Introduction
Estrogens are important regulators of cell growth
and differentiation in a variety of tissues, including normal
mammary epithelium (1). In
addition, estrogens play an important role in breast cancer
promotion and progression (2).
This is based on observations that tumors regress in response to
oophorectomy or after hormone therapies, such as the anti-estrogen
tamoxifen or aromatase inhibitors (AI) that block estrogen
biosynthesis in post-menopausal women (3–5).
Estrogens affect cellular processes by binding to
their cognate receptors, estrogen receptor (ER)α and β, which
function as transcription factors mediating the mitogenic effects
of estrogen. ERα expression in normal breast epithelium is
generally low; however, significantly higher expression has been
reported in pre-malignant lesions (6), with the majority of breast tumors
expressing both receptors (7,8).
Since prolonged endogenous estrogen exposure is a potential risk
factor for invasive breast cancer (9), overexpression of ERα or the emergence
of mutated receptors could be early events in tumor progression
(10,11). Fuqua et al (12) identified an A to G somatic mutation
at ERα nucleotide 908 (A908G) from several usual ductal
hyperplasias (early pre-malignant lesions), resulting in a lysine
to arginine transition at residue 303 (K303R). To our knowledge, no
other ERα mutation has been identified in more than a few
invasive breast cancers (13–17).
The K303R ERα mutation has been found to
confer resistance to tamoxifen treatment (11,18).
Recently, this mutation was investigated in stably wild-type or
K303R ERα-overexpressing MCF-7 cells, transfected with an aromatase
expression vector, stimulated with androstenedione (an aromatase
substrate), with or without the AI anastrozole. Anastrozole
treatment decreased androstenedione-stimulated growth of the
wild-type cells, whereas K303R-expressing cells were resistant to
the inhibitory effect of the drug (19). These findings suggest that, since
K303R-mutant cells may escape from growth inhibition when treated
with AIs, genetic assays for the mutation might offer a novel
predictive marker for hormonal response (19).
The aim of this study was to verify whether the
presence of the K303R ERα mutation is associated with
response to 3-month neoadjuvant treatment with anastrozole
(Arimidex) in a cohort of post-menopausal breast cancer
patients.
Materials and methods
Patients
All tumor samples and clinical data were collected
with approval of the Fondazione ‘Salvatore Maugeri’ Ethics
Committee and with the informed consent of the patients. The 37
patients enrolled in this study between July 2004 to November 2007
were post-menopausal and had breast cancer stage T2 or T3, tumor
size >2.5 cm, variable lymph node status and no distant
metastasis. All of the tumors were HER2/neu− and
ER+/PgR+, apart from two that were
ER+/ PgR− (Table
I). The patients received neoadjuvant therapy with anastrozole
(Arimidex™; Astra Zeneca) 1 mg/day for 3 months. Clinical response
was evaluated by serial tumor clinical examination and mammary
ultrasound bidimensional measurements, performed by a single
operator (L.R.) before, during and after treatment. Patients with a
decrease in tumor volume ≥30%, according to RECIST criteria
(20), were classified as
responders.
 | Table I.Pathophysiological characteristics and
percentage of response to therapy of the studied patients. |
Table I.
Pathophysiological characteristics and
percentage of response to therapy of the studied patients.
| Patient | Age (years) | Initial size
(mm) | Tru-Cut
| Surgical sample
| Stage
| Lymph nodes
| % Response | ERα status |
|---|
| ER (%) | PgR (%) | ki67 (%) | Size (mm) | Histotype | Grading | ER (%) | PgR (%) | ki67 (%) | T | N | M | Positive no. | Total no. |
|---|
| 10050 | 80 | 35 | 60 | 80 | 8 | 37 | IDC | 2 | 80 | 40 | 5 | 2 | 1 | 0 | 1 | 13 | −6 | WT |
| 10055 | 80 | 27 | 60 | 70 | 10 | 25 | IDC | 2 | 80 | 40 | 8 | 2 | NV | 0 | NV | NV | 7 | WT |
| 10041 | 76 | 36 | 70 | 50 | 5 | 33 | IDC | 2 | 70 | 50 | 5 | 2 | 1 | 0 | 3 | 13 | 8 | WT |
| 10038 | 77 | 30 | 80 | 70 | 20 | 26 | ILC | 3 | 70 | 30 | 15 | 1C | 0 | 0 | 0 | 12 | 13 | WT |
| 10051 | 70 | 29 | 80 | 0 | 10 | 25 | ILC | 2 | 80 | 5 | 5 | 2 | 1 | 0 | 1 | 20 | 14 | WT |
| 10022 | 79 | 26 | 70 | 80 | 5 | 22 | IDC | 2 | 80 | 60 | 5 | 2 | NV | 0 | NV | NV | 15 | WT |
| 10027 | 78 | 45 | 80 | 30 | 40 | 38 | IDC | 3 | 80 | 30 | 40 | 2 | 0 | 0 | 0 | 6 | 16 | WT |
| 10054 | 52 | 37 | 80 | 80 | 8 | 30 | ILC | 2 | 60 | 40 | 40 | 1C | 1 | 0 | 1 | 14 | 19 | WT |
| 10052 | 80 | 25 | 80 | 80 | 8 | 20 | IDC | 2 | 80 | 60 | 5 | 1C | 1 | 0 | 1 | 7 | 20 | NV |
| 10024 | 75 | 31 | 80 | 80 | 5 | 24 | IDC | 2 | 80 | 60 | 5 | 1C | 0 | 0 | 0 | 3 | 23 | WT |
| 10003 | 84 | 30 | 90 | 40 | 5 | 23 | IDC | 2 | 90 | 40 | 5 | 2 | 1 | 0 | 2 | 6 | 23 | WT |
| 10018 | 74 | 30 | 80 | 60 | 8 | 23 | ILC | 2 | 80 | 20 | 10 | 2 | 1 | 0 | 2 | 15 | 23 | WT |
| 10000 | 70 | 53 | 70 | 40 | 10 | 40 | IDC | 3 | 70 | 20 | 8 | 3 | 1 | 0 | 14 | 14 | 25 | WT |
| 10028 | 73 | 26 | 80 | 70 | 5 | 19 | IDC | 1 | 80 | 60 | 5 | 2 | 1 | 0 | 2 | 10 | 27 | WT |
| 10013 | 71 | 25 | 80 | 70 | 5 | 18 | IDC | 2 | 80 | 70 | 5 | 1C | 0 | 0 | 0 | 6 | 28 | WT |
| 10029 | 61 | 25 | 80 | 70 | 5 | 18 | IDC | 2 | 80 | 70 | 8 | 1C | 1 | 0 | 6 | 16 | 28 | WT |
| 10064 | 83 | 25 | 80 | 80 | 5 | 18 | IDC | 1 | 80 | 40 | 5 | 1C | NV | 1 | NV | NV | 28 | NV |
| 10056 | 75 | 39 | 70 | 10 | 15 | 28 | IDC | 1 | 80 | 10 | 8 | 2 | NV | 0 | 0 | 22 | 28 | WT |
| 10004 | 82 | 25 | 80 | 80 | 5 | 17 | ILC | 2 | 80 | 80 | 5 | 2 | 0 | 0 | 0 | 4 | 32 | NV |
| 10040 | 72 | 28 | 80 | 80 | 5 | 19 | ILC | 2 | 80 | 70 | 5 | 1C | 0 | 0 | NV | NV | 32 | WT |
| 10026 | 76 | 30 | 80 | 80 | 10 | 20 | IDC | 2 | 90 | 70 | 10 | 2 | NV | 0 | NV | NV | 33 | WT |
| 10035 | 73 | 26 | 70 | 70 | 5 | 17 | IDC | 2 | 80 | 60 | 5 | 1C | 1 | 0 | 2 | 8 | 35 | WT |
| 10049 | 74 | 43 | 80 | 20 | 15 | 28 | ILC | 3 | 80 | 20 | 15 | 2 | 1 | 0 | 1 | 2 | 35 | NV |
| 10010 | 66 | 25 | 70 | 90 | 8 | 16 | ILC | 2 | 80 | 80 | 8 | 1C | 0 | 0 | 0 | 8 | 36 | WT |
| 10001 | 79 | 30 | 70 | 10 | 5 | 19 | ILC | 2 | 80 | 10 | 5 | 2 | 0 | 0 | 0 | 4 | 37 | WT |
| 10017 | 73 | 26 | 90 | 90 | 40 | 16 | IDC | 2 | 90 | 80 | 10 | 1C | 0 | 0 | 0 | 12 | 38 | WT |
| 10043 | 71 | 38 | 70 | 70 | 5 | 23 | ILC | 1 | 70 | 60 | 5 | 1C | 1 | 0 | 1 | 13 | 39 | WT |
| 10007 | 75 | 35 | 80 | 80 | 5 | 21 | ILC | 2 | 80 | 60 | 5 | 2 | NV | 0 | NV | NV | 40 | NV |
| 10048 | 58 | 35 | 90 | 5 | 30 | 21 | ILC | 2 | 90 | 5 | 15 | 2 | 1 | 0 | NV | NV | 40 | NV |
| 10032 | 72 | 32 | 80 | 80 | 5 | 19 | ILC | 2 | 80 | 50 | 5 | 2 | 0 | 0 | 0 | 1 | 41 | WT |
| 10039 | 85 | 27 | 80 | 80 | 10 | 16 | ILC | 3 | 80 | 70 | 5 | 1C | NV | 0 | NV | NV | 41 | WT |
| 10025 | 75 | 41 | 80 | 90 | 5 | 23 | IDC | 1 | 80 | 80 | 5 | 2 | 0 | 0 | 0 | 12 | 44 | WT |
| 10037 | 65 | 25 | 80 | 80 | 5 | 14 | IDC | 2 | 80 | 70 | 5 | 1C | 0 | 0 | 0 | 6 | 44 | WT |
| 10047 | 76 | 28 | 80 | 40 | 10 | 15 | ILC | 2 | 80 | 80 | 5 | 1C | 0 | 0 | 0 | 6 | 46 | WT |
| 9999 | 78 | 40 | 80 | 70 | 5 | 21 | IDC | 2 | 80 | 40 | 5 | 2 | 1 | 0 | 8 | 9 | 48 | WT |
| 10030 | 85 | 29 | 80 | 10 | 8 | 15 | IDC | 2 | 80 | 10 | 8 | 1C | 1 | 0 | 7 | 16 | 48 | WT |
| 10061 | 79 | 27 | 90 | 90 | 10 | 13 | ILC | 2 | 90 | 80 | 10 | 1C | NV | 0 | NV | NV | 52 | NV |
Sequence analysis
Total DNA was extracted from 37 formalin-fixed,
paraffin-embedded breast cancer biopsies using the High Pure PCR
Template Preparation kit (Roche Molecular Biochemicals, Basel,
Switzerland), according to the manufacturer's instructions.
SNaPshot primer extension sequencing was performed
as described by Herynk et al (11).
For dye-labeled terminator sequencing, all DNA
samples were amplified by PCR with primers for an ERα exon 4
fragment (216 bp), including the site of the mutation.
Oligonucleotide sequences were PCR-ERαF, 5′-GACCGAA
GAGGAGGGAGAAT-3′ and PCR-ERαR, 5′-GGAATAGAGT ATCGGGGGCT-3′. PCR was
carried out in a reaction volume of 25 μl, containing ∼100
ng of genomic DNA, 50 mM KCl, 10 mM Tris-Hcl (pH 8.3), 1.5 mM
MgCl2, 0.1 ng/μl bovine serum albumin (BSA), 200
μM dNTPs, 0.3 μM each primer and 0.1 U/μl
Taq polymerase. Amplification consisted of an initial
denaturation at 95°C for 5 min, followed by 35 cycles of 1 min at
95°C, 45 sec at 58°C and 1 min at 72°C, with a final extension at
72°C for 7 min.
PCR products were purified by Wizard SV Gel and the
PCR Clean-Up System (Promega, Madison, WI, USA) and sequenced using
the Big Dye Terminator V3.1 Cycle Sequencing kit and Prism Model
3730XL DNA Analyzer (Applied Biosystems, Foster City, CA, USA).
Results
Using the response criteria as described in
Materials and methods, 19 patients were classified as responders
and 18 as non-responders (Table
I). Only 1 patient (10050) showed disease progression during
the treatment.
K303R mutation analysis
As Herynk et al argued that SNaPshot primer
extension sequencing is more sensitive than dye-labeled terminator
sequencing for the detection of the ERα K303R mutation
(11), an identical approach on an
initial group of 10 DNA samples was attempted. Unfortunately,
interpretation of the SNaPshot primer extension sequencing results
was very difficult, as a very high background was present (data not
shown). We then decided to design a new forward primer for PCR
amplification, as the Forward PCR ERα 1 (described by Herynk et
al) was too near to the site of the mutation for a clear
sequencing. Thus, Reverse PCR ERα 2 was used (11). All DNAs were amplified by PCR and
sequenced as described in Materials and methods. Seven DNA samples
showed no suitable PCR product or had no results in sequencing.
Forward and reverse sequences were readable for 30 out of the 37
DNA samples, and all showed absence of the K303R mutation. An
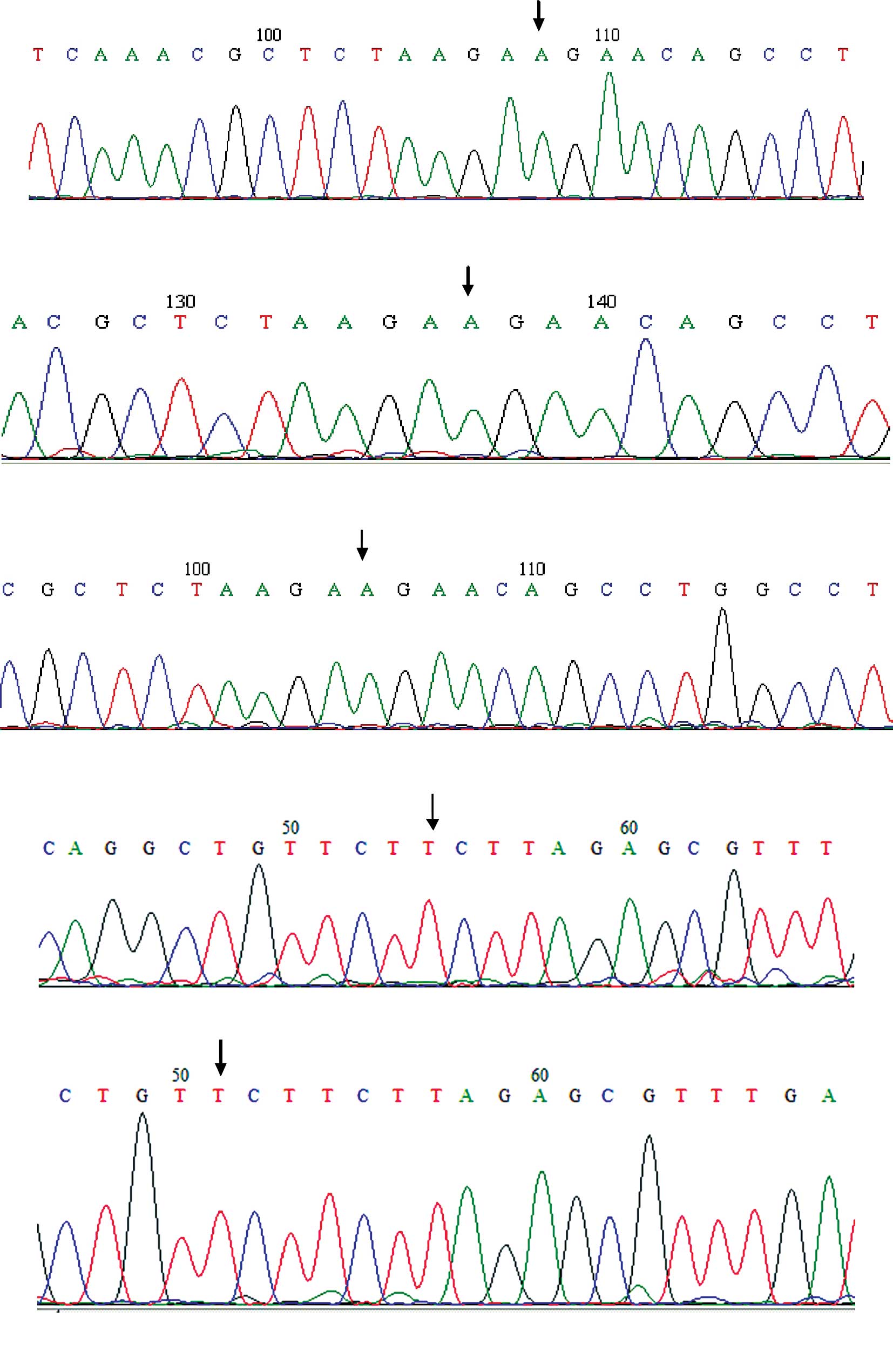
example of sequencing is shown in Fig.
1.
Discussion
A somatic mutation at the nucleotide 908 of
ERα (A908G) has been identified in pre-malignant breast
lesions and invasive breast cancers (11,12).
This mutation, resulting in a lysine to arginine transition at
residue 303 (K303R), confers hyper-sensitivity to estrogen and
resistance to tamoxifen (11,18,21).
Giordano et al hypothesized that the mutant K303R ERα
provides a proliferative advantage to breast tissue through a
continuous mitogenic stimulus, even during phases of low
circulating hormone (as menopause) and demonstrated that its
expression conferred resistance to the aromatase inhibitor
anastrozole in vitro (19).
The present study is the first to investigate K303R
ERα mutations in a cohort of post-menopausal breast cancer
patients treated with anastrozole neoadjuvant therapy. The purpose
of this study was to verify whether the K303R ERα mutation
is associated with response to treatment. Notably, no DNA extracted
from the post-treatment biopsies demonstrated the alteration,
neither in tumors responding to therapy, nor in non-responding
tumors.
The absence of the K303R ERα mutation in our
tumor cohort could be attributed to the relatively limited number
of samples. Nevertheless, this alteration was either not detected
in invasive cancer in four other studies (13–16),
or was found with a low frequency (37 of 653 breast tumors, 5.7%)
(17). Herynk et al argued
that dye-labeled terminator sequencing was not adequate for the
detection of the A908G ERα mutation, and that the alteration
was detected at a high frequency in invasive breast tumors using
only primer extension sequencing (SNaPshot) (11). We attempted the same approach on an
initial group of 10 DNA samples. Unfortunately, interpretation of
the results was very difficult, as a very high background was
present; therefore, dye-labeled terminator sequencing with a
different forward primer was used. With this approach, very clean
sequences and low background were obtained. Conway et al
attributed inferior sensibility to automated sequencing due to
suppression of peaks, particularly G following A (17). Thus, we used Big Dye terminators
and sequenced in the two senses, where the mutated sequence
transforms a T into a C. No mutation was able to be found.
An alternative explanation could be that the in
vivo K303R ERα mutation has biological significance in
only a subset of breast cancers, as hypothesized by Conway et
al (17). In that study, mixed
lobular/ductal tumors were more likely than ductal tumors to carry
the K303R ERα mutation; no tumor in our cohort showed
similar characteristics, as they were all lobular or ductal
carcinomas. Moreover, K303R ERα mutation-positive breast
cancer was significantly associated with longer duration and recent
use of oral contraceptives (OCs) and OC use has been more strongly
associated with the development of lobular and mixed lobular/ductal
breast tumors (22,23). In our cohort, all of our patients
were post-menopausal, and 31 out of the 37 (84%) were treated at an
age older than 70 years; in Italy, the use of OCs was introduced in
1971, so it is likely that many of our patients never practiced
their use.
Acknowledgements
We thank Dr Annalisa Lanza from
Fondazione Maugeri for sample storage and delivery.
References
|
1.
|
Chalbos D, Vignon F, Keydar I and
Rochefort H: Estrogens stimulate cell proliferation and induce
secretory proteins in a human breast cancer cell line (T47D). J
Clin Endocrinol Metab. 55:276–283. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Russo J and Russo IH: The role of estrogen
in the initiation of breast cancer. J Steroid Biochem Mol Biol.
102:89–96. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Beatson G: On the treatment of inoperable
cases of carcinoma of the mamma: suggestions for a new method of
treatment, with illustrative cases. Lancet. 147:101–107. 1986.
|
|
4.
|
Smith IE and Dowsett M: Aromatase
inhibitors in breast cancer. N Engl J Med. 348:2431–2442. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Osborne CK: Tamoxifen in the treatment of
breast cancer. N Engl J Med. 339:1609–1618. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Allred DC, Mohsin SK and Fuqua SA:
Histological and biological evolution of human premalignant breast
disease. Endocr Relat Cancer. 8:47–61. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Elledge RM and Fuqua SAW: Estrogen and
progesterone receptors. Diseases of the Breast. Harris JR, Lippman
ME, Morrow M, et al: Lippincott, Williams & Wilkins;
Philadelphia: pp. 471–488. 2000
|
|
8.
|
Hopp TA, Weiss HL, Parra IS, Cui Y,
Osborne CK and Fuqua SA: Low levels of estrogen receptor β protein
predict resistance to tamoxifen therapy in breast cancer. Clin
Cancer Res. 10:7490–7499. 2004.
|
|
9.
|
Pike MC, Spicer DV, Dahmoush L and Press
MF: Estrogens, progestins, normal breast cell proliferation, and
breast cancer risk. Epidemiol Rev. 15:17–35. 1993.PubMed/NCBI
|
|
10.
|
McGuire WL, Chamness GC and Fuqua SA:
Estrogen receptor variants in clinical breast cancer. Mol
Endocrinol. 5:1571–1577. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Herynk MH, Parra I, Cui Y, Beyer A, Wu MF,
Hilsenbeck SG and Fuqua SA: Association between the estrogen
receptor α A908G mutation and outcomes in invasive breast cancer.
Clin Cancer Res. 13:3235–3243. 2007.
|
|
12.
|
Fuqua SA, Wiltschke C, Zhang QX, Borg A,
Castles CG, Friedrichs WE, Hopp T, Hilsenbeck S, Mohsin S,
O'Connell P and Allred DC: A hypersensitive estrogen receptor-α
mutation in premalignant breast lesions. Cancer Res. 60:4026–4029.
2000.
|
|
13.
|
Tebbit CL, Bentley RC, Olson JA Jr and
Marks JR: Estrogen receptor α (ESR1) mutant A908G is not a common
feature in benign and malignant proliferations of the breast. Genes
Chromosomes Cancer. 40:51–54. 2004.
|
|
14.
|
Tokunaga E, Kimura Y and Maehara Y: No
hypersensitive estrogen receptor-α mutation (K303R) in Japanese
breast carcinomas. Breast Cancer Res Treat. 84:289–292. 2004.
|
|
15.
|
Davies MP, O'Neill PA, Innes H and Sibson
DR: Hypersensitive K303R oestrogen receptor-α variant not found in
invasive carcinomas. Breast Cancer Res. 7:R113–R118. 2005.
|
|
16.
|
Zhang Z, Yamashita H, Toyama T, Omoto Y,
Sugiura H, Hara Y, Haruki N, Kobayashi S and Iwase H: Estrogen
receptor α mutation (A-to-G transition at nucleotide 908) is not
found in different types of breast lesions from Japanese women.
Breast Cancer. 10:70–73. 2003.
|
|
17.
|
Conway K, Parrish E, Edmiston SN, Tolbert
D, Tse CK, Geradts J, Livasy CA, Singh H, Newman B and Millikan RC:
The estrogen receptor-α A908G (K303R) mutation occurs at a low
frequency in invasive breast tumors: results from a
population-based study. Breast Cancer Res. 7:R871–R880. 2005.
|
|
18.
|
Cui Y, Zhang M, Pestell R, Curran EM,
Welshons WV and Fuqua SA: Phosphorylation of estrogen receptor α
blocks its acetylation and regulates estrogen sensitivity. Cancer
Res. 64:9199–9208. 2004.
|
|
19.
|
Giordano C, Cui Y, Barone I, Ando S,
Mancini MA, Berno V and Fuqua SA: Growth factor-induced resistance
to tamoxifen is associated with a mutation of estrogen receptor
alpha and its phosphorylation at serine 305. Breast Cancer Res
Treat. 119:71–85. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Therasse P, Arbuck SG, Eisenhauer EA,
Wanders J, Kaplan RS, Rubinstein L, Verweij J, van Glabbeke M, van
Oosterom AT, Christian MC and Gwyther SG: New guidelines to
evaluate the response to treatment in solid tumors. European
Organization for Research and Treatment of Cancer, National Cancer
Institute of the United States, National Cancer Institute of
Canada. J Natl Cancer Inst. 92:205–216. 2000. View Article : Google Scholar
|
|
21.
|
Barone I, Cui Y, Herynk MH,
Corona-Rodriguez A, Giordano C, Selever J, Beyer A, Andò S and
Fuqua SA: Expression of the K303R estrogen receptor-α breast cancer
mutation induces resistance to an aromatase inhibitor via addiction
to the PI3K/Akt kinase pathway. Cancer Res. 69:4724–4732. 2009.
|
|
22.
|
Conway K, Parrish E, Edmiston SN, Tolbert
D and Tse CK: Risk factors for breast cancer characterized by the
estrogen receptor alpha A908G (K303R) mutation. Breast Cancer Res.
9:R362007. View
Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Li CI, Malone KE, Porter PL, Weiss NS and
Tang MT: Reproductive and anthropometric factors in relation to the
risk of lobular and ductal breast carcinoma among women 65–79 years
of age. Int J Cancer. 107:647–651. 2003.PubMed/NCBI
|