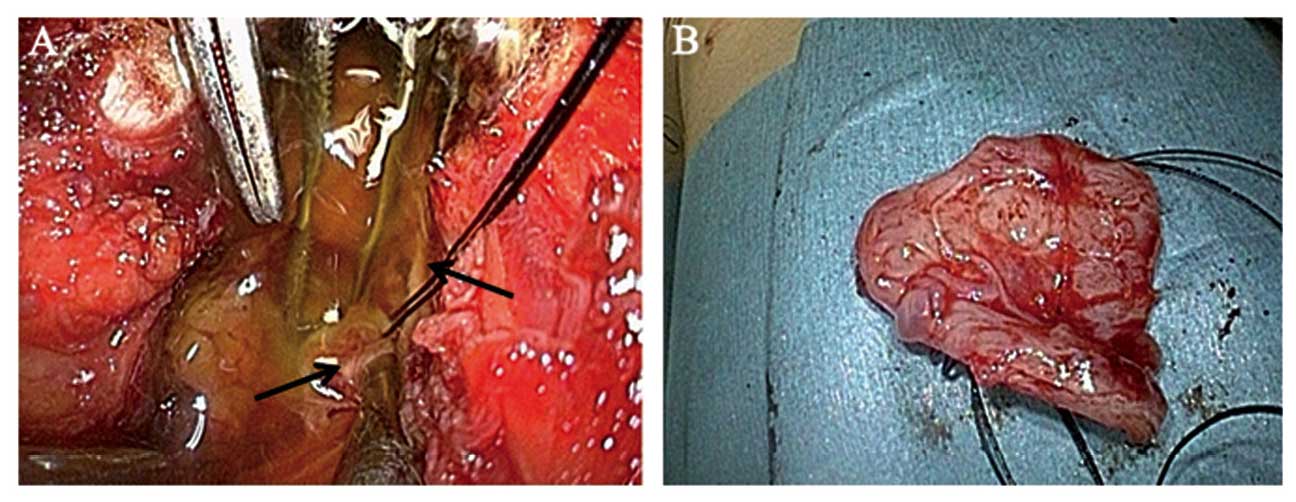
pericardial effusion. Surgical biopsy of the pericardium and
drainage were performed through a subxiphoid approach under general
anesthesia to make a definitive diagnosis. The thickened
pericardium densely adhered to the epicardium on the left side;
pericardial biopsy was performed on the right side, with drainage
of 300 ml of straw-colored mucinous fluid. Scattered whitish
nodules were identified on the right side of the epicardium
(Fig. 3). For continuous
postoperative drainage and sclerosing therapy, a single chest tube
was inserted in the pericardial cavity through a separate stab
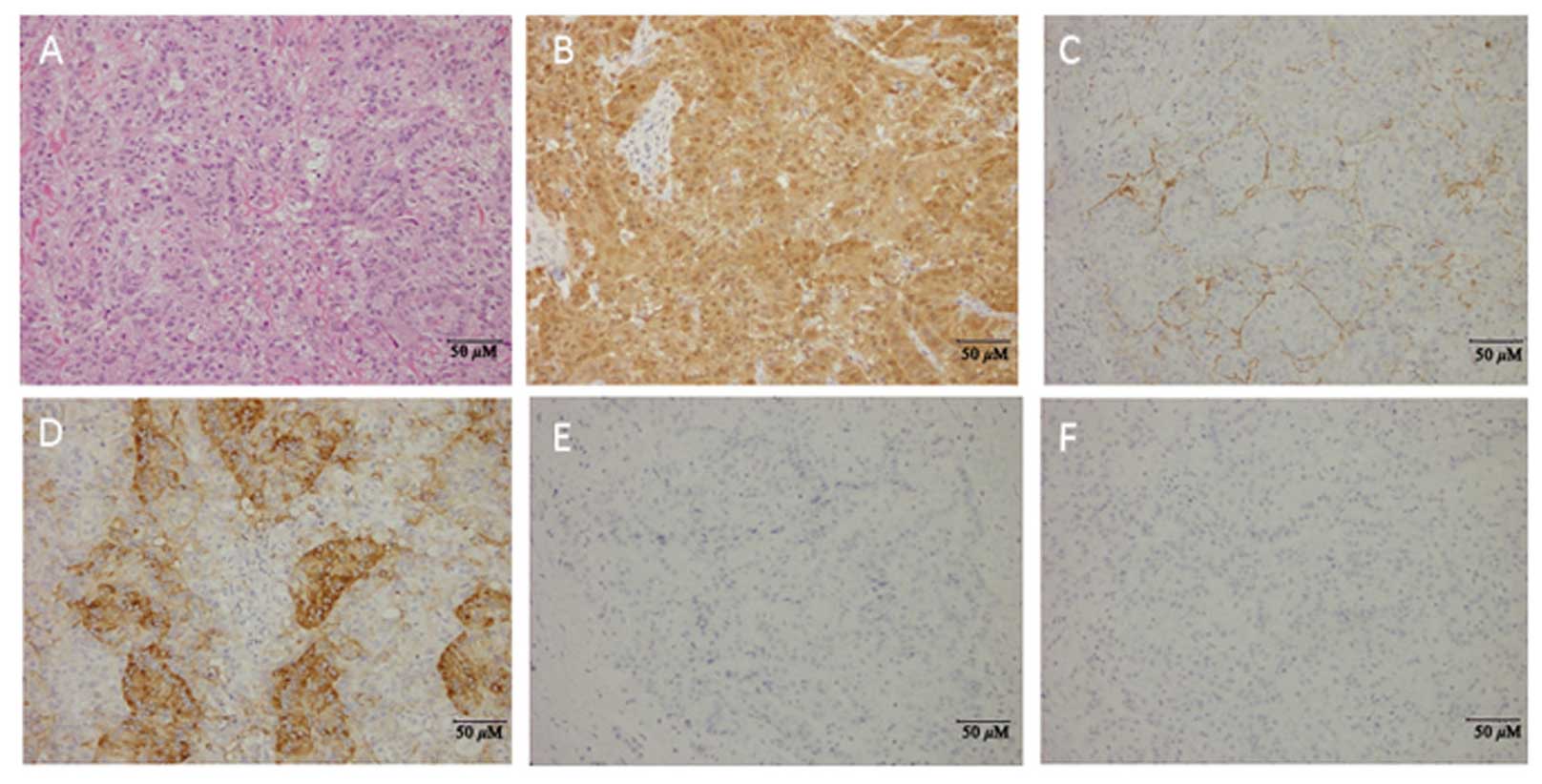
incision. Histopathological examination revealed diffuse
infiltration of the pericardium by PMPM, which consisted of
epithelioid cells with abundant eosinophilic cytoplasm and large
round nuclei with prominent nucleoli, arranged in a
tubular-papillary pattern (Fig. 4A).
Immunohistochemistry was positive for calretinin, D2-40 and Hector
Battifora mesothelial cell-1, whereas it was negative for
carcinoembryonic antigen (CEA) and thyroid transcription factor-1
(TTF-1) (Fig. 4B-F). Finally, the
patient was diagnosed with PMPM of epithelioid type.
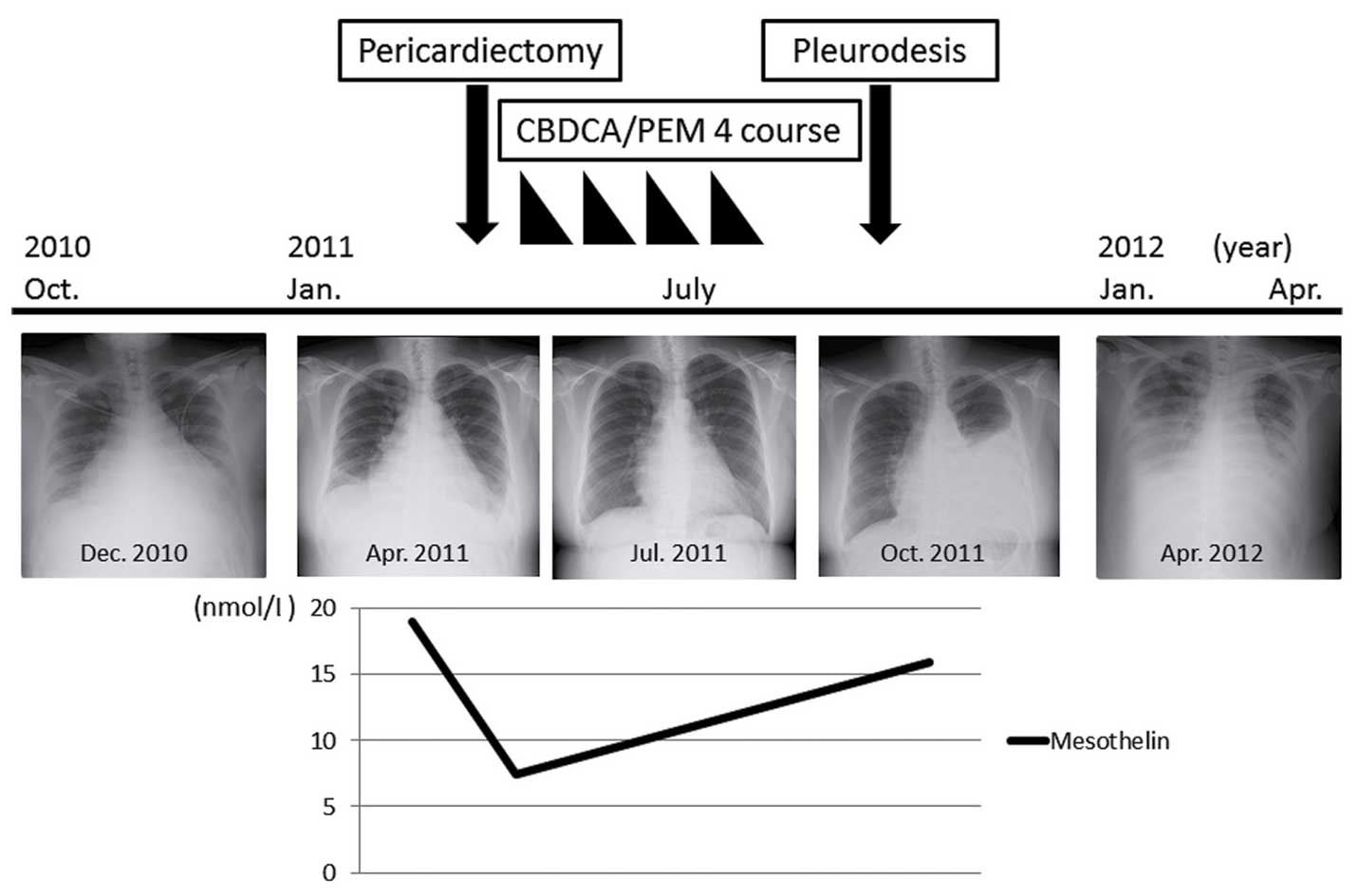
The patient's condition temporarily improved and the
serum mesothelin levels decreased during chemotherapy with
carboplatin (area under the curve of 5.0) and pemetrexed (500
mg/m2) with usual vitamin supplementation every 28 days
for a 4-week cycle. However, her condition worsened, with elevated
serum mesothelin levels, immediately after chemotherapy. As a
result, the patient received no further chemotherapy apart from
pleurodesis, due to the massive pleural effusion. Eventually the
patient succumbed to cardiac tamponade 18 months after the initial
onset of the symptoms (Fig. 5).
Discussion
PMPM is an extremely rare malignancy, with an
incidence of ~0.0022% according to one of the largest necropsy
series (1). PMPM is associated with
a wide patient age range (1–79 years) and the male-to-female ratio
is 2:1 (2). PMPM is less
significantly associated with exposure to asbestos compared with
pleural malignant mesothelioma (2).
In the present case, the patient had no history of asbestos
exposure. Furthermore, there was no evidence of the presence of
asbestos bodies on histological examination at autopsy.
PMPM is often discovered at a late stage during the
clinical course, or at autopsy (3).
The diagnostic yield of pericardial fluid cytology is only 24% of
the PMPM cases (4). Therefore, the
majority of the patients with PMPM are diagnosed using either
surgical or autopsied specimens. The differential diagnosis of
tumors affecting the pericardium includes metastases to the
pericardium from cancer in other organs, hematological
malignancies, melanoma and, rarely, primary cardiac tumors. The
histological and immunohistochemical studies of pericardial
mesothelioma resemble those of pleural mesothelioma.
Immunohistochemically, negativity for adenocarcinoma markers, such
as CEA and TTF-1, and positivity for mesothelial markers, such as
calretinin and cytokeratins 5/6, are useful in differentiating
mesotheliomas from adenocarcinomas (5). In the present case, adenocarcinoma was
initially suspected on the basis of cytology results. However, the
histological and immunohistochemical findings led to a definitive
diagnosis of epithelioid type PMPM.
It was recently suggested that FDG-PET for PMPM may
be useful for disease staging and preoperative evaluation (6). In the present case, an accumulation of
FDG was detected, corresponding with the pericardial tumor.
Furthermore, mesothelin is currently considered to be the best
novel serum biomarker of malignant pleural mesothelioma. According
to a meta-analysis on the efficacy of serum mesothelin in patients
with pleural malignant mesothelioma, the sensitivity and
specificity of mesothelin ranged widely from 19 to 68% and from 88
to 100%, respectively (7). Our
patient presented with an increase in serum mesothelin levels at
initial diagnosis of PMPM. Subsequently, the serum mesothelin level
exhibited a trend towards a decrease after chemotherapy, whereas it
increased along with a deterioration of the patient's general
condition. We hypothesized that the serial changes of the serum
mesothelin level may be correlated with the onset of PMPM and the
disease status.
Treatment guidelines for PMPM have not yet been
established. In fact, PMPMs are treated with a palliative approach
based on surgery, chemotherapy and radiotherapy. Therefore, the
median survival for PMPM patients diagnosed antemortem is <4
months. Surgical resection is one of the treatment options for
localized disease. However, it is difficult to remove the tumor
completely, as the majority of the patients with PMPM are already
at an advanced stage at the time of diagnosis (8). Radiation therapy has little effect on
PMPM, but has been used as adjuvant therapy in patients with PMPM
undergoing incomplete resection (9).
Finally, combination chemotherapy with cisplatin and pemetrexed has
demonstrated prolonged survival in pleural malignant mesothelioma
and is also considered as first-line treatment in PMPM (1,10). Our
patient was administered a combination of carboplatin and
pemetrexed, rather than cisplatin, to reduce cardiac burden
Consequently, our patient survived for 18 months, which was longer
compared with cases reported in previous studies.
In conclusion, we herein present an extremely rare
PMPM case with increased levels of serum mesothelin diagnosed by
surgical pericardial resection. It is crucial to make a timely
definitive diagnosis and administer treatment during the early
stages of PMPM.
Acknowledgements
The authors would like to thank Dr Riphe Park
(Department of Cardiovascular Medicine, Toho University Omori
Medical Center) for the support in treating the patient. We are
grateful to Dr K. Shibuya for the advice and analysis of the
patient's pathology (Department of Pathology, Toho University Omori
Medical Center, Tokyo, Japan).
References
|
1
|
Santos C, Montesinosa J, Castañera E, Sole
JM and Baga R: Primary pericardial mesothelioma. Lung Cancer.
60:291–293. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Patel J and Sheppard MN: Primary malignant
mesothelioma of the pericardium. Cardiovasc Pathol. 20:107–109.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Suman S, Schofield P and Large S: Primary
pericardial mesothelioma presenting as pericardial constriction: A
case report. Heart. 90:e42004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nilsson A and Rasmuson T: Primary
pericardial mesothelioma: Report of a patient and literature
review. Case Rep Oncol. 2:125–132. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Travis WD, Brambilla E, Muller-Hermelink
HK and Harris CC: Pathology and Genetics. Tumors of the Lung,
Pleura, Thymus and Heart, World Health Organization Classification
of Tumours. IARC Press; Lyon: pp. 249–287. 2004
|
|
6
|
Aga F, Yamamoto Y, Norikane T and
Nishiyama Y: A case of primary pericardial mesothelioma detected by
18F-FDG PET/CT. Clin Nucl Med. 37:522–523. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hollevoet K, Reitsma JB, Creaney J,
Grigoriu BD, Robinson BW, Scherpereel A, Cristaudo A, Pass HI,
Nackaerts K, Rodríguez Portal JA, et al: Serum mesothelin for
diagnosing malignant pleural mesothelioma: An individual patient
data meta-analysis. J Clin Oncol. 30:1541–1549. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kaul TK, Fields BL and Kahn DR: Primary
malignant pericardial mesothelioma: A case report and review. J
Cardiovasc Surg(Torino). 35:261–267. 1994.PubMed/NCBI
|
|
9
|
Vigneswaran WT and Stefanacci PR:
Pericardial mesothelioma. Curr Treat Options Oncol. 1:299–302.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Vogelzang NJ, Rusthoven JJ, Symanowski J,
Denham C, Kaukel E, Ruffie P, Gatzemeier U, Boyer M, Emri S,
Manegold C, et al: Phase III study of pemetrexed in combination
with cisplatin versus cisplatin alone in patients with malignant
pleural mesothelioma. J Clin Oncol. 21:2636–2644. 2003. View Article : Google Scholar : PubMed/NCBI
|