1. Introduction
Urinary tract infections (UTI) are a common
condition. Among them, cystitis is the most frequent disease.
Treatment for UTI/cystitis ranges from over-the-counter medications
to antibiotics if the cause is an infection. In such patients,
antimicrobials are often inappropriately prescribed, and their use
is associated with the selection of antimicrobial-resistant
organisms colonizing or infecting the urinary tract (1).
The rise in antimicrobial resistant organisms is
becoming a relevant clinical and public health issue. Worldwide, it
has been estimated that ~10,000,000 deaths by 2050 will be
attributable to antibiotic resistance (2).
Recent guidelines from the main international
associations, such as the American and Canadian Urological
Associations, and the Society of Urodynamics, Female Pelvic
Medicine and Urogenital Reconstruction (3), and the recent scientific literature
(4,5) have underlined the importance of
limited antibiotic use and of new research on molecules that
interact with bacterial load or virulence mechanisms of
uropathogens for the treatment of UTI.
Among these molecules, several studies have
identified D-mannose (6-12),
which is characterized by a non-pharmacological, non-metabolic,
non-bacteriostatic or bactericidal, but biomechanical mechanism of
action and does not affect antibiotic resistance (13).
2. Mechanism of action of D-mannose in the
prevention and treatment of UTI
D-mannose is a monosaccharide naturally produced by
the body from glucose. It is present in the body cells and in some
foods. D-mannose differs from glucose by inversion of one of the
four chiral centers of the molecule, precisely that on the carbon
atom in the position 2. D-mannose is the ‘C-2 epimer’ of glucose
(14,15).
At least 90% of ingested D-mannose is absorbed in
the upper part of the intestine. Its peculiarity is that despite it
being a simple molecule, this sugar is not metabolized by the
organism. Consequently, it is not stored in the liver or other
organs, but it is excreted unconverted into the urine via the
kidneys. About 60 min after ingestion, it arrives unchanged in the
urinary tract. D-mannose also has no effect on human metabolism
after long-term use (16,17).
The most common agent of cystitis is the
uropathogenic E. coli (UPEC). UPEC adheres to urothelial
cells mainly through the interaction between FimH (a fimbrial
adhesin) and mannosylated uroplakin proteins, which are the main
determinants of the urovirulence of UPEC (2).
Several studies have shown that, in the urine, E.
Coli attaches to D-mannose. This mechanism is based on the
structural similarity between D-mannose and urothelial mannosylated
receptors exposed by the epithelium of the urinary tract.
Consequently, D-mannose prevents FimH-mediated bacterial adhesion
to the bladder wall through a competitive inhibition mechanism
(18).
Since the process of bacterial adhesion on the
urothelial cell's surface is a determinant of the start of UTI,
D-mannose was shown to be effective in treating UTIs caused by
E. Coli (7,10). D-mannose exerts a urothelial barrier
function, inhibiting the adhesion of bacteria to the urothelium.
Binding free D-mannose, bacteria are blocked in the urine and then
eliminated by the urinary tract (19). As a consequence of this mechanism,
in vivo and in vitro studies have demonstrated that
mannose-like molecules lower bacterial load 2-4 fold in the urinary
tract and in the bladder (20).
This effect is also present in the case of
concurrent antibiotic therapy. In fact, D-mannose has no
bacteriostatic and/or bactericidal activity and does not modify the
bacterial cell, thus it does not interfere with the action of
antibiotics (2,21).
Furthermore, it has been suggested that the dosages
of D-mannose used in clinical practice does not affect E. coli
metabolism and growth and does not modify bacterial adhesiveness
causing FimH variants. All these characteristics underline the fact
that the long term use of D-mannose is safe (18).
Several studies, reviews and meta-analyses have
shown that D-mannose use lowers the risk of recurrent (r)UTI
(22,23). Less data has been published
regarding the role of D-mannose in the treatment of UTI/cystitis
related symptoms. In particular, to the best of our knowledge, no
reviews have been published on the role of D-mannose in the
treatment of acute UTI/cystitis, i.e., on the use of d-mannose not
as prevention of recurrence but as treatment of acute symptoms.
In this paper, we have performed a systematic review
of the available data on the effect of D-mannose on the typical
symptoms of UTI/cystitis given alone or in association with other
compounds.
3. Literature search methodology
Literature search
We searched PubMed (National Library of Medicine,
Washington, DC) and EMBASE databases from 1990 to January 2022
using combinations of the key words: ‘mannose’, ‘mannoside’,
‘D-mannose’, ‘bacteriuria’, ‘pyuria’, ‘pyelocystitis’, ‘cystitis’,
‘urinary tract infections’, ‘cystalgia, recurrent next urinary
tract infection’ with the appropriate Boolean modifiers (limits:
full article, human, English). After the original search, we
reviewed the reference lists of the identified articles to identify
other pertinent studies.
Two authors reviewed the papers and independently
identified the eligible articles for the systematic review and
extracted the data. Any disagreement was solved after discussion
with a third reviewer.
Studies were considered if they met all the
following criteria: Clinical studies, studies reporting original
data, studies reporting the number of patients using D-mannose
alone or in association with other treatments, studies reporting
number of patients with symptoms of UTI/cystitis at trial entry and
after the follow-up period. Reviews, commentaries, and case reports
were excluded.
This systematic review was performed according to
the PRISMA (Preferred Reporting Items for Systematic reviews and
Meta-Analyses) guidelines (24,25)
and registered in the PROSPERO database (registration no.
CRD42022303244).
Data extraction
A PICOS (Patient, Intervention, Comparator, Outcome,
Study) structure was used for defining the study questions and the
inclusion/exclusion criteria. The question was: ‘Is D-mannose
effective in the treatment of symptoms of UTI/cystitis?’ (Table I).
 | Table IPICOS criteria for inclusion and
exclusion of studies. |
Table I
PICOS criteria for inclusion and
exclusion of studies.
| Parameter | Inclusion
criteria | Data
extraction |
|---|
| Patient | Women with symptoms
of low urinary tract infection/cystitis | Location, age, type
of patients |
| Intervention | D-mannose | Dose and
duration |
| Comparator | No treatment | Group
definition |
| Outcome | Reduction of
symptoms | Number of cases,
type of assessment |
| Study | Cross-sectional,
cohort, case–control studies, clinical trials | Type of study
design |
For each study, the following information was
extracted: First author's last name; year of publication; country
of origin; design of the study; number of subjects treated with
D-mannose; age if present; criteria for study entry; type or
severity of symptoms at study entry and at follow up visit; type
and dose of drug; and length of follow-up.
To evaluate the effect of D-mannose on the symptoms
of UTI /cystitis, from the studies that presented data on long-term
follow-up, we only considered information obtained during the
evaluation of first symptoms after study entry.
Quality assessment
The quality of the studies included in the review
was evaluated using the Newcastle-Ottawa scale (NOS) (26). Studies were evaluated according to
three broad categories: Selection of study groups, comparability of
study groups, and assessment of outcome (cohort studies) or
ascertainment of exposure (case-control studies). The maximum score
was 9.
For Randomized Controlled Trials (RCTs), the Revised
Cochrane risk-of-bias tool for randomized trials was used (27).
4. Systematic review
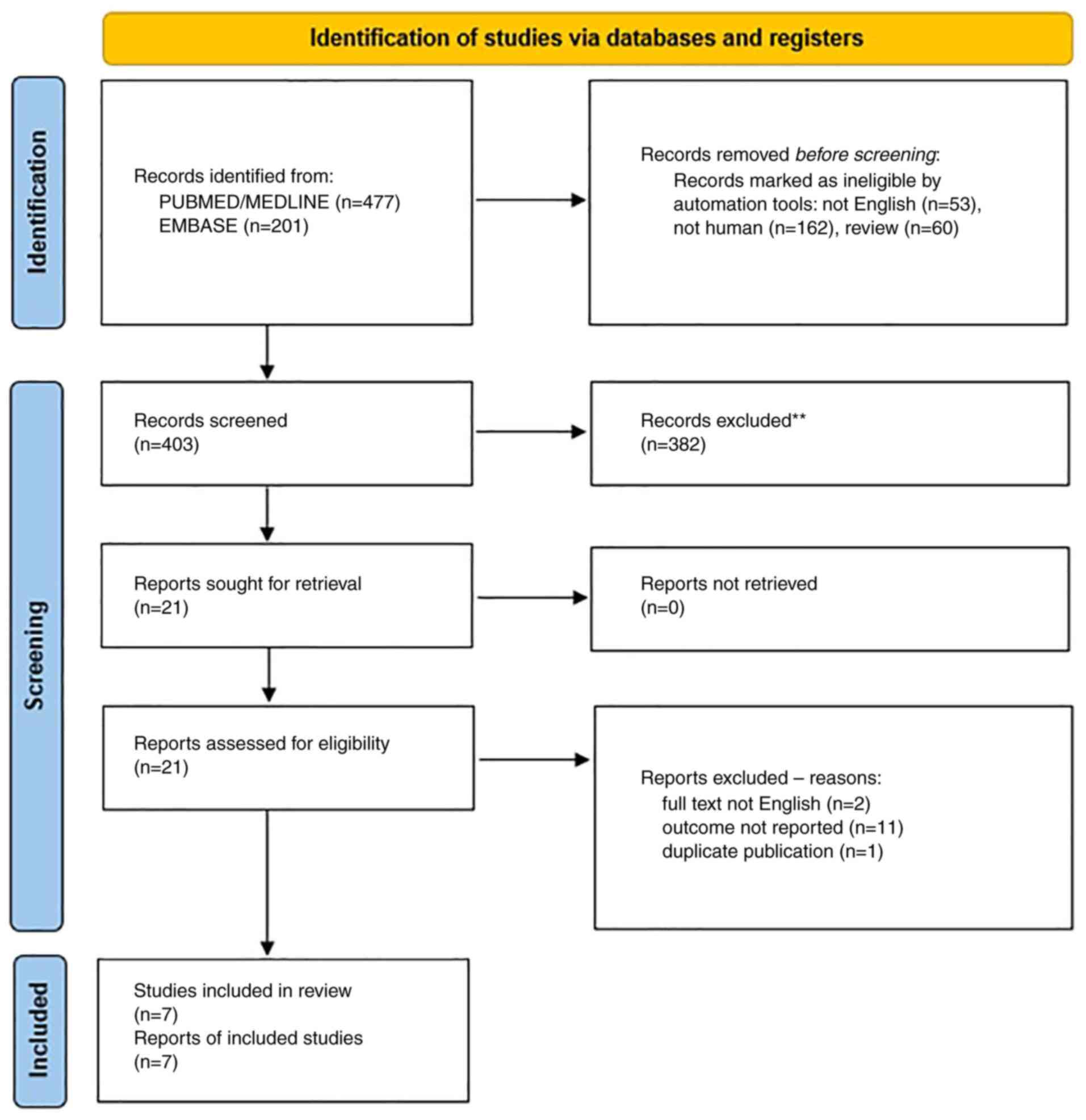
Our search retrieved 477 abstracts from
PubMed/MEDLINE, and 201 from Embase (Fig. 1). After reviewing the abstracts, a
total of 21 publications were identified that were fully read. Two
studies were excluded as the full text was not in English (28,29),
11 studies (19,30-39)
were excluded as they did not report the data on symptoms of
UTI/cystitis, but only the risk of recurrent UTI or the frequency
of UTI in patients submitted to urodynamics, and one (40) study was excluded as it reported a
Visual Analogic Scale (VAS) evaluation of symptoms 12 months after
study entry, but included women with rUTI without any specific
indication of symptoms of UTI/cystitis at study entry.
A total of 7 studies were identified (6-12).
Their primary methodological characteristics are presented in
Table II. A total of four studies
were prospective uncontrolled studies (7,8,10,11),
one was a retrospective chart review case-controlled study
(9) and two were randomized
controlled trials (6,12). All but one of the studies (10), included only women. The sample size
ranged from 33-93 subjects.
 | Table IIMain characteristics of selected
studies. |
Table II
Main characteristics of selected
studies.
| First author,
year | Study design | Country | Cohort | Inclusion
criteria | Sample size, n | Drug doses | Control group | Follow-up | Mode of symptoms
quantification | (Refs.) |
|---|
| Porru et al,
2014 | Randomized cross
over trial | Italy | Women aged ≥18
years (range 22-54) | Acute symptomatic
cystitis and history of rUTI | 46 | Oral D-mannose 1 g
three times a day, for 2 weeks, and subsequently 1 g twice a day
for 22 weeks | 5-day antibiotic
therapy with TMP/SMX 160 mg/800 mg twice a day, followed by a
single dose at bedtime for 1 week each month in the following 23
weeks | 24 weeks | VAS | (6) |
| Vicariotto,
2014 | Prospective
uncontrolled | Italy | Premenopausal women
aged >18 years | Acute uncomplicated
cystitis diagnosed by urine dipstick testing and an evaluation of
the presence of typical symptoms | 33 | 250 mg D-mannose/
500 mg of a high PACs cranberry extract/2.5 billion live cells
L. plantarum LP01/1 billion viable cells, L.
paracasei LPC09/1 billion viable cells S.
thermophilus ST10, 250 mg tara gum. 2 doses/day for 1
month, then 1 sachet/day for 1 month. | - | 30 weeks | UTI-Symptoms
Assessment Questionnaire | (7) |
| Domenici et
al, 2016 | Prospective
uncontrolled | Italy | Women aged 18-65
years | Acute cystitis
and/or history of rUTIs | 43 | D-mannose (1.5 g),
sodium bicarbonate, sorbitol and silicon dioxide twice daily for 3
days and then once a day for 10 days. | - | 15 weeks | UTI-Symptoms
Assessment Questionnaire | (8) |
| Marchiori and
Zanello, 2017 | Retrospective case
control study | Italy | Women with a
diagnosis of breast cancer treated with aromatase inhibitors or
tamoxifen or LHRH analogs | Women with rUTI
complaining urogenital discomfort | 60 (Group 1 40
Group 2: 20) | D-mannose 500 mg,
N-acetylcysteine 100 mg and Morinda citrifolia fruit extract 200 mg
(NDM) 1 vial every 12 h for 60 days and then 1 vial every 24 h for
4 months, associated with antibiotic therapy (see Group 2) | Antibiotic therapy,
depending on microbial sensitivity (Fosfomycin, 3 g per day for 2
days or nitrofurantoin 1 cprs 100 mg three times a day for 6 days
or ciprofloxacin 1,000 RM or prulifloxacin 600 mg 1 cps/day for 6
days) | 2 months | Verbal rating scale
ranking form 0 (absence of symptoms) to 4 (severe symptom) | (9) |
| Del Popolo and
Nelli, 2018 | Prospective
uncontrolled | Italy | Men and women
attending a neuro-urologic clinic | Symptomatic UTI and
history of rUTI | 78 patients (17
men) 39 patients had neurogenic bladder | 5-days regimen with
a tid oral combination of 1,000 mg of D-mannose plus 200 mg of dry
willow extract (salicin) followed by bid 7-days with 700 mg of
D-mannose plus 50 mg (1x109 CFU) of Lactobacillus acidophilus
(La-14). | - | 2
weeksa | VAS | (10) |
| Pugliese et
al, 2020 | Prospective
uncontrolled | Italy | Women (mean ± SD
Age 38±11.2) | Women with urinary
symptoms suggestive of UTI | 33 | D-mannose 2
g/fructo- oligosaccharide 1 g/pomegranate extract 250 mg (with 70%
titration of ellagic acid 175 mg)/Lactobacillus plantarum (Lp115 ≥
2 billion colony-forming unit) 2 times daily for 5 days and then
once a day for 10 days | - | 15 days | Acute Cystitis
Symptoms Score | (11) |
| Rădulescu et
al, 2020 | Randomized control
trial | Romania | Non-pregnant,
healthy women aged 18-60 years (mean ± SD age 39.77±10.36
years) | Uncomplicated lower
urinary tract infection | 93 | D-mannose 1 gr/400
mg cranberry extract for 7 days plus TMP-SMX | TMP-SMX alone for 7
days. | 7 days | 7 items
questionnaire (dysuria, increased urinary frequency/ pollakiuria,
urinary urgency, hematuria, hypogastric pain, lumbar pain, vesical
tenesmus) and 3 degrees of intensity (absent, moderate,
severe). | (12) |
With regard to the study drug, D-mannose was given
alone in only two studies (6,8). In
the study by Del Popolo and Nelli (10), D-mannose was given alongside dry
willow extract (salicin) in the first phase of the study and with
Lactobacillus acidophilus in the second phase. In the other studies
D-mannose was given alongside several other compounds, including
cranberry extract, Morinda citrifolia fruit extract,
pomegranate extract, fructo-oligosaccharides, lactobacilli, and
N-acetylcysteine.
Four studies evaluated the changes in symptoms in
the short term (7-15 days) (8,10-12),
one after 30 days (7), one after 60
days (9) and one after 24 weeks
(6).
Quality of selected studies
Considering the observational studies using the NOS
tool, study quality was constantly 5/9. There was the possibility
that some evaluation using the NOS quality items was debatable
(i.e., if the sample size was too little to control for important
factors or if a not exposed cohort did exist). The two trials
(6,12) had a low risk of bias according to
the Cochrane risk of bias tool (Table
III).
 | Table IIIEvaluation of the study quality
according to the Newcastle-Ottawa Scale (cohort studies) or
Cochrane risk of bias (randomized clinical trials). |
Table III
Evaluation of the study quality
according to the Newcastle-Ottawa Scale (cohort studies) or
Cochrane risk of bias (randomized clinical trials).
| Cohort
studya | Question # | Selection | Question # | Comparability | Question # | Outcome (Cohort
studies) | Study quality | (Refs.) |
|---|
| Vicariotto,
2014 | 1 | * | 1 | - | 1 | - | 5/9 | (7) |
| | 2 | - | 2 | - | 2 | * | | |
| | 3 | * | | | 3 | * | | |
| | 4 | * | | | | | | |
| Domenici et
al, 2016 | 1 | * | 1 | - | 1 | - | 5/9 | (8) |
| | 2 | - | 2 | - | 2 | * | | |
| | 3 | * | | | 3 | * | | |
| | 4 | * | | | | | | |
| Marchiori and
Zanello, 2017 | 1 | * | | | 1 | - | 5/9 | (9) |
| | 2 | - | 1 | - | 2 | * | | |
| | 3 | * | 2 | - | 3 | * | | |
| | 4 | * | | | | | | |
| Del Popolo and
Nelli, 2018 | 1 | * | | | 1 | - | 5/9 | (10) |
| | 2 | - | 1 | - | 2 | * | | |
| | 3 | * | 2 | - | 3 | * | | |
| | 4 | * | | | | | | |
| Pugliese et
al, 2020 | 1 | * | 1 | | 1 | - | 5/9 | (11) |
| | 2 | - | 2 | - | 2 | * | | |
| | 3 | * | | - | 3 | * | | |
| | 4 | * | | | | | | |
| Randomized clinical
trialsb | | | | | | | Overall risk of
bias | |
|
Porru et
al, 2014 | Randomization: low
risk | | | | Low | (6) |
| | Assignment to
intervention: low risk | | | | | |
| | Adhering to
intervention: low risk | | | | | |
| | Missing outcome:
low risk | | | | | |
| | Measure of outcome:
low risk | | | | | |
| | Selection of
results: low risk | | | | | |
|
Rădulescu
et al, 2020 | Randomization: some
concerns | | | | Low | (12) |
| | Assignment to
intervention: some concerns | | | | | |
| | Adhering to
intervention: low risk | | | | | |
| | Missing outcome:
low risk | | | | | |
| | Measure of outcome:
low risk | | | | | |
| | Selection of
results: low risk | | | | | |
To facilitate the reading of the systematic review,
we summarized the main results of the selected studies in Table IV.
 | Table IVResults of selected
studiesk. |
Table IV
Results of selected
studiesk.
| Study, year | Methods for
evaluation of the symptoms | Suprapubic
pain | Dysuria | Frequent
voiding | Urgency | Hematuria | Overall symptoms
evaluation | (Refs.) |
|---|
| Porru et al,
2014 | VAS | | | | | | | (6) |
|
Before
D-mannose | | 4.1
(1.1)c | | 7.1
(1.1)b | 4.6 (1.1) | | | |
|
After
D-mannose | | 2.2
(0.5)a | | 4.7
(1.0)a | 2.6
(0.7)a | | | |
| Vicariotto,
2014 | UTI-SAQ | | | | | | | (7) |
|
Baseline | | 1.39 | 2.03 | 2.18 | 2.15 | 0.61 | | |
|
Day 30 | | 0.97a | 1.36a | 1.70a | 1.64a | 0.58 | | |
| Domenici et
alb,h, 2016 | UTI-SAQ | | | | | | | (8) |
|
Baseline | | 1.47 (0.95) | 1.60 (±1.00) | 2.16 (1.52) | 1.73 (0.92) | 0.34 (0.90) | | |
|
Day 15 | | 0.15
(0.36)a | 0.31
(0.47)a | 0.60
(0.63)a | 0.23
(0.43)a | 0.10 (0.45) | | |
| Marchiori and
Zanello, 2017 | VRS | | | | | | | (9) |
| Cases | | | | | | | | |
|
Baseline | | 32.5%
(13/40)e,j | | 62.5% (25/40) | 37.5%
(15/40)f | | | |
|
2
months | | 25% (10/40) | | 5% (2/40) | 0% (0/40) | | | |
| Control group | | | | | | | | |
|
Baseline | | 50% (10/20) | | 35% (7/20) | 20% (4/20) | | | |
|
2
months | | 45% (9/20) | | 25% (5/20) | 5% (1/20) | | | |
| Del Popolo and
Nelli, 2020 | VAS | | | | | | | (10) |
| Neurogenic bladder
group | | | | | | | | |
|
Baseline | | | | 14.0
(2.6)d | | | 8.07
(1.70)h | |
|
2 weeks | | | | 6.9
(1.3)a | | | 4.74
(2.07)a | |
| Non neurogenic
bladder group | | | | | | | | |
|
Baseline | | | | 15(3)b | | | 7.21 (1.9) | |
|
2 weeks | | | | 8(3)a | | | 3.74
(3.12)a | |
| Pugliese et
al, 2020 | ACSS | | | | | | | (11) |
|
Baseline | | | | | | | 11.5i (95% CI, 10.5-12.6) | |
|
15 days | | | | | | | 4.9a (95% CI 4.0-5.9) | |
| Rădulescu et
all, 2020 | 3 degrees | | | | | | | (12) |
| Cases | questionnaire | | | | | | | |
|
Baseline | | 72.9
(35/48)g,j | 60.4 (29/48) | 85.4 (41/48) | 89.6% (43/48) | 10.4% (5/48) | | |
|
7 days | | 2.1 (1/48) | 0% (0/48) | 2.1% (1/48) | 0% (0/48) | 0% (0/48) | | |
| Control group | | | | | | | | |
|
Baseline | | 86.7% (39/45) | 55.6% (25/45) | 82.2% (37/45) | 80% (36/45) | 22.2% (10/45) | | |
|
7 days | | 2.2% (1/45) | 6.7% (3/45) | 8.9% (4/45) | 4.4% (2/45) | 0% (0/45) | | |
Porru et al (6) conducted a randomized cross-over trial
including women with an acute symptomatic UTI and three or more
rUTIs during the 12 months before study entry. A total of 60 women
were randomly divided into an antibiotic treatment with
trimethoprim/sulfamethoxazole or oral D-mannose three times a day
group, for 2 weeks (phase of the study considered in this review).
The primary endpoint, which was out of the scope of this review,
was the evaluation of the elapsed time to recurrence. We included
the secondary endpoints, which were bladder pain (VASp) and urinary
urgency (VASu). Mean VASp score, mean VASu score, and average
number of 24-h voiding events decreased significantly after 2 weeks
of treatment. Methods for taking into account the period effect
were not clearly stated. The authors did not report any adverse
events.
Vicariotto (7)
reported the results of a small prospective observational study.
Eligible for the study were 33 premenopausal, nonpregnant women
diagnosed with acute uncomplicated cystitis. Patients were given a
compound including D-mannose, cranberry dry extract,
exopolysaccharides produced by Streptococcus thermophilus ST10,
tara gum, Lactobacillus plantarum, and Lactobacillus
paracasei, two doses per day for 1 month. At baseline and in
the 30 day visits the following symptoms were evaluated: Dysuria,
frequent voiding, urgency, and suprapubic pain. A statistically
significant improvement was observed.
Domenici et al (8) reported an observational prospective
study. A total of 43 women with acute cystitis were included.
D-mannose was administered twice daily for 3 days and then once a
day for 10 days. Patients' symptoms, the therapeutic effects and
quality of life (QoL) were evaluated clinically using a validated
questionnaire (UTISA) (41) The
mean UTISA scores significantly improved for most symptoms between
baseline and follow up visits.
Marchiori and Zanello (9) studied the effectiveness of D-mannose
in association with N-acetylcysteine and Morinda citrifolia
fruit extract (DNM) plus antibiotic therapy, in recurrent cystitis.
A total of 60 women with breast cancer and recurrent cystitis were
analyzed retrospectively. Of these, 40 patients received antibiotic
therapy plus DNM and 20 women antibiotics alone for 6 months. After
2 months of study entry, women treated with DNM plus antibiotic
therapy exhibited a more prominent improvement in urgency,
frequency, urge, incontinence, bladder and urethral pain in
comparison to women treated with antibiotics alone.
Del Popolo and Nelli (10) reported an observational uncontrolled
prospective study including 85 patients (68 women and 17 men) with
symptomatic UTI and a history of rUTI. A total of 78 patients
received a 5-day regimen consisting of thrice daily oral D-mannose
and dry willow extract (salicin), followed by a 7-day regimen of
twice daily D-mannose and Lactobacillus acidophilus.
Patients' symptoms were evaluated using a 3-day bladder diary and a
VAS, 15 days after study entry. VAS scores decreased from 8.07±1.70
to 4.74±2.07 (P=0.001) in non-neurological patients (39 subjects,
group A) and from 7.21±1.90 to 3.74±3.12 (P=0.001) in the
neurological patients (39 subjects, group B). A significant
reduction in daily frequency was noted in both groups, from 14±3 to
7±3 (P=0.001) in group A and from 15±3 to 8±3 (P=0.001) in group
B.
D-mannose, pomegranate extract, prebiotics and
probiotics were given twice daily for 5 days and then once a day
for 10 days to 33 women (mean age 38.1±11.2 years) with urinary
symptoms suggestive of an UTI, in a study conducted by Pugliese
et al (11). Antibiotics
were permitted on a clinical basis. Changes in patients' symptoms
were evaluated using the Acute Cystitis Symptom Score (42,43) at
baseline (T0), and 15 (T1) and 30 (T2) days later. For the purpose
of this review, the results at T1 were considered. At T1, all or
most of the symptoms disappeared in 10 women (30.3%), whereas they
persisted or worsened in 7 patients (21.2%). Mean scores for
typical symptoms (i.e. frequency, urgency, pain or burning with
urination, pain in the suprapubic area, feeling of incomplete
bladder emptying, and gross hematuria) decreased significantly from
11.5 [95% confidence interval (CI) 10.5-12.6)] to 4.9 (95% CI
4.0-5.9) (P<0.0001). For differential symptoms (i.e., lower back
pain, vaginal discharge, urethral discharge, fever, and chills),
mean scores decreased from 3.1 (95% CI 2.6-3.6) to 0.6 (95% CI
0.3-0.9) (P<0.0001). The QoL mean score also decreased from 7.2
(95% CI 6.7-7.7) to 4.0 (95% CI 3.3-4.6) (P<0.0001). Antibiotics
were given to six patients. No adverse events were reported.
Rădulescu et al (12) reported a randomized study including
93 non-pregnant healthy women (mean age of 39.77±10.36 years)
diagnosed with uncomplicated lower UTI. Patients were given
antibiotics alone or in association with D-mannose plus cranberry
extract for 7 days. Co-administration of D-mannose plus cranberry
extract was not associated to statistically significant differences
in symptoms, except for urinary urgency/pollakiuria (P=0.024).
5. Discussion
This systematic review is, to the best of our
knowledge, the first attempt to systematically review the published
data on the effect of D-mannose in the treatment of acute symptoms
of UTI/cystitis. The results of this evaluation suggest that, in
women with symptoms of UTI/cystitis, treatment with D-mannose alone
or in association with other compounds is useful for lowering the
intensity of symptoms both in the short and middle-term for all
typical symptoms, except hematuria.
However, this finding has several limitations and
should be considered cautiously. First, the limited data do not
provide the opportunity for analysis in detail of the role of
different doses of D-mannose or the effect of D-mannose alone or in
association with other compounds. Furthermore, most data were
derived from uncontrolled studies. In general, the findings from
this analysis are based on an extremely limited number of studies
with small sample sizes.
The populations included in the considered studies
are markedly heterogeneous. For example, the study conducted by Del
Popolo and Nelli (10) included men
and women with neurological problems. Otherwise, the majority of
studies included only women in the premenopausal period (6,7,11).
Furthermore, as the methods of evaluation of symptoms differed, any
comparison among studies of the magnitude of the effect of
D-mannose is not feasible. Moreover, we also must take into account
that the majority of included studies were conducted in Italy. That
is, it is conceivable that only studies showing positive results
were published in an international journal, whereas negative
findings were instead published in local journals (44). Finally, limiting our analysis to
publications in English language journals may have reduced the
completeness of information, causing bias.
A strength of this analysis is that previous reviews
and meta-analyses considered the role of D-mannose in the
prevention of UTI recurrence (22,23),
whereas we summarized the available evidence regarding its use in
the treatment of UTI symptoms. However, from a biological point of
view, our findings have a rationale, since D-mannose binds to the
tip of type 1 pili and saturates adhesin FimH, blocking bacterial
adhesion to the urothelium and the consequent urothelial invasion
(45).
All these considerations were suggested to explain
the well-documented role of D-mannose in lowering the risk of
recurrent infections in women with rUTI. Less clear is the
potential mechanism of D-mannose on UTI/cystitis symptoms. However,
the fact that D-mannose interacts with bacteria to promote UPEC
excretion may explain a faster resolution of symptoms (18).
We were not able to analyze the effect of D-mannose
on different uropathogenetic agents separately. However, although
type 1 fimbriae were extensively studied in E. coli, type 1 pili
were documented in several other uropathogens commonly involved in
rUTIs (23). Furthermore, recently,
Zhang et al (46) suggested
that D-mannose could play a role as an immune modulator. It was
shown that prolonged exposure to D-mannose did not select FimH
variants that modify bacterial adhesiveness after D-mannose
removal, further indicating that it does not exert
‘antibiotic-like’ activity (21).
Along this line, we have observed similar effects in short and
middle-term treatments.
In conclusion, despite the limitations, consistent
results among all studies give strong support to the general
findings. Although the biological and clinical explanations of our
results are not entirely clear, observational studies and clinical
trials consistently suggest that D-mannose may be useful in the
treatment of UTI/cystitis symptoms. Its non-pharmacological,
non-metabolic, non-bacteriostatic or bactericidal, but
biomechanical mechanism of action, and the fact that it does not
affect antibiotic resistance may support the use of D-mannose in
the treatment of UTI/cystitis.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in the published article.
Authors' contributions
FP and FF designed the study. FC, SC and GE searched
the literature, selected the papers and extracted the data. SC and
ER confirm the authenticity of all the raw data. FP and ER wrote
the paper. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Abbo LM and Hooton TM: Antimicrobial
stewardship and urinary tract infections. Antibiotics (Basel).
3:174–192. 2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Sarshar M, Behzadi P, Ambrosi C, Zagaglia
C, Palamara AT and Scribano D: FimH and Anti-Adhesive therapeutics:
A disarming strategy against uropathogens. Antibiotics (Basel).
9(397)2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Anger J, Lee U, Ackerman AL, Chou R,
Chughtai B, Clemens JQ, Hickling D, Kapoor A, Kenton KS, Kaufman
MR, et al: Recurrent uncomplicated urinary tract infections in
women: AUA/CUA/SUFU guideline. J Urol. 202:282–289. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Taich L, Zhao H, Cordero C and Anger JT:
New paradigms in the management of recurrent urinary tract
infections. Curr Opin Urol. 30:833–837. 2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Barea BM, Veeratterapillay R and Harding
C: Nonantibiotic treatments for urinary cystitis: An update. Curr
Opin Urol. 30:845–852. 2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Porru D, Parmigiani A, Tinelli C, Barletta
D, Choussos D, Di Franco C, Bobbi V, Bassi S, Miller O, Gardella B,
et al: Oral D-mannose in recurrent urinary tract infections in
women: A pilot study. J Clin Urol. 7:208–213. 2014.
|
|
7
|
Vicariotto F: Effectiveness of an
association of a cranberry dried extract, D-mannose and the three
microorganisms Lactobacillus plantarum LP01, Lactobacillus.
paracasei LPC09 in women affected by cystitis: A pilot study. J
Clin Gastroenterol. 48 (Suppl 1):S96–S101. 2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Domenici L, Monti M, Bracchi C, Giorgini
M, Colagiovanni V, Muzii L and Benedetti*Panici P: D-mannose: A
promising support for acute urinary tract infections in women. A
pilot study. Eur Rev Med Pharmacol Sci. 20:2920–2925.
2016.PubMed/NCBI
|
|
9
|
Marchiori D and Zanello PP: Efficacy of
N-acetylcysteine, D-mannose and morinda citrifolia to treat
recurrent cystitis in breast cancer survivals. In Vivo. 31:931–936.
2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
del*Popolo G and Nelli F: Recurrent
bacterial symptomatic cystitis: A pilot study on a new natural
option for treatment. Arch Ital Urol Androl. 90:101–103.
2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Pugliese D, Acampora A, Porreca A, Schips
L and Cindolo L: Effectiveness of a novel oral combination of
D-Mannose, pomegranate extract, prebiotics and probiotics in the
treatment of acute cystitis in women. Arch Ital Urol Androl.
92:34–38. 2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Rădulescu D, David C, Turcu FL, Spătaru
DM, Popescu P and Văcăroiu IA: Combination of cranberry extract and
D-mannose-possible enhancer of uropathogen sensitivity to
antibiotics in acute therapy of urinary tract infections: Results
of a pilot study. Exp Ther Med. 20:3399–3406. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Zalewska-Piątek BM and Piątek RJ:
Alternative treatment approaches of urinary tract infections caused
by uropathogenic Escherichia coli strains. Acta Biochim Pol.
66:129–138. 2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Sharma V, Ichikawa M and Freeze HH:
Mannose metabolism: More than meets the eye. Biochem Biophys Res
Commun. 453:220–228. 2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Steinhardt RG, Calvin AD and Dodd EA:
Taste-Structure correlation with alpha-D-Mannose and
beta-D-Mannose. Science. 135:367–368. 1962.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Hu X, Shi Y, Zhang P, Miao M, Zhang T and
Jiang B: D -Mannose: Properties, production, and applications: An
overview. Compr Rev Food Sci Food Saf. 15:773–785. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Alton G, Kjaergaard S, Etchison JR, Skovby
F and Freeze HH: Oral ingestion of mannose elevates blood mannose
levels: A first step toward a potential therapy for
carbohydrate-deficient glycoprotein syndrome type I. Biochem Mol
Med. 60:127–133. 1997.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Scaglione F, Musazzi UM and Minghetti P:
Considerations on D-mannose mechanism of action and consequent
classification of marketed healthcare products. Front Pharmacol.
12(636377)2021.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Kranjčec B, Papeš D and Altarac S:
D-mannose powder for prophylaxis of recurrent urinary tract
infections in women: A randomized clinical trial. World J Urol.
32:79–84. 2014.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Klein T, Abgottspon D, Wittwer M, Rabbani
S, Herold J, Jiang X, Kleeb S, Lüthi C, Scharenberg M, Bezençon J,
et al: FimH antagonists for the oral treatment of urinary tract
infections: From design and synthesis to in vitro and in vivo
evaluation. J Med Chem. 53:8627–8641. 2010.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Scribano D, Sarshar M, Prezioso C,
Lucarelli M, Angeloni A, Zagaglia C, Palamara AT and Ambrosi C:
D-Mannose treatment neither affects uropathogenic Escherichia coli
properties nor induces stable fimH modifications. Molecules.
25(316)2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Lenger SM, Bradley MS, Thomas DA, Bertolet
MH, Lowder JL and Sutcliffe S: D-Mannose vs other agents for
recurrent urinary tract infection prevention in adult women: A
systematic review and meta-analysis. Am J Obstet Gynecol.
223:265.e1–265.e13. 2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
de Nunzio C, Bartoletti R, Tubaro A,
Simonato A and Ficarra V: Role of D-mannose in the prevention of
recurrent uncomplicated cystitis: State of the art and future
perspectives. Antibiotics (Basel). 10(373)2021.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Liberati A, Altman DG, Tetzlaff J, Mulrow
C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J
and Moher D: The PRISMA statement for reporting systematic reviews
and meta-analyses of studies that evaluate health care
interventions: Explanation and elaboration. PLoS Med.
6(e1000100)2009.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Moher D, Liberati A, Tetzlaff J and Altman
DG: PRISMA Group. Preferred reporting items for systematic reviews
and meta-analyses: The PRISMA statement. PLoS Med.
6(e1000097)2009.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Wells GA, Shea B, O'Connell D, Peterson J,
Welch VL, et al: Newcastle-Ottawa Scale for assessing the quality
of nonrandomized studies in Meta-analysis. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp,
2019.
|
|
27
|
Sterne JAC, Savović J, Page MJ, Elbers RG,
Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge
SM, et al: RoB 2: A revised tool for assessing risk of bias in
randomised trials. BMJ. 366(l4898)2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Kuzmenko AV, Kuzmenko VV and Gyaurgiev TA:
Use of D-mannose in the prevention of recurrent lower urinary tract
infection in women. Urologiia. 3:128–132. 2020.PubMed/NCBI(In Russian).
|
|
29
|
Salinas-Casado J, Méndez-Rubio S,
Esteban-Fuertes M, Gómez-Rodríguez A, Vírseda-Chamorro M,
Luján-Galán M, Iglesias-García C and Rituman G: Large study (283
women) on the effectiveness of Manosar®: 2 g of
d-mannose + 140 mg of proanthocyanidins (PAC), of prolonged
release. Arch Esp Urol. 73:491–498. 2020.PubMed/NCBI(In English, Spanish).
|
|
30
|
Altarac S and Papeš D: Use of D-mannose in
prophylaxis of recurrent urinary tract infections (UTIs) in women.
BJU Int. 113:9–10. 2014.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Lopes De Carvalho L, Francavilla G, Motta
R and Brichetto G: D-mannose, cranberry and Vitamin C are effective
in preventing urinary tract infections in multiple sclerosis
subjects. Mult Scler. 18:2012.
|
|
32
|
Efros M, Bromberg W, Cossu L, Nakeleski E
and Katz AE: Novel concentrated cranberry liquid blend, UTI-STAT
with proantinox, might help prevent recurrent urinary tract
infections in women. Urology. 76:841–845. 2010.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Genovese C, Davinelli S, Mangano K,
Tempera G, Nicolosi D, Corsello S, Vergalito F, Tartaglia E,
Scapagnini G and Di*Marco R: Effects of a new combination of plant
extracts plus d-mannose for the management of uncomplicated
recurrent urinary tract infections. J Chemother. 30:107–114.
2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Madhavan K, Rustagi S, Jena R, Singh UP,
Ansari MS, Srivastava A, Kapoor R and Sureka SK: A prospective
randomized study to define the role of low dose continuous
prophylactic antibiotics and anti-adherence agents in altering the
microbial colonization related to indwelling double-J stents. Asian
J Urol. 8:269–274. 2021.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Milandri R, Maltagliati M, Bocchialini T,
Del Prete C, Bianchi G, Rocco BM and Micali S: Effectiveness of
D-mannose, Hibiscus sabdariffa and Lactobacillus plantarum therapy
in prevention of infectious events following urodynamic study.
Urologia. 86:122–125. 2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Murina F, Vicariotto F and Lubrano C:
Efficacy of an orally administered combination of Lactobacillus
paracasei LC11, cranberry and D-mannose for the prevention of
uncomplicated, recurrent urinary tract infections in women.
Urologia. 88:64–68. 2021.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Palleschi G, Carbone A, Zanello PP, Mele
R, Leto A, Fuschi A, Al Salhi Y, Velotti G, Al Rawashdah S, Coppola
G, et al: Prospective study to compare antibiosis versus the
association of N-acetylcysteine, D-mannose and Morinda citrifolia
fruit extract in preventing urinary tract infections in patients
submitted to urodynamic investigation. Arch Ital Urol Androl.
89:45–50. 2017.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Russo E, Montt Guevara M, Giannini A,
Mannella P, Palla G, Caretto M, Pancetti F, Genazzani AD and
Simoncini T: Cranberry, D-mannose and anti-inflammatory agents
prevent lower urinary tract symptoms in women undergoing prolapse
surgery. Climacteric. 23:201–205. 2020.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Phé V, Pakzad M, Haslam C, Gonzales G,
Curtis C, Porter B, Chataway J and Panicker JN: Open label
feasibility study evaluating D-mannose combined with home-based
monitoring of suspected urinary tract infections in patients with
multiple sclerosis. Neurourol Urodyn. 36:1770–1775. 2017.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Mainini G, Passaro M, Schiattarella A,
Franciscis P, Donna MCD and Trezza G: Prevention and treatment of
cystitis during menopause: Efficacy of a nutraceutical containing
D-mannose, inulin, cranberry, bearberry, Olea europaea, Orthosiphon
and Lactobacillus acidophilus. Prz Menopauzalny. 19:130–134.
2020.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Clayson D, Wild D, Doll H, Keating K and
Gondek K: Validation of a patient-administered questionnaire to
measure the severity and bothersomeness of lower urinary tract
symptoms in uncomplicated urinary tract infection (UTI): The UTI
symptom assessment questionnaire. BJU Int. 96:350–359.
2005.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Alidjanov JF, Abdufattaev UA, Makhsudov
SA, Pilatz A, Akilov FA, Naber KG and Wagenlehner FM: New
self-reporting questionnaire to assess urinary tract infections and
differential diagnosis: Acute cystitis symptom score. Urol Int.
92:230–236. 2014.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Alidjanov JF, Naber KG, Abdufattaev UA,
Pilatz A and Wagenlehner FM: Reliability of symptom-based diagnosis
of uncomplicated cystitis. Urol Int. 102:83–95. 2019.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Grégoire G, Derderian F and Le*Lorier J:
Selecting the language of the publications included in a
meta-analysis: Is there a tower of babel bias? J Clin Epidemiol.
48:159–163. 1995.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Bouckaert J, Berglund J, Schembri M, De
Genst E, Cools L, Wuhrer M, Hung CS, Pinkner J, Slättegård R,
Zavialov A, et al: Receptor binding studies disclose a novel class
of high-affinity inhibitors of the Escherichia coli FimH adhesin.
Mol Microbiol. 55:441–455. 2005.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Zhang M, Wang G, Lai F and Wu H:
Structural characterization and immunomodulatory activity of a
novel polysaccharide from lepidium meyenii. J Agric Food Chem.
64:1921–1931. 2016.PubMed/NCBI View Article : Google Scholar
|