Introduction
Colorectal cancer (CRC) ranks as the third leading
cause of cancer-related mortality in developed regions, with ~1.2
million new cases diagnosed globally each year (1). Bone metastases constitute 6% of all CRC
metastases (2). Notably, ~22% of
patients exhibit distant metastases at the time of diagnosis, and
70% develop these during follow-up (3).
Despite their low incidence rate, bone metastases
signify a worsened oncological prognosis. Metastasectomy may be a
therapeutic option for singular and well-localized lesions;
however, generally, treatment pursues a palliative goal. This
involves systemic chemotherapy (with or without targeted therapy),
radiotherapy, or the pharmacological use of bisphosphonates
(4).
The present study describes the case of a patient
who underwent surgery for stage IV rectal adenocarcinoma and
developed a single bone metastasis at the costal level 79 months
following the initial surgery. The description of this clinical
case reminds us that despite a long follow-up period and radical
surgery, the clinical monitoring of patients with metastatic rectal
cancer should continue. On the other hand, the present study also
describes the use of a bilaminar mesh (Ventralight ST
Bard®) for repairing the defect in the chest wall
following bone metastasectomy.
Case report
A 61-year-old male patient with a history of
dyslipidemia underwent surgery at the Príncipe of Asturias Teaching
Hospital (Madrid, Spain) in October, 2013 for T3N0M1 rectal
adenocarcinoma, which involved low anterior resection and liver
metastasectomy from segment V. The pathological report indicated a
poorly differentiated colorectal adenocarcinoma, with no lymph node
involvement, perineural or lymphovascular invasion, or tumor
deposits. The resection margins were free of tumor involvement.
KRAS was determined to have a wild-type expression (data not
shown). In relation to the metastasectomy, the colorectal origin
was confirmed, with no involvement of the surgical margins.
Adjuvant treatment included six cycles of the XELOX
scheme (intravenous oxaliplatin 130 mg/m2 (day 1)
followed by oral capecitabine 1,000 mg/m2 twice daily
(day 1, evening, to day 15, morning). On May, 2020, the patient
returned to Príncipe of Asturias Teaching Hospital reporting rib
cage pain, which led to the discovery of a bone metastasis. An
initial radiological study was performed consisting of a
posteroanterior chest X-ray that revealed the alteration at the
cortical level of the anterior arch of the right ninth rib in
relation to lytic tumor. A complementary study was performed, using
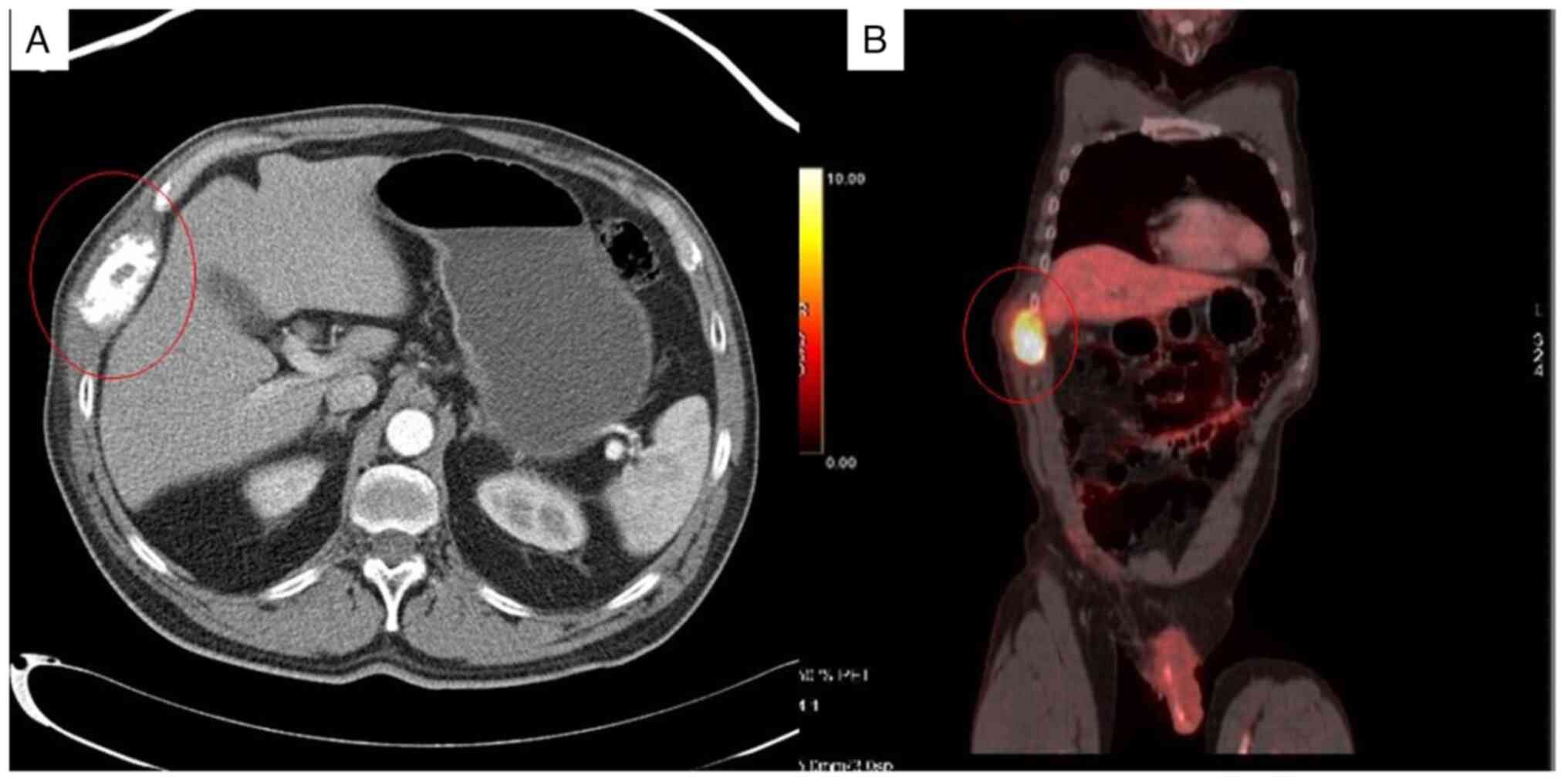
thoraco-abdominopelvic computed tomography and positron emission
tomography and computed tomography, which revealed a hypermetabolic
lesion at the level of the right chest wall with destruction of the
arch of the ninth rib accompanied by a soft tissue mass measuring
69x44x67 mm with pathological uptake (SuvMax of 26.6) without other
metabolically significant lesions (Fig.
1). Following this series of diagnostic tests, a biopsy
confirmed colorectal adenocarcinoma. The patient received
radiotherapy and the CAPOX regimen (130 mg/m2
oxaliplatin on day 1 and 2,000 mg/m2 oral capecitabine
on days 1-14, every 3 weeks) with a subsequent positron emission
tomography and computed tomography revealing no evidence of tumor
progression. Following successful treatment, surgery was performed
in May, 2021, involving the resection of the costal wall lesion and
chest wall repair with a bilaminar mesh (Ventralight
ST®, Bard).
On May 2020, he reported pain at the rib cage level
in relation to the appearance of a soft tissue tumor. A biopsy was
performed using core needle biopsy which revealed bone tissue
infiltrated by an adenocarcinoma of colorectal origin, with a focal
and weak expression for CK7 and an intense expression of CK20 and
CDX2. Following the diagnosis, treatment with radiotherapy and a
systemic treatment regimen was carried out according to the CAPOX
scheme (capecitabine and oxaliplatin) (eight cycles) and subsequent
re-evaluation using a new positron emission tomography and computed
tomography scan that revealed no evidence of tumor progression.
An immunohistochemical analysis was also performed
on the samples (thickness of 3 microns). The sections were embedded
in paraffin for processing. The primary antibody dilution used was
as follows: CK7 (factory pre-diluted; cat. no. GA619; clone
D5/16B4; Agilent Technologies, Inc.; 23˚C, 12.5 min); CK20 (factory
pre-diluted; cat. no. GA777; clone KS 20.8; Agilent Technologies,
Inc.; 21˚C, 18 min); CDx2 (factory pre-diluted; cat. no. GA080;
clone DAk-CDX2; Agilent Technologies, Inc.; 23˚C. 15 min). The
secondary antibody dilution was performed by pre-dilution with
biotin/streptavidin (cat. no. GV800; Agilent Technologies, Inc);
also at 23˚C for 15 min. The counterstain used was
hematoxylin/eosin (Abbey Color, Inc., with processing at 23˚C for 7
min). All samples were observed using a brightfield microscope
(Nikon Eclipse 50i; Nikon Corporation) at x100 magnification
Following the completion of the treatment and given
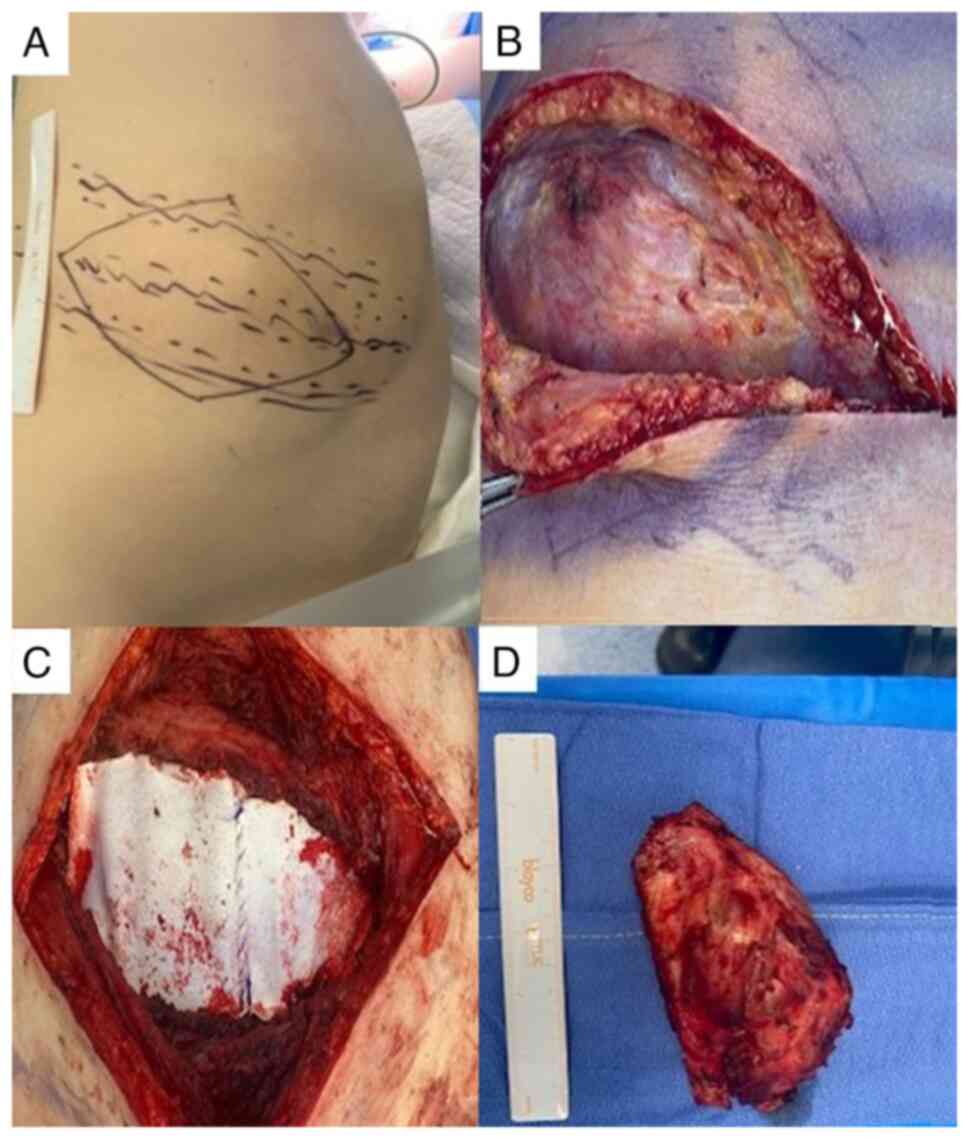
the good response, surgery was performed in May, 2021, performing a
right posterolateral thoracotomy. A tumor of the costal wall was
observed involving the eighth and ninth right costal arch without
diaphragmatic or pulmonary involvement. A complete resection of the
lesion was performed en bloc, including the middle third of
the eighth and ninth right ribs, repairing the chest wall using
bilaminar mesh (Bard®) and placing an endothoracic tube.
The patient was discharged on the fifth post-operative day
(Fig. 2).
Following the intervention, clinical follow-up was
maintained, presenting multiple pulmonary recurrence on a thoracic
CT-scan in July 2022. Systemic treatment was initiated according to
the CAPOX scheme with bevacizumab (6 cycles) with a good response,
maintaining no evidence of recurrence until September, 2023.
Post-treatment follow-up revealed pulmonary
recurrence in July, 2022, prompting systemic treatment with CAPOX
and bevacizumab (7.5 mg/kg intravenous), resulting in no recurrence
until the last follow-up in September, 2023.
Discussion
CRC primarily metastasizes to the liver, but it can
also spread to other parts of the body, including the lungs,
peritoneum and, less commonly, the bones. Bone metastases represent
6% of all metastases from CRC (2).
When CRC spreads to the bones, it can be challenging to treat and
often indicates an advanced stage of the disease.
Bone tissue is a rigid, yet dynamic organ subject to
continuous processes of formation and repair throughout life. Bone
remodeling, predominantly a metabolic process in adults, determines
the structure and function of the skeleton. Consequently, bone
tissue integrity requires coordinated activity between cells
responsible for bone formation (osteoblasts) and bone resorption
(osteoclasts) (5). In the clinical
case reported herein, it is noteworthy that metachronous metastatic
involvement occurred at 79 months following the initial surgical
intervention. This may be explained by a mechanism of hematogenous
dissemination with localization of quiescent tumor cells at the
bone level.
Bone is a frequent site of metastatic involvement in
various solid organ cancers (breast, lung, thyroid, prostate or
kidney cancer). The most frequent location is the pelvis (35.3%),
followed by the lumbar spine (25%), thoracic vertebrae (21.6%),
sacrum (20.6%) and ribs (14.7%) (1).
Secondary bone involvement can be found in lung cancer in up to
40-50% or up to 60-70% in prostate cancer. However, bone as the
location of metastases from gastrointestinal malignancies
represents <10% of cases (6).
The incidence of bone metastases of colorectal
origin is variable (0.96-11.1%), being more frequent in patients
with a diagnosis of rectal cancer, likely influenced by its
vascularization, explaining hematogenous dissemination as the most
frequent mechanism (1). However, a
higher incidence has been described in autopsies, ranging between
8.6 and 27% (7).
The presence of bone metastases, unlike the lung or
liver location, is usually symptomatic, with local pain symptoms
due to bone tissue destruction, causing irritation of the
periosteum and/or nerve injury, potentially leading to pathological
fractures (6). Therefore, unlike
metastases located in the lungs or liver, bone metastases are
diagnosed earlier.
In their study Zhenghong et al (1) described a greater association of bone
metastases in patients with a poor degree of tumor differentiation,
similar to the patient in the present study. Other factors related
to the development of bone metastases are rectal origin, lymph node
involvement and the presence of lung metastases (8).
In the patient in the present study,
immunohistochemical analysis was key for the diagnosis, since the
bone location and the time of appearance (at 79 months since the
initial intervention) rendered colorectal origin unlikely.
Cytokeratin (CK)20 is an intermediate filament
protein found in the cytoskeleton of epithelial cells. In tumor
pathology, CK20 is a critical immunohistochemical marker frequently
expressed in CRC. Its expression, along with other markers, can
help confirm a colorectal origin for metastatic tumors when the
primary site is unknown.
The majority of primary lung adenocarcinomas are
CK7-positive and CK20-negative (CK7+/CK20-).
This CK7+/CK20- pattern can help distinguish
primary lung adenocarcinomas from metastatic colorectal
adenocarcinomas, which are typically CK20+ and
CK7(9). In the case described
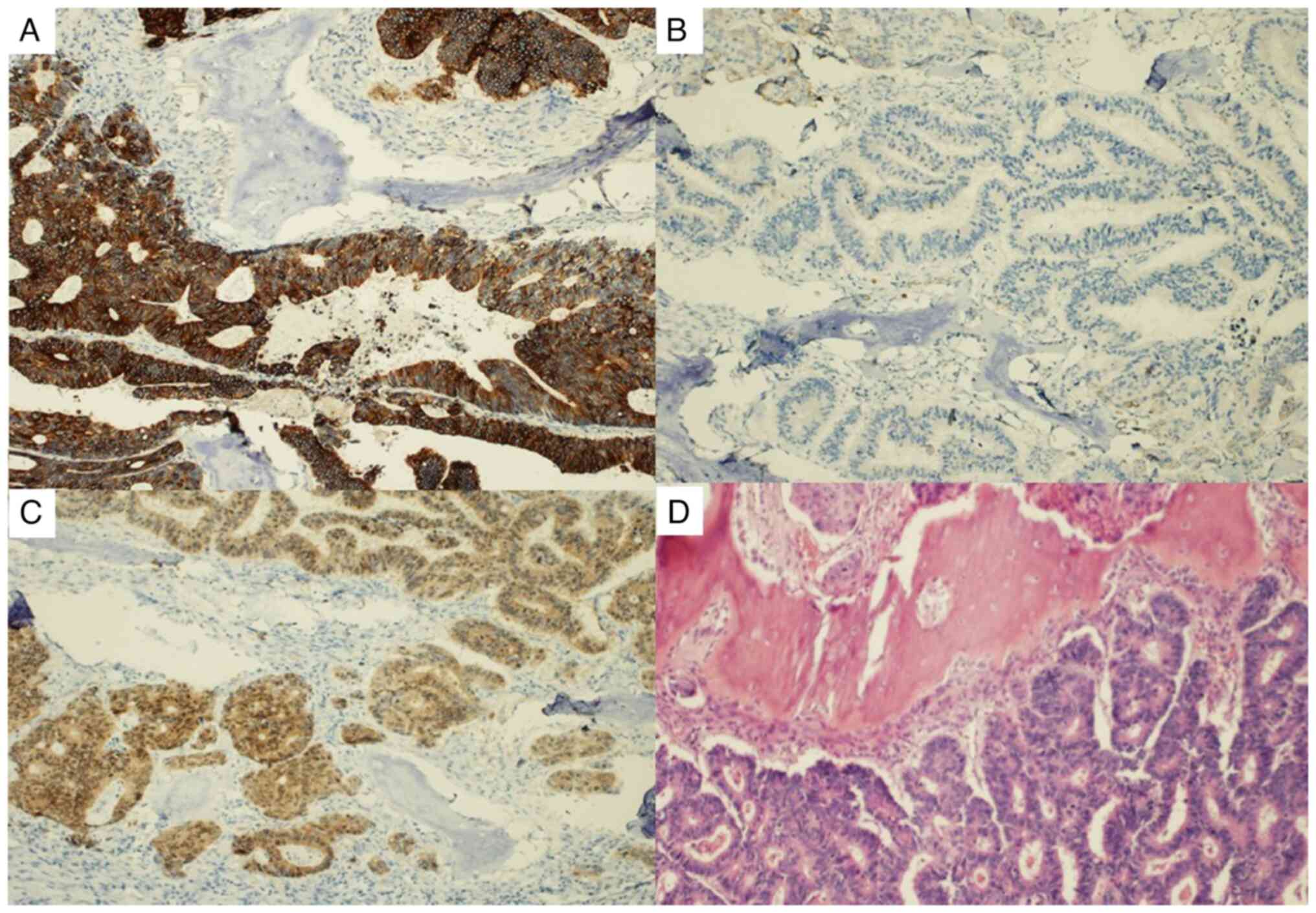
herein, the expression of CK7 was very weak and focal compared to
CK20 and CDX2, which exhibited intense positivity (Fig. 3). In cases of metastatic carcinoma
where the primary site is unknown, the expression pattern of CK7
and CK20 may be useful. For instance, if a metastatic tumor in the
lung is CK20+/CK7-, one would consider
potential primary sites, such as the colorectum. On the other hand,
a CK20-/CK7+ pattern in a lung tumor would be
more supportive of a primary lung origin (though other primary
sites can also have this pattern).
The immunohistochemical profile of CRC is positive
for CDK20 and CDX2, while it tends to be negative for CDK7. This
association can be observed in Fig.
3, noting how in the bone tissue, the CDK20 marker is negative,
whereas it exhibits positivity in the bone fragment infiltrated by
the tumor. The same is true for CDX2 expression, negative in the
bone tissue and positive in the colorectal tissue. CDX2 is a
nuclear transcription factor primarily expressed in the cells of
the intestinal epithelium, both in the small and large intestine.
It plays a role in the development and differentiation of these
cells. In the context of CRC, CDX2 is of interest as the loss of
CDX2 expression in primary CRC has been shown to be associated with
a worse prognosis (10). According
to Cowan et al (11), this
marker is expressed in ~12% of patients diagnosed with lung cancer,
and its expression can be variable. In the context of prostatic
carcinoma, the utility and prevalence of CDX2 expression are not
widely recognized or documented (12).
The management of bone metastasis is an essential
aspect of cancer care, particularly since the bone is a common site
for metastasis for various primary tumors such as breast, prostate,
lung, thyroid and kidney cancer (2).
The effective treatment of bone metastasis can improve the quality
of life of patients, and can decrease pain and reduce
complications.
The treatment of bone metastases should include the
administration of systemic chemotherapy and surgical resection when
possible due to location. In the patient in the present study,
given the absence of metastatic disease at other levels and the
possibility of performing a resection with free surgical margins,
this was performed. Treatment with radiotherapy was also performed
with the aim of improving symptoms and reducing tumor size.
Previous studies have revealed that treatment with
radiotherapy reduces bone pain and prevents pathological fractures,
in addition to increasing survival in metachronous bone metastases
(4). In the patient described
herein, the aim of pre-operative radiotherapy was to reduce bone
pain. Furthermore, the patient received six cycles of chemotherapy
according to the CAPOX regimen with bevacizumab. According to the
literature, the use of this type of systemic treatment regimen in
patients with metastatic colorectal cancer is effective in terms of
survival without compromising the quality of life of patients
(13).
The 5-year survival rate of patients with lung or
liver metastases is 8 and 16.9%, respectively. Some researchers
have determined that, in the case of bone metastases, the 5-year
survival is 3.4% (14). The presence
of bone metastasis, together with age, location in the ascending
colon, a high degree of differentiation, lymph node involvement and
high pre-operative carcinoembryonic antigen levels have been
described as poor prognostic factors in relation to the survival of
patients undergoing surgery for CRC (15).
KRAS mutations occur in 40-50% of patients diagnosed
with CRC (16). In previous studies,
the authors described a negative association between patients with
mutated KRAS with a diagnosis of CRC and the presence of peritoneal
or liver metastases in terms of survival (17,18). In
the patient in the present study, the KRAS determination
corresponded to KRAS wild-type, providing a better prognosis and a
better response to the administered systemic therapy.
In conclusion, CRC predominantly metastasizes to the
liver and lungs, with bone metastasis being relatively rare. Its
development is indicative of a poor prognosis. A comprehensive,
multi-disciplinary approach is crucial in managing bone metastasis,
considering the primary cancer type, stage and the performance
status of the patient. The case presented herein emphasizes the
importance of continued clinical monitoring and tailored treatment
strategies for patients with metastatic CRC.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
All authors (FMM, BMG, MDA, EOM, CVM, ABM, AQV, RJM,
RDA, ILU and AGC) contributed to the diagnosis and treatment of the
patient, and in the design of the study. FMM was a major
contributor to the writing of the manuscript. BMG and FMM confirm
the authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study followed international and
national regulations and was in agreement with the Declaration of
Helsinki, and ethical principles. The patient signed an informed
consent form before the surgery was performed.
Patient consent for publication
The patient provided written informed consent for
the publication of any data and/or accompanying images, before the
surgery was performed. Patients have a right to anonymity and
privacy, and authors have a legal and ethical responsibility to
respect this right.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Zhenghong Zhu Z, Guoweijian Zhangning,
Caiyunyun Yingjiangshan and Xiaomi : Retrospective study of
predictors of bone metastasis in colorectal cancer patients. J Bone
Oncol. 9:25–28. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kobayashi Y, Shida D, Boku N, Yasui K,
Nakamura Y, Kudose Y, Imaizumi J and Kanemitsu Y: Prognostic
factors of bone metastases from colorectal cancer in the era of
targeted therapy. Dis Colon Rectum. 66:401–409. 2023.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Rocha MM, Dariva I, Zornoff GC, De
Laurentis GS, Mendes GC, Santana MG, de Miguel GC, Ferreira RS,
Sciani JM and Priolli DG: A new therapeutic approach for bone
metastasis in colorectal cancer: Intratumoral melittin. J Venom
Anim Toxins Incl Trop Dis. 28(e20210067)2022.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Ma CX, Guan X, Wei R, Wang S, Quan JC,
Zhao ZX, Chen HP, Liu Z, Jiang Z and Wang XS: The distinction of
clinicopathological characteristics, treatment strategy and outcome
in colorectal cancer patients with synchronous vs. metachronous
bone metastasis. Front Oncol. 10(974)2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Fornetti J, Welm AL and Stewart SA:
Understanding the bone in cancer metastasis. J Bone Miner Res.
33:2099–2113. 2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Hong S, Youk T, Lee SJ, Kim KM and Vajdic
CM: Bone metastasis and skeletal-related events in patients with
solid cancer: A Korean nationwide health insurance database study.
PLoS One. 15(e0234927)2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Suresh Babu MC, Garg S, Lakshmaiah KC,
Babu KG, Kumar RV, Loknatha D, Abraham LJ, Rajeev LK, Lokesh KN,
Rudresha AH and Rao SA: Colorectal cancer presenting as bone
metastasis. J Cancer Res Ther. 13:80–83. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Li A, Käsmann L, Rades D and Fu C: A
scoring system to predict the development of bone metastasis after
radical resection of colorectal cancer. Anticancer Res.
37:5169–5172. 2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Ilieva N, Tashkova D, Staykov D, Serteva
D, Feodorova Y, Mehterov N, Mollova A and Bachurska S:
Immunohistochemical expression of CK20, CK7, and CDX2 in colorectal
carcinoma in correlation with pathomorphological characteristics.
Folia Med (Plovdiv). 64:214–220. 2022.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Korphaisarn K, Sukhokanjanachusak K,
Pongpaibul A, Chinswangwatanakul V and Akewanlop C: Loss of CDX-2
expression is an independent poor prognostic biomarker in patients
with early-stage deficient mismatch repair colorectal cancer. Asia
Pac J Clin Oncol. 18:249–258. 2022.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Cowan ML, Li QK and Illei PB: CDX-2
expression in primary lung adenocarcinoma. Appl Immunohistochem Mol
Morphol. 24:16–19. 2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Girardo C, Fakhri N, Khokhar H, Jetly R
and Bhalla R: CDX2 expression in primary and metastatic prostatic
adenocarcinoma: A retrospective study. Am J Clin Pathol. 154 (Suppl
1):S59–S60. 2020.
|
|
13
|
Simkens LH, van Tinteren H, May A, ten
Tije AJ, Creemers GJ, Loosveld OJ, de Jongh FE, Erdkamp FL, Erjavec
Z, van der Torren AM, et al: Maintenance treatment with
capecitabine and bevacizumab in metastatic colorectal cancer
(CAIRO3): A phase 3 randomised controlled trial of the Dutch
colorectal cancer group. Lancet. 385:1843–1852. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Wang H, Shan X, Zhang M, Qian K, Shen Z
and Zhou W: Nomograms for predicting overall survival in colorectal
cancer patients with metastasis to the liver, lung, bone, and
brain. Cancer Causes Control. 34:1059–1072. 2023.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Liu Z, Xu Y, Xu G, Baklaushev VP,
Chekhonin VP, Peltzer K, Ma W, Wang X, Wang G and Zhang C: Nomogram
for predicting overall survival in colorectal cancer with distant
metastasis. BMC Gastroenterol. 21(103)2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Park HS, Chun YJ, Kim HS, Kim JH, Lee CK,
Beom SH, Shin SJ and Ahn JB: Clinical features and KRAS mutation in
colorectal cancer with bone metastasis. Sci Rep.
10(21180)2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Díez-Alonso M, Mendoza-Moreno F,
Gómez-Sanz R, Matías-García B, Ovejero-Merino E, Molina R,
Soto-Schütte S, San Juan A and Gutierrez-Calvo A: Prognostic value
of KRAS gene mutation on survival of patients with peritoneal
metastases of colorectal adenocarcinoma. Int J Surg Oncol.
2021(3946875)2021.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Díez-Alonso M, Mendoza-Moreno F,
Jiménez-Álvarez L, Nuñez O, Blazquez-Martín A, Sanchez-Gollarte A,
Matías-García B, Molina R, San-Juan A and Gutierrez-Calvo A:
Prognostic factors of survival in stage IV colorectal cancer with
synchronous liver metastasis: Negative effect of the KRAS mutation.
Mol Clin Oncol. 14(93)2021.PubMed/NCBI View Article : Google Scholar
|