Introduction
Cancer incidence is increasing globally (1), and this has caused an increase in the
search for anticancer therapies (2).
Among the adverse effects exerted by anticancer drugs (3), only a few are manageable, and the
majority of these agents significantly affect the quality of life
of the patients even after the completion of the chemotherapy
regime. One such adverse effect is ‘chemobrain’, where a severe
decline in cognitive functions are manifested. This manifestation
adversely affects the memory, learning ability, ability to process
information and day-to-day activities. The main underlying reasons
for this condition are reported to be oxidative stress due to
chemotherapy, apoptosis, chemotherapy-induced inhibition of
neuronal proliferation and differentiation and activation of
microglia (4,5). These are associated with chromatin
remodelling which causes aberrant expression of neurotrophic
proteins in the brain (6). Although
the incidence and actuality of ‘chemobrain’ have been a subject of
deliberation, recent studies have confirmed that it is not only
applicable to a majority of patients but also measurable in terms
of decrease in quality of life and outcomes of the adverse effects.
As many as 70% of patients who have undergone cancer chemotherapy
have reported that these effects persist long after the termination
of treatment, especially in patients suffering from breast
(7), ovarian (8) and prostate cancer (9).
Natural products have attracted the significant
attention of the scientific community for protection against
‘chemobrain’. A few such promising products include mangiferin from
mango pulp (10), curcumin (11), and brahmi (12). In the present study, a combination of
such natural products, which are commercially available as an
ayurvedic proprietary balya/poshak Mulmina™ Mango was assessed. The
combination of Mangifera indica fruit pulp, Centella
asiatica whole plant extract, Curcuma longa rhizome
extract along with various vitamins and minerals makes Mulmina™
Mango a potential candidate for preventive care in the case of
‘chemobrain’.
Materials and methods
Animals
Forty male Swiss Albino mice (8-10 weeks, 20-30 g
initial weight) were obtained from the inbred strains of the
Central Animal Research Facility (CARF), Manipal Academy of Higher
Education, Manipal, India. All of the experiments were performed
after approval from the Institutional Animal Ethics Committee of
Manipal Academy of Higher Education (approval no.
IAEC/KMC/115/2018) in accordance with the guidelines set out by
them, following the Committee for the Purpose of Control and
Supervision of Experiments on Animals (CPCSEA) guidelines and in
compliance with the National Institutes of Health Guide for Care
and Use of Laboratory Animals (https://grants.nih.gov/grants/olaw/guide-for-the-care-and-use-of-laboratory-animals.pdf).
The animals were housed in groups of 8, under controlled laboratory
conditions, maintained on a 12-h day and night cycle. Food and
water were available ad libitum.
Chemicals and reagents
All the reagents used for the study were of
analytical grade. Thiobarbituric acid, trichloroacetic acid,
disodium hydrogen phosphate and, sodium dihydrogen phosphate were
obtained from Sigma-Aldrich Co. LLC (Merck KGaA) and the
chemotherapeutic drugs were of pharmaceutical grade.
Cyclophosphamide (Endoxan-N), methotrexate (Biotrexate) and
5-flurouracil (Fluracil) were obtained from a local registered
pharmacy. ELISA kits were obtained from Thermo Fisher Scientific
Inc.
Drug treatments
Animals were randomly assigned into 5 groups (n=8):
Group I: Normal control; Group II: CMF
(cyclophosphamide+methotrexate+5-fluorouracil); Group III: Mulmina™
40; Group IV: Mulmina™ 80; Group V: donepezil (a medication used to
treat Alzheimer's disease). All of the treatments were administered
orally (p.o.) whereas chemotherapy was given through an
intraperitoneal (i.p.) route. Chemotherapy consisted of
cyclophosphamide (50 mg/kg), methotrexate (5 mg/kg) and
5-fluorourcail (5 mg/kg) (CMF). Mulmina™ 40 group received 40
ml/kg/day of Mulmina™ + CMF and Mulmina™ 80 group received 80
ml/kg/day of Mulmina™ + CMF. These doses were converted from human
equivalent dose as per FDA HED conversion table. Donepezil was
administered at a dose of 2 mg/kg p.o. suspended in the vehicle
0.25% w/v carboxymethylcellulose (CMC) along with CMF. The normal
control group received 10 ml/kg CMC p.o. and 0.1 ml of saline i.p.
to keep the conditions of animal handling similar across the groups
and did not receive CMF. Chemotherapy was given in three cycles
over a period of 21 days (once a week for three weeks) and the
treatments were given every day for the same 21-day period.
The health and general behaviour of the animals were
monitored twice daily, once in the morning and once in the evening.
On the days of chemotherapy, animals were closely observed for 3 h
post administration. On all other days, animals were observed for 1
h after administration of the standard drug as well as Mulmina™.
The animals were administered with the standard drug and Mulmina™
between 8-9 a.m. in the morning and between 4-5 p.m. in the
evening.
On completion of the chemotherapy cycle, the open
field test was performed to evaluate any loss of locomotor activity
following which, the water maze test was performed. After the water
maze test, the mice were anesthetised with thiopentone sodium (25
mg/kg, i.v.), and blood was withdrawn via retro-orbital puncture
for biochemical estimations. Later, the same animals were
sacrificed with carbon dioxide (CO2) overdose (flow rate
of 20% chamber volume per min) as per BU ASC guidelines (13), and brain tissue was collected for
biochemical estimations.
Behavioral parameters Open field
test
The open field test (OFT) was used to assess the
effect of the treatment groups on exploratory behavior of the
animals. Locomotor activity (LMA) was assessed in mice individually
placed into a clean, novel glass jar (30x30x60 cm). The open field
was divided into nine virtual quadrants (10x10 cm) and LMA was
measured by counting the number of line crossings, latency to first
line crossing, centre square entries, time spent in the centre
square and, time spent in the periphery over a 5-min period. The
apparatus was cleaned with 70% ethanol between experiments as
previously described (14).
Morris water maze test
The maze was a circular pool with a diameter of 150
cm with an escape platform of 40 cm height. The pool was virtually
divided into 4 quadrants: North-west, north-east, south-west and
south-east using ANY-maze software (Stoelting Co.). The escape
platform was kept 2 cm below the water and was 10 cm in dimension.
The pool was filled with fresh water every day before experiments
and maintained at the temperature of 27˚C. The parameters observed
were escape latency and retention time as previously described
(15).
Estimation of antioxidant parameters
in brain homogenate Catalase
Catalase activity was measured in the whole brain
homogenate. A total of 0.1 ml of supernatant was added to a cuvette
containing 1.9 of 50 mM phosphate buffer (pH 7.0). The reaction was
started by the addition of 1 ml freshly prepared 10 mM
H2O2. The rate of decomposition of
H2O2 was measured spectrophotometrically from
the changes in absorbance at 240 nm. The activity of catalase was
expressed as µmol of H2O2 decomposed/min/mg
protein as described (16).
Lipid peroxidation
The TBA-TCA-HCl reagent was sonicated and mildly
heated to dissolve the components. Thereafter, 0.5 ml of brain
homogenate and 1 ml of TBA-TCA reagent was heated at 80˚C for 10
min. The reaction mixture was centrifuged at 4˚C at 2,000 x g for
20 min. The amount of malondialdehyde (MDA) was measured
spectrophotometrically at 532 nm as previously described (17).
Estimation of hematological
parameters
Estimation of the hematological parameters was
performed using veterinary blood cell counter (PCE-210 Fully
Automatic Blood Cell Counter, ERMA Inc., Tokyo, Japan). The tested
parameters were red blood cell count (106/µl),
haemoglobin (g/dl), white blood cell count (103/µl),
number of monocytes (103/µl), lymphocytes
(103/µl), granulocytes (103/µl) and platelets
(103/µl).
Estimation of IL-6, TNF-α and
BDNF
ELISAs were performed for these parameters as per
the manufacturer's instructions. ELISA kit for interleukin (IL)-6
(E-EL-M0044), tumor necrosis factor (TNF)-α (E-EL-M0049), and
brain-derived neurotrophic factor (BDNF) (E-EL-M0203) were obtained
from Elabscience Inc.
Statistical analysis
Data are expressed as mean ± SEM (n=8) and were
analyzed using GraphPad Prism version 8.4 (GraphPad Software,
Inc.). All the parameters in the study were analyzed by one-way
analysis of variance (ANOVA) followed by Bonferroni as the post-hoc
test. The difference between the two groups was considered
statistically significant at P<0.05.
Results
Memory and locomotor activity
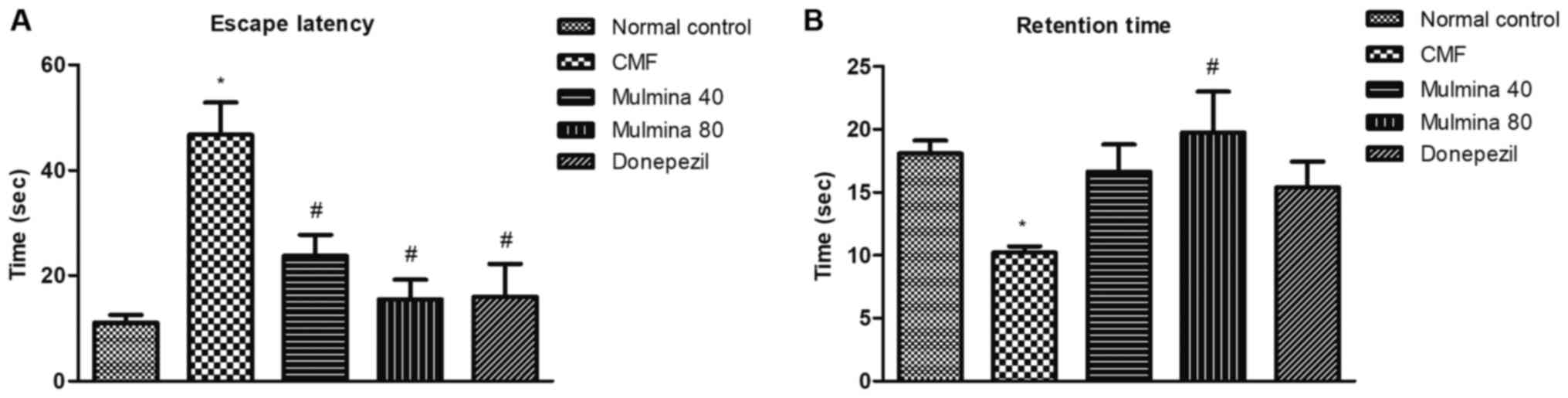
Assessment of memory by escape latency (ELT) and retention time
(RT) using the Morris water maze
ELT is the amount of time each animal takes to reach
the island zone for the first time during the retention trial. The
difference between the normal control (11±1.4 sec) and CMF (47±6.1
sec) groups was found to be significant (P<0.05). Low dose [40
ml/kg/day (24±4.0 sec)] as well as high dose [80 ml/kg/day (16±3.7
sec)] of Mulmina™ significantly reduced the escape latency compared
to the CMF group. Similarly, donepezil (16±6.3 sec) was also found
to be significant in reducing the escape latency (P<0.05)
compared to the CMF group (Fig. 1A).
RT is the amount of time the animal spends in D-quadrant during the
duration of the retention trial which was 60 sec. The CMF (10±0.49
sec) group was found to be significantly different from the normal
control (18±1.1 sec) (P<0.05). Although the low dose of Mulmina™
(17±2.2 sec) and donepezil (15±2.0 sec) did not show a significant
difference, a high dose of Mulmina™ (20±3.3 sec) was found to be
significant at P<0.05 compared to the CMF group (Fig. 1B).
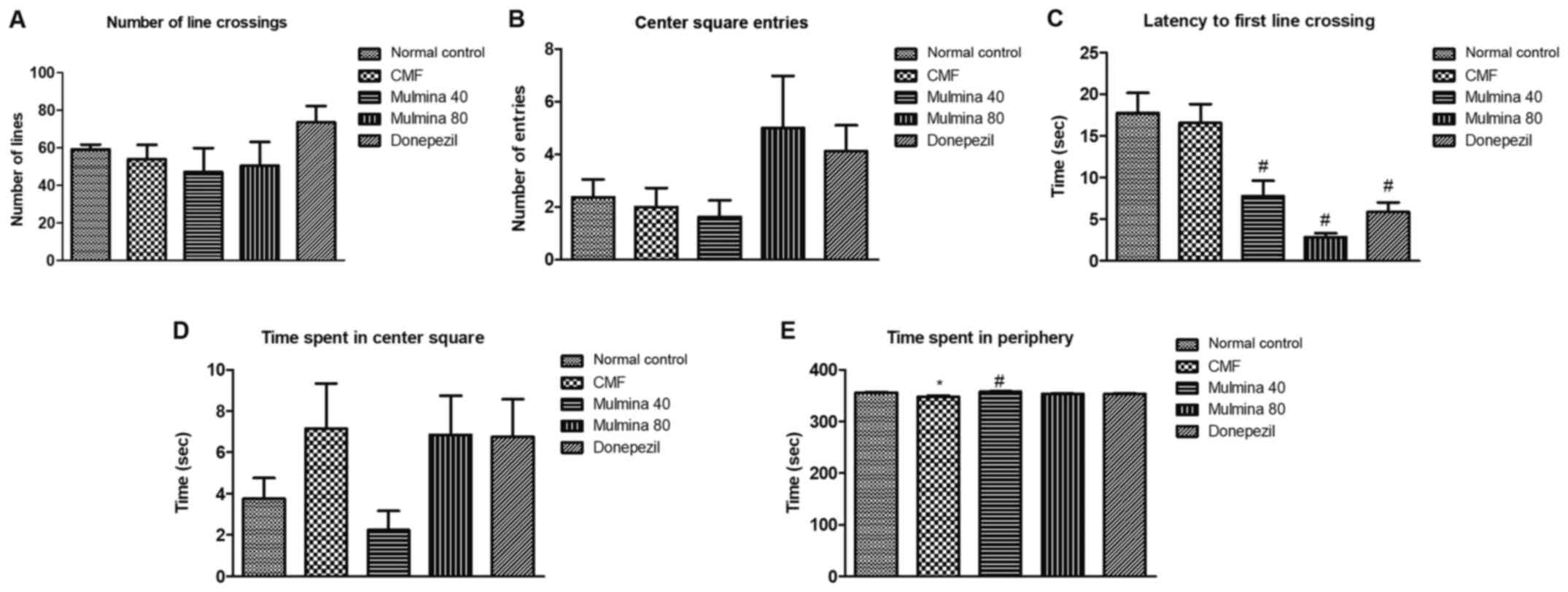
Assessment of locomotor activity by
open field test
The number of line crossings (LC) is the number of
lines the animal crosses while exploring the arena which is divided
into nine equal squares and reflects upon the locomotor activity of
the animal directly. No significant difference was observed between
the groups in regards to LC (Fig.
2A). Center square entry (CS) is the number of times an animal
enters into the center square in the arena while exploring it. No
statistically significant difference was observed in terms of CS
(Fig. 2B). Latency to first line
crossing (FLC) is the measure of the time taken by an animal to
cross the first line after being introduced into the arena and is
measured in sec. The low (7.8±1.9 sec) as well as the high dose of
Mulmina™ (2.9±0.46 sec) and donepezil (5.9±1.2 sec) significantly
reduced the FLC when compared to the CMF group (17±2.3 sec)
(P<0.05) (Fig. 2C). Time spent in
the center square (CST) is the amount of time an animal spends in
the center square while exploring the open field arena and is
measured in sec. No significant difference was observed between the
groups in regards to CST (Fig. 2D).
Time spent in the periphery (PT) is the amount of time an animal
spends in the periphery/along the walls of the open field arena and
is measured in sec. The CMF (347±2.9 sec) group spent significantly
more amount of time in the periphery as compared to the normal
group (356±1.0 sec) (P<0.05) while Mulmina™ 40 (358±0.92 sec)
treated animals spent significantly lesser time in the periphery as
compared to the CMF group (347±2.9 sec) (Fig. 2E).
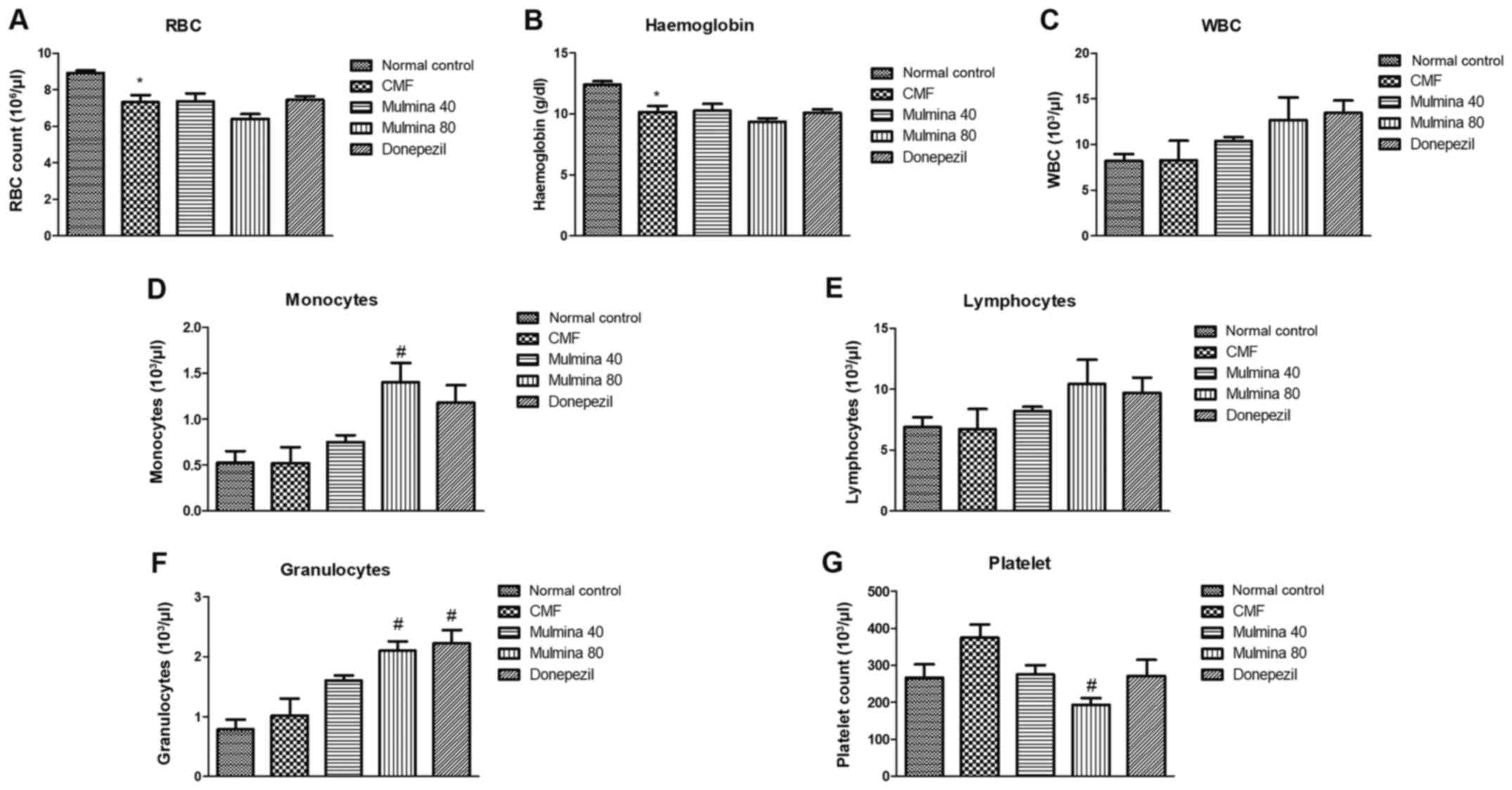
Hematological parameters Effect of
test drugs on the complete blood cell count
The trend observed in red blood cell (RBC) count and
hemoglobin content was almost similar whereas the RBC count and
hemoglobin were found to be significantly reduced in the CMF group
(7.31±0.39 106/µl; 12.38±0.30 g/dl) compared to the
normal control (8.92±0.14 106/µl; 6.84±0.63 g/dl) but
changes in any other group were statistically insignificant
(Fig. 3A and B). CMF treatment increased the total WBC
count in all the groups compared to the normal control group. This
difference, although evident, was not found to be significant
(Fig. 3C). The mice showed an
elevation in monocyte count (103/µl) in all the groups
upon treatment with CMF. This elevation was statistically
significant in the high dose Mulmina™ group (1.40±0.81) when
compared to the normal control (0.52±0.12) (P<0.05) (Fig. 3D). No statistical significance in
lymphocyte count was observed among the treatment groups and CMF
when compared to the normal control (Fig. 3E). CMF administration increased the
granulocyte count (103/µl) among all the test groups but
only the high dose of Mulmina™ (2.10±0.15) and donepezil
(2.22±0.22) showed a statistically significant increase in the
count when compared to the normal control (0.78±0.16) (P<0.05)
(Fig. 3F). Following administration
of CMF, the platelet count (103/µl) was observed to be
elevated in the CMF group, but it was not significant
statistically. Among the test groups, the high dose of Mulmina™
group showed a significant decrease in platelet count when compared
to the normal control (P<0.05) (Fig.
3G).
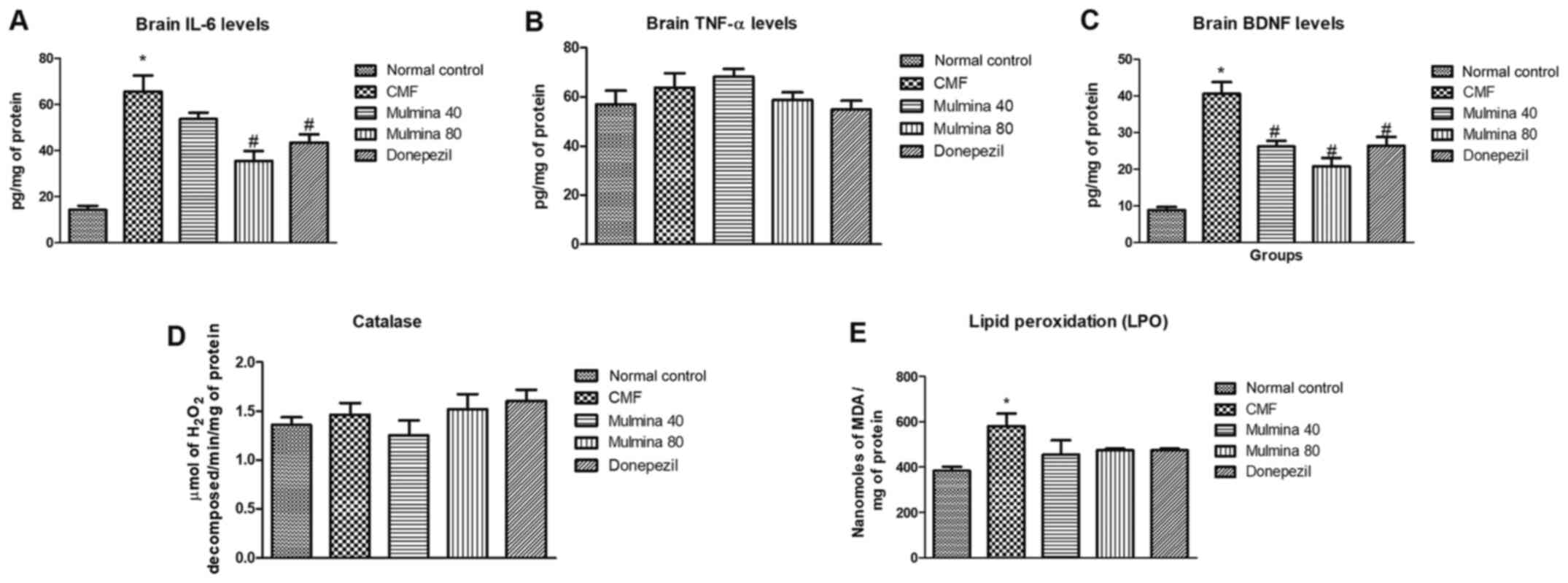
Biochemical parameters Effect of test
drugs on the levels of cytokines in the mouse brain
Interleukin (IL)-6 is predominantly a
pro-inflammatory cytokine whose expression as well as released
levels have been reported to increase in acute as well as chronic
inflammatory conditions and is a well-known player in
neuroinflammatory conditions. For brain IL-6, the difference
between the normal control (14±1.8) and CMF group (66±7.0) was
found to be significant (P<0.05). Although the low dose of
Mulmina™ (54±2.7) did reduce the level of IL-6, the difference was
not statistically significant. The high dose group (35±4.4) showed
a significant reduction (P<0.0.05) as well as the donepezil
group (44±3.5) (P<0.05) (Fig.
4A). Tumor necrosis factor (TNF)-α is a cytokine involved in
systemic inflammation and a key mediator in acute phase reaction of
inflammation. We did not find any significant difference between
the groups with respect to brain TNF-α levels (Fig. 4B).
Effect of test drugs on BDNF in the
brain
Brain derived neurotrophic factor (BDNF) is a
neuronal growth factor which regulates the survival of existing
neurons as well as growth and differentiation of new neurons and
synapses. We observed a significant (P<0.05) increase in BDNF in
the CMF group (41±3.2) when compared to the normal control
(8.9±0.81). This increase was in line with the earlier published
report which showed that activation of microglial cells for an
extended time triggers a release of BDNF (18). This long-term activation of microglia
was seen in the CMF model. All the treatment groups were able to
decrease this elevation in BDNF showing a reduction in inflammation
indirectly. Low (26±1.6) as well as high (21±2.3) dose of Mulmina™
(26±1.6) and donepezil (26±2.4) were significantly able to reduce
the BDNF levels as compared to CMF group (P<0.05) (Fig. 4C).
Effect of test drugs on the level of
oxidative stress in the brain
Catalase is an endogenous antioxidant enzyme and its
levels can be estimated spectrophotometrically by assessing the
rate of degradation of an externally added known quantity of
hydrogen peroxide to tissue supernatants. No significant difference
was observed in regards to the catalase levels among the groups
(Fig. 4D). Lipid peroxidation (LPO)
is the process by which free radicals scavenge the electrons from
lipids in cell membranes causing a destructive damage to tissue.
The CMF (581±55) group showed a significant increase in lipid
peroxidation confirming the injury, compared to the normal control
(383±19) group. There was a decrease in the levels of LPO among the
treatment groups as well, but the differences were not
statistically significant (Fig.
4E).
Discussion
In the present study, mice were challenged with
cyclophosphamide, methotrexate and 5-fluorouracil (CMF)
chemotherapy. The effect of proprietary ayurvedic balya/poshak
Mulmina™ Mango against chemotherapy-induced cognitive decline
(CICD) was investigated. In the majority of cases, ‘chemobrain’
presents itself with cognitive dysfunction along with elevated
pro-inflammatory cytokines in the brain, increased neuronal cell
death and a severe decline in neurogenesis. Three cycles, over a
period of 21 days of CMF chemotherapy were able to elicit these
effects thus establishing the validity of the model. Two doses of
Mulmina™ i.e. 40 and 80 mg/kg/day were chosen for this study which
were directly converted from the human dose and administered to
mice through an oral route every day in equally divided doses. When
put through the Morris water maze, the mice treated with both doses
of Mulmina™ showed significant improvement in spatial memory of the
animals.
The Morris water maze results can be affected
positively as well as negatively if the locomotion of animals is
disturbed. To assure that the results obtained from the Morris
water maze were dependant on the effect of test drugs only, we
accessed the locomotion of animals using the Open Field Test which
concluded that all the animals were equal in cognitive abilities
before the beginning of the water maze test.
To correlate the behavioral data with biochemical
and hematologic parameters, a battery of assays and estimations
were performed. In regards to the hematological parameters, the CMF
group showed a decline in red blood count (RBC) count which was not
increased by any of the treatments. With respect to total and
differential white blood cell (WBC) count, our results indicated an
increase in total white blood cell count as well as in monocytes,
lymphocytes, and granulocytes throughout the test groups, except
for the normal control, which is a clear indicator of exacerbated
inflammatory reaction in the body. On estimation of some common
cytokines involved in the condition, the levels of IL-6 were highly
elevated in the brain in the CMF group and the high dose of
Mulmina™ significantly restored the levels of pro-inflammatory
cytokines. We also evaluated basic markers of oxidative stress in
the brain. Lipid peroxidation was significantly increased in the
CMF group. However, Mulmina™ did not restore the brain lipid
peroxidation levels induced by CMF treatment.
We employed donepezil as our standard drug in the
present study, and it was observed that there were no significant
differences between the effects shown by Mulmina™ and donepezil in
the study. Mulmina™ contains fruit pulp of Mangifera indica,
whole plant extract of Centella asiatica and, rhizome powder
of Curcuma longa. Mangiferin, the main constituent of
Mangifera indica, has been reported to possess antioxidant,
anti-inflammatory, immunomodulation and anti-apoptosis activities.
Since mangiferin can cross the blood-brain barrier, it can
potentially modulate the damage caused by chemotherapy in brain as
well as the periphery (19).
Centella asiatica contains a triterpenoid called asiatic
acid. Asiatic acid has demonstrated the ability to reduce oxidative
stress and inflammation in rat brain (20). Curcumins in Curcuma longa are
also known to reduce the inflammation as well as oxidative stress
through inhibition of apoptosis, TNF-α, iNOS, RNS, COX-2, and LOX
(21).
Although it was confirm in the present study that
Mulmina™ has some effect against CMF-mediated CICD, further
research is warranted, especially at the molecular level to come to
a definitive conclusion with respect to the alterations in proteins
in apoptotic/pyroptotic pathways, potency and the possible
long-term curative/prophylactic beneficial effects of Mulmina™.
Acknowledgements
The authors would like to acknowledge Manipal
Academy of Higher Education for providing the infrastructure
required to carry out the work. We also acknowledge the
collaboration between Manipal Academy of Higher Education and
Griffith University, Gold Coast, Australia, from which the animal
model was developed and validated by Dr Devinder Arora, Senior
Lecturer, School of Pharmacy and Pharmacology, Griffith University,
QLD 4222, Australia.
Funding
The project was supported by Juggat Pharma, Jagdale
Industries Pvt. Ltd., Bengaluru.
Availability of data and materials
The datasets used during the present study are
available from the corresponding author upon reasonable
request.
Authors' contributions
Conception and design of the study was carried out
by JM, KN along with RNJ and SMA. Data acquisition was conducted by
MK, NR and KG. Data analysis and interpretation was carried out by
MK, KG, PGN, JM and KN. Critical intellectual input during the
study was accomplished by KN and KVR. Draft preparation was carried
out by MK, NR, PGN, JM and KN. All authors read and approved the
manuscript and agree to be accountable for all aspects of the
research in ensuring that the accuracy or integrity of any part of
the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
All guidelines dictated by the Institutional Animal
Ethics Committee of Manipal Academy of Higher Education (MAHE)
(Manipal, Karnataka, India) (approval no. IAEC/KMC/115/2018) and
Committee for the Purpose of Control and Supervision of Experiments
on Animals (CPCSEA) were followed while conducting the animal
experiments.
Patient consent for publication
Not applicable.
Competing interests
The authors do declare a competing interest. This
research was a collaborative project between the Department of
Pharmacology, Manipal College of Pharmaceutical Sciences, Manipal
Academy of Higher Education, Manipal and Juggat Pharma, Jagdale
Industries Pvt. Ltd., Bengaluru. Jagdale Industries sponsored the
study and were involved in the design of the study for the purposes
of assessing their marketable product. However, the work was
carried out at the Department of Pharmacology, Manipal College of
Pharmaceutical Sciences where the study was independently performed
in an unbiased manner and data were reported based on the
experimental observations.
References
|
1
|
You W and Henneberg M: Cancer incidence
increasing globally: The role of relaxed natural selection. Evol
Appl. 11:140–152. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Hu Q, Sun W, Wang C and Gu Z: Recent
advances of cocktail chemotherapy by combination drug delivery
systems. Adv Drug Deliv Rev. 98:19–34. 2016.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Nurgali K, Jagoe RT and Abalo R:
Editorial: Adverse effects of cancer chemotherapy: Anything new to
improve tolerance and reduce sequelae? Front Pharmacol.
9(245)2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Aluise CD, Sultana R, Tangpong J, Vore M,
St Clair D, Moscow JA and Butterfield DA: Chemo brain (chemo fog)
as a potential side effect of doxorubicin administration: Role of
cytokine-induced, oxidative/nitrosative stress in cognitive
dysfunction. Adv Exp Med Biol. 678:147–156. 2010.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Staat K and Segatore M: The phenomenon of
chemo brain. Clin J Oncol Nurs. 9:713–721. 2005.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Briones TL and Woods J:
Chemotherapy-induced cognitive impairment is associated with
decreases in cell proliferation and histone modifications. BMC
Neurosci. 12(124)2011.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Jim HS, Phillips KM, Chait S, Faul LA,
Popa MA, Lee YH, Hussin MG, Jacobsen PB and Small BJ: Meta-analysis
of cognitive functioning in breast cancer survivors previously
treated with standard-dose chemotherapy. J Clin Oncol.
30:3578–3587. 2012.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Correa DD and Hess LM: Cognitive function
and quality of life in ovarian cancer. Gynecol Oncol. 124:404–409.
2012.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Chao HH, Uchio E, Zhang S, Hu S, Bednarski
SR, Luo X, Rose M, Concato J and Li CS: Effects of androgen
deprivation on brain function in prostate cancer patients-a
prospective observational cohort analysis. BMC Cancer.
12(371)2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Wattanathorn J, Muchimapura S, Thukham-Mee
W, Ingkaninan K and Wittaya-Areekul S: Mangifera indica fruit
extract improves memory impairment, cholinergic dysfunction, and
oxidative stress damage in animal model of mild cognitive
impairment. Oxid Med Cell Longev. 2014(132097)2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Wu A, Ying Z and Gomez-Pinilla F: Dietary
curcumin counteracts the outcome of traumatic brain injury on
oxidative stress, synaptic plasticity, and cognition. Exp Neuro.
197:309–317. 2006.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Gray NE, Harris CJ, Quinn JF and
Soumyanath A: Centella asiatica modulates antioxidant and
mitochondrial pathways and improves cognitive function in mice. J
Ethnopharmacol. 180:78–86. 2016.PubMed/NCBI View Article : Google Scholar
|
|
13
|
BU ASC Euthanasia Guidelines, 2019.
Available at: urihttps://www.bu.edu/researchsupport/compliance/animal-care/working-with-animals/euthanasia/euthanasia-guidelines/simplehttps://www.bu.edu/researchsupport/compliance/animal-care/working-with-animals/euthanasia/euthanasia-guidelines/
(Accessed on August 14, 2020).
|
|
14
|
Biesmans S, Meert TF, Bouwknecht JA, Acton
PD, Davoodi N, De Haes P, Kuijlaars J, Langlois X, Matthews LJ, Ver
Donck L, et al: Systemic immune activation leads to
neuroinflammation and sickness behavior in mice. Mediators Inflamm.
2013(271359)2013.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Vorhees CV and Williams MT: Morris water
maze: Procedures for assessing spatial and related forms of
learning and memory. Nat Protoc. 1:848–858. 2006.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Goldblith SA and Proctor BE: Photometric
determination of catalase activity. J Biol Chem. 187:705–709.
1950.PubMed/NCBI
|
|
17
|
Ohkawa H, Ohishi N and Yagi K: Assay for
lipid peroxides in animal tissues by thiobarbituric acid reaction.
Anal Biochem. 95:351–358. 1979.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Gomes C, Ferreira R, George J, Sanches R,
Rodrigues DI, Gonçalves N and Cunha RA: Activation of microglial
cells triggers a release of brain-derived neurotrophic factor
(BDNF) inducing their proliferation in an adenosine A2A
receptor-dependent manner: A2A receptor blockade prevents BDNF
release and proliferation of microglia. J Neuroinflammation.
10(16)2013.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Feng ST, Wang ZZ, Yuan YH, Sun HM, Chen NH
and Zhang Y: Mangiferin: A multipotent natural product preventing
neurodegeneration in Alzheimer's and Parkinson's disease models.
Pharmacol Res. 146(104336)2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Ahmad Rather M, Justin-Thenmozhi A,
Manivasagam T, Saravanababu C, Guillemin GJ and Essa MM: Asiatic
acid attenuated aluminum chloride-induced tau pathology, oxidative
stress and apoptosis via AKT/GSK-3β signaling pathway in wistar
rats. Neurotox Res. 35:955–968. 2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Ghasemi F, Shafiee M, Banikazemi Z,
Pourhanifeh MH, Khanbabaei H, Shamshirian A, Amiri Moghadam S,
ArefNezhad R, Sahebkar A, Avan A and Mirzaei H: Curcumin inhibits
NF-kB and Wnt/β-catenin pathways in cervical cancer cells. Pathol
Res Pract. 215(152556)2019.PubMed/NCBI View Article : Google Scholar
|