Introduction
Hypertension is a chronic disease, and its major
characteristics include increasing arterial blood pressure, which
often causes heart, brain, kidney and other vital organ diseases
and corresponding consequences (1,2). The Tiangou
Jiangya capsule is composed of Gastrodia elata, Uncaria
rhynochophylla, Eucommia, Scutellaria
baicalensis, Cortex moutan and Pearl, and represses hyperactive
liver yang and sedative, which was used to treat mild to moderate
hypertension (1,2).
Currently, Chinese herbal monomers are the subject
of Traditional Chinese Medicine (TCM) pharmacokinetic studies
(3–5).
However, as the efficacy of TCM is attributed to the overall effect
of various compounds, investigating only one ingredient is not
considered to be beneficial to TCM research (6). In the present study, the effective
ingredients of the Tiangou antihypertensive capsule were
determined. The ingredients were used as a reference value to
prepare the equivalent concentration of the extract solution. This
solution was then used to examine the effects of complex
prescription on concentration-time curve and the pharmacokinetic
parameters of baicalin, rhynochophylline and gastrodin. The results
provided valuable data for the further study of the Tiangou Jiangya
capsule.
Materials and methods
Instrumentation
Instruments used in the present study were: D2100
Hitachi high-performance liquid chromatography (HPLC), AE240
electronic balance (Mettler-Toledo Instruments Co., Ltd., Shanghai,
China), HSC-12A Organomation (Beijing Zhuoxin Weiye Technology Co.,
Ltd., Beijing, China), Nitrogen (Jinan Dehui Gas Co., Ltd., Jinan,
China), XK96-B fast mixer (Jiangsu Xinkang Medical Instrument Co.,
Ltd., Jiangsu, China), KQ-250E ultrasonic cleaning instrument
(Kunshan Ultrasonic Instrument Co., Ltd., Kunshan, China), LDZ4-0.8
medical centrifuge (Beijing Medical Centrifuge Factory, Beijing,
China), TGL-16G high speed centrifuge (analysis of Shanghai Medical
Instrument Factory, Shanghai, China) and Eppendorf tube (JiNan
YongNen Technology Co., Ltd., JiNan, China).
Reagents and chemicals
Reagents used in the present study included, heparin
sodium injection (lot no. 10067; Changzhou Qianhong Biochemical
Pharmaceutical Co., Ltd., Changzhou, China); chloral hydrate (lot
no. 20081021; Tianjin Guangfu Institute Fine Chemical Research,
Tianjin, China); acetonitrile (Shandong Yuwang Industrial Co., Ltd.
Jinan, China); and methanol (Tianjin Siyou Fine Chemicals Co.,
Ltd., Tianjin, China). Acetonitrile and methanol were
chromatographically pure, while other reagents were analytically
pure, and water was Wahaha Purified water (Hangzhou Wahaha Group
Co., Ltd., Zhejiang, China).
The chemicals included in the study were: Baicalin
reference standard (98% purity, lot no. 110715-201016; National
Institutes for Food and Drug Control, Beijing, China); rutin (98%
purity, lot no. 10080-200707; National Institutes for Food and Drug
Control); rhynochophylline (98% purity, lot no. 20120426 Shanghai
Standard Source Leaves Biological Technology Co., Ltd., Shanghai,
China); gastrodin (98% purity, lot no. 110807-200205; National
Institutes for Food and Drug Control); and the Tiangou
antihypertensive capsule (home made, containing 0.7%
rhynochophylline, 5.6% gastrodin, 7.9% baicalin, and 1.2%
baicalein).
Experimental animals
Fifty-four male Sprague-Dawley (SD) (production
license no. SCXK-Lu-20130001), with a body weight of 220±20 g, were
purchased from the Experimental Animal Center of Shandong Lukang
(Shandong, China). The 54 rats were randomly divided into 9 groups
(n=6 rats/group) labelled A-I. The procedures involving animals and
their care were conducted in accordance with NIH guidelines
(publication no. 85-23, revised 1996) and were approved by the
Animal Care and Use Committee of the Shandong University of
Traditional Chinese Medicine.
HPLC
For baicalin, analysis separation was performed on a
Shimadzu ODS (4.6×250 mm, 5 µm) maintained at 25°C.
Methanol-water-phosphoric (47:53:0.2) was used as the mobile phase
at 1.0 ml/min. Solution (20 µl) was injected into the system. The
detector wavelength was 280 nm (7).
Rutin was used as the internal standard substance.
For gastrodin, analysis separation was performed on
a Shimadzu ODS (4.6×250 mm, 5 µm) maintained at
30°C.Acetonitrile-0.05% acid water solution (3:97) was used as the
mobile phase at 1.0 ml/min and 20 µl solution was injected into the
system. The detector wavelength was 220 nm (7). Phloroglucinol was used as the internal
standard substance.
For rhynochophylline, analysis separation was
performed on a Shimadzu ODS (4.6×250 mm, 5 µm) maintained at 25°C.
Methanol-water (55:45) (containing 0.01 mol/l triethylamine, acetic
acid adjust solution pH 7.5) was used as the mobile phase at 1.0
ml/min. Solution (20 µl) was injected into the system, and the
detector wavelength was 254 nm (8).
Preparation of standard and quality
control samples
The stock solutions of baicalin, rhynochophylline
and gastrodin were prepared in 10 ml methanol at a concentration of
500 µg/ml. A series of standard working solutions were prepared by
the diluted proper stock solutions with methanol. Calibration
working solutions were prepared by adding the diluted working
standard solutions into blank rat plasma (5/95, v/v), and the
resulting concentrations of baicalin, gastrodin and
rhynochophylline are shown in Table I,
respectively.
 | Table I.Regression equation and LOD of the
analytes in rat plasma. |
Table I.
Regression equation and LOD of the
analytes in rat plasma.
| Compound | Regression
equation | Correlation
coefficient (r) | Linear range
(µg/ml) | LOD (ng/ml) |
|---|
| Baicalin | y=0.052x-0.026 | 0.9995 | 0.44–28.00 | 1.85 |
| Gastrodin | y=0.0219x-0.0138 | 0.9991 | 0.13–13.00 | 4.31 |
| Rhynochophylline | y=0.0091x+0.0002 | 0.9993 | 0.04–2.34 | 2.92 |
The quality control samples were prepared at three
concentrations of 0.87, 3.62 and 13.72 µg/ml for baicalin; 0.13,
8.17 and 13.00 µg/ml for gastrodin; and 0.04, 0.51 and 2.54 µg/ml
for rhynochophylline. The solutions were stored at 4°C in a
refrigerator and brought to room temperature before use. The
results are shown in Table II.
 | Table II.Summary of precision, accuracy and
recovery in rat plasma (n=6). |
Table II.
Summary of precision, accuracy and
recovery in rat plasma (n=6).
| Analytes | Concentration
(µg/ml) | Intra-day precision
(%) | Inter-day precision
(%) | Accuracy (%) | Recovery (%) |
|---|
| Baicalin | 0.87 | – | – | – |
98.85 |
|
| 3.62 | 4.80 | 4.03 | 3.4 | 100.08 |
|
| 13.72 | – | – | – |
98.73 |
|
| 0.13 | – | – | – |
99.77 |
| Gastrodin | 8.17 | 2.18 | 3.12 | 2.39 | 102.17 |
|
| 13.00 | – | – | – | 100.34 |
|
| 0.04 | – | – | – |
97.71 |
| Rhynochophylline | 0.51 | 4.26 | 3.42 | 4.89 |
99.09 |
|
| 2.54 | – | – | – | 101.18 |
Selectivity
Blank plasma spiked with the internal standard and
standard substance, and the rat plasma sample collected after oral
administration of the Tiangou antihypertensive capsule solution
were prepared, respectively. The samples were treated with
precipitate protein, and the compounds in plasma were
quantitatively determined by the abovementioned analysis methods.
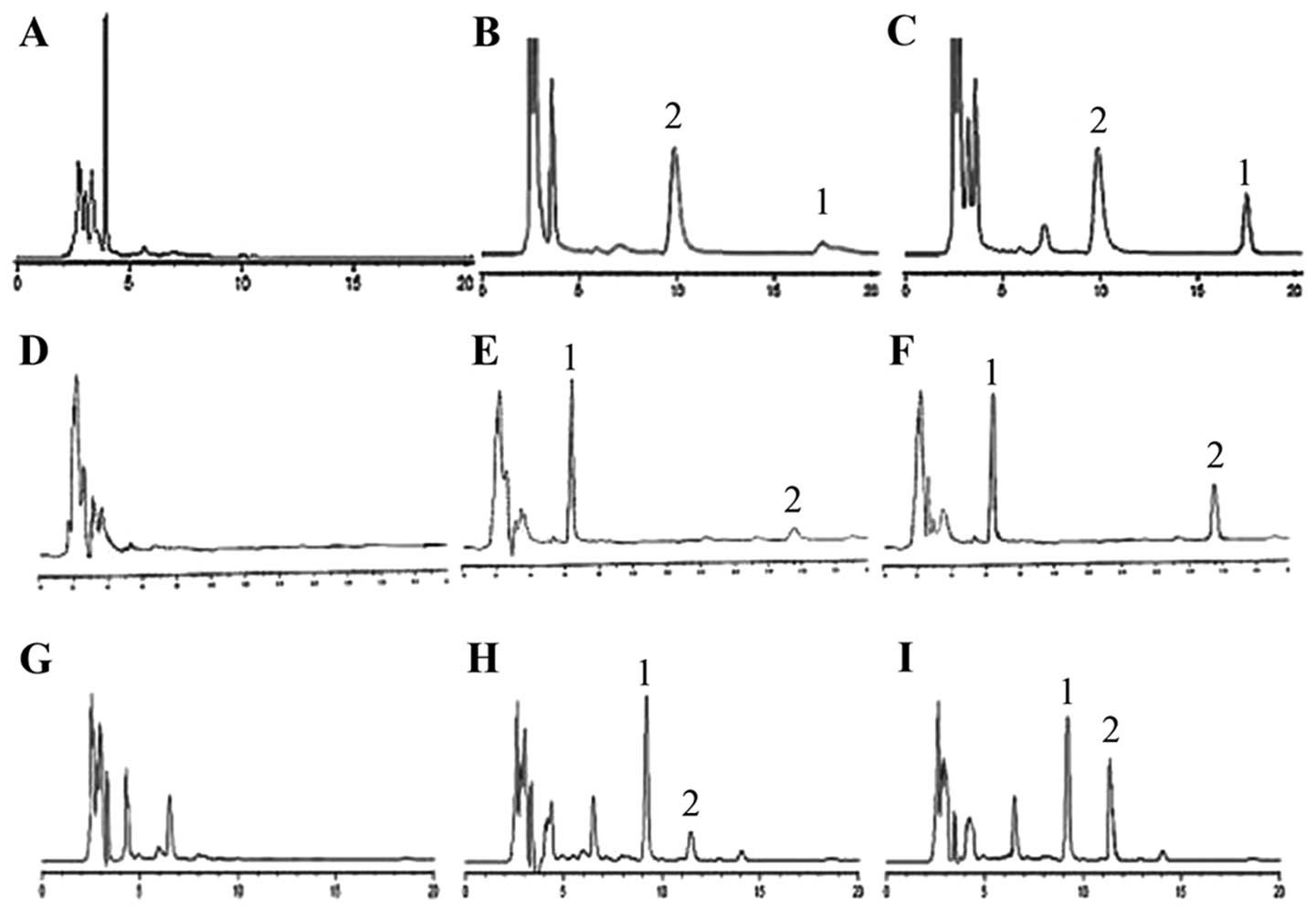
The HPLC spectra of plasma samples are shown in Fig. 1. No endogenous interference was
observed at retention time of the analytes and internal
standard.
Preparation of the solution for animal
oral administration
Tiangou antihypertensive capsule powders were
reconstituted in 0.5% CMC-Na solution at a concentration of 0.1
g/ml. HPLC was used to determine the content of baicalin, gastrodin
and rhynochophylline in the Tiangou antihypertensive capsule. The
data were used as a benchmark to prepare single herb extract
solution. Baicalin and Baicalein standard were reconstituted in
0.5% CMC-Na solution at a concentration of 5.0 mg/ml.
A low concentration of rhynochophylline in the
samples or low sensitivity of the detector towards the
rhynochophylline may explain the non-detection. For preparation of
the extract, 10 g powder of the Tiangou antihypertensive capsule
was extracted with methanol under sonication for 30 min and
filtrated. The extracting solution was centrifuged at 500 × g for
10 min and dried using nitrogen. The residue was reconstituted in
0.5% CMC-Na solution at a concentration of 0.1 g/ml.
Oral administration
A total of 54 male SD rats with a body weight of
220±20 g were randomly divided into 9 groups. The rats were
acclimatized for 5 days, and fasted with free access to water until
12 h prior to the experiment. The rats were administered orally
with the abovementioned drug solutions, and the doses are shown in
Tables III–V.
 | Table III.Initial oral administration
dosage. |
Table III.
Initial oral administration
dosage.
| Groups | Tiangou
anti-hypertensive capsule (ml) | Scutellaria
baicalensis extract (ml) | Gastrodia
Rhizoma extract (ml) |
|---|
| Low dose (A) | 1.5 | – | – |
| Mid dose (B) | 3.0 | – | – |
| High dose (C) | 6.0 | – | – |
| Scutellaria
control (D) | – | 0.5 | – |
| Gastrodia
elata control (E) | – | – | 1.8 |
 | Table V.Oral administration dosage. |
Table V.
Oral administration dosage.
| Group | Aharpleaf uncaria
stem with Hooks extract (ml) | Tiangou
antihypertensive capsule (ml) |
|---|
| H | – | 0.6 |
| I | 0.6 | – |
Collection of blood samples
After administration, the rats were anaesthetized
with 10% Chlorali hydras. Samples were collected from the
suborbital veniplex prior to administration and at 5, 10, 20, 30,
60, 90, 120, 180, 240, 360, 480, 600 and 720 min after
administration. The plasma was placed into a tube in which there
was heparin and was centrifuged at 850 × g for 10 min. Supernatant
(0.2 ml) was transferred to a sample tube and stored at −20°C in
the refrigerator until analysis.
Preparation of rat plasma sample
Baicalin plasma treatment method
Plasma was defrosted at 25°C, and 200 µl of plasma
sample was transferred into an Eppendorf tube. Subsequently, 50 µl
of rutin solution and 50 µl of sodium dihydrogen phosphate solution
(1 mol/l) were added and vortex-mixed for 1 min. Methanol (100 µl)
and acetonitrile (300 µl) were added, vortex-mixed for 1 min and
centrifuged at 850 × g for 10 min to precipiate protein. The
supernatant was evaporated to dryness under a gentle stream of
nitrogen at 25°C. The residue was dissolved in 100 µl mobile phase
and filtered with a 0.22 µm microporous membrane. Sample solution
(20 µl) was injected into the chromatographic system for
analysis.
Gastrodin plasma treatment method
Plasma was defrosted at room temperature and 200 µl
of plasma sample was transferred into an Eppendorf tube.
Subsequently, 100 µl mobile phase and 100 µl phloroglucinol
solution were added and the mixture was vortex-mixed for 1 min.
Acetonitrile (1 ml) was then added, the mixture was vortex-mixed
for 1 min and centrifuged at 9,000 × g for 10 min to precipitate
the protein. The supernatant was dried under a gentle stream of
nitrogen at 25°C. Then, 200 µl of mobile phase was used to dissolve
the residue, which was centrifuged at 850 × g for 10 min, and a
volume of 20 µl sample solution was injected into the
chromatographic system for analysis.
Rhynochophylline plasma treatment method
Plasma was defrosted at room temperature and 200 µl
of plasma sample was transferred into an Eppendorf tube.
Carbamazepine solution (50 µl), 200 µl (0.1 mol/l) NaOH solution,
and 1 ml ether were added, the mixture was vortex-mixed for 3 min
and centrifuged at 500 × g for 10 min. The organic layer was then
transferred into another tube and the above procedures were
repeated. The organic phase was combined and dried under nitrogen.
Methanol (100 µl) was used to dissolve the residue which was
centrifuged at 850 × g for 10 min, and then 20 µl solution was
injected into the chromatographic system for analysis.
The contents of three compounds in plasma were
quantitatively determined by the above-mentioned analysis method.
The concentration-time curve was then drawn and was used to analyze
the absorption mechanism of such compounds.
Results and Discussion
Concentration-time curve and pharmacokinetic
parameter of baicalin. The content of baicalin in groups A-D was
determined by the abovementioned analysis method. The
concentration-time curves of baicalin were drawn as follows: Time
as x-coordinate and the concentration of baicalin in plasma as
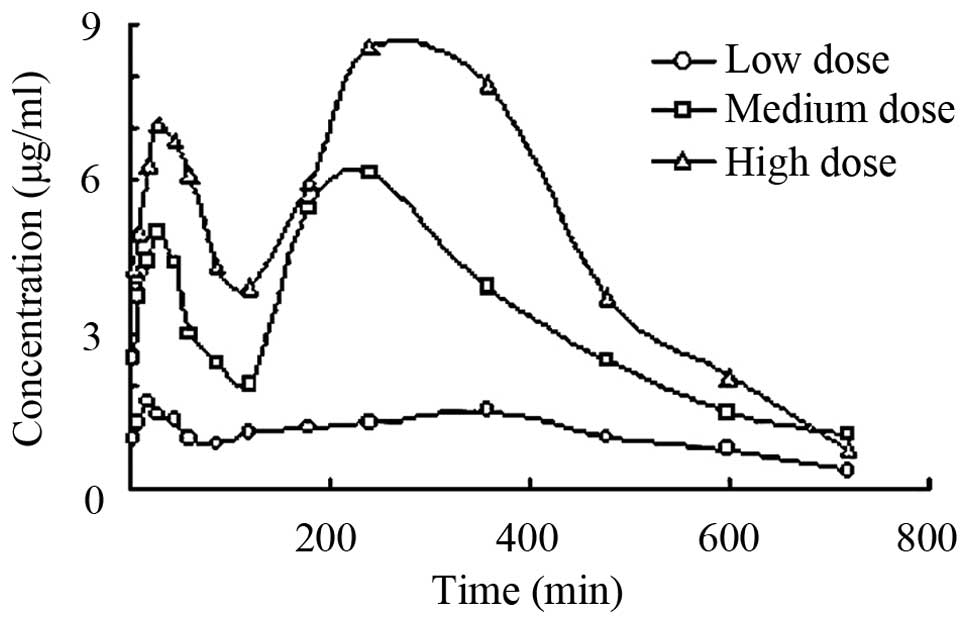
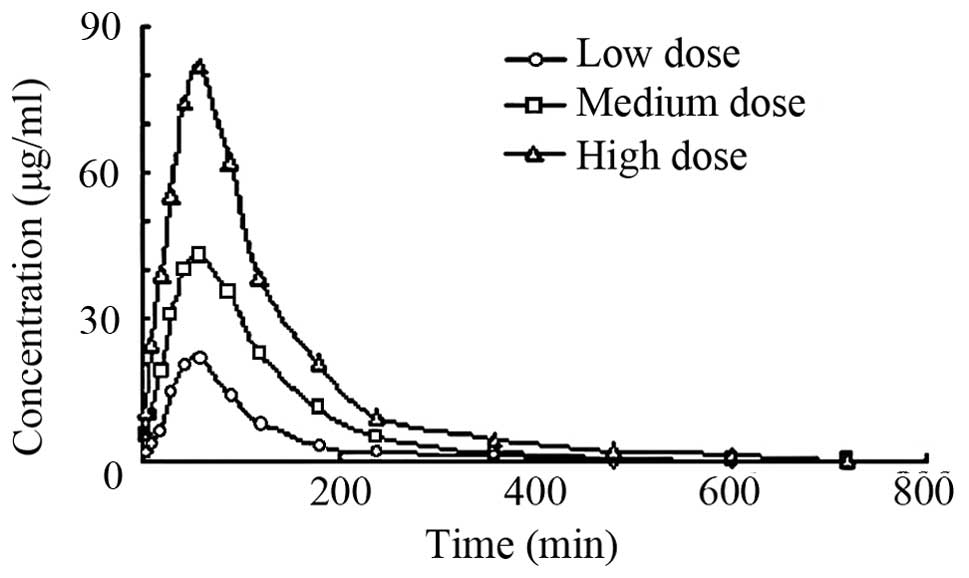
y-coordinate. Fig. 2 shows the
concentration-time curve of baicalin in rats after oral
administration of a low-, medium- and high-dose of the Tiangou
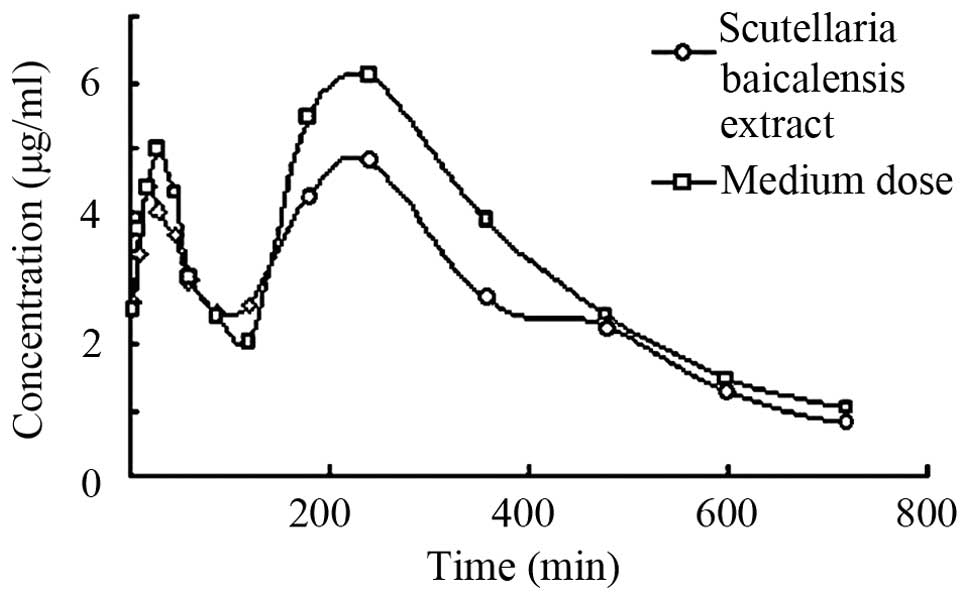
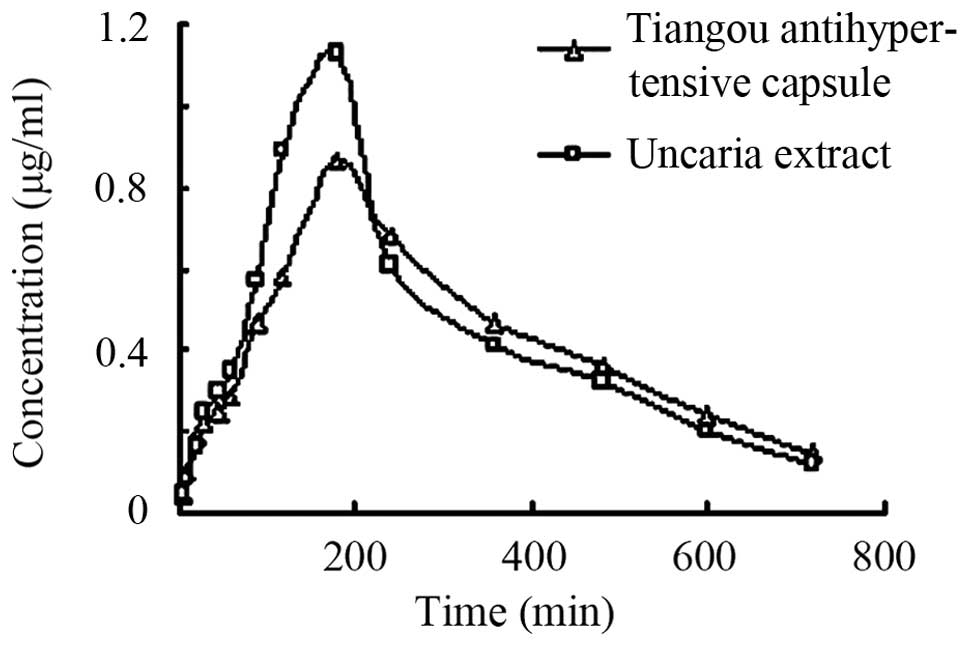
antihypertensive capsule. Fig. 3 shows
the concentration-time curve of baicalin in plasma of rats after
oral administration of the Scutellaria baicalensis extract
and a medium dose of the Tiangou antihypertensive capsule.
As shown in Figs. 2 and
3, as well as in Table VI, no significant difference was found
for the peak concentration time of baicalin between the groups. We
obtained a dose-dependent plasma concentration profile as well as a
peak concentration of baicalin. The results suggest that the
absorption of baicalin was attributed to passive absorption. The
peak concentrations of the medium-dose group were 4.592±0.485 and
5.793±0.395 µg/ml and the peak concentration of the extract were
5.021±0.531 and 6.858±0.834 µg/ml. The t-test indicated no
significant difference for the area under the curve (AUC) of
baicalin between the medium-dose group and Scutellaria
baicalensis extract group (P>0.05). These results showed
that the Tiangou antihypertensive capsule did not influence
baicalin absorption, but made the drug effect more mild, and
maintained the plasma concentration of baicalin to avoid a
peak-valley phenomenon.
 | Table VI.Pharmacokinetics parameters of
baicalin with low, medium and high dose of Tiangou antihypertensive
capsule (n=6). |
Table VI.
Pharmacokinetics parameters of
baicalin with low, medium and high dose of Tiangou antihypertensive
capsule (n=6).
| Parameter | Low dose | Medium dose | High dose | Scutellaria
baicalensis extract |
|---|
|
T1max/min | 28.4±4.082 | 28.3±3.174 | 30.0±1.136 | 28.6±2.798 |
|
T2max/min | 280.0±21.967 | 283.0±24.32 | 300.0±45.92 | 271±34.45 |
|
C1max/µg/ml | 2.223±0.441 | 4.592±0.485 | 6.906±0.475 | 5.021±0.531 |
|
C2max/µg/ml | 1.799±0.176 | 5.793±0.395 | 7.89±1.407 | 6.858±0.834 |
|
AUC(0-t)/µg/ml·h | 907.61±103.58 |
2,194.84±153.38 |
3,394.69±368.61 |
2,201.32±156.41 |
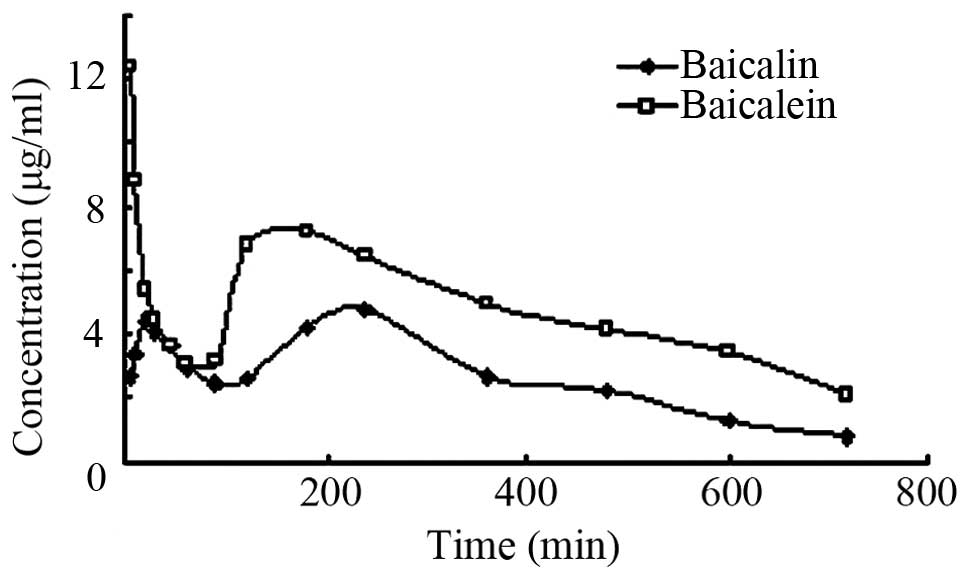
The content of baicalin in groups F and G was
determined by the above-mentioned analysis method. Fig. 4 and Table
VI show that baicalin exhibited a bimodal phenomenon in the
concentration-time curve, which was consistent with previous
findings (9,10). In the concentration-time profile of the
baicalein group, the first peak occurred at 5 min. The peak
concentration was much greater than that of the baicalin group.
Both of them exhibited a bimodal phenomenon in concentration-time
profile.
Blood samples of rats did not contain any baicalein
(11); nevertheless, an amount of
baicalin was detected in the samples. The result of the experiment
showed that baicalin has a high bioavailability and faster
absorption. Previous findings indicate that the bacterium in
intestinal was able to hydrolyze baicalin to baicalein (12). Only in this manner, could baicalin be
absorbed and this metabolic process is an important stage for
baicalin absorption (12). The
intestinal absorption experiment showed that baicalin has a high
absorption rate in duodenum and cecum (13). It may be a cause of the bimodal
phenomenon after oral administration baicalin. As shown in Figs. 5–7, the
concentration-time curve of gastrodin and rhynochophylline do not
have a bimodal phenomenon. Therefore, the bimodal phenomenon caused
by baicalin inhibition of gastric emptying may be excluded. By
intravenous injection, the bimodal phenomenon was observed in the
concentration-time curve. If rat biliary tract is ligated, the
concentration-time curve does not observe the bimodal phenomenon.
This phenomenon probably occurs due to distribution re-absorption
and enterohepatic circulation. The present results therefore showed
that, Tiangou antihypertensive capsule did not have any effect on
baicalin absorption, making the drug effect more mild and
maintaining the concentration of baicalin in vivo more
steady. These findings are consistent with those of previous
investigation (14).
Concentration-time curve and
pharmacokinetic parameter of gastrodin
The content of gastrodin in groups A, B, C and E was
determined by the abovementioned analysis method. The
concentration-time curves of gastrodin were drawn as follows: Time
as x-coordinate and the concentration of gastrodin in plasma as
y-coordinate. Fig. 3 shows the
concentration-time curve of gastrodin after oral administration of
low, medium and high dose of Tiangou antihypertensive capsule.
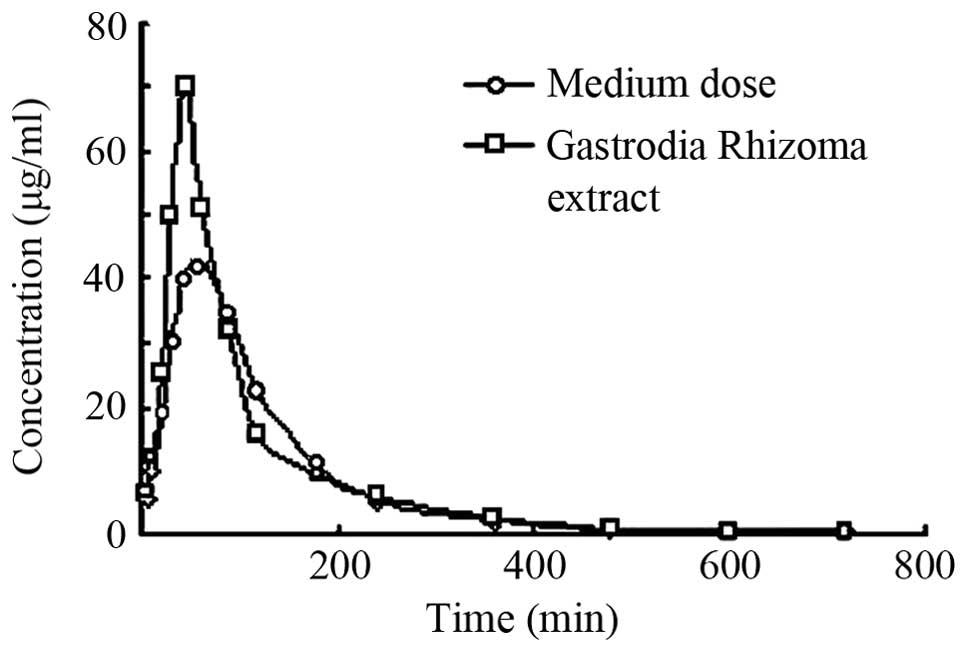
Fig. 5 shows the concentration-time
curve of gastrodin after oral administration of the Gastrodia
Rhizoma extract and medium dose of the Tiangou antihypertensive
capsule.
Figs. 5 and 6, and Table
VIII show that, AUC and Cmax were proportional to
the dose administered. Compared with the Gastrodia Rhizoma
extract, Cmax and Ke of gastrodin in the
Tiangou antihypertensive capsule groups were decreased while the Vd
was significantly increased. However, no significant difference was
found for AUC of gastrodin between the two groups.
 | Table VIII.Pharmacokinetic parameters of
gastrodin after ig three dosage levels of Tiangou antihypertensive
capsule and Gastrodia Rhizoma extract. |
Table VIII.
Pharmacokinetic parameters of
gastrodin after ig three dosage levels of Tiangou antihypertensive
capsule and Gastrodia Rhizoma extract.
|
| Dose |
|
|---|
|
|
|
|
|---|
| Parameter | Low dose | Mid | High | Extract |
|---|
|
Ke/min | 0.017±0.001 | 0.0151±0.003 | 0.013±0.001 | 0.0204±0.004 |
|
Ka/min | 0.032±0.003 | 0.035±0.005 | 0.031±0.007 | 0.036±0.003 |
|
Tmax/min | 42.005±2.918 | 43.971±2.973 | 47.673±4.537 | 36.670±3.098 |
|
AUC/µg·ml−1·min | 2,263.1±411.7 | 5,187.2±871.5 | 10,261±354.2 | 5,462.1±281.2 |
|
V/ml·mg−1 | 0.225±0.033 | 0.223±0.041 | 0.248±0.037 | 0.161±0.029 |
Gastrodin in rats is consistent with a
one-compartment model (15,16). The peak of gastrodin occurred within 45
min, and rapid decay occurred after 1 h, indicating that gastrodin
was not stored in the body, has a high absorption speed in the
intestinal tract and exerts a rapid effect. The results
demonstrated that the complex prescription exerted a certain effect
on the absorption, distribution and elimination of gastrodin. The
complex prescription delayed the absorption and excretion of
gastrodin, prolonged the residence time of gastrodin in
vivo, and could promote the distribution of gastrodin in
vivo, but it did not have significant influence on
bioavailability.
Concentration-time curve of
rhynochophylline
As the data show in Table
IX, compared with the Uncaria rhynchophylla extract
group, the Ka and Ke of rhynochophylline were
significantly decreased in the complex prescription group. However,
there was effect on AUC. By contrast, the Vd of the Tiangou
antihypertensive capsule group was significantly increased.
 | Table IX.Pharmacokinetic parameters of
rhynochophylline in Tiangou antihypertensive capsule and Gambir
plant extract (n=6). |
Table IX.
Pharmacokinetic parameters of
rhynochophylline in Tiangou antihypertensive capsule and Gambir
plant extract (n=6).
| Parameter | Tiangou
antihypertensive capsule | Gambir plant
extract |
|---|
|
Ke/h | 0.2643±0.0132 | 0.2909±0.028 |
|
Ka/h | 0.4965±0.0438 | 0.6274±0.1528 |
|
Tmax/h | 2.7242±0.1282 | 2.3994±0.2295 |
| AUC/µg/ml/h | 5.1222±0.7919 | 5.1432±0.4537 |
| V/ml/ml | 13.5863±0.7832 | 11.1187±0.8734 |
In consideration of the low content of
rhynchophylline in powder and extract (17), we should set up a single experimental
group. Summing up the above, on the premise of without influence
drug absorption, complex prescriptions could avoid peak-valley
phenomenon during taking medicine. And make the plasma
concentratioin more smoothly. Comparison of the Vd we found Tiangou
antihypertensive capsule could promote the in vivo
distribution of rhynochophylline.
In conclusion, the results of the present study have
shown that, the Tiangou antihypertensive capsule made the plasma
concentration of effective components more stable. It was
consistent with the medication principle of smooth reduction.
Complex prescription did not have a significant influence on the
total absorption rate of single composition, respectively. The peak
concentration of gastrodin occurred at an early stage, the peak
concentration of rhynochophylline and baicalin occur subsequently,
and the tmax of rhynochophylline is close to the
tmax of baicalin. It may be associated with the
pharmacological action of the synergistic antihypertensive effect,
which remains to be investigated in future studies.
References
|
1
|
Wang YL, Li LJ and Zhang W: In vivo
pharmacokinetics of paeonol from Tiangou Jiangya capsule in rats.
Chin Exper Trad Med Formulae. 18:114–116. 2012.
|
|
2
|
Lou HW, Li LJ and Li J: Studies on
absorption kinetics of rhynchophylline and isorhynchophylline of
Tiangou Jiangya capsules in rats intestinal. Chin Exper Trad Med
Formulae. 19:150–153. 2013.
|
|
3
|
Hu KJ, Wang YH and Wang D: The inhibited
effect of chlorogenic acid from the honeysuckle on virus in vitro.
Information Trad Chin Med. 27:27–28. 2010.
|
|
4
|
Hou Y, Zhu X and Cheng G: Study on the
anti inflammatory mechanism of baicalin. J Acta Pharmacol Sin.
35:161–164. 2010.
|
|
5
|
Xiong Y, Fu YY, Kuang NZ, et al: Study on
activity and mechanism of baical in against candida albicans. Chin
Pharmacol Bull. 20:1404–1407. 2004.
|
|
6
|
Lin L, Liu JX, Zhang Y, Lin CR and Li LQ:
Effect of different combinations of compound Danshen preparation on
pharmacokinetic parameters of salvianolic acid B in rat plasma.
Zhongguo Zhong Yao Za Zhi. 33:2683–2687. 2008.(In Chinese).
PubMed/NCBI
|
|
7
|
National Pharmacopoeia Committee, .
Pharmacopoeia of the People's Republic of China. Chemical Industry
Press; Beijing: pp. 3012015
|
|
8
|
Wang J, Liu ZH, Long Y and Xin LN: HPLC
determination of rhynchophylline and isorhynchophylline in Anshen
Yangxue oral liquid. Zhong Yao Cai. 32:1134–1137. 2009.(In
Chinese). PubMed/NCBI
|
|
9
|
Zhang J, Zhang S, Teng S and Zhai L: An
LC-MS/MS method for simultaneous determination of four flavonoids
from semen oroxyli in rat plasma and its application to a
pharmacokinetic study. J Chromatogr B Analyt Technol Biomed Life
Sci. 1020:96–102. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tong L, Wan M, Zhang L, Zhu Y, Sun H and
Bi K: Simultaneous determination of baicalin, wogonoside,
baicalein, wogonin, oroxylin A and chrysin of Radix scutellariae
extract in rat plasma by liquid chromatography tandem mass
spectrometry. J Pharm Biomed Anal. 70:6–12. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen H, Li Z, Li YJ, Wu XW, Wang SR, Chen
K, Zheng XX, Du Q and Tang D: Simultaneous determination of
baicalin, oroxylin A-7-O-glucuronide and wogonoside in rat plasma
by UPLC-DAD and its application in pharmacokinetics of pure
baicalin, Radix Scutellariae and Yinhuang granule. Biomed
Chromatogr. 29:1819–1825. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Noh K, Kang Y, Nepal MR, Jeong KS, Oh G,
Kang MJ, Lee S, Kang W, Jeong HG and Jeong TC: Role of intestinal
microbiota in baicalin-induced drug interaction and its
pharmacokinetics. Molecules. 21:337–340. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li LJ, Li J and Lou HW: Study on in situ
intestinal absorption of baicalin contained in Tiangou Jiangya
capsules. Zhongguo Zhong Yao Za Zhi. 38:894–898. 2013.(In Chinese).
PubMed/NCBI
|
|
14
|
Xiao L, Wang F, Li HD and Zhao XY:
Pharmacokinetic study on baicalin of Qingkailing injection in rats.
Zhongguo Zhong Yao Za Zhi. 32:2534–2538. 2007.(In Chinese).
PubMed/NCBI
|
|
15
|
Cai Z, Hou S, Li Y, Zhao B, Yang Z, Xu S
and Pu J: Effect of borneol on the distribution of gastrodin to the
brain in mice via oral administration. J Drug Target. 16:178–184.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li LJ and Yan R: Pharmacokinetics of
gastrodin from Tiangou Jiangya capsule in rats. J. Zhongguo Zhong
Yao Za Zhi. 39:2964–2967. 2014.(In Chinese).
|
|
17
|
Ge Z, Xie Y, Liang Q, Wang Y and Luo G:
Pharmacokinetic Comparative Study of Gastrodin and Rhynchophylline
after Oral Administration of Different Prescriptions of Yizhi
Tablets in Rats by an HPLC-ESI/MS Method. Evid Based Complement
Alternat Med. 2014:1672532014.PubMed/NCBI
|