Introduction
Chronic prostatitis/chronic pelvic pain syndrome
(CP/CPPS) is a complex condition for which the etiological
determinants are still poorly defined. Its prevalence ranges
between 2.2 and 13.8%, depending on specific populations, countries
and surveys (1,2). CP/CPPS is characterized by a wide
array of symptoms, including pain in the pelvic region, irritative
and/or obstructive voiding symptoms, other pelvic disturbances,
sexual dysfunction and psycho-social maladjustment amongst others
(3).
Due to the fact that every patient with CP/CPPS
presents a unique, individual profile of symptoms defining the
syndrome, phenotypic characterization is required to profile the
clinical features of each single subject, and to direct symptomatic
therapy. For this reason, a novel diagnostic/therapeutic algorithm
known as UPOINT was created, addressing six major phenotypic
domains of CP/CPPS, specifically the urinary (U), psycho-social
(P), organ-specific (O), infection (I), neurological/systemic (N)
and muscular tenderness (T) domains. UPOINT, with or without an
additional domain addressing sexual dysfunction (‘UPOINTS’), has
been initially validated in two cohort studies, performed on
North-American (4) and European
patients (5).
The major finding of the first North-American
validation study was the correlation between the number of UPOINT
domains and the total score of the National Institutes of
Health-Chronic Prostatitis Symptom Index (NIH-CPSI) measured in
each patient (4). Whereas a strong
correlation was also observed in the European cohort (5), certain discrepancies between the two
studies were evident. In the study by Shoskes et al, the
severity of the symptoms in the patients with a positive infection
domain was not significantly different from the symptom scores of
patients without evidence of prostatic infection (4). Conversely, in the study by Magri
et al, patients with a positive infection domain showed
higher symptom scores, measured using the NIH-CPSI (5). This suggested the existence of
different populations of patients, exhibiting different degrees of
symptom severity, depending on the presence or absence of
infection. Recently, we have investigated the distribution of
NIH-CPSI symptom scores in a large cohort of patients from
different countries globally (6).
Graphic representations of the total NIH-CPSI symptom score
distributions showed irregular patterns (6), possibly due to multiple overlapping
peaks, which suggested the existence of different subpopulations of
patients showing various degrees of symptom severity.
In the present study, we report the results of a
retrospective analysis of a population of 935 Italian patients who
had been included in the large international studies mentioned
previously (5,6). The aim of the present study was to
investigate in greater detail the extent to which a positive
UPOINTS infection domain may modify the distribution patterns of
NIH-CPSI symptom scores in this patient population.
Patients and methods
Clinical database
The present retrospective observational study was
performed on a database of 935 Italian patients, representing a
subset of a total population of 1,563 subjects from Europe and
North America enrolled in two recent, ethically approved
international studies investigating NIH-CPSI and the UPOINT(S)
system (5,6). This subset of patients was diagnosed
and treated by one urologist (VM) in a single specialized, second
referral prostatitis center in Italy (Urology and Urological
Sonography Secondary Care Clinic, Azienda Ospedaliera Istituti
Clinici di Perfezionamento, Milan). Patients provided written
consent for the handling and anonymous publication of their
data.
The database used for this subset analysis included
demographic data, diagnosis [CP/CPPS, category IIIa (inflammatory)
versus IIIb (non-inflammatory)], UPOINT phenotypes and the scores
of the NIH-CPSI questionnaire.
Inclusion criteria and diagnostic
procedures
CP/CPPS was diagnosed according to the NIH criteria
(7). Inclusion/exclusion criteria
for the study, as well as the items by which patients were
allocated as positive for the individual UPOINT domains, have been
described in the main study report (5). Exclusion criteria included a
diagnosis of acute or chronic bacterial prostatitis (CBP; NIH
categories I or II, NIH criteria) (7).
The presence of bacteria or pathogens in the
prostate was assessed with the ‘4-glass’ lower urinary tract
segmented test according to Meares and Stamey, based on the
collection and culture of i) first-voided (VB1), ii) midstream
(VB2) and iii) post-massage voided urine (VB3) specimens, and iv)
of expressed prostatic secretions (EPS) collected during prostatic
massage (8). A one-log ratio
between bacterial counts in VB3 versus VB2 or VB1 was the cutoff
value defining “prostatic infection (8) and a positive UPOINTS infection
domain.
CP/CPPS symptoms were evaluated by asking the
patients to complete the NIH-CPSI questionnaire, an internationally
validated clinical tool measuring disease symptoms and
characteristics (9). The NIH-CPSI
measures combined aspects of the three most important symptom
domains of CP/CPPS, with higher scores indicating higher disease
severity. These symptom domains include pain (location, frequency
and severity; score range, 0 to 21), voiding problems (irritative
and obstructive symptoms; score range, 0 to 10), and impact of the
disease on the quality of life of patients (QoL; score range, 0 to
12), with a total score ranging from 0 to 43.
Statistics
χ2 analysis was used to evaluate
differences in the proportions of patients with and without a
positive infection domain for each score of the NIH-CPSI
questionnaire (total score, and pain, void and QoL sub-scores).
The median was used as a measure of central tendency
for the scores and sub-scores of the NIH-CPSI. The upper and lower
quartiles were calculated to evaluate the dispersion of the data.
The Mann-Whitney-Wilcoxon test was used to analyze unpaired
differences in NIH-CPSI scores between patient groups.
Due to its large sample size, the present study was
adequately powered (1−β=90%) to detect clinically relevant changes
(6 points) in the total scores of the NIH-CPSI, with a 5% α error
probability. The results of all statistical tests were considered
significant in the presence of an α error <0.05. Data were
analyzed with the XLStatistics 5.71 program (©Rodney
Carr; http://www.deakin.edu.au/~rodneyc/).
Results
NIH-CPSI scores and distribution patterns
in patients with or without evidence of infection
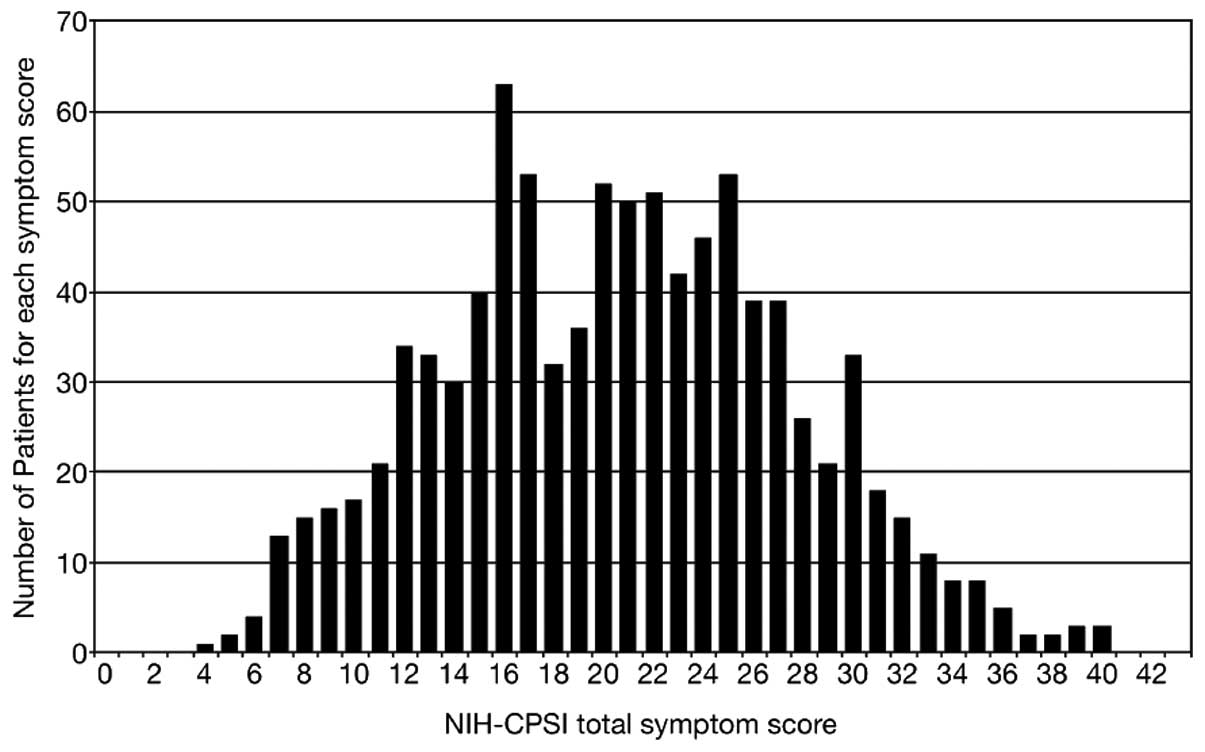
Fig. 1 shows the
distribution pattern of total NIH-CPSI scores in the total CP/CPPS
patient population. The distribution appears to be non-unimodal and
slightly right-skewed (moment coefficient of skewness, 0.11:
‘approximately symmetric’ by definition). Table I shows the median values of the
NIH-CPSI total score and symptom domain subscores, and the
statistical analysis of the differences between cohorts of patients
with or without evidence of prostatic infection (i.e. a positive or
negative UPOINTS infection domain, respectively). The pain and QoL
subdomain scores, and the total score of the NIH-CPSI were
significantly different between these cohorts. The voiding symptom
scores of the NIH-CPSI did not differ between cohorts.
 | Table I.Median values of the NIH-CPSI total
score and symptom domain subscores, and statistical analysis of
differences between cohorts of patients with category III CP/CPPS
with or without evidence of prostatic infection, defined according
to NIH criteria (7). |
Table I.
Median values of the NIH-CPSI total
score and symptom domain subscores, and statistical analysis of
differences between cohorts of patients with category III CP/CPPS
with or without evidence of prostatic infection, defined according
to NIH criteria (7).
| NIH-CPSI score | Total patient
population
| Patients with
evidence of infection
| Patients without
evidence of infection
|
|---|
| Median | Upper and lower
quartiles | Median | Upper and lower
quartiles | Median | Upper and lower
quartiles |
|---|
| 1 - pain domain | 9 | 7–12 | 11 | 8–14 | 9a | 7–11 |
| 2 - voiding symptom
domain | 4 | 2–6 | 4 | 2–7 | 4b | 2–6 |
| 3 - QoL impact
domain | 8 | 5–9 | 8 | 6–10 | 7a | 5–9 |
| Total score
(1+2+3) | 21 | 16–26 | 24 | 18–29 | 20c | 15–24 |
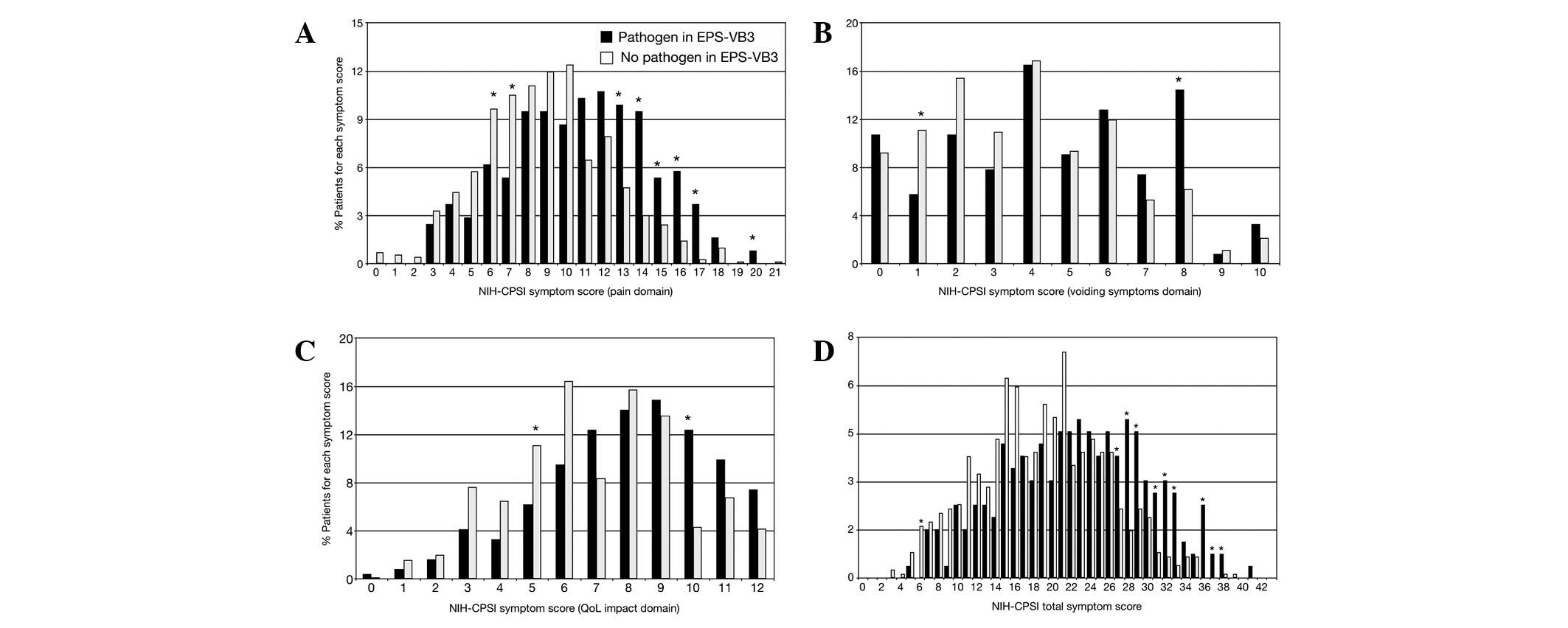
Fig. 2 shows the
distribution patterns of NIH-CPSI scores in patients with a
positive or negative infection domain of the UPOINTS phenotyping
system. In the graph showing the pain domain (Fig. 2A), the distribution pattern of
patients with a positive infection domain is shifted to the right,
i.e. toward more severe pain symptom scores. Significantly higher
proportions of patients with infection showed symptom scores as
high as 13–17, compared with patients showing no evidence of
bacterial infection in the prostate.
Since the pain domain is the component of greater
weight in the NIH-CPSI scoring system, the previously described
difference between ‘infected’ versus ‘non-infected’ patients is
reflected in the distribution of the NIH-CPSI total scores, as
shown in Fig. 2D. At symptom
scores >27, patients with a positive infection domain were
predominantly present in significantly higher proportions, compared
with subjects without evidence of infection. These differences were
less evident in the two other sections of the NIH-CPSI test, i.e.
the voiding and QoL domains. However, significantly higher numbers
of infected patients were still observed at higher scores in these
two domains (Fig. 2B and C).
Impact of infection on NIH-CPSI scores in
phenotypic clusters of CP/CPPS patient
Since the distribution of data shown in Fig. 2 indicated the existence of two
distinct populations with differing symptom scores, and due to the
fact that infection was the discriminant element in this respect,
we investigated whether it was also possible to observe these
differences in the CP/CPPS phenotype clusters reported by Davis
et al in their recent UPOINT(S) cluster analysis (10). In this previous study, it was shown
that patients with CP/CPPS tended to segregate into six different
clusters, each containing patients showing high frequencies of
specific UPOINTS domains (10).
These clusters were denominated: i) prostate infection-related
sexual dysfunction (high frequencies of O, I and S domains); ii)
widespread symptoms (high frequencies of U, O and N domains); iii)
psychosocially complex lower urinary tract symptoms (LUTS) with
sexual dysfunction (high frequencies of P and S domains); iv)
psychosocially complex LUTS without sexual dysfunction (high
frequencies of U and P domains); v) muscle
tenderness/fibromyalgia-like symptoms (high frequency of T domain)
and vi) uncomplicated LUTS (increased frequencies of U and O
domains). The infection domain of the UPOINT system was present at
very high frequency (80% of patients) only in the first
cluster.
NIH-CPSI scores were calculated subsequent to the
classification of the present patient population according to the
clusters described by Davis et al (10). As shown in Table II, patients belonging to the
‘prostate infection-related sexual dysfunction’ cluster showed
significantly higher pain, voiding symptom, QoL and total scores of
the NIH-CPSI, compared with all other clusters. Table II also shows that the scores of a
modified group, i.e. the ‘prostate infection-related sexual
dysfunction’ cluster lacking all patients with evidence of
infection, were significantly lower than the ones shown by the
original cluster, and were virtually equivalent to those shown by
the other clusters, which were also lacking high proportions of
infected patients.
 | Table II.Median values (and interquartile
range) of the NIH-CPSI total score and symptom domain subscores,
and statistical analysis of differences between cohorts of patients
with category III CP/CPPS divided by cluster according to Davis
et al (10). Median scores
of group ‘1*’, a subgroup of cluster 1 devoid of
patients showing evidence of infection, defined according to NIH
criteria (7), are also
presented. |
Table II.
Median values (and interquartile
range) of the NIH-CPSI total score and symptom domain subscores,
and statistical analysis of differences between cohorts of patients
with category III CP/CPPS divided by cluster according to Davis
et al (10). Median scores
of group ‘1*’, a subgroup of cluster 1 devoid of
patients showing evidence of infection, defined according to NIH
criteria (7), are also
presented.
| NIH-CPSI score | Cluster
classification
|
|---|
| 1 | 1* | 2 | 3 | 4 | 5 | 6 |
|---|
| 1 - pain domain | 12 (5) | 9a (4) | 9a (5) | 9a (5) | 10a (4) | 10a (4) | 9a (5) |
| 2 - voiding symptom
domain | 6 (3.75) | 5a (2.00) | 4a (4.00) | 4a (4.00) | 6b (2.00) | 4a (4.00) | 5a (4.00) |
| 3 - QoL impact
domain | 9 (4) | 8a (3) | 8a (3) | 8a (3) | 9b (4) | 8a (4) | 8a (3) |
| Total score
(1+2+3) | 28 (9.75) | 22a (9.00) | 21a (10.00) | 22a (9.00) | 24a (7.50) | 22a (10.00) | 22a (9.00) |
Discussion
Category III CP/CPPS, and its inflammatory and
non-inflammatory (categories IIIa and IIIb, respectively) variants,
have been originally defined, diagnosed and investigated as a
chronic prostatitis syndrome involving pelvic pain and voiding
symptoms in the presence of sterile microbiological cultures of
expressed prostatic secretions and/or urine specimens following
prostatic massage (11). Over
time, this original definition has evolved, and experts have
proposed a model whereby the prostate of patients with CP/CPPS may
be colonized by a variety of organisms, including either
traditional uropathogens (such as E.coli, Klebsiella
spp., other Enterobacteriaceae and Enterococci),
viz the etiological agents of acute and chronic bacterial
prostatitis, or bacteria causing disease in other distant or
adjacent organs (including coagulase-negative Staphylococci,
Streptococci, Chlamydia trachomatis and
Mycoplasmata) that have not yet been unanimously classified
as prostatic pathogens (12). This
model, also based on the results of a study showing similar
infection frequencies in symptomatic versus asymptomatic patients,
has led to a tentative definition of ‘bacterial CP/CPPS’ (13: see
reply to published letter).
According to this theory, these bacteria do not act
as determinants of pathogenicity in category III CP/CPPS. Instead,
they represent ‘bystander flora’, whose pharmacological eradication
would not be likely to result in symptom remission. Using this
model, a diagnosis of category II CBP is only possible in cases of
a documented history of relapsing episodes of urinary tract
infections (UTIs) originating from an infected prostate. This
concept has led to the inclusion of an infection domain into the
UPOINT algorithm, a system recently proposed to improve the
phenotypic profiling of patients with CP/CPPS (4).
However, this concept has not been unanimously
applied in clinical practice, and other clinicians and scientists
have maintained that the presence of pathogens in the prostatic
secretions of chronically symptomatic patients may be etiologically
linked to the clinical manifestation of category II CBP, even in
the absence of a long, documented history of repeated flare-ups of
the infection (14,15). According to this view, a
symptomatic episode of clinical chronic prostatitis with evidence
of uropathogens in the prostate may represent the initial phase of
a history of CBP, and there is therefore a requirement for it to be
diagnosed and treated accordingly. Antibacterial treatment of such
patients has been associated with symptom remission in numerous
cases (14,15). For clarity, it is necessary to
state that in the present study CP/CPPS was diagnosed according to
the ‘bystander flora’ hypothesis, and patients not showing a
documented history of repeated episodes of UTIs stemming from a
chronically infected prostate were classified as being affected by
category III CP/CPPS.
In the first validation study by the authors who
proposed the new UPOINT system, there was a failure to demonstrate
the influence of pathogens cultured in prostate-specific specimens
on CP/CPPS clinical symptoms, as assessed using the NIH-CPSI
questionnaire (4). By contrast,
data obtained by our research group suggested that the presence of
microorganisms may influence the severity of clinical symptoms of
CP/CPPS. In patients showing evidence of infection at the prostatic
level, the mean total NIH-CPSI symptom scores were significantly
increased by ∼20% (5), when
compared with scores from uninfected patients. This was
demonstrated in a cohort of 937 individuals, predominantly second
referral Italian patients; however, it was not observed in second
referral German patients (5).
Notably, the difference observed in the second referral patients
has been recently demonstrated by a prospective study performed on
a cohort of ∼400 Chinese patients, further validating the modified
UPOINTS system (16). In addition,
the mean total NIH-CPSI symptom scores in the Chinese cohort were
increased by 20–25% in patients with a positive infection domain of
the UPOINTS system, including a sexual dysfunction domain (16).
Since this evidence suggests that the presence of
pathogens is correlated with the deterioration of CP/CPPS symptoms,
the present subset study analyzed in greater detail the symptom
profile of a cohort of patients with CP/CPPS who had previously
been included in two studies aimed at investigating the NIH-CPSI in
patients with CP/CPPS, and at validating the UPOINT algorithm
(5,6).
The patients were divided into two groups, depending
on whether the prostate-specific specimens of patients symptomatic
for CP/CPPS were positive or negative for cultures. The median
NIH-CPSI scores, as well as pain and QoL subdomain scores, were
significantly higher in the patients with a positive UPOINT
infection domain, compared with patients with no evidence of
infection (Table I). An exception
was represented by the voiding score of NIH-CPSI, which is known to
be the least responsive among all NIH-CPSI domains.
Despite the large size of the present patient
population, which made the clinical relevance of the statistical
significance of these differences debatable, the statistically
significant, 4-point difference observed between the ‘infection’
and the ‘non-infection’ groups (median scores, 24 versus 20 points;
Table I) corresponded to the
commonly accepted minimum value of a clinically appreciable
difference in total NIH-CPSI scores (17).
Notably, the analysis of symptom score distributions
resulted in a right-shift of the curve representing pain symptoms
and total NIH-CPSI scores in patients with evidence of prostatic
infection (Fig. 2A and D). This
evidence suggested the existence of populations showing different
degrees of symptom severity, depending on the presence/absence of a
concomitant prostatic infection.
Data from of a historical cohort of 157 Hellenic
patients from our research group supported this finding: Patients
showing the presence of pathogens in prostate-specific specimens
(assessed with the Meares and Stamey segmented test) had higher
NIH-CPSI symptom scores, compared with patients without evidence of
infection (NIH-CPSI total score median difference: 7 points;
P<0.0001, Mann-Whitney-Wilcoxon test; analysis not shown).
Subsequently, it was investigated whether it was
possible to reproduce differential symptom scores in the two
populations (infected versus non-infected) when the patient
population was divided according to the six phenotypic clusters of
patients with CP/CPPS, as described by Davis et al (10). The ‘prostate infection-related
sexual dysfunction’ cluster, including the highest proportion of
patients with evidence of infection (80%), scored the highest
number of points of the NIH-CPSI among all the clusters. To assess
the influence of the infection domain on the severity of the
symptoms exhibited by the patients, all subjects with evidence of
infection were withdrawn from the prostate ‘infection-related
sexual dysfunction’ cluster. This modified cluster showed symptom
scores significantly less severe than those of the original
cluster. Furthermore, the scores were comparable with those of the
five other clusters.
Another recent study investigated the separation of
patients with CP/CPPS into distinct phenotype clusters (18). According to this study, it was
possible to divide the patients into two major clusters. One
cluster included patients with increased frequencies of U, O and T
domains, while the other showed a high frequency of P, I and N
domains of UPOINT [notably, the ‘UOT’ cluster in our study cohort
represented the most prominent group of patients (data not shown)].
Separation of the present patient population into the ‘UOT’ and
‘PIN’ clusters resulted in significantly higher NIH-CPSI symptom
scores in the latter cluster (median total NIH-CPSI scores: 29
versus 25 in the ‘UOT’ cluster; P<0.05, Mann-Whitney Wilcoxon
test), which contained higher proportions of subjects showing
evidence of infection (detailed analysis not shown). Furthermore,
when patients with evidence of infection were withdrawn from the
cluster, NIH-CPSI scores decreased significantly [median total
NIH-CPSI score: 24 (detailed analysis not shown)].
In combination, the results of the present study
strengthened the hypothesis that the presence of pathogens in the
prostate gland may exert a significant effect on the clinical
presentation of patients affected by CP/CPPS, and that the UPOINTS
infection domain represents an important determinant of the
severity of CP/CPPS symptoms in clusters of patients characterized
by a positive infection domain of the UPOINTS system. This evidence
implies that, if pathogens are partly responsible for a
deterioration of symptoms in CP/CPPS, antibacterial agents may
represent an important therapeutic tool for the treatment of
patients showing evidence of prostate colonization by uropathogens
or by other suspiciously pathogenic bacteria. A retrospective
analysis is in progress and a prospective study is being planned to
assess the efficacy of antibacterial therapy in this specific
category of patients.
References
|
1.
|
Bartoletti R, Cai T, Mondaini N, et al:
Prevalence, incidence estimation, risk factors and characterization
of chronic prostatitis/chronic pelvic pain syndrome in urological
hospital outpatients in Italy: results of a multicenter
case-control observational study. J Epidemiology and evaluation of
chronic pelvic pain syndrome in men Urol. 178:2411–2415. 2007.
|
|
2.
|
Schaeffer AJ: Epidemiology and evaluation
of chronic pelvic pain syndrome in men. Int J Antimicrob Agents.
31(Suppl 1): S108–S111. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Habermacher GM, Chason JT and Schaeffer
AJ: Prostatitis/chronic pelvic pain syndrome. Annu Rev Med.
57:195–206. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Shoskes DA, Nickel JC, Dolinga R and Prots
D: Clinical phenotyping of patients with chronic
prostatitis/chronic pelvic pain syndrome and correlation with
symptom severity. Urology. 73:538–542. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5.
|
Magri V, Wagenlehner F, Perletti G,
Schneider S, Marras E, Naber KG and Weidner W: Use of the UPOINT
chronic prostatitis/chronic pelvic pain syndrome classification in
European patient cohorts: sexual function domain improves
correlations. J Urol. 184:2339–2345. 2010. View Article : Google Scholar
|
|
6.
|
Wagenlehner FM, van Till JW, Magri V,
Perletti G, Houbiers JG, Weidner W and Nickel JC: National
Institutes of Health Chronic Prostatitis Symptom Index (NIH-CPSI)
symptom evaluation in multinational cohorts of patients with
chronic prostatitis/chronic pelvic pain syndrome. Eur Urol.
63:953–959. 2013. View Article : Google Scholar
|
|
7.
|
Krieger JN, Nyberg L Jr and Nickel JC: NIH
consensus definition and classification of prostatitis. JAMA.
282:236–237. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Stamey TA: Prostatitis. J R Soc Med.
74:22–40. 1981.
|
|
9.
|
Litwin MS, McNaughton-Collins M, Fowler
FJ, et al: The National Institutes of Health chronic prostatitis
symptom index: development and validation of a new outcome measure.
Chronic Prostatitis Collaborative Research Network. J Urol.
162:369–75. 1999. View Article : Google Scholar
|
|
10.
|
Davis SN, Binik YM, Amsel R and Carrier S:
Is a sexual dysfunction domain important for quality of life in men
with urological chronic pelvic pain syndrome? Signs “UPOINT” to
yes. J Urol. 189:146–151. 2013.PubMed/NCBI
|
|
11.
|
Nickel JC, Downey J, Johnston B and Clark
J; Canadian Prostatitis Research Group: Predictors of patient
response to antibiotic therapy for the chronic prostatitis/chronic
pelvic pain syndrome: a prospective multicenter clinical trial. J
Urol. 165:1539–1544. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Nickel JC, Alexander RB, Schaeffer AJ,
Landis JR, Knauss JS and Propert KJ; Chronic Prostatitis
Collaborative Research Network Study Group: Leukocytes and bacteria
in men with chronic prostatitis/chronic pelvic pain syndrome
compared to asymptomatic controls. J Urol. 170:818–822. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Magri V and Perletti G: Re: How does the
pre-massage and post-massage 2-glass test compare to the
Meares-Stamey 4-glass test in men with chronic prostatitis/chronic
pelvic pain syndrome? J Urol. 180:1571–1572. 2008. View Article : Google Scholar
|
|
14.
|
Magri V, Trinchieri A, Ceriani I, Marras E
and Perletti G: Eradication of unusual pathogens by combination
pharmacological therapy is paralleled by improvement of signs and
symptoms of chronic prostatitis syndrome. Arch Ital Urol Androl.
79:93–98. 2007.PubMed/NCBI
|
|
15.
|
Magri V, Montanari E, Škerk V, Markotić A,
Marras E, Restelli A, Naber KG and Perletti G:
Fluoroquinolone-macrolide combination therapy for chronic bacterial
prostatitis: retrospective analysis of pathogen eradication rates,
inflammatory findings and sexual dysfunction. Asian J Androl.
13:819–827. 2011. View Article : Google Scholar
|
|
16.
|
Zhao Z, Zhang J, He J and Zeng G: Clinical
utility of the UPOINT phenotype system in Chinese males with
chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS): a
prospective study. PLoS One. 8:e520442013. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Nickel JC, Krieger JN, McNaughton-Collins
M, et al: Alfuzosin and symptoms of chronic prostatitis-chronic
pelvic pain syndrome. N Engl J Med. 359:2663–2673. 2008. View Article : Google Scholar
|
|
18.
|
Samplaski MK, Li J and Shoskes DA:
Clustering of UPOINT domains and subdomains in men with chronic
prostatitis/chronic pelvic pain syndrome and contribution to
symptom severity. J Urol. 188:1788–1793. 2012. View Article : Google Scholar : PubMed/NCBI
|