1. Introduction
Myeloperoxidase (MPO) belongs to the
peroxidase-cyclooxygenase subgroup of the heme peroxidase family of
enzymes. It is secreted by neutrophils and monocytes where it plays
a crucial role in innate immunity (1,2). In
the microbicidal system of phagocytes, MPO catalyzes the formation
of hypochlorous acid (HOCl) which is considered one of the
strongest oxidant molecules produced in the human body (3). Despite its crucial role in immune
defense against pathogens, MPO and its oxidative products react
with various lipids, proteins, and nucleic acids causing some
detrimental effects in host tissues that are usually associated
with ongoing inflammatory states such as atherosclerosis (3). Immunohistochemical analysis reported
the presence of MPO in atheroma plaques of human patients and
several studies have also shown its co-localization with
HOCl-modified low-density lipoproteins (LDLs) in human
atherosclerotic lesions where it can be found in both vascular
cells and in extracellular spaces (4,5). Thus,
MPO and its downstream products, mainly HOCl, are known to be
implicated in the pathophysiology of atherosclerotic diseases
(6,7). Despite the fact that several processes
are reported to be responsible for the oxidation of LDL both in
in vitro and in vivo settings, LDL that is oxidized
by MPO remains the most physiologically relevant type of oxidized
LDL (oxLDL) with considerable evidence confirming this theory. LDL
that is modified by the MPO system affects multiple cells that are
found in the atheroma plaques; this includes endothelial cells
(ECs), macrophages, and smooth muscle cells (7). In this review, we describe the
involvement of MPO in health and disease with an emphasis on the
role of MPO oxLDL (Mox-LDL) in macrophage and endothelial
dysfunction (ED) in the context of atherosclerosis. Overall, this
work provides better insights into the crucial role that Mox-LDL
plays during atherogenesis, as the ultimate driver of the disease,
by presenting an up-to-date review on the different pathways that
involve Mox-LDL in both EC and macrophage models of
atherosclerosis. This may also pave the way for future research
tackling MPO and its oxidation byproducts as important disease
biomarkers and targets for novel therapeutic approaches.
2. Heme peroxidase family and MPO
Heme peroxidases
Heme peroxidases are a ubiquitous group of enzymes
with robust oxidoreductase activity. They contain a heme group,
which acts as a redox cofactor for catalyzing the hydrogen peroxide
(H2O2) mediated one- and two-electron
oxidation reactions of numerous organic and inorganic molecules.
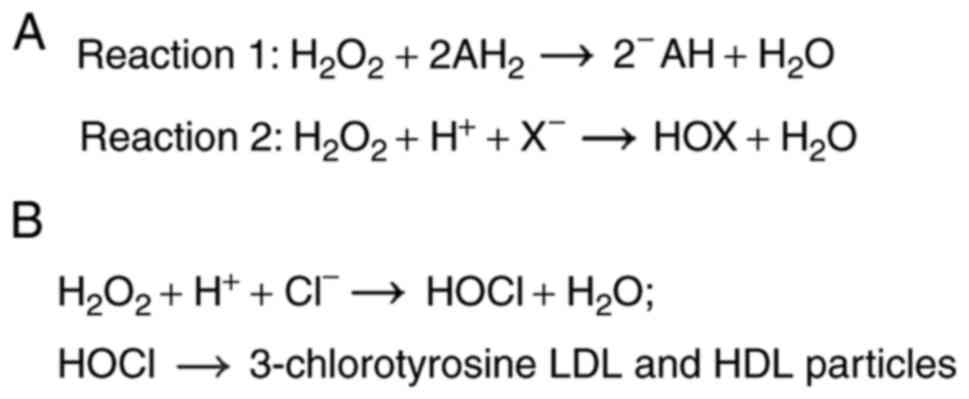
One-electron donors such as organic molecules (AH2) are
oxidized into their corresponding radicals (AH) and
H2O2 is reduced to water (Reaction 1)
(1). Meanwhile, two-electron donors
such as halides (X) are oxidized into their respective hypohalous
acids (HOX) and H2O2 is also reduced into
water (Reaction 2) (1). The heme
peroxidase superfamily was previously classified according to the
species origin of the enzymes into ‘animal’ and ‘non-animal’
superfamilies; however, this was found to be somewhat misleading
since many of these enzymes were reported to be present in all
kingdoms (8). Accordingly, the heme
peroxidase superfamily is better categorized on the basis of
differences in enzymatic activities into four subgroups (8). These four subgroups include the
peroxidase-catalase subgroup, the peroxidase-cyclooxygenase
subgroup, the peroxidase-chlorite dismutase superfamily, and the
peroxidase-peroxygenase superfamily (8). Particularly relevant to this review is
the peroxidase-cyclooxygenase subgroup. This subgroup is
characteristically unique since its heme group is
post-translationally modified, and bound via ester bonds molded by
the highly conserved aspartic acid and glutamic acid residues
(8). All enzymes of this subgroup
catalyze both reactions 1 and 2, with halide oxidation being the
more dominant physiological enzymatic activity (Fig. 1A) (8). This subgroup enlists MPO, eosinophil
peroxidase (EPO), lactoperoxidase (LPO), and thyroid peroxidase
(TPO) as its members (8). MPO, EPO,
and LPO are integral mediators of the innate immune system.
Conversely, thyroid peroxidase catalyzes the synthesis of the
thyroid hormones thyroxine (T4) and triiodothyronine (T3).
MPO is one of the most prominent enzymes of the
peroxidase-cyclooxygenase subgroup and is encoded by a gene located
on the long arm of chromosome 17 (17q) with a size of 14 kb
(9). Synthesis of MPO is exclusive
to cells of the myeloid lineage, and it commences during
myelopoiesis in the bone marrow in promyelocytes and promonocytes
(10). During the differentiation
of the myeloid precursors into neutrophils or monocytes, MPO mRNA
levels progressively decrease until they reach a point where MPO
synthesis is terminated in the fully differentiated monocytes and
neutrophils (11). The mature form
of MPO is present mainly in the azurophilic granules of neutrophils
and to a lesser extent in the lysosomes of monocytes and MPO
species isolated from human plasma including both precursor and
mature forms of the enzyme (12).
The three-dimensional structure of MPO forms a cationic surface (pI
>10) at physiological pH, with numerous arginine and lysine
residues, and this enables MPO to bind to electronegative molecules
such as lipoproteins, proteoglycans, and bacterial surfaces
(6).
Role of MPO in the host defense
MPO is a crucial mediator of the intracellular
microbicidal system of phagocytes and the host innate immune
system. After synthesis, MPO is stored in the azurophilic granules
of resting neutrophils and it is inactivated through inactivated
when it is captured by anionic proteoglycans accompanied by the low
pH milieu (2). Once a neutrophil
becomes activated by ingesting a pathogen, the nicotinamide adenine
dinucleotide phosphate (NADPH) oxidase enzyme system is activated
and recruited to the internal phagolysosomal membrane surface;
concurrently MPO is also released into the phagosome. The majority
of cellular MPO is released into the phagosome; however, some MPO
is released into the extracellular environment via degranulation or
by association with neutrophil extracellular traps (NETs) (6). In circulation, the enzyme is
transported by albumin and neutrophil microparticles (9). Inside the phagosome, the activity of
the NADPH oxidase results in the formation of a superoxide anion
(O2-), which is converted to H2O2
by superoxide dismutase (SOD). MPO can couple
H2O2 with halides/pseudohalides to catalyze
the formation of powerful reactive oxygen intermediates such as
HOCl, hypobromous acid (HOBR), and hypothiocyanous acid (HOSCN).
Although chlorine has the lowest reactivity to MPO among the
halides, it is considered to be the primary physiological substrate
of MPO, due to its high concentration in tissues (10). The unique product of the
MPO/H2O2/Cl- system is the potent
antimicrobial oxidant hypochlorous acid/hypochlorite. HOCl is a
unique product of MPO, since the latter is the only human enzyme
with the ability to produce HOCl at physiological chloride
concentrations (Fig. 1B) (9). HOCl is able to initiate modification
reactions targeting DNA, lipids, and lipoproteins (13). Other notable MPO primary and
secondary oxidant products include nitrous radical (NO•)
and peroxynitrite (ONOO-), chloramines, hydroxyl
radicals (HO·), and ozone (O3). The canonical role of
MPO is thought to be the mediation of the host defense against
invading pathogens such as fungi and bacteria, although this role
of MPO varies among different species (3). In fact, MPO knock-out mice were found
to be more susceptible to infections (3). On the other hand, humans with complete
and partial MPO-deficiency live normally with rare cases of
persistent infections with Candida albicans reported
(2).
Role of MPO in inflammation and
disease
MPO has been implicated to varying extents in the
pathogenesis of several diseases (Table
I). These diseases are usually associated with chronic or acute
inflammatory states. The detrimental effect of MPO on host tissue
is usually mediated by MPO-derived oxidants, which interact with
nucleic acids, lipids, and proteins causing deleterious effects.
First, MPO is implicated in the pathogenesis of several cancer
types such as bladder, breast, colon, larynx, lung, leukemia, and
stomach. The carcinogenic role of MPO is mediated by MPO oxidative
products which are genotoxic and mutagenic. The role of
MPO-downstream oxidants in cancer has been reviewed in depth
elsewhere (9,11). MPO also plays a role in
neurodegenerative diseases such as Parkinson's disease, Alzheimer's
disease, and multiple sclerosis; this role is articulately reviewed
in (3). Furthermore, reports have
also demonstrated the involvement of MPO in vasculitis, renal
illnesses, lung inflammation, rheumatoid arthritis, colitis,
pancreatitis, cystic fibrosis, liver diseases, periodontitis,
sinusitis, and inflammatory bowel disease (2). MPO is also established to play a role
in all stages of atherosclerosis, and this is particularly relevant
to the present review.
 | Table IStudies assessing the role of
myeloperoxidase in inflammation and disease. |
Table I
Studies assessing the role of
myeloperoxidase in inflammation and disease.
| Disease | (Refs.) |
|---|
| Inflammatory
diseases | |
|
Vasculitis | (2,9,11) |
|
Rheumatoid
arthritis | (2,3,9,11) |
|
Colitis | (2,9) |
|
Pancreatitis | (2) |
|
Periodontitis | (2) |
|
Sinusitis | (2) |
|
Inflammatory
bowel syndrome | (2,11) |
|
Lung
inflammation | (2,3,11) |
| Neurodegenerative
diseases | |
|
Parkinson's
disease | (2,3,9,11) |
|
Alzheimer's
disease | (2,3,9,11) |
|
Multiple
sclerosis | (2,3,9,11) |
| Cancer | |
|
Lung
cancer | (9,11) |
|
Ovary
cancer | (9) |
|
Bladder
cancer | (9,11) |
|
Liver
cancer | (9) |
|
Stomach
cancer | (9) |
|
Colon
cancer | (11) |
|
Lung
cancer | (9,11) |
|
Larynx
cancer | (11) |
|
Breast
cancer | (11) |
|
Pancreatic
cancer | (11) |
|
Myeloid
leukemia | (11) |
| Cardiovascular
diseases | |
|
Coronary
artery disease | (2,3,9,11) |
|
Myocardial
infarction | (3,11) |
|
Heart
failure | (11) |
3. MPO and atherosclerosis
Source of MPO in atherosclerosis
MPO is pervasive inside atherosclerotic lesions. MPO
has been extensively shown to be present inside human
atherosclerotic tissues (4). Inside
vascular walls and atherosclerotic lesions, there exist different
sources of MPO. For example, during inflammatory states, MPO is
secreted by neutrophils into the circulation. Subsequently, MPO
binds to ECs through electrostatic interactions which leads to the
internalization of MPO into the atherosclerotic lesions in vascular
walls by endocytosis (9).
Nevertheless, macrophages are thought to be the primary source of
MPO inside atherosclerotic lesions. Maturation of monocytes into
macrophages is linked to a loss of MPO expression; however,
granulocyte-macrophage colony-stimulating factor (GM-CSF) has been
found to selectively regulate the ability of macrophages to express
MPO in human atherosclerotic lesions (14). Furthermore, it was demonstrated that
macrophages present in the earlier stages of atherosclerosis
minimally express MPO, whereas macrophages present in the late
stages of atherosclerosis highly express it (14). Immunohistochemical evidence showed
that antihuman MPO antibodies co-localized with antimacrophage
antibodies, and this immunostaining of MPO was particularly
prominent in the atherosclerotic lesions of the shoulder region
(4). Therefore, both local
secretion by macrophages and transcytosis of intraluminal MPO are
thought to be the primary sources of MPO in atherosclerotic lesions
(15).
Role of MPO in atherosclerosis
Numerous studies including in vitro
experiments and pathophysiological observations have implicated MPO
and its downstream oxidative products in the pathogenesis of
atherosclerosis. MPO contributes to atherosclerosis by oxidizing
LDL, impairing high-density lipoprotein (HDL) function, reducing
the bioavailability of nitric oxide (NO), causing ED, generating a
thrombogenic environment, activating matrix metalloproteinases
(MMPs), and inactivating tissue inhibitors of MMPs (TIMPs), and
recruiting leukocytes, consequently destabilizing the
atherosclerotic plaque (6,15).
High-density lipoprotein is renowned for its
athero-protective characteristics including its antioxidant,
anti-inflammatory, and antithrombotic properties. A reputed
contribution of MPO to atherosclerosis involves targeting and
modifying apolipoprotein A-I (apoA-I) of HDL resulting in the
attenuation of the athero-protective role of HDL. MPO binds to
helix 8 on apoA-I, and converts HDL into a dysfunctional form
(16). As previously mentioned, MPO
forms the highly reactive HOCl, which will chlorinate electron-rich
residues such as lysine and tyrosine, thus forming MPO-specific
3-chlorotyrosine (Fig. 1B)
(17). Moreover, MPO oxidative
byproducts are capable of producing 3-nitrotyrosine in vitro
(18). 3-chlorotyrosine and
3-nitrotyrosine were found to be abundant in apoA-I of HDL
recovered from atherosclerotic lesions (19). Furthermore, the levels of
3-chlorotyrosine and 3-nitrotyrosine in serum HDL of individuals
with cardiovascular disease (CVD) are markedly higher than in serum
HDL of healthy individuals (18-20).
In addition, HDL recovered from human aortic atherosclerotic
lesions had substantially higher levels of 3-chlorotyrosine in
comparison to HDL obtained from plasma (20). Similarly, the mean levels of
3-nitrotyrosine in HDL obtained from human aortic atherosclerotic
intima are significantly higher when compared to circulating HDL
(18). However, it is important to
note that 3-nitrotyrosine is not unique to MPO like
3-chlorotyrosine, since there are other pathways independent of MPO
that also produce 3-nitrotyrosine in atherosclerotic lesions
(15). Modified HDL is also
pro-inflammatory, and it induces the expression of vascular cell
adhesion molecule 1 (VCAM1) in ECs (6). Moreover, several studies suggest that
MPO modification of HDL attenuates the athero-protective properties
of HDL, primarily by hindering the interaction of HDL ATP binding
cassette subfamily A member 1 (ABCA1) and ATP binding cassette
subfamily G member 1 (ABCG1), consequently impairing the reverse
cholesterol efflux process (11,15,21).
Interestingly, it was found that HDL-bound MPO retains its
enzymatic activity, and binding to HDL may protect MPO from
cellular uptake and degradation (15).
There is ample evidence showing that MPO and its
downstream oxidative products cause ED. First, several peroxidases
such as MPO act as a catalytic sink for NO, since NO is a substrate
for the one-electron reactions of the peroxidases (9,11).
Similarly, downstream oxidative products of MPO can also scavenge
NO (15). Furthermore, HOCl
chlorinates the nitrogen atoms of arginine the substrate for NO
formation, thereby inhibiting nitric oxide synthetase (NOS)
(22,23). On a similar note, HOCl oxidizes
endothelial NOS (eNOS) and uncouples it, turning eNOS into a
superoxide-generating enzyme (24).
Scavenging NO and inhibiting its formation ultimately causes ED,
which initiates atherosclerosis (25). MPO may also promote EC apoptosis and
desquamation in vitro, which provides a clue into its
possible role in provoking plaque erosion and thrombus formation
in vivo (6). In line with
these aforementioned observations, serum MPO levels were found to
be associated with ED in humans (26). In addition, MPO contributes to a
thrombogenic setting by stimulating ECs to secrete tissue factors
and by priming platelet aggregation (6). MPO also promotes atherosclerotic
plaque destabilization by activating MMPs and inactivating TIMPs.
In fact, HOCl that is produced by MPO has been found to activate
MMP-7 in vitro by oxidizing and converting the thiol residue
of the cysteine switch to sulfinic acid (27). Moreover, HOCl was shown to oxidize
the N-terminal cysteine of TIMP-1, thereby rendering it inactive
(28). Atherosclerosis is an
inflammatory disorder, and there is increasing evidence revealing a
role of MPO-derived products in the perpetuation of inflammation
and atherosclerosis by leukocyte recruitment. MPO can contribute to
leukocyte recruitment through several mechanisms. First, as
previously mentioned, MPO acts as a catalytic sink for NO, and NO
has anti-adhesive properties, thus consuming NO promotes the
adhesion of leukocytes to the endothelial layer (21). Under physiological conditions, there
exists an electrostatic repulsion between the negatively charged
endothelium glycocalyx and the negatively charged surfaces of
polymorphonuclear neutrophils (PMNs). In inflamed tissues, the
cationic MPO is released, and MPO binds to the negatively charged
endothelium causing a reduction in the negative surface charge
(21). It seems that MPO favors
binding to the heparan sulfate glycosaminoglycans, which constitute
a major building block of the endothelial glycocalyx (29). This decrease in the negative charge
could allow for a new electrostatic attraction between the
negatively charged PMN and the now positively charged endothelium,
thereby facilitating the binding, rolling, adhesion, and the
transmigration of the PMNs into the surrounding tissue and
perpetuation of inflammation (21).
Furthermore, MPO produces HOSCN, and HOSCN is known to induce the
expression of EC surface adhesion molecules (21). MPO also induces EC production of
cytokines and chemokines such as IL-6 and IL-8, which enhance
inflammation and recruit leukocytes (21).
MPO as a biomarker for CVDs
Several studies have established a relationship
between varying levels of MPO and CVD, independent of classical
risk factors. For instance, elevated levels of functional MPO per
ml of blood and per leukocyte were linked with the risk of coronary
artery disease (CAD) (30).
Similarly, individuals with the-463 G/A polymorphism of the MPO
gene exhibit elevated levels of MPO, and this is associated with an
increased risk of CAD (31). On the
contrary, cardiovascular problems appear to be very rare among
individuals with MPO deficiency (32). Likewise, individuals having a
promoter polymorphism linked with a 2-fold reduction in MPO
expression appear to be protected from CVD-like problems (33). There is also considerable evidence
supporting the role of MPO in the prediction of long-term risk of
cardiovascular mortality in patients with cardiovascular problems
(6). For example, after a follow-up
of 13 years, the top tertile of MPO levels predicted approximately
twice the risk of CVD mortality in comparison with patients in the
lowest tertile of MPO levels (34).
4. Oxidized LDL (oxLDL) and
atherosclerosis
oxLDL
Atherosclerosis is a progressive chronic
inflammatory disorder characterized by the interplay of multiple
risk factors. High plasma LDL cholesterol levels are one of the
most well-established risk factors underlying the development of
atherosclerosis and cardiovascular problems. Conversely, low plasma
LDL levels are associated with low cardiovascular risk. Indeed,
individuals with exceedingly low LDL levels generally do not
develop clinically relevant atherosclerosis, irrespective of the
presence of other risk factors (35). However, approximately half of the
patients with cardiovascular problems have normal levels of
cholesterol (36). This might be
related to the fact that lipoprotein quality is crucial in
mediating atherogenesis; in fact, LDL is post-translationally
modified in several processes such as glycation, glycosylation,
carbamylation, glycoxidation, acetylation, methylation, ethylation,
and oxidation (10,36). Interestingly, one of the most
significant LDL modifications is oxidation, which results in the
generation of an inflammatory form known as highly oxLDL (7). The oxidation of LDL occurs along an
extensive and continuous spectrum. This spectrum ranges from the
minimally oxLDL to the highly oxLDL (37). The oxidation of LDL usually begins
in the plasma where minimally oxLDL can be detected (38). Measuring the degree of LDL oxidation
is usually determined by a fluorometric Thiobarbituric acid
reactive substances assay. The degree of oxidation of minimally
oxLDL is normally <10 nmol/mg LDL protein; meanwhile, the oxLDL
value is usually >30-40 nmol/mg LDL protein (37). These two forms are
characteristically very distinct. Minimally oxLDL is oxidized to an
extent where it can still be recognized by native LDL receptors and
not by scavenger receptors (37).
Conversely, oxLDL is oxidized to a level where apolipoprotein B-100
(apoB-100) is modified such that it is no longer able to be
recognized by the native LDL receptor (7). Indeed, oxLDL is recognized, bound, and
internalized by receptor-mediated endocytosis through multiple
scavenger receptors. These scavenger receptors include class A
scavenger receptor type 1 and type 2 (SR-A1 & SR-A2), class B
scavenger receptor type 1 and class B scavenger receptor type 2
(SR-B1 and SR-B2), macrosialin/CD68, scavenger receptor for
phosphatidylserine and oxidized lipoprotein (SRPSOX), and
lectin-like oxLDL receptor-1 (LOX-1) (37,39,40).
Unlike the native LDL receptor, scavenger receptors are not
down-regulated when the cholesterol content of the cell increases
(40). Oxidative modification of
LDL plays a substantial role in atherosclerosis, and may therefore
be a plausible target for interventions to slow the development of
atherosclerosis. In fact, several epidemiological studies have
found an association between circulating oxLDL levels and clinical
cardiovascular events. For example, circulating oxLDL was found to
be a sensitive marker for CAD where it correlates with the
Framingham risk factors (41).
Circulating oxLDL is also associated with other risk factors of
atherosclerosis such as diabetes, fatty liver, and obesity
(40). A myriad of studies have
identified the presence of oxLDL in atherosclerotic lesions
(36). Inside atherosclerotic
plaques, uptake of oxLDL by receptor-mediated endocytosis through
scavenger receptors converts macrophages to foam cells, which
constitute the hallmark of atherosclerosis. Besides inducing foam
cell formation, oxLDL is pro-inflammatory and it has a wide range
of biological effects and properties that contribute to the
development of atherosclerosis, which is eloquently summarized in
(38,42,43).
Mechanisms of LDL oxidation
There exist several mechanisms for the oxidative
modification of LDL. Different cells, enzymes, and cations are
involved in the oxidation of LDL. In vitro studies have
demonstrated that cells such as fibroblasts, monocytes, smooth
muscle cells, macrophages, neutrophils, and ECs can oxidize LDL
(44). LDL is oxidized by various
enzyme systems such as xanthine oxidase, NADPH oxidase,
lipoxygenase, and MPO (10).
Moreover, Copper (Cu2+) and Iron (Fe3+)
dependent oxidation of LDL has been widely used in experiments;
however, these mechanisms require high concentrations of the
respective metal ions at the site of oxidation and they should not
be considered physiologically relevant (10). Some oxidative mechanisms target the
lipid portion of LDL including copper and lipoxygenases, whereas
other mechanisms target primarily the protein fraction of LDL such
as MPO (10). Particularly relevant
to this review is the modification by MPO which forms Mox-LDL.
5. MPO modified LDL
Evidence of the formation of MPO
modified LDL in atherosclerosis
The exact processes underlying the formation of
oxLDL in vivo are still widely debated among scientists.
However, recently, there has considerable evidence confirming the
in vivo oxidative modification of apoB-100 of LDL by the
MPO/H2O2/Cl- system, and the
ability of MPO to bind to LDL and to co-precipitate with it
(45). The cationic properties of
MPO suggest that MPO binds to the negatively charged amino acids of
apoB-100(9). MPO is predicted to
strongly bind to and adsorb the NH2-βα1 domain of
apoB-100 since this domain is exposed to the outside surface of LDL
(10). In fact, the probable site
of interaction between apoB-100 and MPO is the amino acid stretch
445-456 (EQIQDDCTGDED) of apoB-100(46). Furthermore, it was found that once
MPO adsorbs the surface of LDL, its chlorinating and peroxidase
activities are increased by 90% (36). Moreover, MPO kinetic reactivity
favors proteins and free amino acids and this will cause MPO to
primarily oxidize the amino acid residues of apoB-100(9). Lipid oxidation of LDL by MPO may also
occur, albeit to a lesser extent, and may be restricted to acidic
conditions with an excess of reactants (10). To study the ability of MPO to
oxidize LDL, researchers use either the
MPO/H2O2/Cl- system or the
MPO/H2O2/Cl- system primary
product ‘HOCl’ which is considered the unique product of this
system; in fact, MPO is the only human enzyme with the ability to
produce HOCl at physiological chloride concentrations (17). However, it is worth noting that HOCl
used as a reactant does not 100% mimic the
MPO/H2O2/Cl- since as mentioned
earlier, MPO adsorbs with a strong interaction with the surface of
LDL (9). Nevertheless, in order to
facilitate the experimental procedure, HOCl is sometimes used as a
reactant instead of the whole
MPO/H2O2/Cl- system. Therefore,
there exist two different forms when referring to LDL oxidized by
MPO: LDL oxidized by the whole
MPO/H2O2/Cl- system and LDL
oxidized directly by HOCl alone. In this review, we will refer to
LDL oxidized by the whole
MPO/H2O2/Cl- system as Mox-LDL,
and LDL oxidized directly by HOCl as HOCl-LDL. One of the earliest
pieces of evidence for LDL oxidation by MPO is the
immunohistochemical evidence showing specific monoclonal antibodies
cross-reacting with human HOCl-LDL but not with Cu2+-,
malondialdehyde-, peroxynitrite-, hemin-, glycated-, acetylated-,
or 4-hydroxynonenal-modified LDL (5). These antibodies were specific to
epitopes present in LDL after treatment with HOCl added alone as a
reagent or generated by the
MPO/H2O2/Cl- system (5). It was also reported that the presence
of HOCl-LDL epitopes increased in conjunction with the severity of
the atherosclerotic lesion (5).
Furthermore, HOCl-LDL epitopes were shown to be present inside and
around monocytes/macrophages, ECs, as well as in the extracellular
matrix (5). Intriguingly, the
staining of HOCl-LDL epitopes was co-localized with MPO and
CD68-positive cells suggesting that monocytes/macrophages are
involved in the production of MPO and MPO-modified epitopes of LDL
in advanced human atherosclerotic lesions (5). Another study reported the use of four
different monoclonal antibodies specifically raised against LDL
oxidized by the MPO (47). These
monoclonal antibodies did not cross-react with neither native LDL,
very-low-density lipoproteins (VLDL) nor hydrogen peroxide, or
copper sulfate oxLDL (Cuox-LDL) (47). Remarkably, these four antibodies
demonstrated reactivity with human atherosclerotic aortic and
carotid tissues (47). Another line
of evidence implicating MPO in the oxidative modification of LDL is
mass spectrometry analysis that revealed a total of 97 peptides of
apoB-100 modified by either HOCl added as a reagent or by the
MPO/H2O2/Cl- system (36). It is important to note that in their
study, the authors found differences in the modified residues of
LDL oxidized by HOCl or by the
MPO/H2O2/Cl- system (36). This same study also demonstrated
that many of the modifications of apoB-100 induced by HOCl
identified in vitro were also present on LDL isolated from
patients who have increased levels of plasma MPO and Mox-LDL
(36). HOCl chlorinates
electron-rich substrates on apoB-100 such as lysine and tyrosine
residues, generating MPO-specific 3-chlorotyrosine.
3-chlorotyrosine is a unique product of MPO-catalyzed oxidation of
LDL, and its levels are undetectable in LDL oxidatively modified by
traditional oxidizing agents such as cations (copper and iron),
enzymes (horseradish peroxidase, lactoperoxidase, and
lipoxygenase), and molecules such as hemin, glucose, and
peroxynitrite (17). Therefore,
3-chlorotyrosine is used as evidence for the in vivo
modification of LDL by MPO (Fig.
1B). Interestingly, gas chromatography-mass spectrometry
analysis revealed that the levels of 3-chlorotyrosine recovered
from atherosclerotic tissue were 6x greater than those recovered
from normal aortic tissue (17). It
was also found that the levels of 3-chlorotyrosine were 30x greater
in LDL obtained from atherosclerotic tissue compared with
circulating LDL (17). LDL
oxidation by the MPO/H2O2/Cl-
system was presumably restricted to the intima; however, there is
recent evidence demonstrating that LDL oxidation by the MPO system
may also occur in the plasma (48).
This hypothesis was based on the fact that MPO is known to adsorb
LDL and the endothelium, and therefore, MPO may oxidize LDL at the
surface of EC in the circulation. In vitro experiments were
performed using EC, which were treated with native LDL, MPO, and
angiotensin II to promote the activity of the NADPH complex
(48). The results revealed that
Mox-LDL production directly correlated with the LDL and MPO
concentrations used in the study (48). These results showed that LDL
oxidation may not be restricted to the intima. Furthermore, it was
reported that physiological serum concentrations of Mox-LDL range
from 10-100 µg/ml (49). LDL can
also be oxidized by MPO-generated species other than HOCl. For
example, in vitro treatment of LDL with reactive nitrogen
species produced by the MPO/H2O2/Nitrite
system yields a nitrated form of LDL (NO2-LDL) (50). This nitrated form of LDL does not
bind to the native LDL receptor or scavenger-receptor class A, but
to CD36 promoting foam cell formation (50). The reactive nitrogen species
produced by the MPO/H2O2/Nitrite convert
tyrosine into 3-nitrotyrosine, which was found to be enriched in
LDL recovered from human atherosclerotic aortic tissue (51). However, it should be noted that the
formation of 3-nitrotyrosine is not unique to MPO since other
mechanisms may produce 3-nitroyrosine as well (15). In addition, it has been shown that
LDL protein carbamylation can be mediated by MPO.
MPO/H2O2 oxidizes thiocyanate into cyanate
which carbamylates lysine residues on LDL (52). It was found that carbamyl-modified
lipoproteins detected in human atheroma plaques co-localized with
MPO (52). Moreover, carbamylation
of LDL generated an atherogenic molecule that could bind with high
affinity to scavenger receptor class A inducing its uptake and foam
cell formation (52). Taken
together this information demonstrates that MPO is highly
implicated in modifying LDL in atherosclerotic lesions. Mox-LDL
interacts with a variety of cells such as macrophages, neutrophils,
T cells, ECs, platelets, and smooth muscle cells. In this review,
we will focus on the interaction of Mox-LDL with macrophages and
ECs.
Macrophage interactions with MPO
modified LDL
The majority of studies on the effect of oxLDL on
monocytes/macrophages have been performed using Cuox-LDL which is
not a physiologically relevant model of oxLDL (53-56);
meanwhile, Mox- LDL is rarely used. In vitro experiments
using murine macrophages (RAW 264.7 cells) showed that HOCl-LDL
possesses pro-inflammatory characteristics that activate monocytes,
promote monocyte/macrophage differentiation, and induce the
expression of CD36 and peroxisome proliferator-activated receptor-γ
(PPAR-γ) (57). Furthermore,
HOCl-LDL was found to strongly induce the expression of the
α-chemokine IL-8 but not the β-chemokine MCP-1 in human monocytes
(58). In addition, HOCl-LDL was
reported to promote apoptosis in THP-1 monocytes through a
caspase-dependent pathway (59).
Moreover, HOCl-LDL inhibited lysosomal cysteine protease cathepsin
B via a chloramine-dependent mechanism (45). CD36 and SR-B1 were identified as
receptors for HOCl-LDL that can take up HOCl-LDL via
receptor-mediated endocytosis (60). HOCl-LDL upregulates the expression
of CD36 via PPAR-γ (57). This
uptake of HOCl-LDL along with the reported protease inhibition
causes lipid accumulation in macrophages and consequently foam cell
formation. Nevertheless, it should be noted that scavenger
receptors that recognize, bind, and take up Mox-LDL on macrophages
are yet to be reported. LDL modified by the whole
MPO/H2O2/Cl- system also has
pro-inflammatory properties that activate macrophages. Treatment of
THP-1 monocytes with Mox-LDL at a physiologically relevant
concentration of 100 µg/ml for 4 h resulted in a 2-fold increase in
the secretion of TNF-α; meanwhile, there was no increase in TNF-α
secretion when the cells were treated with native LDL, albumin, or
Mox-albumin (61). This shows the
specificity of Mox-LDL in the induction of TNF-α release (61). Additionally, incubating RAW264.7 and
PBMC-derived macrophages with Mox-LDL resulted in an increase in
reactive oxygen species (ROS) production (62). Furthermore, it has been reported
that Mox-LDL treatment resulted in overexpression of the
antioxidant genes Gclm and Heme oxygenase-1 (HO-1), which are
induced by the activation of the transcription factor nuclear
factor erythroid 2-related factor 2(62). The effect of Mox-LDL on macrophages
can be further elucidated by the ability of Mox-LDL to form a more
pronounced foam cell phenotype than is possible via Cuox-LDL in
THP-1 cells (63). In this context,
it was found that Mox-LDL induced an unfolded protein response
(UPR), apoptosis, and autophagy in THP-1 cells (63). Recently, our research group explored
the potential role of Mox-LDL in the polarization and
repolarization of macrophages by evaluating the in vitro
effects of Mox-LDL on the polarization of resting M0-macrophages,
as well as on the repolarization of M1- and M2-macrophages using a
well-established model of human THP-1-derived macrophages. Our
results showed that the surface expression of CD80 was highly
significant in M1 macrophages when compared to all other subsets.
Similarly, CD209 was significantly expressed in M2 macrophages when
compared to the other subtypes. Although IL-6 release was minimal
in all macrophage types except in the M1 macrophages where it was
upregulated as expected, the same M1 macrophage subtype also showed
a surprising and significant increase in IL-10 release (64). In our model, physiological
concentrations of Mox-LDL had no significant effects on CD80/CD209
expression or IL-6/IL-10 release in Mox-LDL treated M0 macrophages
(64). However, treatment of THP-1
M1 macrophages with Mox-LDL had a significant negative effect on
IL-10 cytokine secretion (64).
Overall, these results highlighted the inability of Mox-LDL to
drive M0 macrophages towards an inflammatory or an alternative
phenotype. More importantly, the data revealed an important role of
Mox-LDL in increasing the pro-inflammatory state in macrophages by
reducing the levels of the anti-inflammatory cytokine IL-10.
In summary, LDL modified by the
MPO/H2O2/Cl- system or directly by
HOCl appears to activate macrophages and induce a stronger reaction
in them compared with other forms of oxLDL.
MPO modified LDL and endothelial
dysfunction
There is a general consensus among scholars
regarding the role of ED in the initiation of atherosclerosis, and
oxLDL has the ability to facilitate ED in various manners (7). Meanwhile, the pro-inflammatory nature
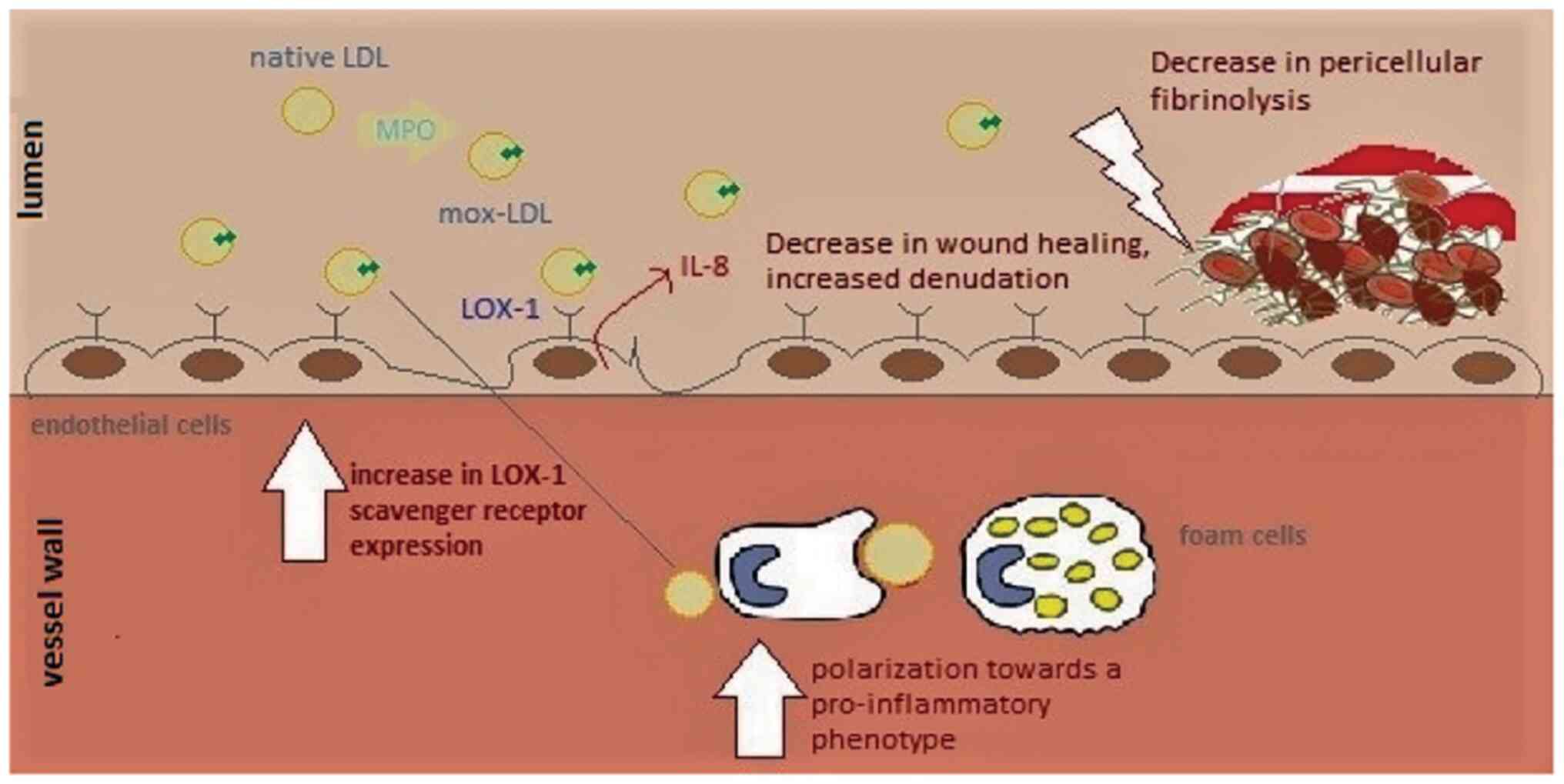
of LDL oxidized by MPO activates EC at multiple levels (7). In Ea.hy 926 endothelial cells, Mox-LDL
treatment induces an increase in the release of IL-8 at
concentrations of 100 and 200 mg/ml (61). Similarly, LDL modified by the
MPO/H2O2/Cl- system was found to
interrupt several EC functions in human umbilical vein endothelial
cells (HUVECs) (65). It was
reported that pathophysiological concentrations of Mox-LDL decrease
tubulogenesis, motility, and wound healing in HUVECs (65). These properties are associated with
in vivo angiogenesis. At the molecular level, it was also
shown that the observed phenotypic effects can be attributed to
miR-22 and the HO-1 gene (Fig. 2)
(65). In addition, ECs are known
for their role in balancing coagulation and fibrinolysis to form a
dynamic equilibrium at their surfaces. Interestingly, Mox-LDL was
found to interfere with this role of ECs. In fact, treatment of
Ea.hy926 endothelial cells with physiological concentrations of
Mox-LDL resulted in an increase in the duration of pericellular
fibrinolysis (Fig. 3) (49). Furthermore, this phenotypic change
was not related to plasminogen activator inhibitor-1 (PAI-1),
tissue plasminogen activator (t-PA), t-PA receptor, or
urokinase-type plasminogen activator (uPA) receptor expression, and
therefore, it was inferred that Mox-LDL delays pericellular
fibrinolysis in a pathway independent of the t-PA- and PAI-1
pathways (49). On a similar note,
Mox-LDL treatment was also found to have no transcriptional effect
on key fibrinolytic factors such as t-PA, uPA, and their receptors
as well the clotting factor XIII in human aortic endothelial cells
(HAECs) (66). Fibrin promotes
atherosclerosis by disorganizing the normal cobblestone arrangement
of EC and causing secretion of IL-8 from ECs (49). Therefore, Mox-LDL may be driving
atherosclerotic plaque formation by delaying fibrinolysis, thus
prolonging fibrin interaction with ECs that augment endothelial
permeability allowing further infiltration and subendothelial
accumulation of lipoproteins (Fig.
3). Moreover, Mox-LDL was found to exert differential effects
on cell viability between HAECs and bovine aortic endothelial (BAE)
cells (66). Indeed, physiological
concentrations of Mox-LDL did not exert any significant effect on
HAEC viability, whereas the same treatment caused significant death
of BAE cells, which is reminiscent of the cytotoxic effect of
Cuox-LDL on endothelial cells (66). With regard to ROS production, oxLDL,
and particularly the non-physiologically relevant model Cuox-LDL,
was reported to increase ROS production in ECs via the receptor
LOX-1(67). Interestingly,
physiologically relevant concentrations of Mox-LDL did not elevate
ROS production in HAECs (66).
HOCl-LDL is also reported to exert adverse effects on ECs.
Interestingly, HOCl-LDL was reported to exert a dose-dependent
anti-proliferative effect on human proximal tubular epithelial
cells (HK-2) (68). Additionally,
genes associated with inflammation, tissue remodeling, reactive
oxygen species metabolism, and cell stress were upregulated in HK-2
cells treated with HOCl-LDL (68).
Even though some of the scavenger receptors for HOCl-LDL on
macrophages are documented, the receptors that recognize Mox-LDL
and enable its uptake by macrophages and ECs are yet to be
identified (10). Scavenger
receptors known for the uptake of HOCl-LDL in macrophages (CD36 and
SR-BI) are also present on the surface of endothelial cells.
However, ECs are not capable of forming foam cells when incubated
with oxLDL (10). Previously, LOX-1
was reported to bind and take up oxLDL in ECs; however, the notion
of whether LOX-1 may function as the receptor for Mox-LDL in EC
remains debated. One study reported that LOX-1 is not potentially a
receptor for Mox-LDL, since antibodies raised against LOX-1 did not
affect IL-8 production induced by Mox-LDL in Ea.hy926(49). Meanwhile, our research group has
reported for the first time that the LOX-1 scavenger receptor might
regulate the inflammatory response of HAECs to physiological levels
of Mox-LDL. It was discovered that Mox-LDL upregulates the
expression of its own receptor in HAECs and induces inflammation in
the cells via LOX-1 in concert with the induction of this receptor
(69). These observations may have
important implications with regard to Mox-LDL-driven ED and provide
an initial hint to the pathways that are initiated by Mox-LDL
during ED and the progression of the atherosclerotic disease
(Fig. 3).
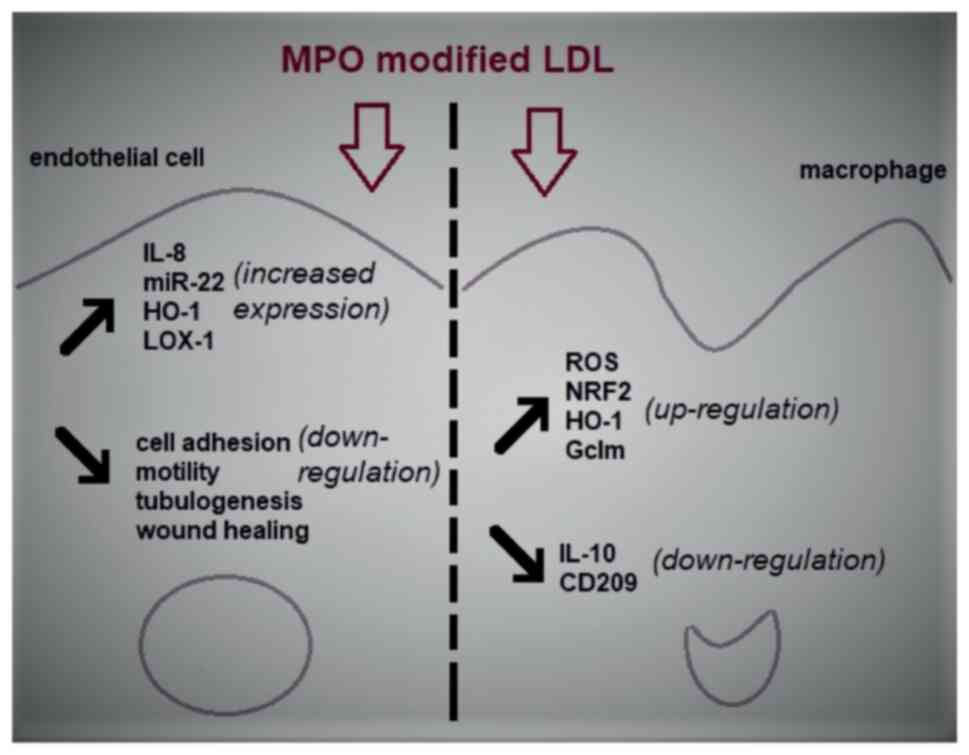 | Figure 2The effects of MPO modified LDL on
endothelial cells and macrophage models of atherosclerosis. LDL
that has been oxidized by MPO is able to induce ED which is
primarily demonstrated by an increase in inflammation and a
decrease in the physiological properties of ECs such as cell
motility and wound healing. Similarly, Mox-LDL is responsible for
the increase in oxidative stress and inflammation in macrophages
through the upregulation of the generation of ROS and the
downregulation of the major anti-inflammatory cytokine IL-10 in
these cells. MPO, myeloperoxidase; LDL, low-density lipoproteins;
ED, endothelial dysfunction; EC, endothelial cell; Mox-LDL, MPO
oxidized LDL; ROS, reactive oxygen species; miR, microRNA; HO-1,
heme oxygenase 1; LOX-1, lectin-like oxLDL receptor-1; NRF2,
nuclear factor erythroid 2-related factor 2; Gclm,
glutamate-cysteine ligase modifier subunit. |
6. Conclusions and future perspectives
In summary, there is a substantial body of evidence
suggesting that MPO is implicated in health and in disease.
Physiologically, MPO plays a role in innate microbial defenses by
catalyzing the formation of powerful reactive oxygen intermediates,
which are potent antimicrobial tools against phagocytosed
pathogens. Beyond the role of MPO in antimicrobial defenses, there
exist other uncharted important physiological roles of MPO
(9). Future research should focus
on exploring these still unknown or incompletely understood roles
in order to fully comprehend the pathophysiological functions of
MPO. In addition, it could be argued that considerably more is
known regarding the pathological role of MPO than its physiological
role. MPO is reported to be implicated in a range of diseases
characterized by an ongoing inflammatory state. In prolonged
inflammatory states, MPO can cause tissue damage through the
chemical modification of molecules as a result of MPO enzymatic
activities and through the action of MPO-generated oxidants. In
such circumstances, targeting and inhibiting MPO may induce a
therapeutic effect. Thus, in the future, it would be an intriguing
prospect to determine whether it is possible to design MPO
inhibitors that are efficient in vivo, and these may be able
to protect against the deleterious effects of MPO while preserving
its physiological role. Additionally, there exist species-specific
differences in the role of MPO in atherosclerosis and therefore,
researchers should be vigilant when translating and interpreting
the role of MPO in atherosclerosis.
On that same note, the ubiquitous presence of MPO
in atherosclerotic lesions along with the newly reported effects of
MPO-modified LDL in this particular context supports the notion
that LDL that is oxidized by MPO should now be considered as the
more pathophysiological model for LDL oxidation and that future
research involving oxLDL should now use this form of modified LDL
as a preferred model to study atherosclerosis. However, despite
recent advancements in the field and the increase in our
understanding of the effects of Mox-LDL in the development of
atherosclerosis, there remain several knowledge gaps pertaining to
the mechanisms regulating the role of Mox-LDL in macrophage and EC
pathobiology during the progression of the disease. In the latter
model, as already mentioned, our recent results provide an updated
knowledge on the signaling pathways that are promoted by Mox-LDL by
providing initial insights into its corresponding scavenger
receptor LOX-1(69). In light of
the latter finding, conceiving anti-atherogenic strategies that
target the Mox-LDL-LOX-1 signaling axis as a therapeutic target may
be extremely valuable in the context of treating atherosclerotic
diseases and alleviating its symptoms. Moreover, the same
strategies could be of an important benefit in the management of
other MPO-related pathologies, which include cancer, and
neurodegenerative and inflammatory diseases.
Nevertheless, these results are still insufficient
and thus, should pave the way for ongoing research to further
elucidate additional signaling pathways and novel functions of
Mox-LDL; this would ultimately direct scientists towards a more
complete understanding of the complex paradigm of ED in
atherosclerosis. Moreover, regarding the effect of Mox-LDL on
macrophage biology, we recently showed that Mox-LDL might play a
potential role in the repolarization of THP-1 macrophages by
increasing their pro-inflammatory state through the downregulation
of the major anti-inflammatory cytokine IL-10(64). Hopefully, these results will also
lead the way to future investigations that will give the scientific
community better insights into the immunomodulatory effects of
Mox-LDL and the instrumental role that this particular form of
modified LDL may play in the progression of atherosclerosis.
Additional data that could be generated at this level are highly
anticipated since they would certainly be crucial in introducing
research strategies that aim to develop therapeutics that target
the major pro-inflammatory immunological processes underlying
Mox-LDL-driven atherogenesis.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
CF drafted the manuscript. JD conceived the review
topic, and wrote and edited the manuscript. Both authors have read
and approved the final manuscript. Data authentication is not
applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Zamocky M, Jakopitsch C, Furtmüller PG,
Dunand C and Obinger C: The peroxidase-cyclooxygenase superfamily:
Reconstructed evolution of critical enzymes of the innate immune
system. Proteins. 72:589–605. 2008.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Arnhold J: The dual role of
myeloperoxidase in immune response. Int J Mol Sci.
21(8057)2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Aratani Y: Myeloperoxidase: Its role for
host defense, inflammation, and neutrophil function. Arch Biochem
Biophys. 640:47–52. 2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Daugherty A, Dunn JL, Rateri DL and
Heinecke JW: Myeloperoxidase, a catalyst for lipoprotein oxidation,
is expressed in human atherosclerotic lesions. J Clin Invest.
94:437–444. 1994.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Malle E, Waeg G, Schreiber R, Gröne EF,
Sattler W and Gröne HJ: Immunohistochemical evidence for the
myeloperoxidase/H2O2/halide system in human
atherosclerotic lesions: Colocalization of myeloperoxidase and
hypochlorite-modified proteins. Eur J Biochem. 267:4495–4503.
2000.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Teng N, Maghzal GJ, Talib J, Rashid I, Lau
AK and Stocker R: The roles of myeloperoxidase in coronary artery
disease and its potential implication in plaque rupture. Redox Rep.
22:51–73. 2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Daher J: Other forms of oxidized LDL:
Emerging functions (Review). World Acad Sci J. 2(4)2020.
|
|
8
|
Zámocký M, Hofbauer S, Schaffner I,
Gasselhuber B, Nicolussi A, Soudi M, Pirker KF, Furtmüller PG and
Obinger C: Independent evolution of four heme peroxidase
superfamilies. Arch Biochem Biophys. 574:108–119. 2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Vanhamme L, Zouaoui Boudjeltia K, Van
Antwerpen P and Delporte C: The other myeloperoxidase: Emerging
functions. Arch Biochem Biophys. 649:1–14. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Delporte C, Van Antwerpen P, Vanhamme L,
Roumeguère T and Zouaoui Boudjeltia K: Low-density lipoprotein
modified by myeloperoxidase in inflammatory pathways and clinical
studies. Mediators Inflamm. 2013(971579)2013.PubMed/NCBI View Article : Google Scholar
|
|
11
|
van der Veen BS, de Winther MP and
Heeringa P: Myeloperoxidase: Molecular mechanisms of action and
their relevance to human health and disease. Antioxid Redox Signal.
11:2899–2937. 2009.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Hansson M, Olsson I and Nauseef WM:
Biosynthesis, processing, and sorting of human myeloperoxidase.
Arch Biochem Biophys. 445:214–224. 2006.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Malle E, Furtmüller PG, Sattler W and
Obinger C: Myeloperoxidase: A target for new drug development? Br J
Pharmacol. 152:838–854. 2007.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Sugiyama S, Okada Y, Sukhova GK, Virmani
R, Heinecke JW and Libby P: Macrophage myeloperoxidase regulation
by granulocyte macrophage colony-stimulating factor in human
atherosclerosis and implications in acute coronary syndromes. Am J
Pathol. 158:879–891. 2001.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Schindhelm RK, van der Zwan LP, Teerlink T
and Scheffer PG: Myeloperoxidase: A useful biomarker for
cardiovascular disease risk stratification? Clin Chem.
55:1462–1470. 2009.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Smith JD: Dysfunctional HDL as a
diagnostic and therapeutic target. Arterioscler Thromb Vasc Biol.
30:151–155. 2010.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Hazen SL and Heinecke JW:
3-Chlorotyrosine, a specific marker of myeloperoxidase-catalyzed
oxidation, is markedly elevated in low density lipoprotein isolated
from human atherosclerotic intima. J Clin Invest. 99:2075–2081.
1997.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Pennathur S, Bergt C, Shao B, Byun J,
Kassim SY, Singh P, Green PS, McDonald TO, Brunzell J, Chait A, et
al: Human atherosclerotic intima and blood of patients with
established coronary artery disease contain high density
lipoprotein damaged by reactive nitrogen species. J Biol Chem.
279:42977–42983. 2004.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zheng L, Nukuna B, Brennan ML, Sun M,
Goormastic M, Settle M, Schmitt D, Fu X, Thomson L, Fox PL, et al:
Apolipoprotein A-I is a selective target for
myeloperoxidase-catalyzed oxidation and functional impairment in
subjects with cardiovascular disease. J Clin Invest. 114:529–541.
2004.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Bergt C, Pennathur S, Fu X, Byun J,
O'Brien K, McDonald TO, Singh P, Anantharamaiah GM, Chait A,
Brunzell J, et al: The myeloperoxidase product hypochlorous acid
oxidizes HDL in the human artery wall and impairs ABCA1-dependent
cholesterol transport. Proc Natl Acad Sci USA. 101:13032–13037.
2004.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Nussbaum C, Klinke A, Adam M, Baldus S and
Sperandio M: Myeloperoxidase: A leukocyte-derived protagonist of
inflammation and cardiovascular disease. Antioxid Redox Signal.
18:692–713. 2013.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yang J, Ji R, Cheng Y, Sun JZ, Jennings LK
and Zhang C: L-arginine chlorination results in the formation of a
nonselective nitric-oxide synthase inhibitor. J Pharmacol Exp Ther.
318:1044–1049. 2006.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zhang C, Reiter C, Eiserich JP, Boersma B,
Parks DA, Beckman JS, Barnes S, Kirk M, Baldus S, Darley-Usmar VM
and White CR: L-arginine chlorination products inhibit endothelial
nitric oxide production. J Biol Chem. 276:27159–27165.
2001.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Stocker R, Huang A, Jeranian E, Hou JY, Wu
TT, Thomas SR and Keaney JF Jr: Hypochlorous acid impairs
endothelium-derived nitric oxide bioactivity through a
superoxide-dependent mechanism. Arterioscler Thromb Vasc Biol.
24:2028–2033. 2004.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Gimbrone MA Jr and García-Cardeña G:
Endothelial cell dysfunction and the pathobiology of
atherosclerosis. Circ Res. 118:620–636. 2006.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Vita JA, Brennan ML, Gokce N, Mann SA,
Goormastic M, Shishehbor MH, Penn MS, Keaney JF Jr and Hazen SL:
Serum myeloperoxidase levels independently predict endothelial
dysfunction in humans. Circulation. 110:1134–1139. 2004.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Fu X, Kassim SY, Parks WC and Heinecke JW:
Hypochlorous acid oxygenates the cysteine switch domain of
pro-matrilysin (MMP-7). A mechanism for matrix metalloproteinase
activation and atherosclerotic plaque rupture by myeloperoxidase. J
Biol Chem. 276:41279–41287. 2001.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Wang Y, Rosen H, Madtes DK, Shao B, Martin
TR, Heinecke JW and Fu X: Myeloperoxidase inactivates TIMP-1 by
oxidizing its N-terminal cysteine residue: An oxidative mechanism
for regulating proteolysis during inflammation. J Biol Chem.
282:31826–31834. 2007.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Klebanoff SJ, Kinsella MG and Wight TN:
Degradation of endothelial cell matrix heparan sulfate proteoglycan
by elastase and the myeloperoxidase-H2O2-chloride system. Am J
Pathol. 143:907–917. 1993.PubMed/NCBI
|
|
30
|
Zhang R, Brennan ML, Fu X, Aviles RJ,
Pearce GL, Penn MS, Topol EJ, Sprecher DL and Hazen SL: Association
between myeloperoxidase levels and risk of coronary artery disease.
JAMA. 286:2136–2142. 2001.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Nikpoor B, Turecki G, Fournier C, Théroux
P and Rouleau GA: A functional myeloperoxidase polymorphic variant
is associated with coronary artery disease in French-Canadians. Am
Heart J. 142:336–339. 2001.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kutter D, Devaquet P, Vanderstocken G,
Paulus JM, Marchal V and Gothot A: Consequences of total and
subtotal myeloperoxidase deficiency: Risk or benefit? Acta
Haematol. 104:10–15. 2000.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Nicholls SJ and Hazen SL: Myeloperoxidase
and cardiovascular disease. Arterioscler Thromb Vasc Biol.
25:1102–1111. 2005.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Heslop CL, Frohlich JJ and Hill JS:
Myeloperoxidase and C-reactive protein have combined utility for
long-term prediction of cardiovascular mortality after coronary
angiography. J Am Coll Cardiol. 55:1102–1109. 2010.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Bentzon JF, Otsuka F, Virmani R and Falk
E: Mechanisms of plaque formation and rupture. Circ Res.
114:1852–1866. 2014.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Delporte C, Boudjeltia KZ, Noyon C,
Furtmüller PG, Nuyens V, Slomianny MC, Madhoun P, Desmet JM, Raynal
P, Dufour D, et al: Impact of myeloperoxidase-LDL interactions on
enzyme activity and subsequent posttranslational oxidative
modifications of apoB-100. J Lipid Res. 55:747–757. 2014.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Yoshida H, Quehenberger O, Kondratenko N,
Green S and Steinberg D: Minimally oxidized low-density lipoprotein
increases expression of scavenger receptor A, CD36, and macrosialin
in resident mouse peritoneal macrophages. Arterioscler Thromb Vasc
Biol. 18:794–802. 1998.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Steinberg D: Low density lipoprotein
oxidation and its pathobiological significance. J Biol Chem.
272:20963–20966. 1997.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Mehta JL, Chen J, Hermonat PL, Romeo F and
Novelli G: Lectin-like, oxidized low-density lipoprotein receptor-1
(LOX-1): A critical player in the development of atherosclerosis
and related disorders. Cardiovasc Res. 69:36–45. 2006.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Gao S, Zhao D, Wang M, Zhao F, Han X, Qi Y
and Liu J: GW28-e0393 Association between circulating oxidized
low-density lipoprotein and atherosclerotic cardiovascular disease:
A meta-analysis of prospective observational studies. J Am Coll
Cardiol. 70 (16 Suppl):C79–C80. 2017.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Holvoet P, Mertens A, Verhamme P, Bogaerts
K, Beyens G, Verhaeghe R, Collen D, Muls E and Van de Werf F:
Circulating oxidized LDL is a useful marker for identifying
patients with coronary artery disease. Arterioscler Thromb Vasc
Biol. 21:844–848. 2001.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Kita T, Kume N, Minami M, Hayashida K,
Murayama T, Sano H, Moriwaki H, Kataoka H, Nishi E, Horiuchi H, et
al: Role of oxidized LDL in atherosclerosis. Ann N Y Acad Sci.
947:199–206. 2001.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Mitra S, Goyal T and Mehta JL: Oxidized
LDL, LOX-1 and atherosclerosis. Cardiovasc Drugs Ther. 25:419–429.
2011.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Yoshida H and Kisugi R: Mechanisms of LDL
oxidation. Clin Chim Acta. 411:1875–1882. 2010.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Carr AC: Hypochlorous acid-modified
low-density lipoprotein inactivates the lysosomal protease
cathepsin B: Protection by ascorbic and lipoic acids. Redox Rep.
6:343–349. 2001.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Sokolov AV, Chekanov AV, Kostevich VA,
Aksenov DV, Vasilyev VB and Panasenko OM: Revealing binding sites
for myeloperoxidase on the surface of human low density
lipoproteins. Chem Phys Lipids. 164:49–53. 2011.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Moguilevsky N, Zouaoui Boudjeltia K, Babar
S, Delrée P, Legssyer I, Carpentier Y, Vanhaeverbeek M and Ducobu
J: Monoclonal antibodies against LDL progressively oxidized by
myeloperoxidase react with ApoB-100 protein moiety and human
atherosclerotic lesions. Biochem Biophys Res Commun. 323:1223–1228.
2004.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Vaes M, Zouaoui Boudjeltia KZ, Van
Antwerpen P, Babar S, Deger F, Neve J, Vanhaeverbeek M and Ducobu
J: ThP15:146 Low-density lipoprotein oxidation by myeloperoxidase
occurs in the blood circulation during hemodialysis.
Atherosclerosis Suppl. 7(525)2006.
|
|
49
|
Zouaoui Boudjeltia K, Daher J, Van
Antwerpen P, Moguilevsky N, Delree P, Ducobu J, Raes M, Badran B,
Vanhaeverbeek M, Brohee D, et al: Exposure of endothelial cells to
physiological levels of myeloperoxidase-modified LDL delays
pericellular fibrinolysis. PLoS One. 7(e38810)2012.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Podrez EA, Schmitt D, Hoff HF and Hazen
SL: Myeloperoxidase-generated reactive nitrogen species convert LDL
into an atherogenic form in vitro. J Clin Invest. 103:1547–1560.
1999.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Leeuwenburgh C, Hardy MM, Hazen SL, Wagner
P, Oh-ishi S, Steinbrecher UP and Heinecke JW: Reactive nitrogen
intermediates promote low density lipoprotein oxidation in human
atherosclerotic intima. J Biol Chem. 272:1433–1436. 1997.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Wang Z, Nicholls SJ, Rodriguez ER, Kummu
O, Hörkkö S, Barnard J, Reynolds WF, Topol EJ, DiDonato JA and
Hazen SL: Protein carbamylation links inflammation, smoking, uremia
and atherogenesis. Nat Med. 13:1176–1184. 2007.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Nicholson AC, Frieda S, Pearce A and
Silverstein RL: Oxidized LDL binds to CD36 on human
monocyte-derived macrophages and transfected cell lines. Evidence
implicating the lipid moiety of the lipoprotein as the binding
site. Arterioscler Thromb Vasc Biol. 15:269–275. 1995.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Isa SA, Ruffino JS, Ahluwalia M, Thomas
AW, Morris K and Webb R: M2 macrophages exhibit higher sensitivity
to oxLDL-induced lipotoxicity than other monocyte/macrophage
subtypes. Lipids Health Dis. 10(229)2011.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Montano EN, Boullier A, Almazan F, Binder
CJ, Witztum JL and Hartvigsen K: Development and application of a
nonradioactive binding assay of oxidized low-density lipoprotein to
macrophage scavenger receptors. J Lipid Res. 54:3206–3214.
2013.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Houben T, Oligschlaeger Y, Bitorina AV,
Hendrikx T, Walenbergh SMA, Lenders MH, Gijbels MJJ, Verheyen F,
Lütjohann D, Hofker MH, et al: Blood-derived macrophages prone to
accumulate lysosomal lipids trigger oxLDL-dependent murine hepatic
inflammation. Sci Rep. 7(12550)2017.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Westendorf T, Graessler J and Kopprasch S:
Hypochlorite-oxidized low-density lipoprotein upregulates CD36 and
PPARgamma mRNA expression and modulates SR-BI gene expression in
murine macrophages. Mol Cell Biochem. 277:143–152. 2005.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Woenckhaus C, Kaufmann A, Bussfeld D,
Gemsa D, Sprenger H and Gröne HJ: Hypochlorite-modified LDL:
Chemotactic potential and chemokine induction in human monocytes.
Clin Immunol Immunopathol. 86:27–33. 1998.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Vicca S, Hennequin C, Nguyen-Khoa T, Massy
ZA, Descamps-Latscha B, Drüeke TB and Lacour B: Caspase-dependent
apoptosis in THP-1 cells exposed to oxidized low-density
lipoproteins. Biochem Biophys Res Commun. 273:948–954.
2000.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Marsche G, Zimmermann R, Horiuchi S,
Tandon NN, Sattler W and Malle E: Class B scavenger receptors CD36
and SR-BI are receptors for hypochlorite-modified low density
lipoprotein. J Biol Chem. 278:47562–47570. 2003.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Boudjeltia KZ, Legssyer I, Van Antwerpen
P, Kisoka RL, Babar S, Moguilevsky N, Delree P, Ducobu J, Remacle
C, Vanhaeverbeek M and Brohee D: Triggering of inflammatory
response by myeloperoxidase-oxidized LDL. Biochem Cell Biol.
84:805–812. 2006.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Calay D, Rousseau A, Mattart L, Nuyens V,
Delporte C, Van Antwerpen P, Moguilevsky N, Arnould T, Boudjeltia
KZ and Raes M: Copper and myeloperoxidase-modified LDLs activate
Nrf2 through different pathways of ROS production in macrophages.
Antioxid Redox Signal. 13:1491–1502. 2010.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Vlaminck B, Calay D, Genin M, Sauvage A,
Ninane N, Zouaoui Boudjeltia K, Raes M and Michiels C: Effects of
copper sulfate-oxidized or myeloperoxidase-modified LDL on lipid
loading and programmed cell death in macrophages under hypoxia.
Hypoxia (Auckl). 2:153–169. 2014.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Bazzi S, Frangie C, Azar E and Daher J:
The effect of myeloperoxidase-oxidized LDL on THP-1 macrophage
polarization and repolarization. Innate Immun. 28(2):91–103.
2022.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Daher J, Martin M, Rousseau A, Nuyens V,
Fayyad-Kazan H, Van Antwerpen P, Courbebaisse G, Martiat P, Badran
B, Dequiedt F, et al: Myeloperoxidase oxidized LDL interferes with
endothelial cell motility through miR-22 and heme oxygenase 1
induction: Possible involvement in reendothelialization of vascular
injuries. Mediators Inflamm. 2014(134635)2014.PubMed/NCBI View Article : Google Scholar
|
|
66
|
El Samad G, Bazzi S, Karam M, Boudjeltia
KZ, Vanhamme L and Daher J: Effect of myeloperoxidase modified LDL
on bovine and human aortic endothelial cells. Exp Ther Med.
18:4567–4574. 2019.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Neri Serneri GG, Coppo M, Bandinelli M,
Paoletti P, Toscano T, Micalizzi E, Chiostri M and Boddi M:
Exaggerated myocardial oxLDL amount and LOX-1 receptor
over-expression associated with coronary microvessel inflammation
in unstable angina. Atherosclerosis. 226:476–482. 2013.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Porubsky S, Schmid H, Bonrouhi M, Kretzler
M, Malle E, Nelson PJ and Gröne HJ: Influence of native and
hypochlorite-modified low-density lipoprotein on gene expression in
human proximal tubular epithelium. Am J Pathol. 164:2175–2187.
2004.PubMed/NCBI View Article : Google Scholar
|
|
69
|
El-Hajjar L, Hindieh J, Andraos R,
El-Sabban M and Daher J: Myeloperoxidase-oxidized LDL activates
human aortic endothelial cells through the LOX-1 scavenger
receptor. Int J Mol Sci. 23(2837)2002.PubMed/NCBI View Article : Google Scholar
|