Introduction
Helicobacter pylori (H. pylori) is a
Gram-negative bacterium that persistently colonizes in the stomachs
of more than half the human population (1). Colonization of the stomach by H.
pylori consistently induces gastric inflammation, known as
superficial chronic gastritis, and is a risk factor for the
development of peptic ulcer disease and gastric malignancies
(2). H. pylori strains are
thought to possess various virulence factors, which contribute to
digestive disease complications (3).
In 2005, it was reported that H. pylori hopQ genotypes are
associated with an increased risk for peptic ulcer disease
(4). H. pylori genomes contain
~30 paralogous hop genes, which encode outer membrane
proteins (5). HopQ is localized at the
surface of H. pylori (6). The
H. pylori outer membrane protein (Hop) family is the largest
and includes adhesions such as BabA (HopS) (7), SabA (HopP) (8), OipA (HopH) (9), AlpAB (HopB and HopC) (10) and HopQ (11). However, nothing is known regarding the
functional properties of HopQ and this study did not provide
further data regarding disease-specific virulence factor of
hopQ. H. pylori hopQ alleles exhibit a high level of
genetic diversity, and two families of hopQ alleles have
been described (type I hopQ and type II hopQ)
(12). Iran is a developing country
with a high prevalence of H. pylori infections, among
symptomatic and asymptomatic individuals, and the prevalence is
≤90% in the northwestern part of the country (13). The aim of the present study was to
determine the association between H. pylori virulence type I
and type II hopQ genotypes and patients with different
gastrointestinal diseases.
Materials and methods
Patients
In the study, patients undergoing upper gastric
endoscopy due to different digestive diseases that visited the
hospitals in Tabriz (Iran) were included. The standard number of
gastric biopsy samples for patients who were suspected of being
infected with H. pylori was obtained for routine culture and
histological investigations. Patient groups according to endoscopic
and pathology findings were: Gastric ulcer (GU), duodenal ulcer
(DU), gastritis and gastric carcinoma (GC). Written informed
consent was obtained from all the patients prior to entering the
study, and the study was approved by the Regional Ethics Committee,
Tabriz University of Medical Sciences (Tabriz, Iran; no. 5/47/1375,
April 4, 2015).
H. pylori isolates and bacterial culture. The
presence of H. pylori was determined by cultures placed on
Brucella agar (Merck KGaA, Darmstadt, Germany) containing 5% sheep
blood and antibiotics such as amphotericin B (5 mg/ml),
trimethoprim lactate (5 mg/l), vancomycin (10 mg/ml) and
polymixin-B (2,500 U/ml). Plates were incubated at 37°C, in 10%
CO2 conditions, and were subsequently identified as
H. pylori, based on: Colony morphology, Gram-staining and
positive oxidase, catalase and urease tests. In the present study,
the H. pylori American Type Culture Collection (ATCC) 43504
strain (ATCC, Manassas, VA, USA) was used as a reference.
DNA extraction and polymerase chain
reaction (PCR)
Bacterial DNA was extracted from single colonies of
H. pylori and their DNA was extracted by sodium dodecyl
sulphate, proteinase K and the cetyltrimethyl ammonium bromide
method (14) and stored at −20°C.
Initially, the PCR assay was used to detect the H.
pylori-specific ureC (glmM) gene for confirmation
of H. pylori strains (15). In
this experiment, hopQ type I and type II genes were detected
by the PCR method (16,17) (Table I),
under the following conditions for both genes: 35 cycles at 94°C
for 60 sec, at 54°C for 45 sec, and at 72°C for 65 sec, and a final
extension at 72°C for 10 min. PCR products were analyzed on 1.5%
agarose and the strains containing the hopQ type I and
hopQ type II genes were used as a positive control.
 | Table I.Amplification primers for
amplification used in the study. |
Table I.
Amplification primers for
amplification used in the study.
| Study, year | DNA region
amplified | Primers | Primer sequence | Refs. |
|---|
| Kidd et al,
1999 | glmM | HP-F |
AAGCTTTTAGGGGTGTTAGGGGTTT | (16) |
|
|
| HP-R |
AAGCTTACTTTCTAACACTAACGC |
|
| Sicinschi et
al, 2012 | Type I
hopQ | HP-F |
CAACGATAATGGCACAAACT | (17) |
|
|
| HP-R |
GTCGTATCAATAACAGAAGTTG |
|
| Sicinschi et
al, 2012 | Type II
hopQ | HP-F |
TCCAATCCAGAAGCGATTAA | (17) |
|
|
| HP-R |
GTTTTAATGGTTACTTCCACC |
|
Statistical analysis
The data obtained were analyzed by SPSS (version 19;
IBM, Corp., Armonk, NY, USA) and the χ2 test and
Fisher's exact test was used to compare the clinical outcomes and
the presence of genes. P<0.05 was considered to indicate a
statistically significant difference.
Results
H. pylori culture positive
From 286 gastric biopsies, 88 (30.76%) were positive
for H. pylori. Of the patients (including 88 H.
pylori-positive) that attended the endoscopy ward, 6 suffered
from anemia (6.8%), 24 from epigastria pain (27.3%), 10 from
gastro-esophageal reflux disease (11.4%), 46 from dyspepsia
(52.3%), and 2 from gastrointestinal bleeding (2.3%). According to
endoscopy and pathology findings, among the remaining 88 H.
pylori culture-positive patients, 10 (11.4%) had GU, 16 (18.2%)
had DU, 36 (41.0%) had gastritis and 26 (29.5%) had GC (Fig. 1).
Using primers for the ureC (glmM)
gene, the PCR product of 294-base pairs was obtained in all 88
strains. The mean age of patients was 40±15 years, ranging from 18
to 84 years and including 40 (45.5%) males and 48 (54.5%) females.
The data showed that of 88 isolates, hopQ type I was present
in 52 (59.1%) isolates and hopQ type II was found in 36
(40.9%) isolates. Table II depicts
the prevalence of these genes in association with the different
disease groups.
 | Table II.Distribution of Helibacter pylori
hopQ genes (type I and II) among the different groups. |
Table II.
Distribution of Helibacter pylori
hopQ genes (type I and II) among the different groups.
| Diseases (no.) | Rate of type I
hopQ, no. (%) | Odd ratio (95%
CI) | Rate of type II
hopQ, no. (%) | Odds ratio (95%
CI) |
|---|
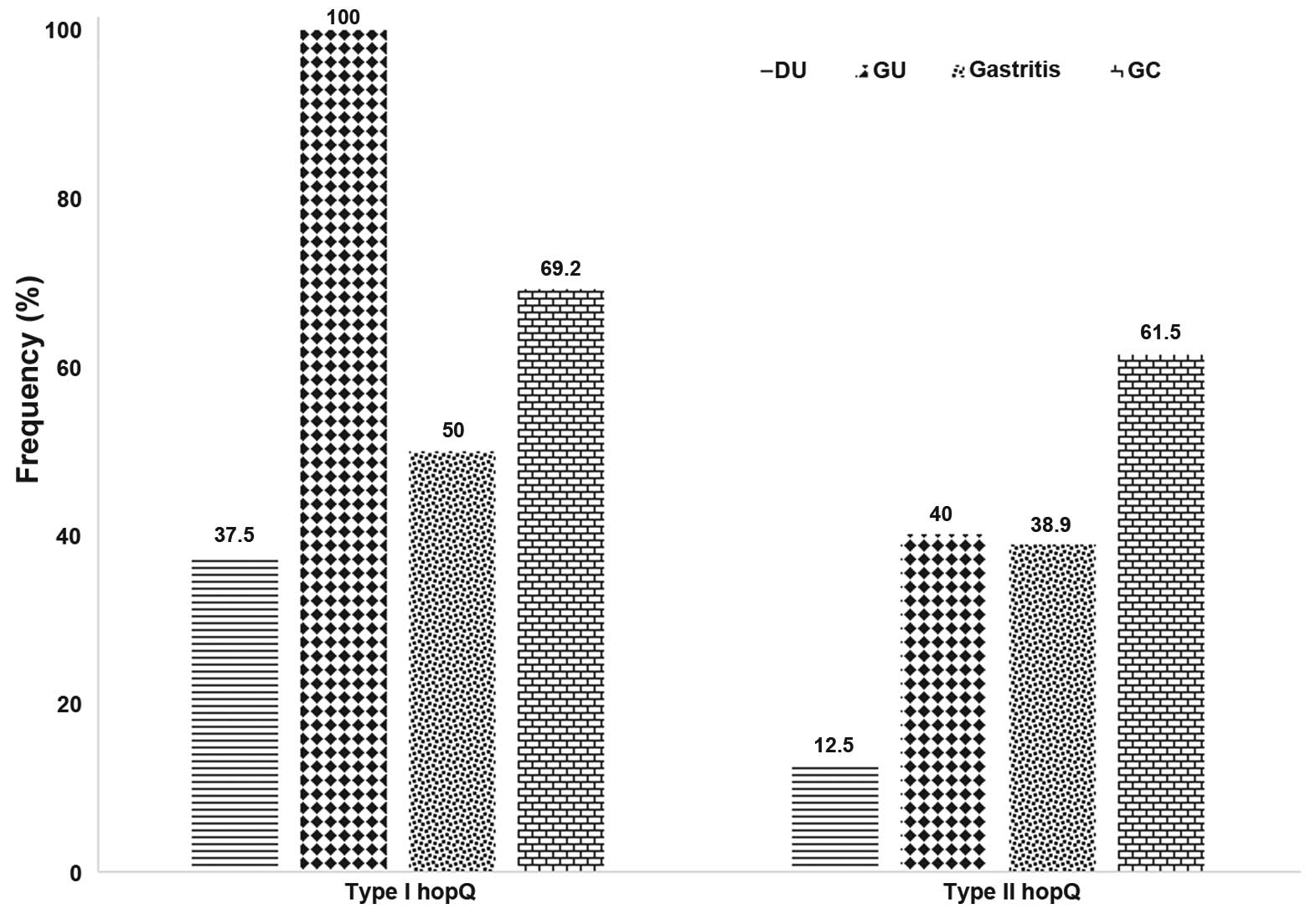
| Gastric carcinoma
(26) | 18 (69.2) | 2.3 (1.3–4.1) | 16 (61.5) | 2.4 (1.1–3.0) |
| Duodenal ulcer
(16) | 6
(37.5) | 0.6 (0.3–1.0) | 2
(12.5) | 0.2 (0.1–0.4) |
| Gastric ulcer
(10) | 10 (100.0) | 2.5 (1.4–4.3) | 4
(40.0) | 0.7 (0.4–1.2) |
| Gastritis (36) | 18 (50.0) | Control group | 14 (38.9) | Control group |
Associations between hopQ types and
disease
There were no statistically significant associations
between age and gender with hopQ types (P>0.05).
Statistical analysis indicated significant associations between
hopQ type I and GC [odds ratio, 2.3; 95% confidence interval
(CI), 1.3–4.1] and GU (odds ratio, 2.5; 95% CI, 1.4–4.3). Type II
hopQ was also found to be significantly predominant in
patients with gastric cancer (odds ratio, 2.4; 95% CI, 1.1–3.0). A
combination of hopQ type I/hopQ type II genotypes
were observed in 36 (40.9%) patients and statistical analysis
demonstrates that there is a significant association between the
simultaneous presence of these two genes (P=0.004).
Discussion
H. pylori cause the most prevalent bacterial
infections globally (18). The
adherence of H. pylori to the gastric mucosa have important
roles in the initial colonization and long-term persistence on the
gastric mucosa as well as in the intensity of the resulting
inflammatory response. Colonization of H. pylori usually
does not result in clinical consequences but can increase the risk
of developing peptic ulcer diseases, gastric adenocarcinoma and
lymphoma (19). It has been suggested
that specific genotyping-based analysis of H. pylori
isolates can be useful for predicting post-infection disorders
(20).
In the present study, the prevalence of H. pylori
hopQ type I and type II genotypes was analyzed in patients with
different gastrointestinal diseases. The predominant genotype In
the Azerbaijan area was the hopQ type I genotype found in 52
(59%) cases and this high rate of hopQ type I was in
contrast to a study, also performed in Iran, that considered the
finding of 52% hopQ type II genotypes as being high
(21). However, the present finding
concerning the frequency of hopQ type I is consistent with a
previous study conducted in the USA (11). Geographical differences in the
distribution of type I and type II hopQ alleles have been noted.
For example, the majority of H. pylori strains isolated in
East Asia are cag PAI-positive and contain type I hopQ
alleles. Type II alleles are commonly identified in H.
pylori strains isolated in Western countries, but are uncommon
among H. pylori strains from East Asia (4).
In the present study, the rate of the hopQ
type II genotype was found most commonly in GC (61.5%). This
finding is similar to results obtained in the North of Iran
(21). The present findings showed
that hopQ type I was most predominant in patients with GU,
which is in disagreement with the study conducted in the North of
Iran (21). To the best of our
knowledge, this is the first study on the prevalence of H.
pylori hopQ alleles, among patient with gastrointestinal
disease in the Azerbaijan area, and this is the second study in
Iran to determine the prevalence and association of these virulence
genes with clinical outcomes. The present findings have shown that
the hopQ type I and type II genotypes are associated with
gastric cancer. The study by Cao and Cover (11) was conducted in the USA and reported
that there was a significant association between the carriers of
H. pylori hopQ type I among the peptic ulcer patients
(11). However, Ohno et al
(22) did not identify an association
between the two hopQ alleles and clinical outcomes. Additionally,
the combination of hopQ type I/hopQ type II genotypes
were evaluated in the present study in association with the
clinical outcomes and statistically significant correlation was
identified between these alleles and disease conditions. It appears
that these two genes are located in one place and may have a
synergistic effect.
The prevalence and association between clinical
outcome and the H. pylori virulence gene was reported to be
different among various countries, regions, ethnicities and patient
groups. The differences between studies may be due to geographical
area, sample size, studied groups, ethnicity, primer sets, PCR
conditions and the variety of bacterial strains. The weakness and
limitations of the study firstly were low sample size and the small
number of studies performed to obtain a whole evaluation on the
prevalence of genes.
If additional studies showed similar results to the
present findings, this may lead to improved diagnostic policies for
clinicians to fight the present doctrine for gastric diseases. In
conclusion, the association between hopQ types I and II, and
gastric cancer outcomes, may suggest that these genes may be
helpful for the prediction of specific-disease risk.
Acknowledgements
The authors are grateful to Dr H. Banazadeh Baghi
for the English editing. The study was financially supported by
Tabriz University of Medical Sciences.
References
|
1
|
Cover TL and Blaser MJ: Helicobacter
pylori in health and disease. Gastroenterology. 136:1863–1873.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cover TL, Berg DE, Blaser MJ and Mobley H:
Helicobacter pylori pathogenesis. Principles of bacterial
pathogenesis. Academic Press. (San Diego, CA). 510–558. 2001.
|
|
3
|
Costa AC, Figueiredo C and Touati E:
Pathogenesis of Helicobacter pylori infection. Helicobacter.
14(Suppl 1): 15–20. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cao P, Lee KJ, Blaser MJ and Cover TL:
Analysis of hopQ alleles in East Asian and Western strains
of Helicobacter pylori. FEMS Microbiol Lett. 251:37–43.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Alm RA, Bina J, Andrews BM, Doig P,
Hancock RE and Trust TJ: Comparative genomics of Helicobacter
pylori: Analysis of the outer membrane protein families. Infect
Immun. 68:4155–4168. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sabarth N, Hurvitz R, Schmidt M,
Zimny-Arndt U, Jungblut PR, Meyer TF and Bumann D: Identification
of Helicobacter pylori surface proteins by selective
proteinase K digestion and antibody phage display. J Microbiol
Methods. 62:345–349. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ilver D, Arnqvist A, Ögren J, Frick IM,
Kersulyte D, Incecik ET, Berg DE, Covacci A, Engstrand L and Borén
T: Helicobacter pylori adhesin binding fucosylated
histo-blood group antigens revealed by retagging. Science.
279:373–377. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mahdavi J, Sondén B, Hurtig M, Olfat FO,
Forsberg L, Roche N, Angstrom J, Larsson T, Teneberg S, Karlsson
KA, et al: Helicobacter pylori SabA adhesin in persistent
infection and chronic inflammation. Science. 297:573–578. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yamaoka Y, Kwon DH and Graham DY: A M(r)
34,000 proinflammatory outer membrane protein (oipA) of
Helicobacter pylori. Proc Natl Acad Sci USA. 97:7533–7538.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Odenbreit S, Till M, Hofreuter D, Faller G
and Haas R: Genetic and functional characterization of the alpAB
gene locus essential for the adhesion of Helicobacter pylori
to human gastric tissue. Mol Microbiol. 31:1537–1548. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cao P and Cover TL: Two different families
of hopQ alleles in Helicobacter pylori. J Clin
Microbiol. 40:4504–4511. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Backert S and Selbach M: Role of type IV
secretion in Helicobacter pylori pathogenesis. Cell
Microbiol. 10:1573–1581. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ghotaslou R, Milani M, Akhi MT, Nahaei MR,
Hasani A, Hejazi MS and Meshkini M: Diversity of Helicobacter
pylori caga and vaca genes and its relationship with clinical
outcomes in Azerbaijan, Iran. Adv Pharm Bull. 3:57–62.
2013.PubMed/NCBI
|
|
14
|
Sambrook J and Russell DW: Molecular
cloning. A laboratory manual (3rd). Cold Spring Harbor Laboratory
Press. (New York, NY). 2001.
|
|
15
|
Espinoza MGC, Vazquez RG, Mendez IM,
Vargas CR and Cerezo SG: Detection of the glmM gene in
Helicobacter pylori isolates with a novel primer by PCR. J
Clin Microbiol. 49:1650–1652. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kidd M, Louw JA and Marks IN:
Helicobacter pylori in Africa: Observations on an ‘enigma
within an enigma’. J Gastroenterol Hepatol. 14:851–858. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sicinschi LA, Correa P, Bravo LE, Peek RM
Jr, Wilson KT, Loh JT, Yepez MC, Gold BD, Thompson DT, Cover TL, et
al: Non-invasive genotyping of Helicobacter pylori cagA,
vacA, and hopQ from asymptomatic children.
Helicobacter. 17:96–106. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rafeey M, Ghotaslou R, Nikvash S and Hafez
AA: Primary resistance in Helicobacter pylori isolated in
children from Iran. J Infect Chemother. 13:291–295. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ghotaslou R, Leylabadlo HE and Asl YM:
Prevalence of antibiotic resistance in Helicobacter pylori:
A recent literature review. World J Methodol. 5:164–174. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Graham DY and Fischbach L: Helicobacter
pylori infection. N Engl J Med. 363:595–596; author reply 596.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Talebi Bezmin, Abadi A and Mohabbati
Mobarez A: High prevalence of Helicobacter pylori hopQ II
genotype Isolated from Iranian patients with gastroduodenal
disorders. J Pathogens. 2014:8424692014.
|
|
22
|
Ohno T, Sugimoto M, Nagashima A, Ogiwara
H, Vilaichone RK, Mahachai V, Graham DY and Yamaoka Y: Relationship
between Helicobacter pylori hopQ genotype and clinical
outcome in Asian and Western populations. J Gastroenterol Hepatol.
24:462–468. 2009. View Article : Google Scholar : PubMed/NCBI
|