Introduction
Nerve cell apoptosis may occur following cerebral
ischemia or reperfusion injury as a result of the expression of
apoptosis-related proteins and a complicated process leading to
physiological and pathological alterations. The brain tissue in the
ischemic area undergoes irreversible damage within a short period
of time and develops foci of infarction. Ischemic semi-diazonal
nerve cells principally undergo delayed apoptotic neuronal death
(1), which is a key factor leading
to motor dysfunction and neurological impairment. Our previous
studies (2,3) indicated that the loci of brain tissue
damage and pathological characteristics in a rat ischemia model
coincided with human cerebral infarction loci and clinical symptoms
with regard to injury as the same target tissue is involved in both
rats and humans. Loci vulnerable to injury were shown to be the
sensitive functional areas of the cortex and hippocampus. Those
studies also demonstrated that the expressed Bax activated
caspase-3, indirectly inhibited Bcl-2 expression, formed Bax-Bcl-2
heterodimers and initiated cell apoptosis following cerebral
ischemia/reperfusion injury. Those results also confirmed the
significance of the cerebral ischemia/reperfusion injury model in
clinical diagnosis. The present study investigated the expression
of caspase-3, Bcl-2 and Bax and neuron apoptosis, as well as the
ethological alterations following cerebral ischemia-reperfusion
injury in a rat model.
Materials and methods
Materials
Sixty healthy, adult, specific pathogen-free (SPF)
Wistar rats, with a body weight of 200–240 g, were supplied by the
Experimental Animal Center of Shandong University, Jinan, China;
the in situ end-labeling TUNEL kit was purchased from Roche
Diagnostics GmbH, Mannheim, Germany; the rabbit anti-rat caspase-3
polyclonal antibody was purchased from Santa Cruz Biotechnology,
Inc. (Santa Cruz, CA, USA) and the rabbit anti-rat Bax or Bcl-2
monoclonal antibodies were obtained from Sigma-Aldrich (St. Louis,
MO, USA); the TRIzol kit, streptavidin-biotin-peroxidase complex
(SABC) immunohistochemistry kit and 3,3′-diaminobenzidine (DAB) kit
were purchased from Wuhan Boshide Biological Engineering Co. Ltd.
(Wuhan, China); a BX20 microscope camera was provided by Olympus
(Tokyo, Japan); a paraffin microtome was obtained from Microm
International GmbH (Walldorf, Germany); the GeneAmp PCR system 2400
was purchased from Perkin Elmer (Waltham, MA, USA); the Bio-Gel
Analysis system was purchased from Bio-Rad (Hercules, CA, USA); and
the HPIAS-1000 Image Analysis system was purchased from Wuhan
Tongji Qianping Image Engineering (Wuhan, China). All animal
experiments were approved by the Medical Ethics Committee of
Qingdao University Medical College, Qingdao, China (QUMC
2011-09).
Animal grouping
Sixty male healthy adult Wistar rats were randomly
assigned into control (n=6), sham operation (n=6) and experimental
(n=48) groups. The experimental group was randomly subdivided into
eight subgroups according to reperfusion time points (3, 6, 12, 24
and 48 h and 3, 7 and 14 days), with 6 rats-subgroup. The animal
models of middle cerebral artery occlusion/reperfusion (MCAO/R)
were performed according to the Zea-Longa method (2).
Establishment of the animal model
The cerebral ischemia rat model was performed
according to the Zea-Longa method (4). Following deep anesthesia of the rat
bodies, the neck was opened in the midline, the right common
carotid artery, external carotid artery (ECA), internal carotid
artery (ICA) and the branches of ECA and ICA were separated, the
ECA was sheared and a monofilament line was inserted to interrupt
the flow through the middle cerebral artery (MCA). Reperfusion was
permitted after 1 h of ischemia. The sham operation group only
underwent separation of the blood vessels without any obstruction.
The rats in the normal control group did not undergo any
operation.
The rats developed a left-sided Horner syndrome.
Flexing or circular crawling of the right anterior limb towards the
right when the tail was lifted was considered a sign of a
successful model. All the experimental procedures were performed in
accordance with animal ethical standards.
Score of neurological function
When the tail was gently lifted 10 cm from the
table, the normal rats extended their two forepaws on the table.
According to the Bederson score standard (5), the scores were classified as 0–3.
Function test of balance and
coordination
The rats were placed on an 80-cm long and 2.4-cm
wide balance beam, situated 10 cm above the ground. The experiment
was performed according to the Altumbabic method (6). According to the Freeney score standard
(7), the scores were classified as
0–5.
Measurement of forelimb placing
Referring to the Bland experiment (8), the investigator held the rat by the
dorsal skin to suspend the limbs, with the vibrissae touching the
table edge to detect ipsilateral forelimb activity. In response to
the vibrissae lightly brushing the edge of the table, the forepaws
of normal rats quickly touched the table, whereas the response of
those with brain ischemic injury were affected to some extent. Each
side of each rat was tested 10 times and the score was based on the
percentage of times the forepaws touched the table edge. According
to the Basso-Beattie-Bresnahan (BBB) score standard (9), the scores were classified as 0–21.
Preparation of paraffin sections
Six rats in each subgroup were deeply anesthetized
by intraperitoneal injection of 10% chloral hydrate (0.6 ml/100 g)
and perfused through the heart with phosphate-buffered solution,
followed by 4% paraformaldehyde solution to fix the brain. The rats
were then decapitated and middle brain tissue was collected. The
tissue was continuously fixed in 4% paraformaldehyde for 2 h,
immersed in double-distilled water for 4 h, underwent conventional
gradient ethanol dehydration, were transparented in dimethylbenzene
and embedded in paraffin. Coronal 6-μm sections through the
cortical area and the hippocampus were successively cut with a
cryostat from the posterior of the optic chiasma and were then
investigated with immunohistochemistry. The positive control
section was provided by an immunohistochemistry agent and the
negative control was set up by 0.01 mol/l PBS replacing the primary
antibody to eliminate false-positive or false-negative results.
Cell apoptosis assay
The TUNEL staining method was employed to detect
cell apoptosis. The procedure was performed according to the
manufacturer’s instructions (Roche Diagnostics GmbH). Following
TUNEL staining, the sections were examined under a light microscope
to observe apoptotic neurons in the cortex and hippocampus.
Immunohistochemistry assay
Rabbit anti-rat caspase-3, Bax and Bcl-2
immunohistochemistry kits were employed to detect the expression of
the three proteins. Assay procedures were performed according to
the manufacturer’s instructions. Following immunohistochemical
staining, the brain tissue sections were placed under the
microscope to observe caspase-3-, Bax- and Bcl-2-positive neurons
in the cortical area and the CA1, stratum lucidum (Slu) and
molecular layer of the dentate gyrus (MoDG) of the hippocampus. The
HPIAS-1000 Image Analysis system was used to calculate the total
number of positive cells in the ischemic cortical area and the
hippocampus.
Semiquantitive reverse transcriptase
polymerase chain reaction (RT-PCR)
The sequences of the caspase-3, Bax, Bcl-2 and
β-actin genes were obtained from GeneBank. RT-PCR primers were
designed by Primer Premier 5.0 software (Premier Biosoft
International, CA, USA) to amplify gene mRNA. Total RNA was
isolated by the TRIzol kit. RT-PCR was performed according to the
manufacturer’s instructions (Perkin Elmer). The RT-PCR product was
analyzed by 1% agarose gel electrophoresis, images were captured by
Bio-Gel analysis system, the absorbance of each band was measured
with the analysis system, compared with the internal control
β-actin and statistical analysis was performed between the two
groups.
Statistical analysis
Data analyses were peformed with SPSS software,
version 14.0 (SPSS Inc., Chicago, IL, USA). Data were compared with
the χ2 test and are expressed as means ± standard
deviation. Analysis of variance (ANOVA) was used to compare
variables within a group and between groups. P<0.05 was
considered to indicate a statistically significant difference.
Results
Neurological function score
Compared to the sham operation group, the resistance
to side push on the paralyzed side was decreased following 3 h of
reperfusion in the experimental subgroup, accompanied by anterior
limb flexing, with a distinct score difference (P<0.01). The
spontaneous rotation accompanied with walking for the 6–48 h
subgroups after reperfusion, showed a distinct difference in the
function scores. The neurological function gradually improved with
reperfusion lasting 3–14 days (Table
I).
 | Table IEthology scores in the
ischemia/reperfusion experimental and sham operation groups at
different time points (mean ± standard deviation). |
Table I
Ethology scores in the
ischemia/reperfusion experimental and sham operation groups at
different time points (mean ± standard deviation).
| | Neurological function
score | Balance beam walking
score | Forelimb placing test
(%) |
|---|
| |
|
|
|
|---|
| Time points | No. | Sham | Experimental | Sham | Experimental | Sham | Experimental |
|---|
| 3 h (I/R) | 6 | 1.0±0.5 | 3.0±0.3a | 1.0±0.4 | 3.0±0.5a | 8.0±0.2 | 5.0±0.2a |
| 6 h (I/R) | 6 | 0.0±0.0 | 2.0±0.4 | 0.0±0.0 | 2.0±0.4 | 8.0±0.3 | 6.0±0.4a |
| 12 h (I/R) | 6 | 0.0±0.0 | 2.0±0.2 | 0.0±0.0 | 2.0±0.3 | 9.0±0.4 | 7.0±0.3a |
| 24 h (I/R) | 6 | 0.0±0.0 | 1.0±0.3 | 0.0±0.0 | 1.0±0.4 | 9.0±0.5 | 7.0±0.5a |
| 48 h (I/R) | 6 | 0.0±0.0 | 1.0±0.2 | 0.0±0.0 | 1.0±0.3 | 9.0±0.3 | 8.0±0.3b |
| 3 d (I/R) | 6 | 0.0±0.0 | 0.0±0.0 | 0.0±0.0 | 0.0±0.0 | 9.0±0.4 | 9.0±0.4 |
| 7 d (I/R) | 6 | 0.0±0.0 | 0.0±0.0 | 0.0±0.0 | 0.0±0.0 | 9.0±0.2 | 9.0±0.5 |
| 14 d (I/R) | 6 | 0.0±0.0 | 0.0±0.0 | 0.0±0.0 | 0.0±0.0 | 9.0±0.3 | 9.0±0.4 |
Balance beam walking test
Compared to the sham operation group, when the
experimental rats walked on a balance beam at 3 h following
reperfusion, the anterior limb on the paralyzed side was not able
to move forward, with a distinct difference in the function scores
(P<0.01). At 6–48 h following reperfusion the rats were not able
to walk on the balance beam, although they were able to sit on it,
with a distinct difference in the scores. The motor ability
significantly improved with reperfusion lasting 3–14 days (Table I).
Measurement of forelimb placing
Compared to the sham operation group, the positive
percentage of the rats that were able to touch the table angle or
edge with their anterior limbs at 3 h following reperfusion was
extremely low (P<0.01), whereas that at 6–48 h reperfusion was
notably increased, with a significant difference in the scores
(P<0.05). The percentage following 3 days of reperfusion was
identical to that of the sham operation and control groups
(Table I).
BBB score
Compared to the sham operation and control groups,
the BBB score was distinctly decreased in the experimental group
following reperfusion for 3–12 h (P<0.01) and 24–48 h
(P<0.05). The differences in the scores were not significantly
different following reperfusion for 3–14 days (P<0.05) (Table II).
 | Table IIBasso, Beattie and Bresnahan (BBB)
score following reperfusion at different time points in each group
(mean ± standard deviation). |
Table II
Basso, Beattie and Bresnahan (BBB)
score following reperfusion at different time points in each group
(mean ± standard deviation).
| | Groups |
|---|
| |
|
|---|
| Time points | No. | Control | Sham | Experimental |
|---|
| 3 h (I/R) | 6 | 20.0±3.0 | 18.0±1.0 | 9.0±2.0a |
| 6 h (I/R) | 6 | 20.0±3.0 | 18.0±1.0 | 11.0±2.0a |
| 12 h (I/R) | 6 | 21.0±4.0 | 19.0±1.0 | 13.0±1.0a |
| 24 h (I/R) | 6 | 20.0±3.0 | 19.0±2.0 | 17.0±1.0b |
| 48 h (I/R) | 6 | 21.0±4.0 | 20.0±2.0 | 18.0±2.0b |
| 3 d (I/R) | 6 | 20.0±3.0 | 20.0±3.0 | 19.0±2.0c |
| 7 d (I/R) | 6 | 20.0±4.0 | 20.0±3.0 | 19.0±3.0c |
| 14 d (I/R) | 6 | 21.0±3.0 | 21.0±4.0 | 20.0±4.0c |
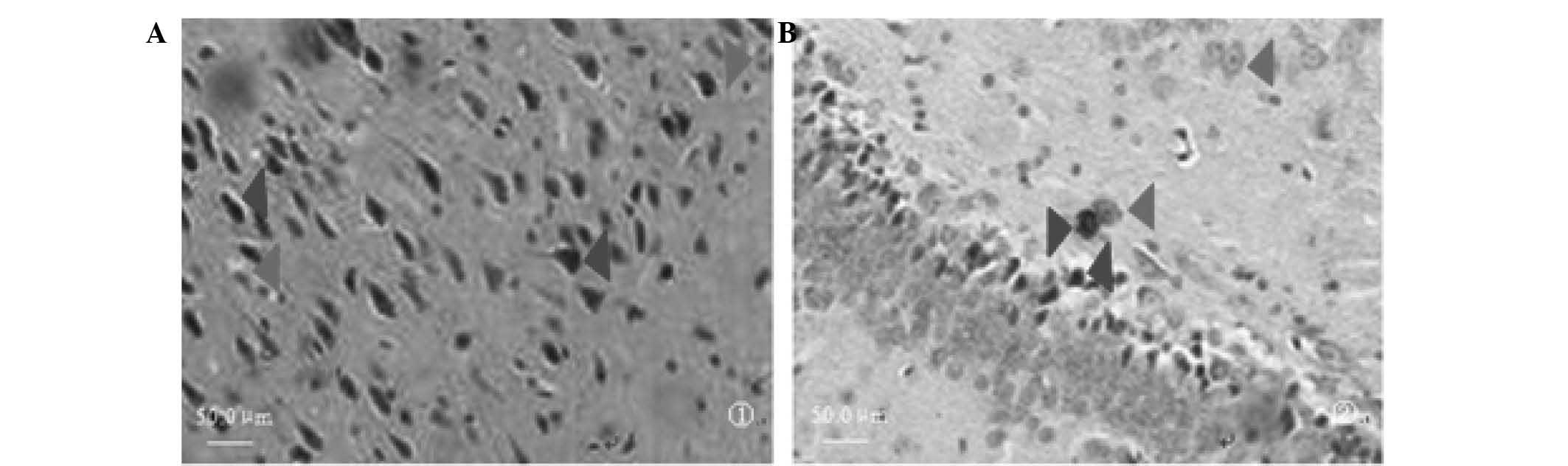
Expression of TUNEL-positive neurons in
ischemic regions
Compared to the sham operation group, the expression
of TUNEL-positive neurons in the cortex and the CA3 region of the
hippocampus was not significantly different (P<0.05) following
reperfusion for 3 h. The expression was markedly increased during
reperfusion for 6 h–7 days, reaching a peak at 48 h (P<0.01).
The number of TUNEL-positive neurons following reperfusion for 14
days did not differ significantly between the groups (P<0.05)
(Table III, Fig. 1A and B).
 | Table IIIExpression of TUNEL-positive neurons
in the cortex and CA3 region of the hippocampus at different time
points following ischemia/reperfusion (mean ± standard
deviation). |
Table III
Expression of TUNEL-positive neurons
in the cortex and CA3 region of the hippocampus at different time
points following ischemia/reperfusion (mean ± standard
deviation).
| Time points | No. | Cortex | Hippocampus |
|---|
|
|
|---|
| Control | Sham | Experimental | Control | Sham | Experimental |
|---|
| 3 h (I/R) | 6 | 5.72±1.10 | 6.30±1.14 | 8.75±1.12a | 5.50±1.21 | 6.21±1.14 | 8.64±1.17a |
| 6 h (I/R) | 6 | 6.36±1.17 | 6.75±1.23 | 13.63±2.27b | 6.21±1.34 | 6.60±1.31 | 13.34±2.83b |
| 12 h (I/R) | 6 | 6.39±1.46 | 7.18±1.35 | 17.42±3.87b | 6.32±1.23 | 7.04±1.41 | 15.17±3.21b |
| 24 h (I/R) | 6 | 7.63±1.35 | 7.57±1.65 | 19.71±4.24b | 7.44±1.25 | 7.11±1.19 | 17.60±4.41b |
| 48 h (I/R) | 6 | 7.27±1.22 | 8.08±1.74 | 23.80±5.20b | 7.15±1.27 | 8.00±1.36 | 21.52±5.71b,c |
| 3 d (I/R) | 6 | 6.84±1.46 | 7.42±1.35 | 16.64±3.56b | 6.70±1.39 | 7.34±1.53 | 15.61±3.52b |
| 7 d (I/R) | 6 | 6.27±1.32 | 7.17±1.43 | 12.32±3.34b | 5.20±1.25 | 7.05±1.48 | 11.31±2.01b |
| 14 d (I/R) | 6 | 5.53±1.28 | 6.28±1.52 | 7.12±1.31 | 5.41±1.14 | 6.21±1.31 | 7.09±1.25 |
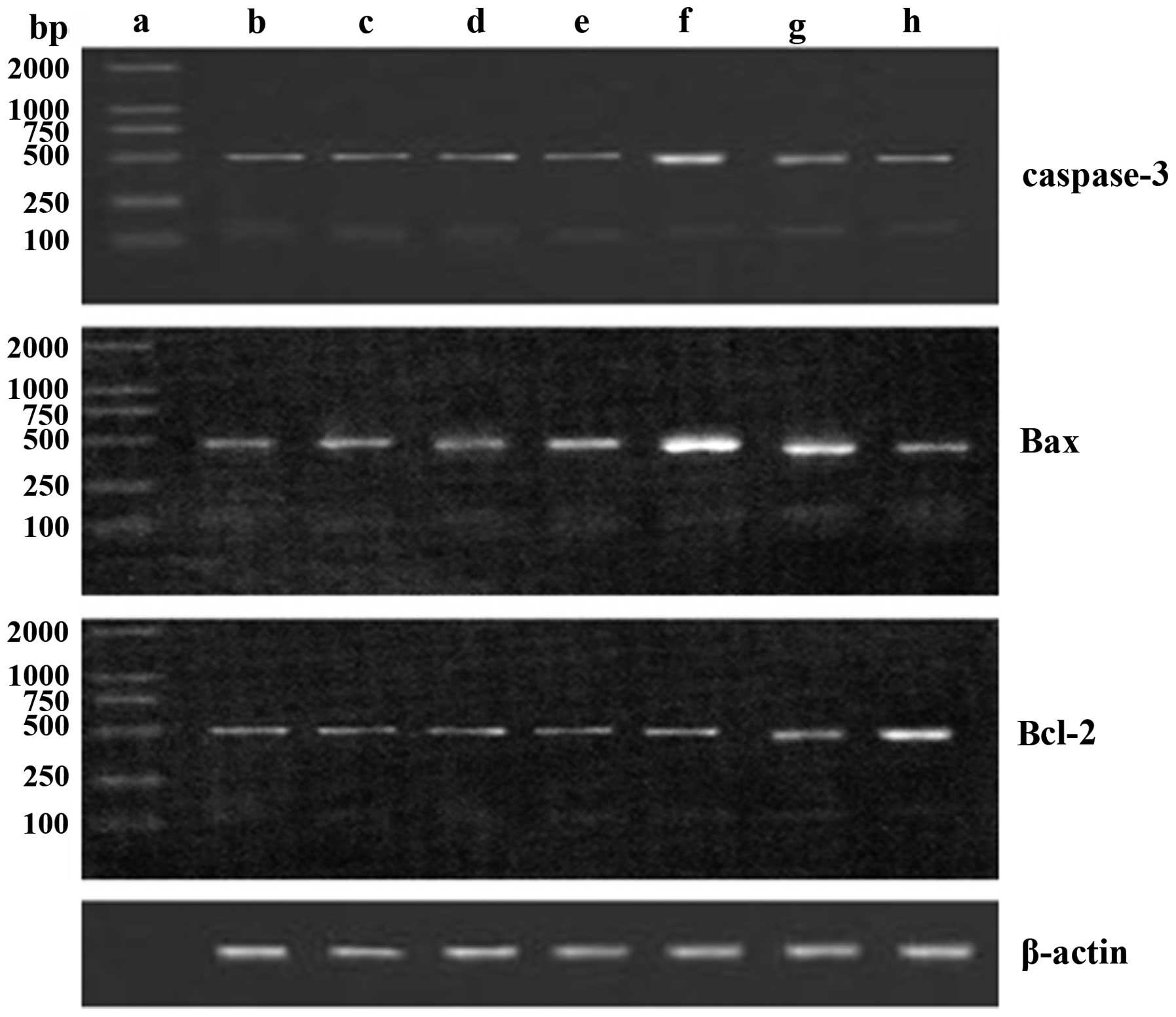
RT-PCR of apoptosis-related gene mRNA in
the cerebral cortex and hippocampus
RT-PCR demonstrated positive caspase-3, Bax, Bcl-2
and β-actin mRNA expression in the cerebral cortex and hippocampus
of the rats in each group (Fig.
2).
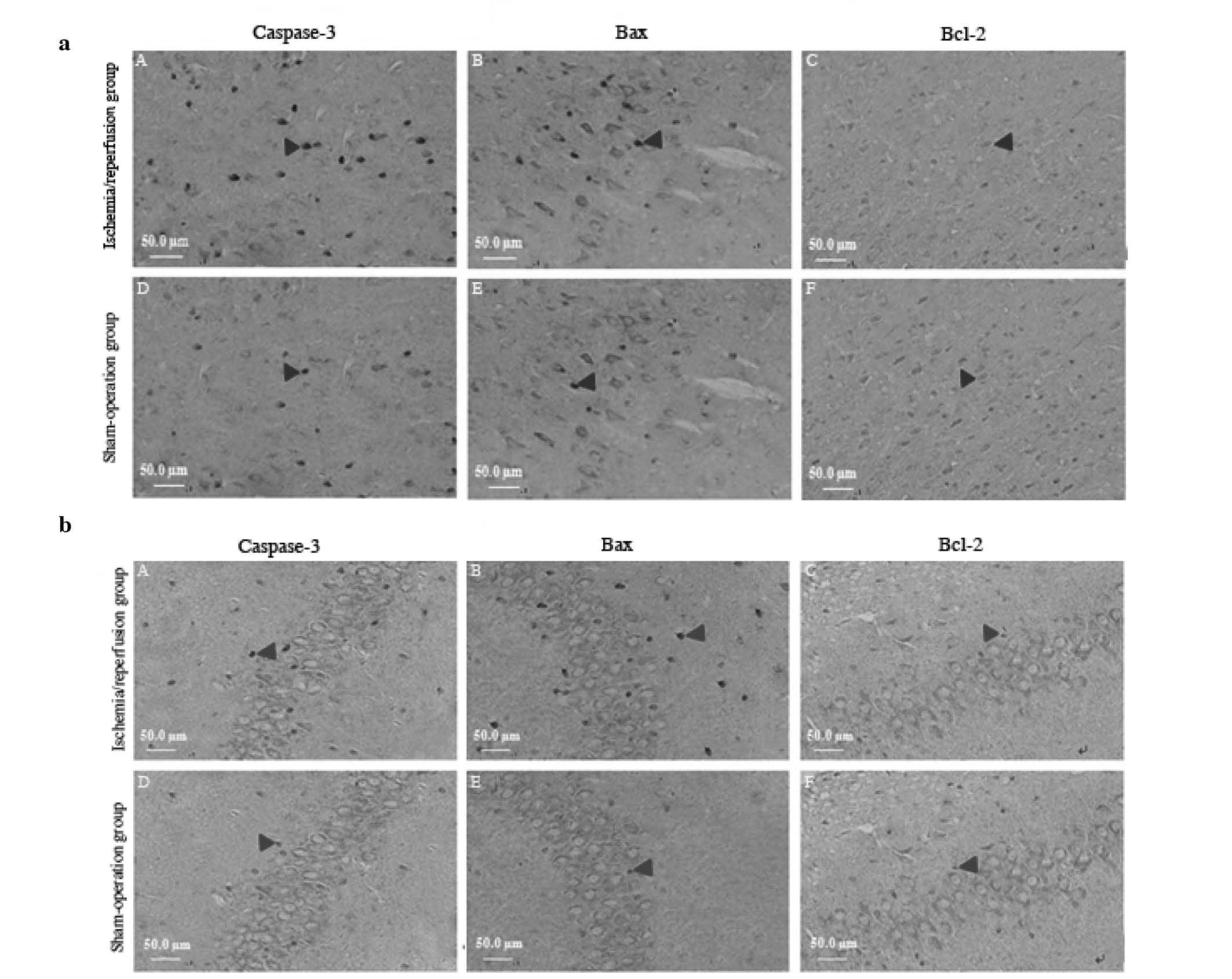
Expression of caspase-3-, Bax- and
Bcl-2-positive neurons in the cerebral cortex and hippocampus
Compared to the sham operation group, the expression
of caspase-3-positive neurons was distinctly different at 3 h
following reperfusion in the cerebral cortex and the CA1 region of
the hippocampus (P<0.05) and was further increased following
reperfusion for 6 h–7 days (Fig. 3aA
and bA), reaching a peak at 48 h (P<0.01), decreasing
approximately to sham operation or normal control group levels at
14 days (Table IV).
 | Table IVExpression of caspase-3-positive
neurons at different time points in the cortex and the hippocampus
following ischemia-reperfusion (mean ± standard deviation). |
Table IV
Expression of caspase-3-positive
neurons at different time points in the cortex and the hippocampus
following ischemia-reperfusion (mean ± standard deviation).
| Time points | No. | Cortex | Hippocampus |
|---|
|
|
|---|
| Control | Sham | Experimental | Control | Sham | Experimental |
|---|
| 3 h (I/R) | 6 | 5.51±1.01 | 6.26±1.10 | 8.80±1.08a | 5.48±1.03 | 6.15±1.06 | 8.71±1.00a |
| 6 h (I/R) | 6 | 5.23±1.24 | 6.13±1.12 | 13.91±2.83b | 5.20±1.26 | 6.08±1.12 | 11.11±2.01b |
| 12 h (I/R) | 6 | 6.38±1.17 | 7.16±1.41 | 15.40±3.15b | 6.34±1.18 | 7.10±1.14 | 13.23±2.23b |
| 24 h (I/R) | 6 | 7.54±1.10 | 8.10±1.26 | 18.25±3.45b | 7.16±1.20 | 8.03±1.23 | 17.60±3.07b |
| 48 h (I/R) | 6 | 7.12±1.41 | 8.12±1.71 | 20.08±3.78b,c | 7.33±1.35 | 8.07±1.54 | 19.10±3.31b,c |
| 3 d (I/R) | 6 | 6.51±1.36 | 7.34±1.50 | 15.67±2.32b | 6.35±1.26 | 7.21±1.27 | 13.09±2.17b |
| 7 d (I/R) | 6 | 5.13±1.25 | 6.23±1.66 | 12.70±2.11b | 6.10±1.27 | 6.19±1.20 | 10.38±2.25b |
| 14 d (I/R) | 6 | 5.49±1.28 | 6.10±1.09 | 8.11±1.19 | 5.05±1.22 | 6.02±1.21 | 8.03±1.12 |
Compared to the sham operation group, the expression
of Bax-positive neurons did not differ significantly at 3 h or 14
days of reperfusion in the cerebral cortex and the Slu of the
hippocampus (P<0.05), although it was higher at 6, 12, 24, 48 h
and 7 days, with a peak at 48 h after ischemia/reperfusion
(P<0.01) (Fig. 3aB and bB,
Table V).
 | Table VExpression of Bax-positive neurons at
different time points in the cortex and the hippocampus following
ischemia-reperfusion (mean ± standard deviation). |
Table V
Expression of Bax-positive neurons at
different time points in the cortex and the hippocampus following
ischemia-reperfusion (mean ± standard deviation).
| Time points | No. | Cortex | Hippocampus |
|---|
|
|
|---|
| Control | Sham | Experimental | Control | Sham | Experimental |
|---|
| 3 h (I/R) | 6 | 5.23±1.22 | 6.47±1.05 | 8.70±1.03a | 5.20±1.28 | 6.35±1.11 | 8.59±1.01a |
| 6 h (I/R) | 6 | 6.27±1.21 | 7.23±1.24 | 11.53±2.11a | 6.21±1.34 | 7.12±1.34 | 10.23±2.07a |
| 12 h (I/R) | 6 | 7.34±1.45 | 8.51±1.51 | 12.33±3.10a | 6.42±1.37 | 8.49±1.08 | 12.23±2.12a |
| 24 h (I/R) | 6 | 6.33±1.37 | 7.71±1.49 | 15.61±3.14b | 5.31±1.27 | 7.60±1.14 | 14.38±3.21b |
| 48 h (I/R) | 6 | 7.46±1.32 | 8.81±1.75 | 21.16±3.24b,c | 6.43±1.43 | 7.27±1.31 | 19.08±3.32b,c |
| 3 d (I/R) | 6 | 6.12±1.54 | 8.37±1.46 | 12.94±3.31a | 6.10±1.35 | 8.02±1.43 | 11.12±3.71a |
| 7 d (I/R) | 6 | 5.37±1.48 | 7.54±1.32 | 10.62±2.15a | 5.28±1.21 | 7.36±1.17 | 9.82±2.20a |
| 14 d (I/R) | 6 | 5.20±1.35 | 6.35±1.06 | 7.18±1.00 | 5.13±1.36 | 6.21±1.06 | 5.02±1.01 |
Compared to the sham operation group, the expression
of Bcl-2-positive neurons did not differ at 3 h or 14 days of
reperfusion in the cerebral cortex and the Slu of the hippocampus
(P<0.05), although it was lower at 6, 12, 24, 48 h and 7 days,
with the lowest at 48 h after ischemia/reperfusion (P<0.01)
(Fig. 3aC and bC, Table VI).
 | Table VIExpression of Bcl-2-positive neurons
at various time points in the cortex and the hippocampus following
ischemia-reperfusion (mean ± standard deviation). |
Table VI
Expression of Bcl-2-positive neurons
at various time points in the cortex and the hippocampus following
ischemia-reperfusion (mean ± standard deviation).
| Time points | No. | Cortex | Hippocampus |
|---|
|
|
|---|
| Control | Sham | Experimental | Control | Sham | Experimental |
|---|
| 3 h (I/R) | 6 | 24.13±1.32 | 23.32±1.21 | 21.13±1.06a | 24.06±1.35 | 23.17±1.28 | 20.08±2.63a |
| 6 h (I/R) | 6 | 21.75±1.54 | 21.47±1.27 | 19.23±3.42a | 21.10±1.51 | 22.23±1.51 | 18.07±2.31a |
| 12 h (I/R) | 6 | 22.21±1.47 | 20.55±1.40 | 18.12±3.24a | 22.18±1.42 | 21.07±1.21 | 16.17±3.07a |
| 24 h (I/R) | 6 | 23.47±1.44 | 21.29±1.42 | 12.73±2.16b | 22.10±1.46 | 20.11±1.32 | 11.62±3.32b |
| 48 h (I/R) | 6 | 21.29±1.35 | 20.15±1.30 | 9.94±2.17b,c | 21.21±1.41 | 20.14±1.26 | 9.75±2.43b,c |
| 3 d (I/R) | 6 | 22.84±1.23 | 21.51±1.34 | 17.41±2.13a | 23.70±1.50 | 21.24±1.33 | 17.12±4.01a |
| 7 d (I/R) | 6 | 23.68±1.58 | 22.23±1.15 | 19.21±3.01a | 22.43±1.57 | 22.20±1.19 | 19.01±2.38a |
| 14 d (I/R) | 6 | 21.36±1.47 | 21.17±1.38 | 20.98±1.04 | 21.21±1.43 | 21.11±1.20 | 20.16±1.31 |
Discussion
It is crucial to establish an MCAO/R animal model
according to the clinical characteristics of a human cerebral
natural infarct. The model characteristics are as follows: i)
single damage mechanism that is easy to study; ii) simple method,
small wound, easy to control condition, stable infarct site, and
distinct symptomatic reaction and high achievement ratio; iii)
uniformity of cerebral infarction and good reproducibility; and iv)
necrotic brain tissue following injury, with a similar
pathophysiological process to clinical cerebral ischemia. In this
study, a rat cerebral ischemia-reperfusion injury model was
established according to the Zea-Longa method (4). Following cerebral ischemia for 1 h,
the rats developed severe nervous and behavioral functional
impairment symptoms, indicating the establishment of a successful
model.
The Bederson (5)
score method was employed for qualitative and semiquantitative
evaluation, with particular emphasis on motor function evaluation.
The balance beam walking assay evaluated motor reaction speed, fine
motor coordination ability and balance under surrounding stimuli.
The anterior limb-placing assay evaluated reaction ability, vision,
touch and proprioceptive sensation. A normal reaction was
considered the anterior limb immediately gripping the tabletop and
the behavioral response of injured rats was retardation. The
results demonstrated that the scores of neurological function and
balance beam walking test were decreased from 3 to 1 at 6–48 h
following cerebral ischemia/reperfusion in experimental rats,
although that of the forelimb placing assay was distinctly
increased at 6–12 h of reperfusion, which indicates that
reperfusion for 6–48 h is the time window causing injury of the
cortical and hippocampal vulnerable regions. Motor behavior
functional impairment was closely correlated with reperfusion
injury.
The order of vulnerability of the hippocampal
subsectors to global ischemia is as follows: CAl,
hilus>CA2>CA3>dentate gyrus (10). A recent study by Xie et
al(11) demonstrated that the
hippocampal CA1 area ipsilateral to the occluded MCA may undergo
delayed neuronal death and the process may be due to the spread of
injury signals through the astrocytic gap junction from the infarct
region to the remote hippocampus. Neurological impairment mediated
by neuron apoptosis was confirmed by the experiment. The results
demonstrated that TUNEL-positive cells reached a peak expression at
48 h following reperfusion in the cortical area and the CA3 of the
hippocampus, which is in accordance with the results of Ou Yang
et al(12) and indicates
that the energy did not exhaust but inhibited protein metabolism,
leading to neuron apoptosis ~48 h after reperfusion. The time point
of 48 h after reperfusion may be the key time circumscription
resulting in pathological changes of the cortex and
hippocampus.
Caspase-3 is an important protein in the apoptotic
enzyme cascade and regulates neuron apoptosis. Our results have
demonstrated that the expression of caspase-3 and neuron apoptosis
reached a peak at ~48 h after reperfusion in the cortex and the CA1
and CA3 regions of the hippocampus. The disposition of the two was
considered a coincidence and was consistent with the experiment of
Choi et al(13). This
finding indicated that ischemia/reperfusion injury enhanced
caspase-3 activity or induced its expression, promoting neuron
apoptosis.
The Bax protein exerts an important effect in the
regulation of cell apoptosis. High Bax expression levels and
formation of homo- or heterodimers with Bcl-2 may lead to cell
death (14,15). It was previously demonstrated that
Bax is highly expressed at 24–72 h following reperfusion (16). Our results have demonstrated that
the expression of Bax was distinctly increased at 48 h following
reperfusion in the cortical area and the Slu of the hippocampus,
which was consistent with those results. The time and disposition
of Bax expression coincided with neuron TUNEL positivity. This
indicated that the expression of Bax altered the balance between
the genes promoting and those inhibiting apoptosis, inducing or
accelerating neuron apoptosis and possibly increasing the activity
of caspase-3.
Bcl-2 is the key cell apoptosis inhibitory protein.
Its regulatory effect depends on its expression, although the
Bcl-2-Bax ratio is also considered a key factor. When the Bcl-2-Bax
ratio increases, cell apoptosis is inhibited and when the ratio is
decreased, apoptosis is promoted. The expression levels of Bcl-2
were low at 48 h following reperfusion in the cortex and the MoDG
of the hippocampus, in accordance with the results of Qin et
al(15). This may be the reason
that the low levels of Bcl-2 expression and the decrease of the
Bcl-2/Bax ratio resulted in neuron apoptosis.
In conclusion, the successful establishment of a
cerebral ischemia/reperfusion injury animal model and the different
expressions of caspase-3, Bax and BcL-2 following
ischemia-reperfusion may provide important reference evidence on
the etiopathogenesis of clinical cerebral thrombosis.
References
|
1
|
Candelario-Jalil E: Injury and repair
mechanisms in ischemic stroke: considerations for the development
of novel neurotherapeutics. Curr Opin Investig Drugs. 10:644–654.
2009.PubMed/NCBI
|
|
2
|
Li Q, Zhang R, Guo YL and Mei YW: Effect
of neuregulin on apoptosis and expressions of STAT3 and GFAP in
rats following cerebral ischemic reperfusion. J Mol Neurosci.
37:67–73. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Li Q, Zhang R, Ge YL, Mei YW and Guo YL:
Effects of neuregulin on expression of MMP-9 and NSE in brain of
ischemia/reperfusion rat. J Mol Neurosci. 38:207–215. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Longa EZ, Weistein PR, Calson S, et al:
Reversible middle cerebral artery occlusion without craniectomy in
rats. Stroke. 20:84–89. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bederson JB, Pitts LH, Tsuji M, et al: Rat
middle cerebral artery occlusion: evaluation of the model and
development of a neurologic examination. Stroke. 17:472–476. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Altumbabic M, Peeling J, Del Bigio MR, et
al: Intracerebral hemorrhage in the rat: effects of hematoma
aspiration. Stroke. 29:1917–1922. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Freeney DM, Boyeson MG, Linn RT, et al:
Responses to cortical injury: I. Methodology and local effects of
contusions in the rat. Brain Res. 211:67–77. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bland ST, Schallert T, Strong R, et al:
Early exclusive use of the affected forelimb after moderate
transient focal ischemia in rat: function and anatomic outcome.
Stroke. 31:1144–1152. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Basso DM, Beattie MS and Bresnahan JC: A
sensitive and reliable locomotor rating scale for open filed
testing in rats. J Neurotrauma. 12:1–21. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Schmidt-Kastner R and Freund TF: Selective
vulnerability of the hippocampus in brain ischemia. Neuroscience.
40:599–636. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Xie M, Yi C, Luo X, et al: Glial gap
junctional communication involvement in hippocampal damage after
middle cerebral artery occlusion. Ann Neurol. 70:121–132. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ou-Yang FL, Zhou XZ, Fang SZ, Cai YQ and
Li H: Long-term behavioral and ultrastructural alterations
following hypoxic-ischemic brain damage in neonatal rats. Zhongguo
Dang Dai Er Ke Za Zhi. 14:380–384. 2012.(In Chinese).
|
|
13
|
Choi JY, Kim BM, Kim YJ, et al:
Hypoxia/reoxygenation-induced cytotoxicity in cultured human
lymphocytes. Biochem Biophys Res Commun. 352:366–371. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Haddad JJ: The role of Bax/Bcl-2 and
pro-caspase peptides in hypoxia/reperfusion-dependent regulation of
MAPK (ERK): discordant proteomic effect of MAPK (p38). Protein Pept
Lett. 14:361–371. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Qin AP, Zhang HL and Qin ZH: Mechanisms of
lysosomal proteases participating in cerebral ischemia-induced
neuronal death. Neurosci Bull. 24:117–123. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xing B, Chen H, Zhang M, Zhao D, Jiang R,
Liu X and Zhang S: Ischemic postconditioning inhibits apoptosis
after focal cerebral ischemia/reperfusion injury in the rat.
Stroke. 39:2362–2369. 2008. View Article : Google Scholar : PubMed/NCBI
|