|
1
|
Tostmann A, Boeree MJ, Aarnoutse RE, de
Lange WC, van der Ven AJ and Dekhuijzen R: Antituberculosis
drug-induced hepatotoxicity: Concise up-to-date review. J
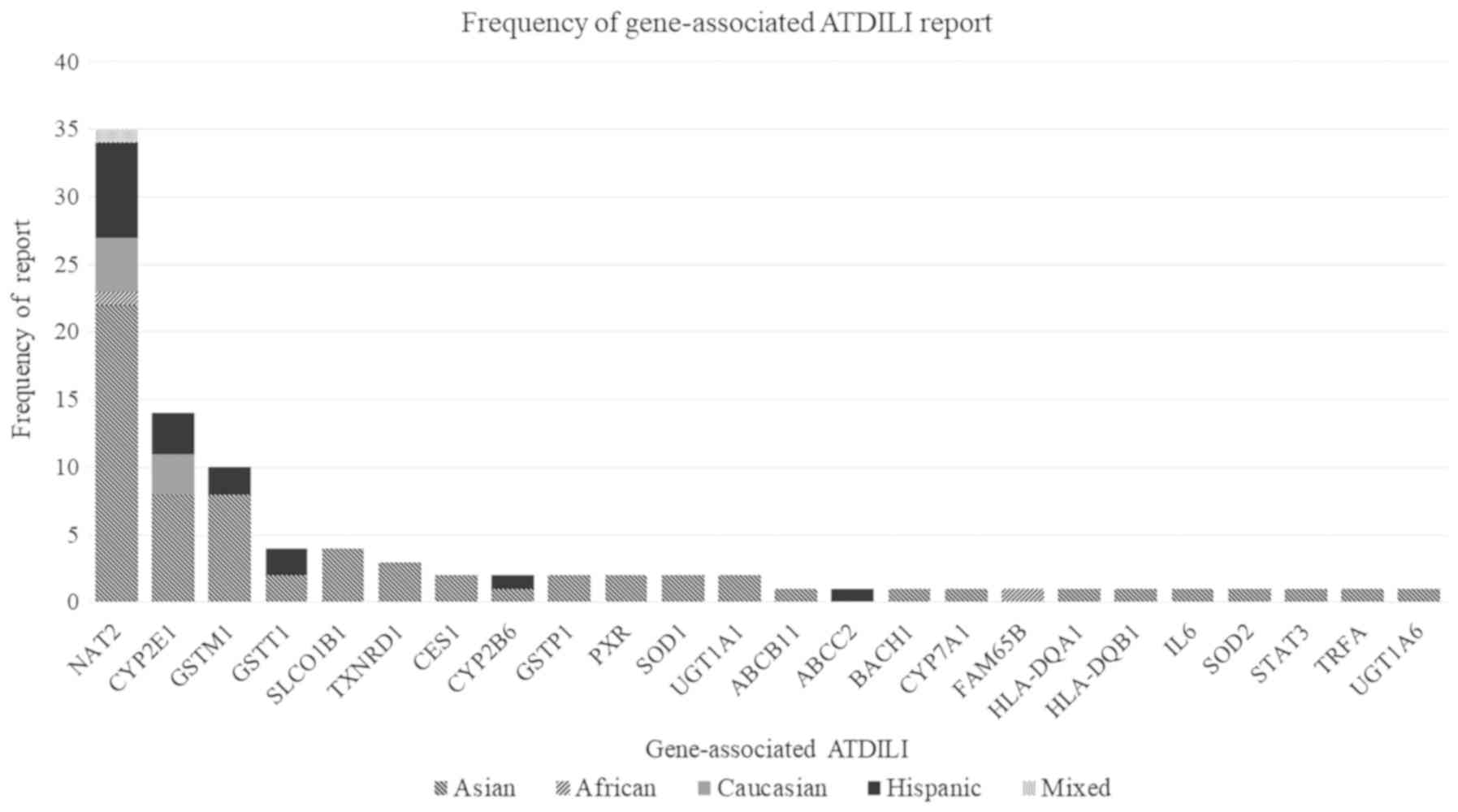
Gastroenterol Hepatol. 23:192–202. 2008.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Ramappa V and Aithal GP: Hepatotoxicity
related to anti-tuberculosis drugs: Mechanisms and management. J
Clin Exp Hepatol. 3:37–49. 2013.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Clare KE, Miller MH and Dillon JF: Genetic
factors influencing drug-induced liver injury: Do they have a role
in prevention and diagnosis? Curr Hepatol Rep. 16:258–264.
2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Bao Y, Ma X, Rasmussen TP and Zhong XB:
Genetic variations associated with anti-tuberculosis drug-induced
liver injury. Curr Pharmacol Rep. 4:171–181. 2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Eaton DL and Bammler TK: Concise review of
the glutathione S-transferases and their significance to
toxicology. Toxicol Sci. 49:156–164. 1999.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Tang N, Deng R, Wang Y, Lin M, Li H, Qiu
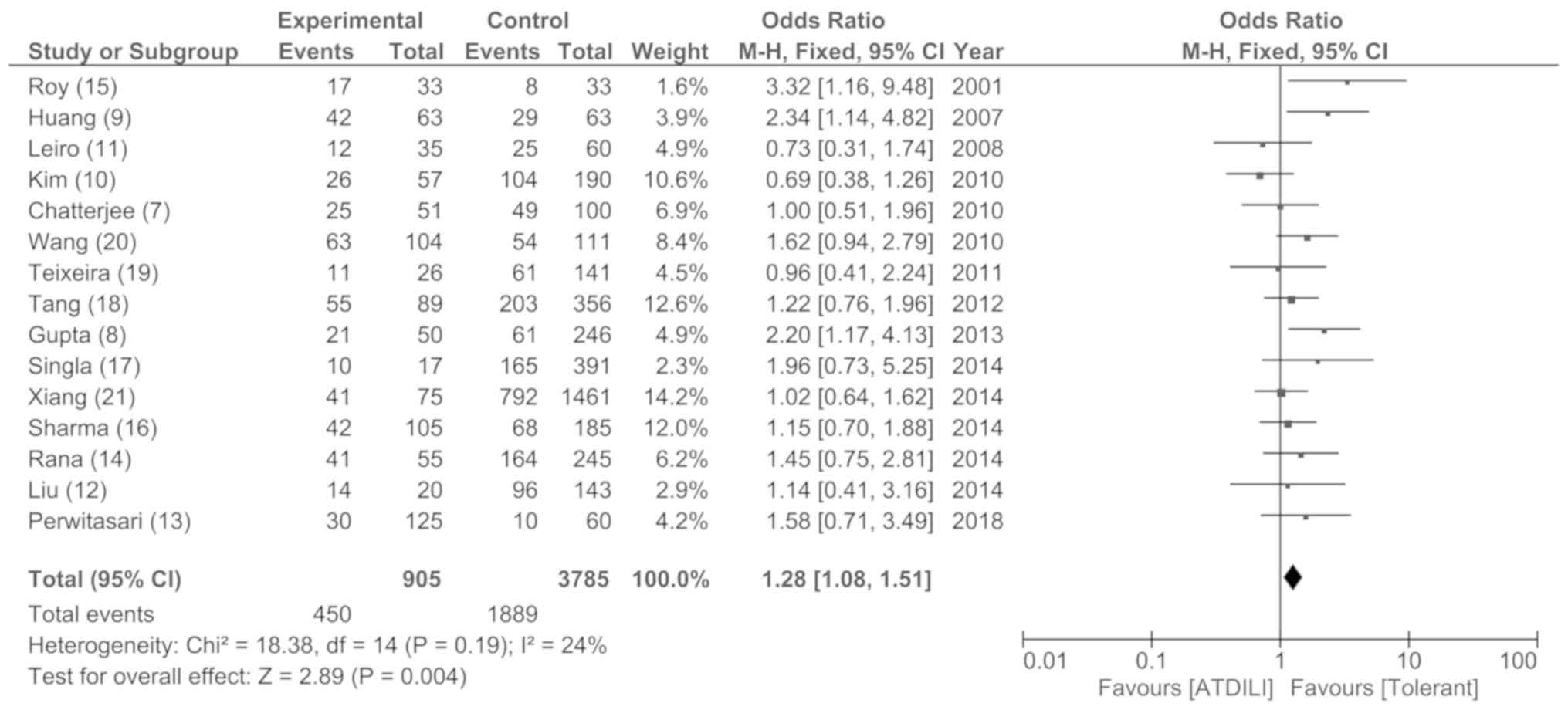
Y, Hong M and Zhou G: GSTM1 and GSTT1 null
polymorphisms and susceptibility to anti-tuberculosis drug-induced
liver injury: A meta-analysis. Int J Tuberc Lung Dis. 17:17–25.
2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Chatterjee S, Lyle N, Mandal A and Kundu
S: GSTT1 and GSTM1 gene deletions are not associated
with hepatotoxicity caused by antitubercular drugs. J Clin Pharm
Ther. 35:465–470. 2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Gupta VH, Singh M, Amarapurkar DN, Sasi P,
Joshi JM, Baijal R, H R PK, Amarapurkar AD, Joshi K and Wangikar
PP: Association of GST null genotypes with anti-tuberculosis
drug induced hepatotoxicity in western indian population. Ann
Hepatol. 12:959–965. 2013.PubMed/NCBI
|
|
9
|
Huang YS, Su WJ, Huang YH, Chen CY, Chang
FY, Lin HC and Lee SD: Genetic polymorphisms of manganese
superoxide dismutase, NAD(P)H: Quinone oxidoreductase, glutathione
S-transferase M1 and T1, and the susceptibility to drug-induced
liver injury. J Hepatol. 47:128–134. 2007.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Kim SH, Kim SH, Yoon HJ, Shin DH, Park SS,
Kim YS, Park JS and Jee YK: GSTT1 and GSTM1 null
mutations and adverse reactions induced by antituberculosis drugs
in koreans. Tuberculosis (Edinb). 90:39–43. 2010.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Leiro V, Fernandez-Villar A, Valverde D,
Constenla L, Vázquez R, Piñeiro L and González-Quintela A:
Influence of glutathione S-transferase M1 and T1 homozygous null
mutations on the risk of antituberculosis drug-induced
hepatotoxicity in a Caucasian population. Liver Int. 28:835–839.
2008.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Liu F, Jiao AX, Wu XR, Zhao W, Yin QQ, Qi
H, Jiao WW, Xiao J, Sun L, Shen C, et al: Impact of glutathione
S-transferase M1 and T1 on anti-tuberculosis drug-induced
hepatotoxicity in Chinese pediatric patients. PLoS One.
9(e115410)2014.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Perwitasari DA, Darmawan E, Mulyani UA,
Vlies PV, Alffenaar JC, Atthobar J and Wilffert B: Polymorphisms of
NAT2, CYP2E1, GST, and HLA related to
drug-induced liver injury in indonesian tuberculosis patients. Int
J Mycobacteriology. 7:380–386. 2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Rana SV, Sharma SK, Ola RP, Kamboj JK,
Malik A, Morya RK and Sinha SK: N-Acetyltransferase 2, cytochrome
P4502E1 and glutathione S-transferase genotypes in antitubercular
treatment-induced hepatotoxicity in North Indians. J Clin Pharm
Ther. 39:91–96. 2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Roy B, Chowdhury A, Kundu S, Santra A, Dey
B, Chakraborty M and Majumder PP: Increased risk of
antituberculosis drug-induced hepatotoxicity in individuals with
glutathione S-transferase M1 ‘null’ mutation. J Gastroenterol
Hepatol. 16:1033–1037. 2001.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Sharma SK, Jha BK, Sharma A, Sreenivas V,
Upadhyay V, Jaisinghani C, Singla R, Mishra HK and Soneja M:
Genetic polymorphisms of CYP2E1 and GSTM1 loci and
susceptibility to anti-tuberculosis drug-induced hepatotoxicity.
Int J Tuberc Lung Dis. 18:588–593. 2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Singla N, Gupta D, Birbian N and Singh J:
Association of NAT2, GST and CYP2E1
polymorphisms and anti-tuberculosis drug-induced hepatotoxicity.
Tuberculosis (Edinb). 94:293–298. 2014.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Tang SW, Lv XZ, Zhang Y, Wu SS, Yang ZR,
Xia YY, Tu DH, Deng PY, Ma Y, Chen DF and Zhan SY: CYP2E1,
GSTM1 and GSTT1 genetic polymorphisms and
susceptibility to antituberculosis drug-induced hepatotoxicity: A
nested case-control study. J Clin Pharm Ther. 37:588–593.
2012.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Teixeira RL, Morato RG, Cabello PH, Muniz
LM, Moreira Ada S, Kritski AL, Mello FC, Suffys PN, Miranda AB and
Santos AR: Genetic polymorphisms of NAT2, CYP2E1 and GST enzymes
and the occurrence of antituberculosis drug-induced hepatitis in
Brazilian TB patients. Mem Inst Oswaldo Cruz. 106:716–724.
2011.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Wang T, Yu HT, Wang W, Pan YY, He LX and
Wang ZY: Genetic polymorphisms of cytochrome P450 and glutathione
S-transferase associated with antituberculosis drug-induced
hepatotoxicity in Chinese tuberculosis patients. J Int Med Res.
38:977–986. 2010.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Xiang Y, Ma L, Wu W, Liu W, Li Y, Zhu X,
Wang Q, Ma J, Cao M, Wang Q, et al: The incidence of liver injury
in uyghur patients treated for TB in xinjiang uyghur autonomous
region, China, and its association with hepatic enzyme
polymorphisms NAT2, CYP2E1, GSTM1 and GSTT1. PLoS One.
9(e85905)2014.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Cai Y, Yi J, Zhou C and Shen X:
Pharmacogenetic study of drug-metabolising enzyme polymorphisms on
the risk of anti-tuberculosis drug-induced liver injury: A
meta-analysis. PLoS One. 7(e47769)2012.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Li C, Long J, Hu X and Zhou Y:
GSTM1 and GSTT1 genetic polymorphisms and risk of
anti-tuberculosis drug-induced hepatotoxicity: An updated
meta-analysis. Eur J Clin Microbiol Infect Dis. 32:859–868.
2013.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Sun F, Chen Y, Xiang Y and Zhan S:
Drug-metabolising enzyme polymorphisms and predisposition to
anti-tuberculosis drug-induced liver injury: A meta-analysis. Int J
Tuberc Lung Dis. 12:994–1002. 2008.PubMed/NCBI
|
|
25
|
Cai L, Cai MH, Wang MY, Xu YF, Chen WZ,
Qin SY, Wan CL and He L: Meta-analysis-based preliminary
exploration of the connection between ATDILI and schizophrenia by
GSTM1/T1 gene polymorphisms. PLoS One.
10(e0128643)2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
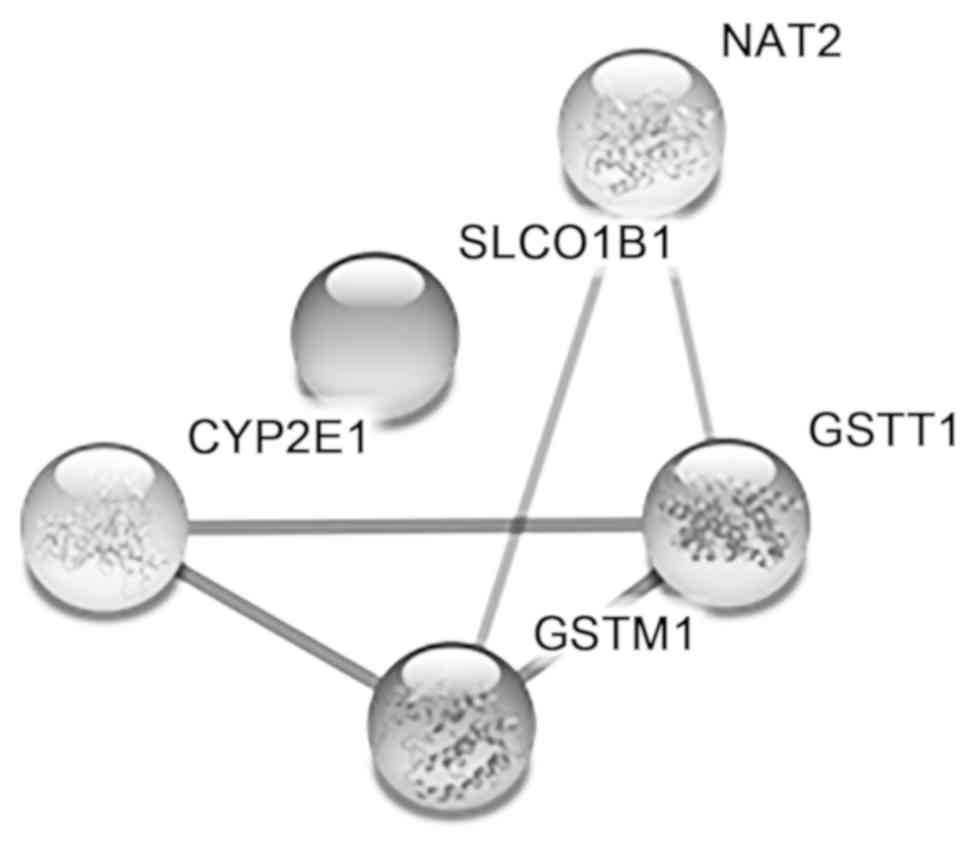
Szklarczyk D, Franceschini A, Wyder S,
Forslund K, Heller D, Huerta-Cepas J, Simonovic M, Roth A, Santos
A, Tsafou KP, et al: STRING v10: Protein-protein interaction
networks, integrated over the tree of life. Nucleic Acids Res.
43:D447–D452. 2015.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Aslam S and Emmanuel P: Formulating a
researchable question: A critical step for facilitating good
clinical research. Indian J Sex Transm Dis AIDS. 31:47–50.
2010.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Grewal A, Kataria H and Dhawan I:
Literature search for research planning and identification of
research problem. Indian J Anaesth. 60:635–639. 2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Wells G, Shea B, O'Connell D, Robertson J,
Peterson J, Welch V, Losos M and Tugwell P: The Newcastle-Ottawa
Scale (NOS) for assessing the quality of nonrandomised studies in
meta-analyses. Ottawa Hospital Research Institute. 2019.
|
|
30
|
Higgins JP and Thompson SG: Quantifying
heterogeneity in a meta-analysis. Stat Med. 21:1539–1558.
2002.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Higgins JP, Thompson SG, Deeks JJ and
Altman DG: Measuring inconsistency in meta-analyses. BMJ.
327:557–560. 2003.PubMed/NCBI View Article : Google Scholar
|
|
32
|
McGill MR and Jaeschke H: Biomarkers of
drug-induced liver injury: Progress and utility in research,
medicine, and regulation. Expert Rev Mol Diagn. 18:797–807.
2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Saukkonen JJ, Cohn DL, Jasmer RM, Schenker
S, Jereb JA, Nolan CM, Peloquin CA, Gordin FM, Nunes D, Strader DB,
et al: An official ATS statement: Hepatotoxicity of
antituberculosis therapy. Am J Respir Crit Care Med. 174:935–952.
2006.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Kanehisa M and Goto S: KEGG: Kyoto
encyclopedia of genes and genomes. Nucleic Acids Res. 28:27–30.
2000.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Wang P, Pradhan K, Zhong XB and Ma X:
Isoniazid metabolism and hepatotoxicity. Acta Pharm Sin B.
6:384–392. 2016.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Roy PD, Majumder M and Roy B:
Pharmacogenomics of anti-TB drugs-related hepatotoxicity.
Pharmacogenomics. 9:311–321. 2008.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Ioannidis JP, Trikalinos TA and Khoury MJ:
Implications of small effect sizes of individual genetic variants
on the design and interpretation of genetic association studies of
complex diseases. Am J Epidemiol. 164:609–614. 2006.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Lucena MI, Andrade RJ, Martínez C,
Ulzurrun E, García-Martín E, Borraz Y, Fernández MC, Romero-Gomez
M, Castiella A, Planas R, et al: Glutathione S-transferase m1 and
t1 null genotypes increase susceptibility to idiosyncratic
drug-induced liver injury. Hepatology. 48:588–596. 2008.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Ginsberg G, Smolenski S, Hattis D, Guyton
KZ, Johns DO and Sonawane B: Genetic polymorphism in glutathione
transferases (GST): Population distribution of GSTM1,
T1, and P1 conjugating activity. J Toxicol Environ
Health B Crit Rev. 12:389–439. 2009.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Chowdhury A, Santra A, Bhattacharjee K,
Ghatak S, Saha DR and Dhali GK: Mitochondrial oxidative stress and
permeability transition in isoniazid and rifampicin induced liver
injury in mice. J Hepatol. 45:117–126. 2006.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Zhai Q, Lu SR, Lin Y, Yang QL and Yu B:
Oxidative stress potentiated by diallylsulfide, a selective CYP2E1
inhibitor, in isoniazid toxic effect on rat primary hepatocytes.
Toxicol Lett. 183:95–98. 2008.PubMed/NCBI View Article : Google Scholar
|