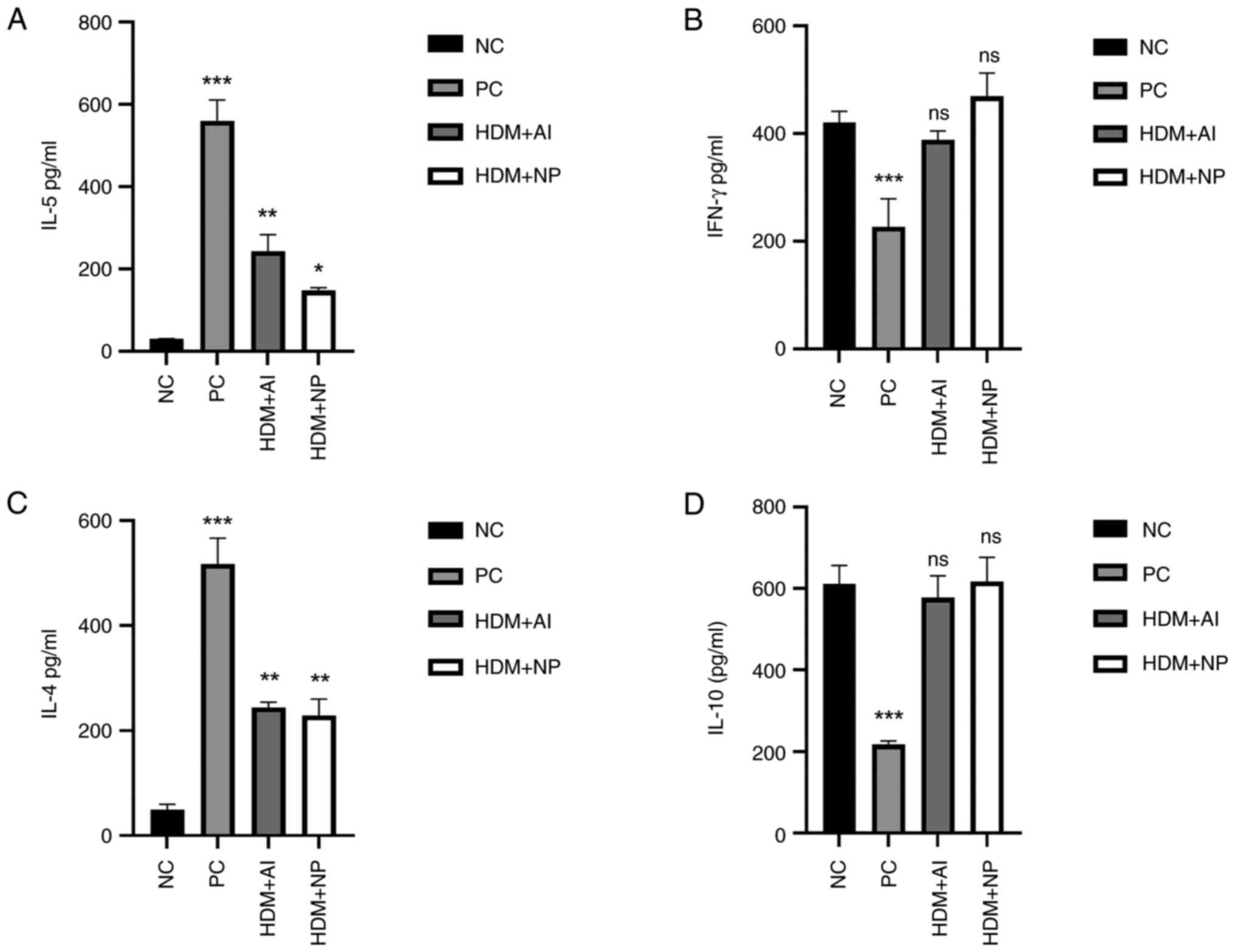
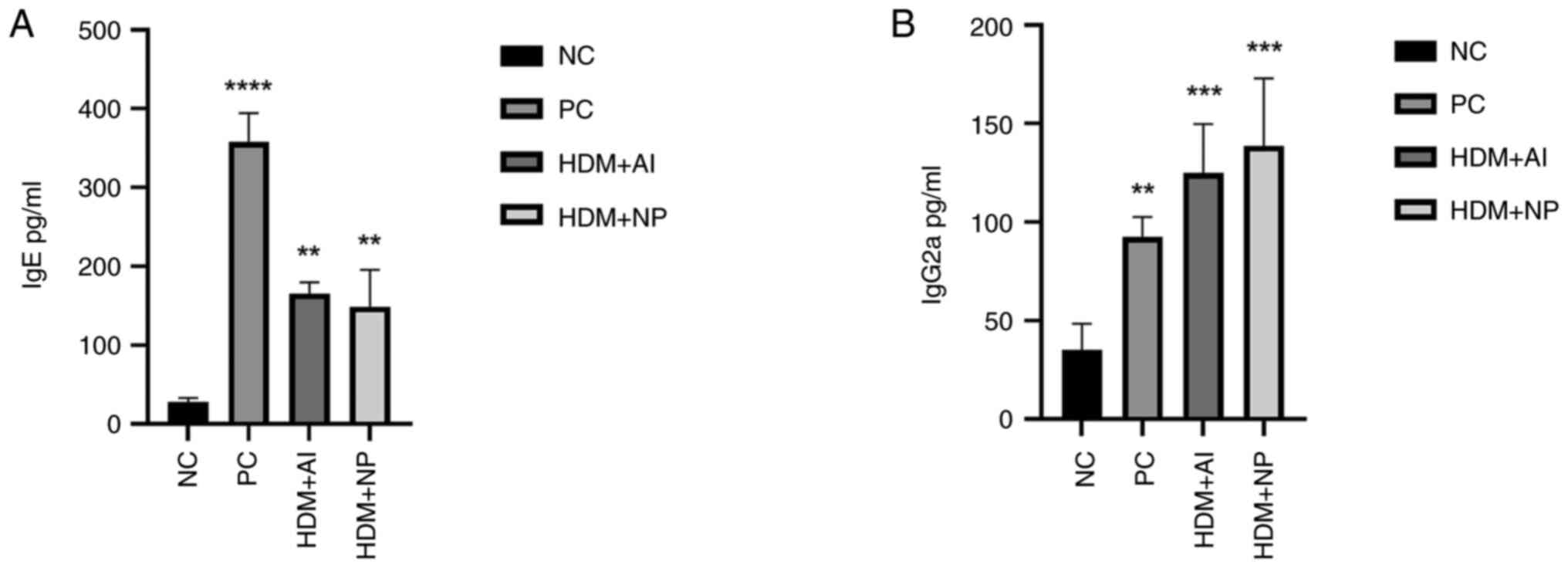
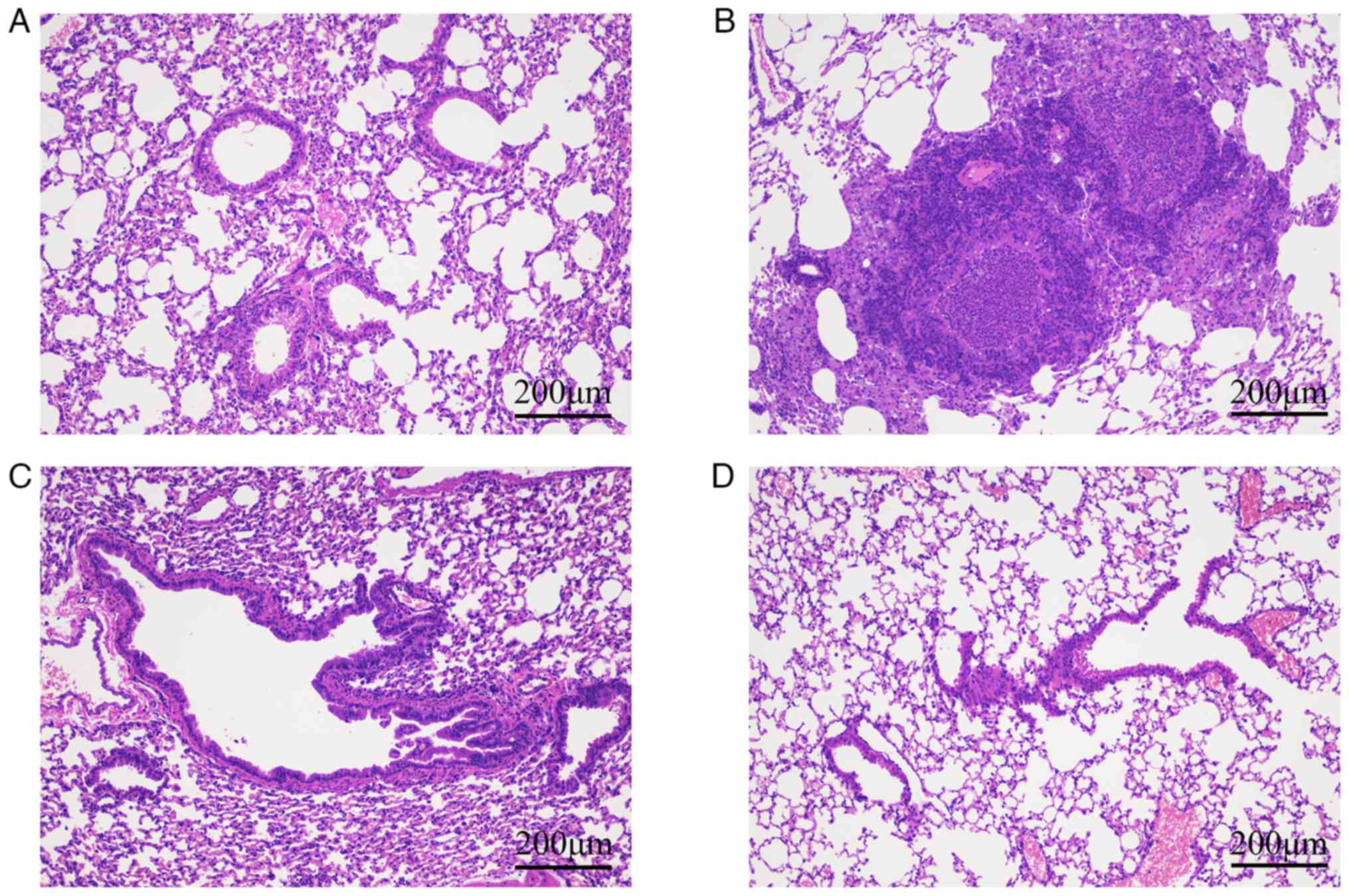
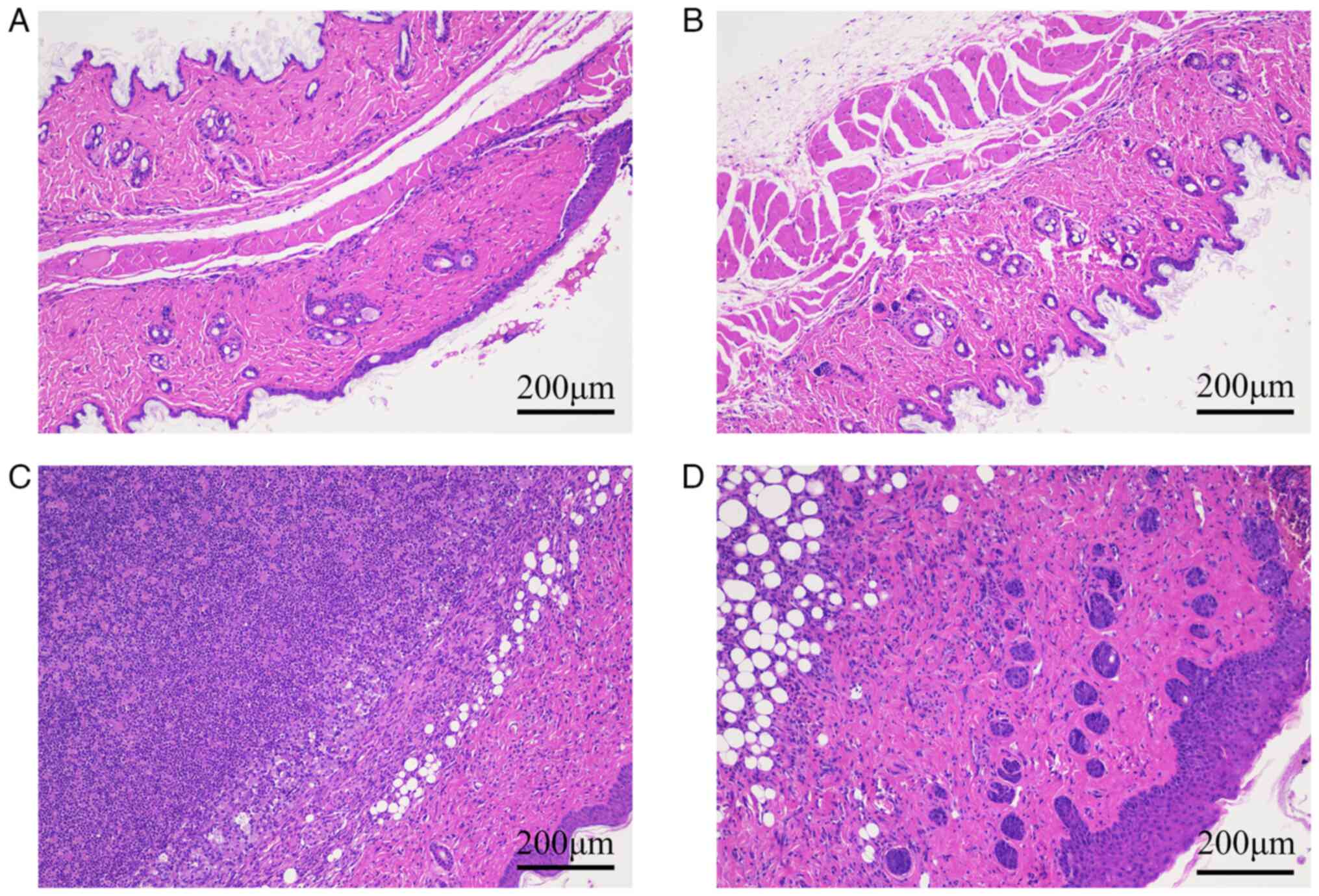
|
1
|
Hoffmann HJ, Valovirta E, Pfaar O,
Moingeon P, Schmid JM, Skaarup SH, Cardell LO, Simonsen K, Larché
M, Durham SR and Sørensen P: Novel approaches and perspectives in
allergen immunotherapy. Allergy. 72:1022–1034. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Hammad H and Lambrecht BN: Barrier
epithelial cells and the control of type 2 immunity. Immunity.
43:29–40. 2015.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Zubeldia JM, Ferrer M, Dávila I and
Justicia JL: Adjuvants in allergen-specific immunotherapy:
Modulating and enhancing the immune response. J Invest Allergol
Clin Immunol. 29:103–111. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
HogenEsch H, O'Hagan DT and Fox CB:
Optimizing the utilization of aluminum adjuvants in vaccines: You
might just get what you want. NPJ Vaccines. 3(51)2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Ashman DM, Frankland AW, Brown HM,
Feinberg JG and Langman MJ: Treatment of house dust allergy with
pyridine extracted alum precipitated mite fortified house dust
(Allpyral). Ann Allergy. 29:578–581. 1971.PubMed/NCBI
|
|
6
|
Moreno C, De San Pedro BS, Millán C,
Panizo C, Martín S and Florido F: Exploratory study of tolerability
and immunological effect of a short up-dosing immunotherapy phase
with a standardised allergen extract derived from pollen of Olea
europaea. Clin Transl Allergy. 5(27)2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Ozden MG, Kefeli M, Aydin F, Senturk N,
Canturk T and Turanli AY: Persistent subcutaneous nodules after
immunotherapy injections for allergic asthma. J Cutan Pathol.
36:812–814. 2009.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Mold M, Shardlow E and Exley C: Insight
into the cellular fate and toxicity of aluminium adjuvants used in
clinically approved human vaccinations. Sci Rep.
6(31578)2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Smith SR and Petillo J: IgE production in
five inbred rat strains following immunization with
alum-precipitated egg albumin. Int Arch Allergy Appl Immunol.
52:21–31. 1976.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Li X, Aldayel AM and Cui Z: Aluminum
hydroxide nanoparticles show a stronger vaccine adjuvant activity
than traditional aluminum hydroxide microparticles. J Control
Release. 173:148–157. 2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Yan S, Rolfe BE, Zhang B, Mohammed YH, Gu
W and Xu ZP: Polarized immune responses modulated by layered double
hydroxides nanoparticle conjugated with CpG. Biomaterials.
35:9508–9516. 2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Oreskovic Z, Nechvatalova K, Krejci J,
Kummer V and Faldyna M: Aspects of intradermal immunization with
different adjuvants: The role of dendritic cells and Th1/Th2
response. PloS One. 14(e0211896)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Hou Y, Wang Y, Tang Y, Zhou Z, Tan L, Gong
T, Zhang L and Sun X: Co-delivery of antigen and dual adjuvants by
aluminum hydroxide nanoparticles for enhanced immune responses. J
Control Release. 326:120–130. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Amini Y, Moradi B and Fasihi-Ramandi M:
Aluminum hydroxide nanoparticles show strong activity to stimulate
Th-1 immune response against tuberculosis. Artif Cells Nanomed
Biotechnol. 45:1331–1335. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Chen Y, Yang F, Yang J, Hou Y, He L, Hu H
and Lv F: Aluminum (oxy) hydroxide nanorods activate an early
immune response in pseudomonas aeruginosa vaccine. ACS Appl Mater
Interfaces. 10:43533–43542. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Hesse L and Nawijn MC: Subcutaneous and
sublingual immunotherapy in a mouse model of allergic asthma.
Methods Mol Biol. 1559:137–168. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Debeuf N, Haspeslagh E, van Helden M,
Hammad H and Lambrecht BN: Mouse models of asthma. Curr Protoc
Mouse Biol. 6:169–184. 2016.PubMed/NCBI View
Article : Google Scholar
|
|
18
|
Li X, Hufnagel S, Xu H, Valdes SA, Thakkar
SG, Cui Z and Celio H: Aluminum (Oxy)hydroxide nanosticks
synthesized in bicontinuous reverse microemulsion have potent
vaccine adjuvant activity. ACS Appl Mater Interfaces.
9:22893–22901. 2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Chevigné A, Campizi V, Szpakowska M,
Bourry D, Dumez ME, Martins JC, Matagne A, Galleni M and Jacquet A:
The Lys-Asp-Tyr triad within the mite allergen Der p. 1 propeptide
is a critical structural element for the pH-dependent initiation of
the protease maturation. Int J Mol Sci. 18(1087)2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
NOON L: Prophylactic inoculation against
hay fever. Int Arch Allergy Appl Immunol. 4:285–288.
1953.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Liu LY, Sedgwick JB, Bates ME, Vrtis RF,
Gern JE, Kita H, Jarjour NN, Busse WW and Kelly EA: Decreased
expression of membrane IL-5 receptor alpha on human eosinophils: I.
Loss of membrane IL-5 receptor alpha on airway eosinophils and
increased soluble IL-5 receptor alpha in the airway after allergen
challenge. J Immunol. 169:6452–6458. 2002.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Karani LW, Tolo FM, Karanja SM and Khayeka
CW: Safety and efficacy of prunus africana and warburgia ugandensis
against induced asthma in BALB/c Mice. European J Med Plants.
3:345–368. 2013.
|
|
23
|
Tibbitt CA, Stark JM, Martens L, Ma J,
Mold JE, Deswarte K, Oliynyk G, Feng X, Lambrecht BN, De Bleser P,
et al: Single-cell RNA sequencing of the T helper cell response to
house dust mites defines a distinct gene expression signature in
airway Th2 cells. Immunity. 51:169–184.e5. 2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Hesse L, van Ieperen N, Habraken C,
Petersen AH, Korn S, Smilda T, Goedewaagen B, Ruiters MH, van der
Graaf AC and Nawijn MC: Subcutaneous immunotherapy with purified
Der p1 and 2 suppresses type 2 immunity in a murine asthma model.
Allergy. 73:862–874. 2018.PubMed/NCBI View Article : Google Scholar
|