Introduction
Cystic lesions are amongst the most common
pathologies diagnosed in the maxillofacial region. They may occur
in facial soft tissues as well as the craniofacial skeleton.
Moreover, these anomalies are characterized by distinct
etiopathogenetic processes (1-3).
The most frequent cystic lesions localized within the jaws are
radicular cysts (RCs), which are associated with the development of
periapical granulomas (PGs) that form as a result of untreated
periapical periodontitis (4). The
second most prevalent type of odontogenic cystic lesion, the
dentigerous cyst (DC), is characterized as a developmental cyst
(5). Despite the distinct
differences in the etiology of the respective pathological lesions,
their growth processes are predominantly associated with resorption
and remodeling of bone tissue (6).
Numerous studies have been conducted to investigate the mechanisms
involved in both types of lesions. MMPs are a group of proteins
potentially involved in cyst formation and bone destruction
(7-11).
This family of zinc-dependent enzymes is responsible for the
degradation of the extracellular matrix (ECM) during various
physiological and pathological processes, including in
embryogenesis, organization of dentin organic matrix, arthritis and
ischemic stroke (12,13). Moreover, proteolytic activity of
these endopeptidases is observed in angiogenesis during wound
healing and cancer metastasis (14,15).
Tissue inhibitors of metalloproteinases (TIMPs) comprise a group of
specific endogenous enzymes regulating the activity of MMPs. Thus,
TIMPs serve a key role in the maintenance of homeostasis and proper
functioning of the body as a whole (16).
MMP-2 and MMP-9 belong to a well-known subgroup of
MMPs termed gelatinases (11,17).
Since some of bone organic components (for example, type I collagen
and fibronectin) are substrates for these endopeptidases, the
possible involvement of MMP-2 (also known as gelatinase A) and
MMP-9 (also known as gelatinase B) in the etiology of cystic
lesions has been investigated (18,19).
Accordingly, cleavage of the organic elements of the ECM in bone
tissue may lead to cyst formation, and gene expression analysis
revealed higher expression of MMP-2, TIMP-1 and TIMP-2 in PGs and
RCs, underlining its significant role in the etiology of these
disorders (20). Ameloblastomas,
which are considered as more expansive pathologies than benign
lesions, are also characterized by higher expression of MMPs
(21). However, the etiopathogenesis
of DCs and MMPs remains unclear (22). Retention cysts (RtCs) are lesions
occurring within the maxillary sinuses and exhibit a lower
destructive potential compared with typical odontogenic cysts
(23). Unfortunately, our knowledge
regarding the involvement of MMPs in the etiology of RtCs is
limited. Searching PubMed with the phrase ‘retention cysts’ and
‘MMP’ returned only 1 article (24).
Furthermore, to the best of our knowledge, there are currently no
studies comparing MMP activity and TIMP levels between RtCs and the
aforementioned odontogenic cysts.
Therefore, the aim of the present study was to
understand and characterize the role of MMP-2, MMP-9 and their
tissue inhibitors, TIMP-1 and TIMP-2, in the pathogenesis of cystic
lesions of the maxillofacial region.
Materials and methods
Patients
A total of 25 patients (16 males and 9 females),
aged 18-66 years old (median age, 43.2), who were diagnosed with
cystic lesions of the maxillofacial region were enrolled in the
present study. In the investigated group of patients, 20 RCs, 7
RtCs and 3 DCs were identified using clinical examination, computed
tomography and/or panoramic X-ray imaging. The higher number of
cysts detected than patients recruited was due to the presence of
multiple comorbidities amongst patients. In addition, 2 separate
RCs were identified in each patient of a group of 3 patients; 1
patient was diagnosed with 2 RCs and 1 RtC; and 1 RC and 1 RtC were
identified in 1 patient. The characteristics of all the lesions
were confirmed by preliminary histopathological examination.
Studies were performed using cystic tissues collected during
surgical treatment conducted at the Department of Maxillofacial
Surgery, Medical University of Lublin. A total of 2 lesions with
inconsistencies between the clinical, histopathological or
radiological diagnoses were excluded from the study. Moreover, due
to the non-specific microscopic characteristics of RtCs,
concomitant histopathological, clinical and radiological
examinations were considered during evaluation for inclusion in the
study (25).
All study protocols were reviewed and approved by
the Ethics Committee at the Medical University of Lublin (approval
no. KE-0254/5/2017). Each patient included in the study provided
signed informed consent. All procedures followed the guidelines
outlined in the Declaration of Helsinki (26). The characteristics of the study group
are summarized in Table I.
 | Table ICharacteristics of the study
group. |
Table I
Characteristics of the study
group.
|
Characteristics | RC, n=20 | RtC, n=7 | DC, n=3 |
|---|
| Average age,
years | 42.4 | 41.6 | 44.3 |
| Sex,
male/female | 12/5 | 4/3 | 1/2 |
| Localization,
maxilla/mandible | 16/4 | - | 1/2 |
| Maxillary sinus,
right/left | - | 2/5 | - |
| Average largest
dimension, mm | 21.8 | 27.2 | 14.3 |
Sample collection and preparation
A sample of venous blood was routinely collected
prior to surgery under general anesthesia. The blood was
centrifuged at 1,800 x g for 20 min at room temperature and 0.5-1.0
ml of the obtained serum was collected in Eppendorf tubes and
stored at -30˚C. The remaining serum was used to perform
biochemical blood tests. Cystic lesions were exposed during
surgery, and cystic fluid was obtained by diagnostic puncture and
aspiration from the cavity using a sterile syringe. Subsequently,
enucleation of the entire cyst capsule was performed. Finally, a
small fragment (0.3-0.5 g) was detached from the capsule using a
sterile surgical blade. All cyst tissue samples were placed in
separate Eppendorf tubes and frozen at -30˚C. The primary part of
the lesion was subjected to histopathological examination at the
Pathomorphology Unit of the Independent Public Teaching Hospital
No. 1 to confirm the initial diagnosis. All subsequent tests were
performed at the Department of Medical Chemistry, Medical
University of Lublin.
The samples detached from the capsules were
homogenized using buffer containing 1% Triton X-100 and centrifuged
at 1,800 x g for 20 min at room temperature. Protein levels in all
supernatant samples were determined using commercial Bradford
reagent (Bio-Rad Laboratories, Inc.) according to manufacturer's
protocol (18 µg/ml). In each examined case, serum and cyst fluid
samples were diluted (1:50) with deionized water.
Zymography
All zymographic procedures (incubation, protein
separation, staining/distaining) were performed at room
temperature. The proteolytic activity of MMP-2 and MMP-9 in the
serum, cyst fluid and cyst capsules were detected and evaluated
using gelatin zymography (27).
Initially, 30 µl study material was mixed with 10 µl sample buffer
with 10% SDS and incubated for 30 min. Subsequently, 20 µl of this
solution was loaded on a 10% SDS-gel with 0.05% gelatine type A
from porcine skin (Sigma-Aldrich; Merck KGaA), and resolved using
SDS-PAGE for 90 min at 30 mA/gel at room temperature. Next, the
resolved proteins were incubated with Tris-HCl buffer (pH 7.2)
containing 10 mM CaCl2, 0.02% NaN3 and 2.5%
Triton X-100 twice for 30 min. Subsequent incubation was performed
for 18 h at 37˚C using Tris-HCl buffer (pH 7.2) containing 10 mM
CaCl2, 0.02% NaN3 and 1% Triton X-100. Gels
were stained with 0.1% Coomassie Blue R-250 in solution of 30%
ethanol and 10% acetic acid for 4 h. Finally, they were destained
twice in solution of 30% ethanol and 10% acetic acid. The emergence
of clear bands against the blue background indicated the activity
of MMP-2 and MMP-9(28).
Investigation using gelatine zymography was performed
simultaneously on two separate gels to avoid inconclusive
results.
Enzymes were identified by comparing their
localization with molecular mass standards (SM0441; Fermentas;
Thermo Fisher Scientific, Inc.) and with standards of both
gelatinases (911-MP and 902-MP; R&D Systems, Inc.).
Quantitative analysis of MMP activity was performed using a
computer scanner (1,200 dpi) and ImageJ 1.51.9 (National Institutes
of Health). The activity of the tested enzymes was expressed as the
optical density (OD) of the substrate lysis zone.
ELISA
TIMP-1 and TIMP-2 levels were detected in serum and
cyst fluid using commercially available ELISA kits (cat. nos.
DMP100 and DMP200; R&D Systems Inc.) according to the
manufacturer's protocol. Both tissue inhibitors were diluted 100
times with the diluent included in the kit. All results were
detected using a microplate reader (Epoch; BioTek Instruments,
Inc.) at a wavelength of 450 nm. Moreover, MMP-9/TIMP-1 and
MMP-2/TIMP-2 ratios, as calculated from zymography and ELISA
results, were expressed in OD/ng/ml.
Statistical analysis
A Kruskal-Wallis test followed by a Dunns' post hoc
test was used for statistical analysis. Data are presented as the
median and the interquartile range. Statistical analysis was
performed using InStat version 3.06 (GraphPad Software, Inc.).
P<0.05 was considered to indicate a statistically significant
difference.
Results
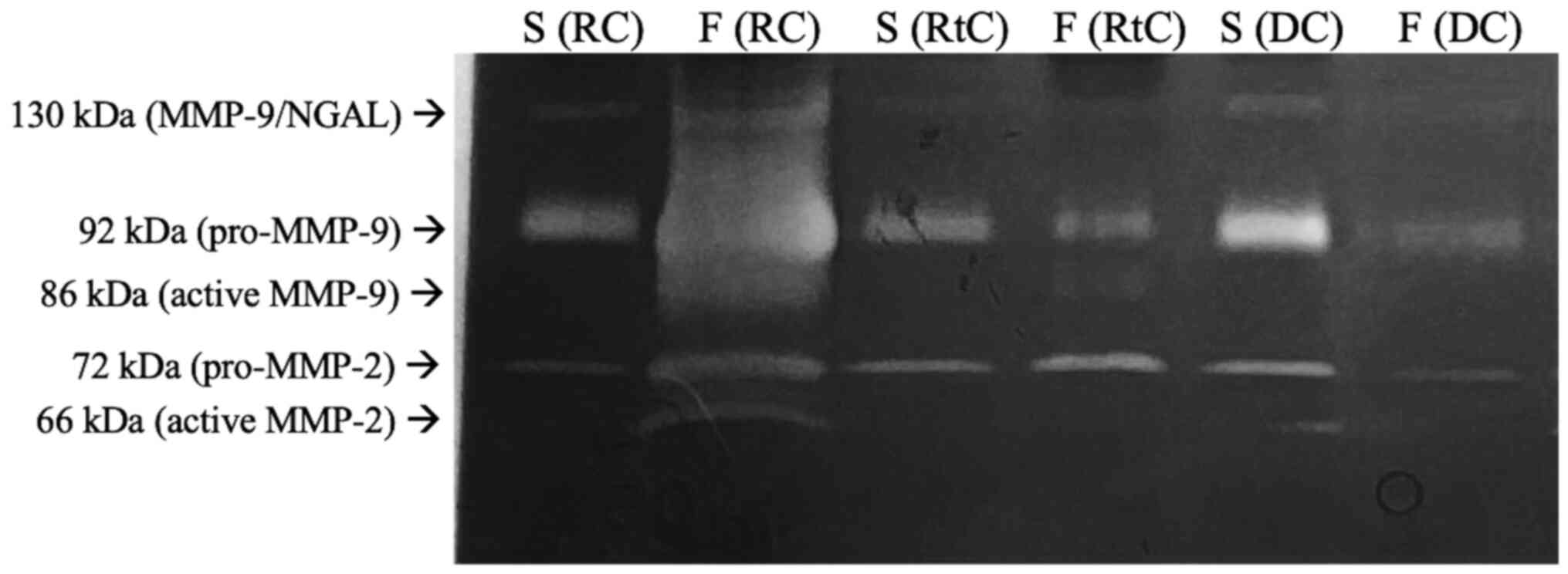
Zymography analysis confirmed the activity of MMP-2
and MMP-9 in all samples analyzed. A representative zymogram
showing the proteolytic activity of both gelatinases in the serum
and cyst fluid of patients with RCs, DCs and RtCs is presented in
Fig. 1. Gelatinolytic activity was
detected at molecular weights of 66 and 86 kDa, corresponding to
the active forms of MMP-2 and MMP-9, respectively. Additionally, a
92 kDa band was present, which corresponds to the latent form of
MMP-9 (pro-MMP-9), and the 72 kDa band was associated with
pro-MMP-2. Moreover, an MMP-9/neutrophil gelatinase-associated
lipocalin heterodimer band was obtained (130 kDa band). Due to the
association between enzymatic activity and brightness of the bands
(28), higher activity of both forms
of MMP-2 and MMP-9 were observed in the RC fluid.
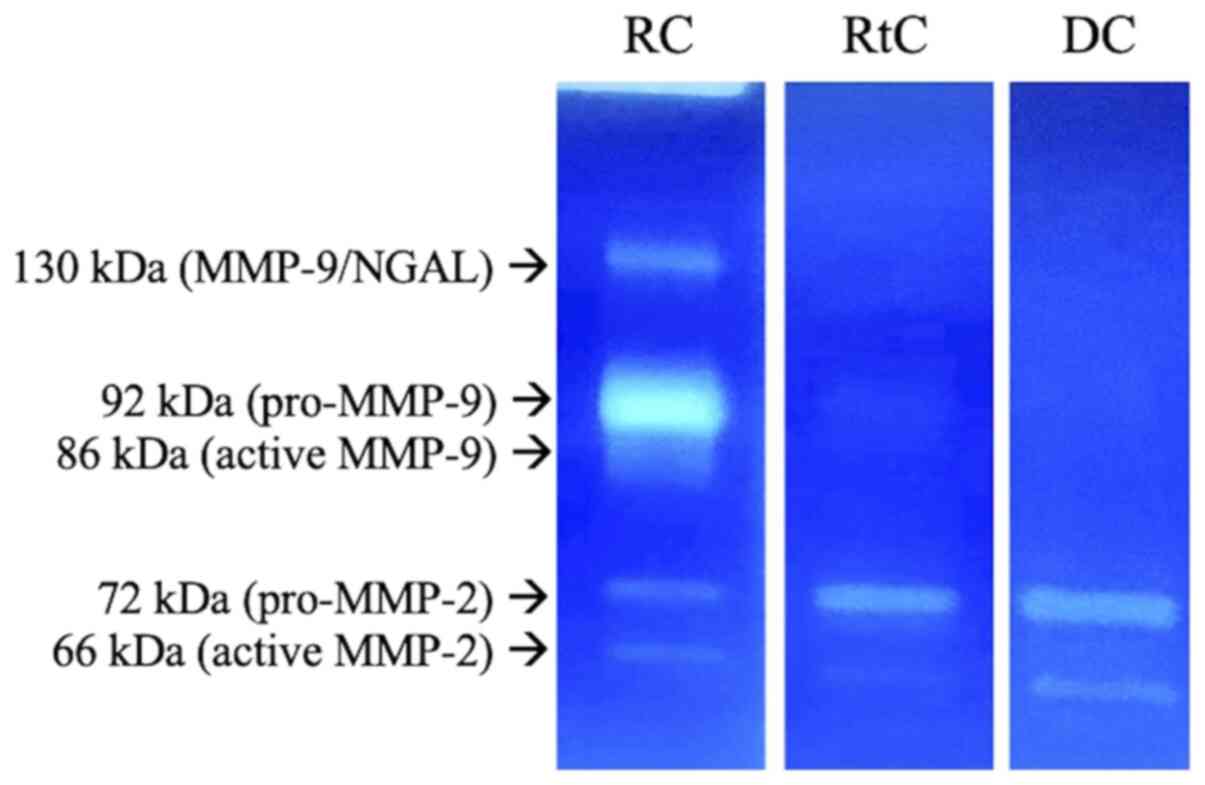
The activity of MMP-2 and MMP-9 in the cyst capsule
is shown in Fig. 2. The activity of
different MMP forms were measured as described above. Of note,
higher activity of pro-MMP-9 (~92 kDa) in the RC capsule was
observed.
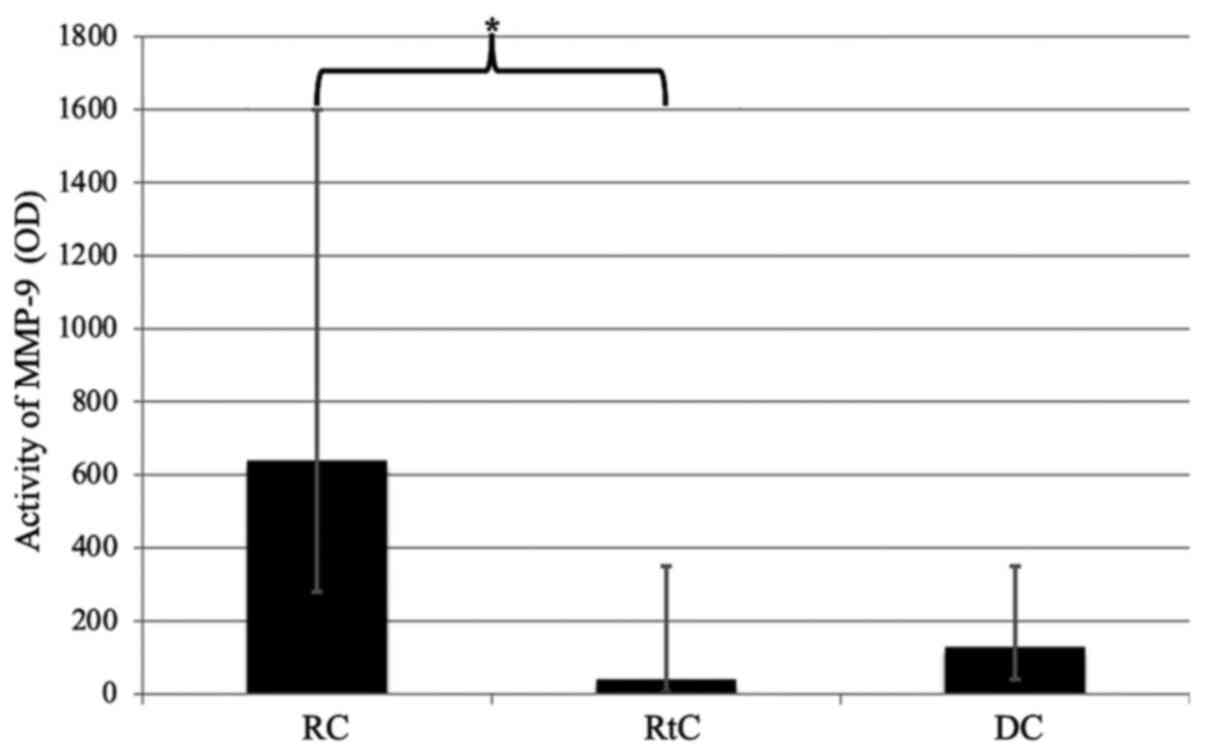
In the present study, significantly increased
activity of MMP-9 was observed in the RC fluid compared with the
RtC fluid (P=0.0065; Fig. 3). No
significant differences were observed between DGs and other types
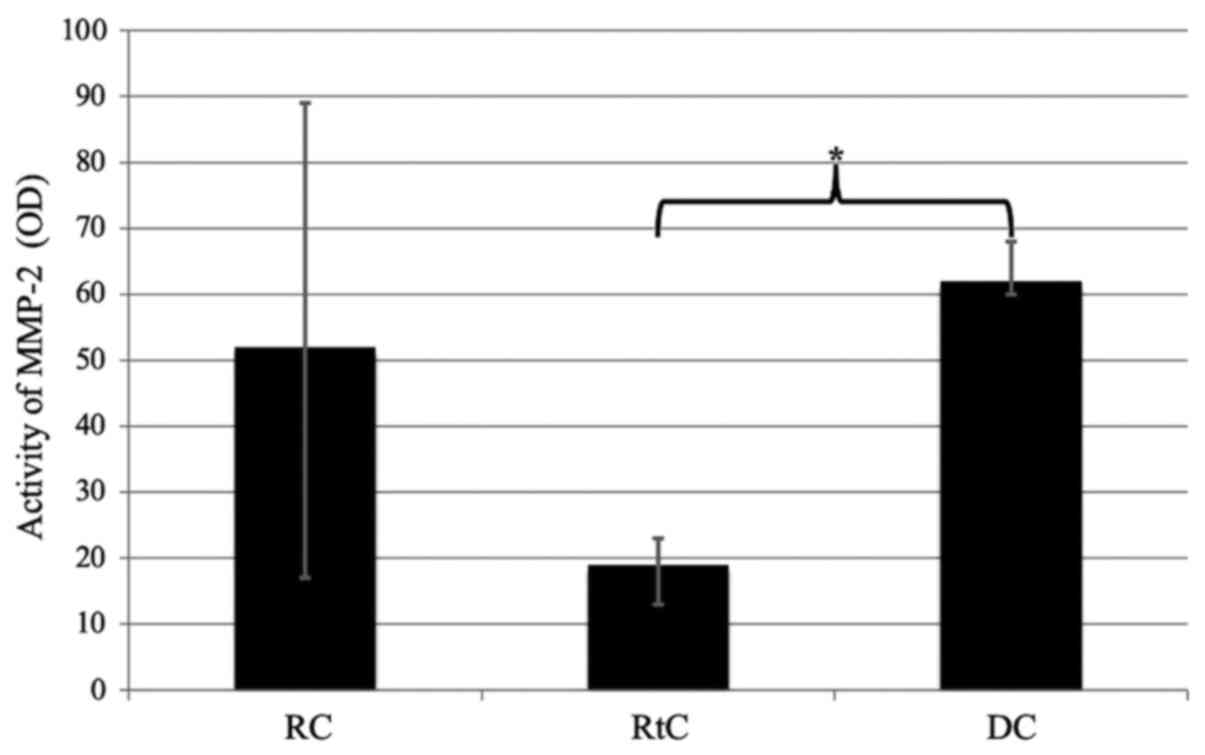
of lesions. Moreover, decreased activity levels of MMP-2 were
observed in the cyst capsule of patients with RtCs, but this
difference was statistically significant only when compared with
DCs (P=0.046; Fig. 4). Of note,
MMP-9 was the most frequently detected MMP in the RC capsule group
(65% of cases). No MMP-9 was detected in any of the patients with
DCs.
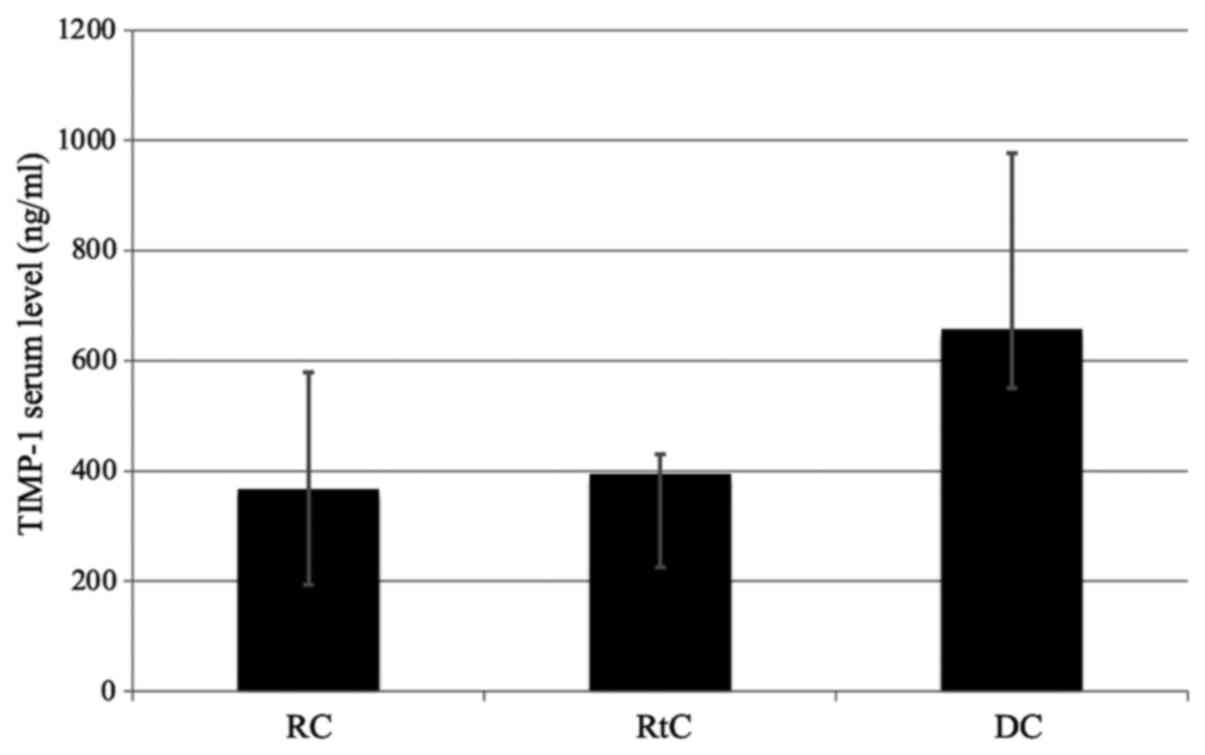
The TIMP-1 serum levels in patients with RCs were
reduced compared with DCs and RtCs, but the difference was not
statistically significant (Fig. 5).
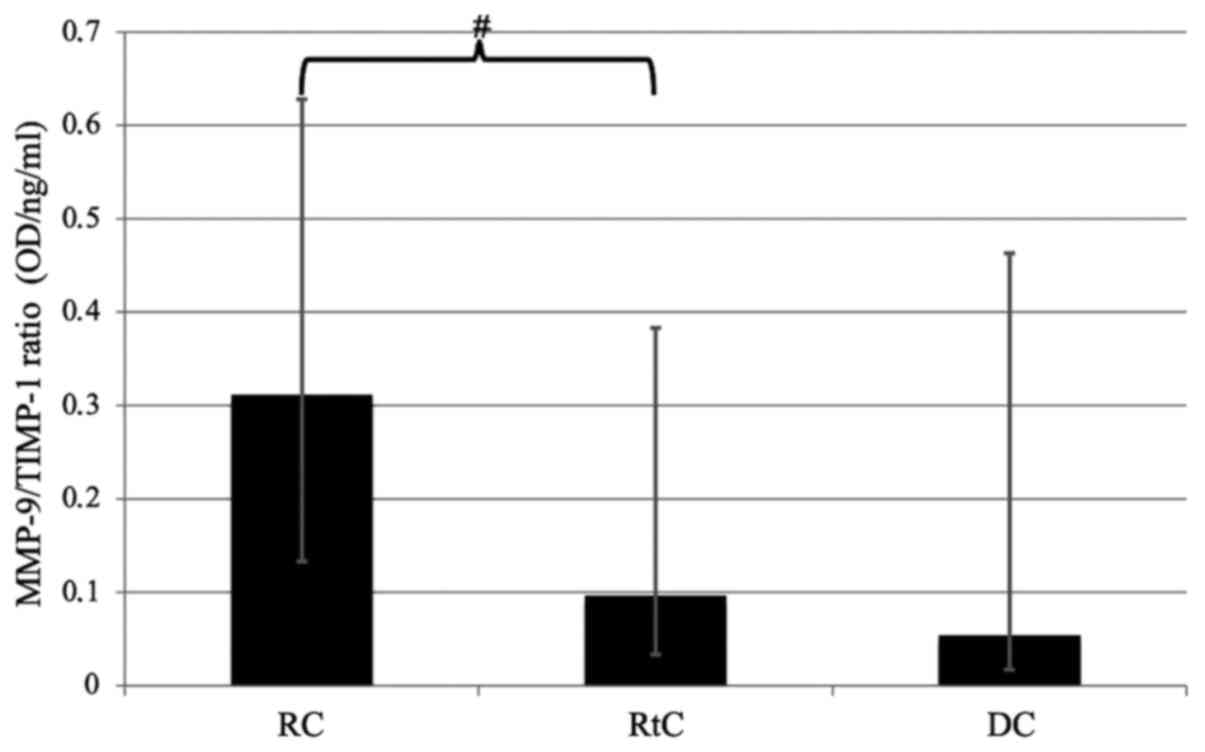
Of note, the MMP-9/TIMP-1 ratio was higher in the fluid from RCs
compared with RtCs and DCs. However, the difference between the
value of the ratio in RCs and RtCs only trended towards statistical
significance (P=0.09; Fig. 6).
Discussion
The biological factors that affect the development
of odontogenic cysts have yet to be clearly determined (29). Due to fact that MMPs are able to
degrade a wide range of substrates within the ECM, they represent
one of the most common subjects of investigation in the context of
elucidating the formation and expansion of cystic lesions localized
in the maxillofacial region. However, current literature comparing
their activity in various components of the maxillofacial cysts, as
well as between different types of cysts, are limited. The
comparisons of MMP activity in the cysts and in other odontogenic
tumors is understandable, given their more aggressive potential.
Ameloblastoma, which is described as a benign, locally aggressive
odontogenic tumor, and odontogenic keratocyst (OKC) are
characterized by higher destructive potentials and a tendency to
recur when compared with RCs and DCs (7). According to the World Health
Organizations recommendations on the reclassification of
keratocystic odontogenic tumors (KCOTs) into the group of OKCs, all
references to KCOT in the present study should be considered as an
OKC (30). It is necessary to
underline that the new classification system does not modify their
clinical and/or pathological status (30). Henriques et al (8) reported significantly higher MMP-9
expression in ameloblastoma and KCOTs compared with that in RCs and
DCs. Furthermore, DCs exhibited slightly higher expression of this
protease compared with RCs. It should be noted that the present
study did not identify MMP-9 activity in the DC capsule, whereas
its activity was detected in RC and RtC. Due to the small number of
patients diagnosed with DC, further studies are necessary in order
to draw more definitive conclusions. Moreover, immunohistochemical
analysis demonstrated higher expression of MMP-9 within the
epithelial, fibrous and vascular components in OKCs compared with
DCs and RCs. Therefore, it was concluded that the levels of
expression of MMP-9 may be associated with the more destructive
potential of OKCs compared with that of other odontogenic cysts
(31). An identical association was
observed between MMP-2 activity and KCOT. Numerous studies have
emphasized the involvement of MMP-2 and other enzymes from the
matrixins group in the process of bone resorption and odontogenic
lesion expansion. However, its activity also significantly affects
KCOT development (32-34).
A genetic study has also shed some light on the aforementioned
conclusions. It was shown that MMP-2 gene polymorphisms may be
involved in the increased aggressiveness of OKCs (29).
Comprehensive assessment of bone tissue homeostasis
and the dynamics of the bone degradation process requires
juxtaposition of both matrixins and their specific tissue
inhibitors. A previous study confirmed increased expression of both
gelatinases, TIMP-1 and TIMP-2, in ameloblastoma tissues (35). Moreover, Pinheiro et al
(36) reported increased activity of
MMP-1, MMP-2 and MMP-9 in particular tumor types. These enzymes, in
addition to their effect on lesion expansion, may underlie the
etiology behind local aggressiveness. In their view, digestion of
ECM results in the release of mitogenic factors that increase the
rate of tumor cell proliferation. Moreover, disturbances in the
expression of the genes encoding both gelatinases may be associated
with the destructive nature of ameloblastomas (29). The comparison of the expression of
matrixins with that of their inhibitors and inducers may provide
further insight with regard to the significance of these enzymes in
the alterations of bone tissue adjacent to the pathological
lesions. Expression of numerous MMPs was confirmed in the
parenchymal and stromal component of calcifying odontogenic cysts
(COCs) (37). Previously, a COC was
considered to be a calcifying cystic odontogenic tumor (30). However, Prosdócimi et al
(38) noticed differences in the
expression levels of MMPs, their inhibitors (TIMPs and the
membrane-anchored glycoprotein RECK) and ECM metalloproteinase
inducers in the epithelium and stroma of COCs. Their results
suggested that these abnormalities in ECM homeostasis may
contribute to bone degradation, and assist in the determination of
the characteristics of the pathological lesions.
Development of PGs and, consequently, RCs is based
on the inflammatory process within the dental pulp (39). Despite the fact that residual RCs
(RRCs) are characterized by an identical etiology as both PGs and
RCs, the inflammatory factor contributing to the expansion of the
lesion is eliminated by extraction of the affected tooth (40). Thus, despite a comparable
histopathological architecture, the biochemical status of the two
lesions may be completely distinct (41). It has been shown that MMP-9 is
expressed in both inflammatory cysts; however, a higher level of
expression was observed in RCs compared with that in RRCs (9). This correlation may suggest that RRCs
have a reduced ability to degrade ECM compared with RCs.
Furthermore, it is noteworthy that RCs were the only typical
inflammatory lesions in the present study. The results of the
present study demonstrated a statistically significantly higher
gelatinolytic activity of MMP-9 in RC fluid and its capsule
compared with that in both DG and RtC. Conversely, none of the
tested samples in the group of patients diagnosed with RC exhibited
prominent activity of MMP-2 compared with the other cysts. This may
suggest a higher effectiveness of MMP-9 in ECM degradation in
inflammatory lesions compared with MMP-2. Previous studies
confirmed the involvement of MMP-9 in the etiology of other
inflammatory processes (42,43). Due to the similarities in the
substrates between MMP-9 and MMP-2, the lack of statistically
significant differences between MMP-2 activity in the serum and
cyst fluid does not exclude the importance of MMP-2 in the
remodeling process of ECM. However, further research is required to
elucidate this association. Regarding the present study, research
conducted by Teronen et al (44) also reported significant results.
Activity of enzymes with a molecular weight of 92 kDa
(corresponding to pro-MMP-9) and 72 kDa (corresponding to
pro-MMP-2) and their tissue inhibitors (TIMP-1 and TIMP-2) was
detected in the fluid and capsule of all odontogenic lesions (RC,
RRC, DG and OKC). However, the dominant gelatinolytic activity in
the cyst capsule belonged to MMP-9. It was noted that ECM
degradation is possible due to partial blocking of MMPs by
endogenous inhibitors. Moreover, they suggested that gelatinases
may be involved in the final stages of this process, which was
initiated by the activity of collagenases (MMP-1 and MMP-8)
(44).
There is currently a lack of sufficient information
on the role of MMPs in RtC development. To the best of our
knowledge, there is only one study that has investigated MMP
expression within the RtC and maxillary sinusitis (MS) fluid, which
demonstrated distinct differences between the two disorders. The
expression of MMP-1, -2, -3, -9 and -10 were found to be higher in
the patients with MS compared with that in RtC (24). There are currently no available
studies regarding the activity of matrixins in the RtC capsule.
Moreover, no studies have been performed comparing MMP activity in
the components of RtC and odontogenic lesions. This is particularly
important due to the fact that the aforementioned lesions are
characterized by diametrically different etiopathogeneses, growth
dynamics and aggressiveness. In the present study, lower MMP-2
activity was observed in the RtC capsule compared with both
odontogenic cysts. However, this result was statistically
significant in relation only to DC. The emergence and expansion of
both RCs and DCs is associated with bone tissue destruction. By
contrast, RtC does not completely fill the cavity of the maxillary
sinus, and no bone damage or bone remodeling of the maxillary sinus
were observed (23). Upon comparing
the reduced activity of MMP-2 in the RtC wall with its clinical
characteristics, it may be inferred that MMP-2 serves an important
role in the ECM degradation process resulting in bone destruction.
Therefore, it may be considered a specific biomarker that may be
used to reflect the tendency of the lesions to cause bone
remodeling, with potential implications in treatment planning or
monitoring of outcomes. However, this hypothesis requires further
investigations with larger cohorts. Other reports have focused on
the pathologies of the paranasal sinuses and nasal cavity, for
example, nasal polyps (NP) and chronic inflammation of the sinuses
and/or nasal cavity. Literature data have reported increased
expression of MMP-9 and TIMP-1 in NP and chronic sinusitis compared
with unaffected sinus mucosa of a control group. This suggests the
involvement of MMP-9 in the pathogenesis of these conditions
(45,46).
As gelatinases are inhibited in vivo by their
specific tissue inhibitors, the MMP-9/TIMP-1 and MMP-2/TIMP-2
ratios were compared in serum and fluids collected from various
types of cysts. The MMP-9/TIMP-1 ratio was only slightly increased
in RC fluid compared with RtC, and this increase trended towards
statistical significance. The obtained results may confirm the role
of MMP-9 in the pathogenesis of this type of cyst. Of note, the
analyzed ratios were based on the inhibitor with the highest
affinity for a given enzyme. However, each of the MMPs can also be
inhibited by other tissue inhibitors, as well as by non-specific
protease inhibitors (47,48). Therefore, conclusions on the actual
in vivo activity of MMPs should be drawn with caution.
It is worth noting that, in several previous
studies, immunohistochemical analysis, ELISA or western blotting
have been used (8,9,42,44). Due
to the characteristics of these methods, it is not possible to
differentiate between the forms (active, latent or dimer) of
specific matrixins. The results obtained in the present study also
provide information regarding the activity of latent forms of MMPs,
which are unable to perform proteolysis in vivo. ELISA and
western blot analysis cannot evaluate the catalytic ability of
MMPs, and do not reflect the dynamics of processes occurring within
the ECM. The use of gelatin zymography provides additional
information, as it also analyzes the latent, inactive in
vivo forms of gelatinases. Matrixin proforms are activated by
SDS as a component of the zymographic gel. In this context, it was
possible to observe the enzymatic activity of pro-MMP-2 and
pro-MMP-9, as shown on representative zymograms. Another advantage
of zymography is also the ability to detect MMPs at low
concentrations (pg/µl) (49).
However, expressing their activity in OD units only indirectly
reflects the concentration of these enzymes. It appears that the
use of gelatin zymography more accurately reflects the dynamics of
processes occurring between the ECM and MMPs.
The findings of the present study confirmed the
involvement of MMPs in the development of cystic lesions within the
maxillofacial region. Despite the promising results, certain issues
require further study to resolve. More detailed research on RtCs is
necessary, as the current state of knowledge on these lesions is
considerably limited. Further study may provide novel insight into
the nature of MMPs, and may also improve the accuracy of the
complicated differential diagnosis of benign, non-odontogenic
cystic lesions of the maxillary sinuses. The juxtaposition of
benign cystic lesion with pathologies characterized by a more
aggressive nature may provide further valuable conclusions.
Moreover, the combination of gelatin zymography with other methods,
such as immunohistochemical assays, may allow for identification of
the cellular components of the lesions that are primarily
responsible for the expression of MMPs.
In conclusion, MMP-9 is involved in the pathogenesis
of RC, based on the observation that it exhibited the highest
degree of activity in the fluids collected from the cyst cavity
when compared with the other studied lesions. It should also be
emphasized that the presence of this gelatinase was confirmed in RC
walls, and the proportion of expression of MMP-9 was highest
amongst all lesions. Conversely, the low activity of MMP-2 in the
RtC walls suggests a limited effect of this gelatinase on the
development of this type of lesion. Thus, both gelatinases may
serve a potential role in the differential clinical diagnosis of
craniofacial cysts and may serve as supplementary elements in
histopathological studies. Furthermore, it appears that MMP-9 may
be of value as a specific biomarker in RC etiology. However, due to
the aforementioned limitations, further, more complex
investigations are required to draw definitive conclusions.
Acknowledgements
Not applicable.
Funding
The present study was funded by the Medical University of
Lublin.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
KK wrote the manuscript and performed the
experiments. DLK performed the experiments, and wrote and reviewed
the manuscript. TT supervised the study, interpreted the data, and
wrote and revised the manuscript. JK conceived the study, wrote and
revised the manuscript, and analyzed the data. All authors read and
approved the final manuscript. KK and JK confirm the authenticity
of all the raw data.
Ethics approval and consent to
participate.
The present study was approved by the Ethical
Committee at the Medical University of Lublin (approval no.
KE-0254/5/2017). Each patient recruited provided singed informed
consent. All procedures were performed in accordance with the
guidelines described in the Declaration of Helsinki.
Patient consent for publication
Not applicable.
Competing interests.
The authors declare that they have no competing
interests.
References
|
1
|
Koivisto T, Bowles WR and Rohrer M:
Frequency and distribution of radiolucent jaw lesions: A
retrospective analysis of 9,723 cases. J Endod. 38:729–732.
2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Lawal A, Adisa A and Sigbeku O: Cysts of
the oro-facial region: A Nigerian experience. J Oral Maxillofac
Pathol. 16:167–171. 2012.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Johnson NR, Gannon OM, Savage NW and
Batstone MD: Frequency of odontogenic cysts and tumors: A
systematic review. J Investig Clin Dent. 5:9–14. 2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Kadam NS, Ataide IN, Raghava P, Fernandes
M and Hede R: Management of large radicular cyst by conservative
surgical approach: A case report. J Clin Diagn Res. 8:239–241.
2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Tortorici S, Amodio E, Massenti MF,
Buzzanca ML, Burruano F and Vitale F: Prevalence and distribution
of odontogenic cysts in Sicily: 1986-2005. J Oral Sci. 50:15–18.
2008.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Rajendra Santosh AB: Odontogenic Cysts.
Dent Clin North Am. 64:105–119. 2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Ali A, Asif M, Ahmad B, Jamal S, Ali I and
Khadim MT: Stromal Expression of CD10 by immunohistochemistry in
odontogenic keratocyst (OKC), dentigerous and radicular cysts and
its correlation with local recurrence and aggressive behaviour.
Asian Pac J Cancer Prev. 20:249–253. 2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Henriques ÁC, Vasconcelos MG, Galvão HC,
de Souza LB and de Almeida Freitas R: Comparative analysis of the
immunohistochemical expression of collagen IV, MMP-9, and TIMP-2 in
odontogenic cysts and tumors. Oral Surg Oral Med Oral Pathol Oral
Radiol Endod. 112:468–475. 2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Ruiz PA, Toledo OA, Nonaka CF, Pinto LP
and Souza LB: Immunohistochemical expression of vascular
endothelial growth factor and matrix metalloproteinase-9 in
radicular and residual radicular cysts. J Appl Oral Sci.
18:613–620. 2010.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Liang H, Xu J, Xue M and Jackson C: Matrix
metalloproteinases in bone development and pathology: Current
knowledge and potential clinical utility. Metalloproteinases Med.
3:93–102. 2016.
|
|
11
|
Kurzepa J, Baran M, Watroba S, Barud M and
Babula D: Collagenases and gelatinases in bone healing. The focus
on mandibular fractures. Curr Issues Pharm Med Sci. 27:121–126.
2014.
|
|
12
|
Cunningham LA, Wetzel M and Rosenberg GA:
Multiple roles for MMPs and TIMPs in cerebral ischemia. Glia.
50:329–339. 2005.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Palosaari H, Wahlgren J, Larmas M, Rönkä
H, Sorsa T, Salo T and Tjäderhane L: The expression of MMP-8 in
human odontoblasts and dental pulp cells is down-regulated by
TGF-beta1. J Dent Res. 79:77–84. 2000.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Shay G, Lynch CC and Fingleton B: Moving
targets: Emerging roles for MMPs in cancer progression and
metastasis. Matrix Biol. 44-46:200–206. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Pardo A and Selman M: MMP-1: The elder of
the family. Int J Biochem Cell Biol. 37:283–288. 2005.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Moore CS and Crocker SJ: An alternate
perspective on the roles of TIMPs and MMPs in pathology. Am J
Pathol. 180:12–16. 2012.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Descamps FJ, Martens E and Opdenakker G:
Analysis of gelatinases in complex biological fluids and tissue
extracts. Lab Invest. 82:1607–1608. 2002.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Hannocks M-J, Zhang X, Gerwien H,
Chashchina A, Burmeister M, Korpos E, Song J and Sorokin L: The
gelatinases, MMP-2 and MMP-9, as fine tuners of neuroinflammatory
processes. Matrix Biol. 75-76:102–113. 2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Pereira Faustino IS, Azevedo RS and
Takahama A Jr: Metalloproteinases 2 and 9 immunoexpression in
periapical lesions from primary endodontic infection: Possible
relationship with the histopathological diagnosis and the presence
of pain. J Endod. 42:547–551. 2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Hadziabdic N, Kurtovic-Kozaric A, Pojskic
N, Sulejmanagic N and Todorovic L: Gene-expression analysis of
matrix metalloproteinases 1 and 2 and their tissue inhibitors in
chronic periapical inflammatory lesions. J oral Pathol Med Off Publ
Int Assoc Oral Pathol Am Acad Oral Pathol. 45:224–230.
2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Bariş E, Sengüven B, Bozkaya S and Oygür
T: Immunohistochemical analysis of matrix metalloproteinases-1,-9
and tenascin in odontogenic lesions. Eur J Inflamm. 12:419–427.
2014.
|
|
22
|
Suojanen J, Lehtonen N, Färkkilä E,
Hietanen J, Teronen O, Sorsa T and Hagström J: Common matrix
metalloproteinases (MMP-8, -9, -25, and -26) cannot explain
dentigerous cyst expansion. J Clin Diagn Res. 8:ZC82–ZC85.
2014.
|
|
23
|
Bal M, Berkiten G and Uyanık E: Mucous
retention cysts of the paranasal sinuses. Hippokratia.
18(379)2014.PubMed/NCBI
|
|
24
|
Kim SM, Eo MY, Cho YJ, Kim YS and Lee SK:
Differential protein expression in the secretory fluids of
maxillary sinusitis and maxillary retention cyst. Eur Arch
Otorhinolaryngol. 274:215–222. 2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Giotakis EI and Weber RK: Cysts of the
maxillary sinus: A literature review. Int Forum Allergy Rhinol.
3:766–771. 2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
World Medical Association Declaration of
Helsinki: Ethical Principles for Medical Research Involving Human
Subjects. 2018.
|
|
27
|
Babula D, Kocot J, Horecka A, Baran M and
Kurzepa J: Different patterns of gelatinolytic activity in
pituitary macro- and microadenomas. Clin Neurol Neurosurg.
158:90–92. 2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Vandooren J, Geurts N, Martens E, Van den
Steen PE and Opdenakker G: Zymography methods for visualizing
hydrolytic enzymes. Nat Methods. 10:211–220. 2013.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Aloka D, Padmakumar SK, Sathyan S,
Sebastian M, Banerjee M and Beena VT: Association of matrix
metalloproteinase 2 and matrix metalloproteinase 9 gene
polymorphism in aggressive and nonaggressive odontogenic lesions: A
pilot study. J Oral Maxillofac Pathol. 23(158)2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Kaczmarzyk T, Stypułkowska J and
Tomaszewska R: Update of the WHO classification of odontogenic and
maxillofacial bone tumours. J Stomatol (Brux). 70:484–506.
2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
de Andrade Santos PP, de Aquino ARL,
Oliveira Barreto A, de Almeida Freitas R, Galvão HC and de Souza
LB: Immunohistochemical expression of nuclear factor κB, matrix
metalloproteinase 9, and endoglin (CD105) in odontogenic
keratocysts, dentigerous cysts, and radicular cysts. Oral Surg Oral
Med Oral Pathol Oral Radiol Endod. 112:476–483. 2011.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Amm HM, Casimir MD, Clark DB, Sohn P and
MacDougall M: Matrix metalloproteinase expression in keratocystic
odontogenic tumors and primary cells. Connect Tissue Res. 55 (Suppl
1):97–101. 2014.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Scariot R, Morosini IC, Torres-Pereira CC,
Amenabar JM, Rebellato NL and Gugisch RC: Immunohistochemical
analysis of metalloproteases in dentigerous cysts, radicular cysts
and keratocystic odontogenic tumors: Systematic review. Stomatos.
18(34)2012.
|
|
34
|
Nadalin MR, Fregnani ER, Silva-Sousa YTC
and da Cruz Perez DE: Presence of myofibroblasts and matrix
metalloproteinase 2 in radicular cysts, dentigerous cysts, and
keratocystic odontogenic tumors: A comparative immunohistochemical
study. J Endod. 38:1363–1367. 2012.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Kumamoto H, Yamauchi K, Yoshida M and Ooya
K: Immunohistochemical detection of matrix metalloproteinases
(MMPs) and tissue inhibitors of metalloproteinases (TIMPs) in
ameloblastomas. J Oral Pathol Med. 32:114–120. 2003.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Pinheiro JJ, Freitas VM, Moretti AI, Jorge
AG and Jaeger RG: Local invasiveness of ameloblastoma. Role played
by matrix metalloproteinases and proliferative activity.
Histopathology. 45:65–72. 2004.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Ribeiro BF, Ferreira de Araújo CR, dos
Santos BR and de Almeida Freitas R: Immunohistochemical expression
of matrix metalloproteinases 1, 2, 7, 9, and 26 in the calcifying
cystic odontogenic tumor. Oral Surg Oral Med Oral Pathol Oral
Radiol Endod. 112:609–615. 2011.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Prosdócimi FC, Rodini CO, Sogayar MC,
Sousa SC, Xavier FC and Paiva KB: Calcifying Cystic Odontogenic
Tumour: Immunohistochemical expression of matrix
metalloproteinases, their inhibitors (TIMPs and RECK) and inducer
(EMMPRIN). J Oral Pathol Med. 43:545–553. 2014.PubMed/NCBI View Article : Google Scholar
|
|
39
|
de Paula-Silva FW, D'Silva NJ, da Silva LA
and Kapila YL: High matrix metalloproteinase activity is a hallmark
of periapical granulomas. J Endod. 35:1234–1242. 2009.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Bava FA, Umar D, Bahseer B and Baroudi K:
Bilateral radicular cyst in mandible: an unusual case report. J Int
Oral Health. 7:61–63. 2015.PubMed/NCBI
|
|
41
|
Muglali M, Komerik N, Bulut E, Yarim GF,
Celebi N and Sumer M: Cytokine and chemokine levels in radicular
and residual cyst fluids. J Oral Pathol Med. 37:185–189.
2008.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Sheen P, O'Kane CM, Chaudhary K, Tovar M,
Santillan C, Sosa J, Caviedes L, Gilman RH, Stamp G and Friedland
JS: High MMP-9 activity characterises pleural tuberculosis
correlating with granuloma formation. Eur Respir J. 33:134–141.
2009.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Zhang P, Wu C, Huang XH, Shen CL, Li L,
Zhang W and Yao CZ: Aspirin suppresses TNF-α-induced MMP-9
expression via NF-κB and MAPK signaling pathways in RAW264.7 cells.
Exp Ther Med. 14:5597–5604. 2017.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Teronen O, Salo T, Konttinen YT, Rifkin B,
Vernillo A, Ramamurthy NS, Kjeldsen L, Borregaard N, Hietanen J and
Sorsa T: Identification and characterization of gelatinases/type IV
collagenases in jaw cysts. J Oral Pathol Med. 24:78–84.
1995.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Lechapt-Zalcman E, Coste A, d'Ortho MP,
Frisdal E, Harf A, Lafuma C and Escudier E: Increased expression of
matrix metalloproteinase-9 in nasal polyps. J Pathol. 193:233–241.
2001.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Watelet JB, Bachert C, Claeys C and Van
Cauwenberge P: Matrix metalloproteinases MMP-7, MMP-9 and their
tissue inhibitor TIMP-1: Expression in chronic sinusitis vs. nasal
polyposis. Allergy. 59:54–60. 2004.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Belotti D, Paganoni P and Giavazzi R: MMP
inhibitors: Experimental and clinical studies. Int J Biol Markers.
14:232–238. 1999.PubMed/NCBI
|
|
48
|
Bourboulia D and Stetler-Stevenson WG:
Matrix metalloproteinases (MMPs) and tissue inhibitors of
metalloproteinases (TIMPs): Positive and negative regulators in
tumor cell adhesion. Semin Cancer Biol. 20:161–168. 2010.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Gogly B, Groult N, Hornebeck W, Godeau G
and Pellat B: Collagen zymography as a sensitive and specific
technique for the determination of subpicogram levels of
interstitial collagenase. Anal Biochem. 255:211–216.
1998.PubMed/NCBI View Article : Google Scholar
|