Introduction
Colon cancer is one of the most common
gastrointestinal sarcomas. The incidence of colon cancer in North
America and Western Europe is higher than that in developing
countries, but there is also a trend to increasing incidences in
developing countries (1,2). The etiology of colon cancer remains
unclear, but is believed to be closely associated with
environmental factors, genetic factors and pre-cancerous diseases.
The survival rate of patients with colon cancer is closely
associated with the stage of the disease, early diagnosis and
treatment, which would also contribute to improvement in survival
and prognostic evaluation. The treatment of colon cancer includes
surgery, chemotherapy, radiotherapy, immunotherapy and biotherapy.
Due to its molecular and cellular diversification, the individual
treatment of colon cancer has attracted much attention. In
addition, >25% of colon cancer cases occur in patients with a
family history of colon cancer. Prevention, monitoring or
preventive treatment of individuals at high risk has become
extremely important (3,4). The discovery of new colon cancer
tumor markers and the mechanism underlying their modality may
provide new targets for diagnosis and treatment.
The expression level and impact of brain-derived
neurotrophic factor (BDNF) has been reported in several types of
carcinomas, although how this protein exerts its effects remains to
be identified (5). BDNF is a
protein composed of 247 amino acids, which was initially separated
and purified from pig brain (6).
It is a member of the nerve growth factor family and is secreted by
the target cells for BDNF, being important to the phenotypic
behavior of neurons or neural system cancers. BDNF mediates
cellular biological effects mainly through a cell surface tyrosine
kinase receptor, tropo-myosin-related kinase B (TrkB). The
extracellular N-terminal binds to BDNF, followed by receptor
autophosphorylation to activate signaling pathways in cells
(7).
In previous years it has been shown that BDNF
expression is associated with different functions in non-neuronal
solid tumors. BDNF expression controls cellular proliferation and
survival, and is also connected to cell invasion via the secretion
of matrix metalloproteinase (8–12).
The PI3K/AKT and ERK pathways in tumor cells activated by BDNF may
result in cells that are not sensitive to chemotherapeutic drugs
(13,14).
As previously reported, screening of clinical
samples has revealed that BDNF is highly expressed in colon
carcinoma compared with non-tumor tissues (5). The present study aimed to investigate
the biological roles of BDNF expression in human colon cancer. We
investigated the expression level and the impact of BDNF in
clinical samples. We observed high expression of BDNF in Caco-2,
HRT18 and RKO human colon cancer cells and therefore anti-BDNF
ribozymes were constructed to knock down the expression of BDNF in
these cell lines. We then investigated the effects of BDNF
knockdown on cellular behavior.
Materials and methods
Patient samples
Colorectal cancer tissues (n=66) and normal
background tissues (n=88) were collected immediately after surgery
and stored in a deep freeze until use. The presence of tumor cells
in the collection tissues was verified by a consultant pathologist
following hematoxylin and eosin (H&E) staining of the frozen
sections. Details of the histology were obtained from pathology
reports and together with basic patient demographics are shown in
Tables I and II. The anonymous breast tissue samples
were obtained within the guidelines of the appropriate ethics
committee [University Hospital of Wales Trust (05/DMD/3562)].
informed patient consent was obtained following guidelines set out
by the Human Tissue Act 2004, UK (http://www.legislation.gov.uk/ukpga/2004/30/contents).
 | Table IMedian values of BDNF expression in
patient samples. |
Table I
Median values of BDNF expression in
patient samples.
| Clinical/pathological
features | Sample (No.) | Median | Q1–Q3 | P-value |
|---|
| Normal | 68 | 0 | 0–7 | |
| Tumor | 88 | 25.5 | 2–438 | <0.0001 |
| Paired N | 58 | 0 | 0–9.4 | |
| Paired T | 64 | 13 | 1–261 | 0 |
| TNM |
| Normal | 68 | 0 | 0–7 | |
| TNM1 | 9 | 163.4 | 39–1545 | 0.0001 |
| TNM2 | 27 | 11 | 1–458 | <0.0001 |
| TNM3 | 25 | 7 | 1–175 | 0.0006 |
| TNM4 | 6 | 244 | 1–939 | 0.0095 |
| TNM2&3 | 52 | 10 | 1–246 | 0.0934 |
| T1234 |
| Normal | 68 | 0 | 0–7 | |
| T-1 | 2 | 1491 | NC | 0.1435 |
| T2 | 9 | 451 | 66–9953 | <0.0001 |
| T3 | 37 | 5 | 0–107 | 0.0001 |
| T4 | 18 | 53 | 6–1058 | <0.0001 |
| T2 | 9 | 451 | 66–9953 | |
| T3 | 37 | 5 | 0–107 | 0.0033 |
| T2 | 9 | 451 | 66–9953 | |
| T3&4 | 55 | 11 | 1–261 | 0.0086 |
| Duke |
| Normal | 68 | 0 | 0–7 | |
| Dukes A | 7 | 74 | 20–451 | 0.0012 |
| Dukes B | 30 | 13 | 2–610 | <0.0001 |
| Dukes C | 31 | 11 | 1–458 | 0.0001 |
| Dukes BC | 61 | 11 | 1–481 | <0.0001 |
| Alive | 33 | 7 | 0–175 | |
| Deceased | 22 | 82 | 7–744 | 0.0554 |
 | Table IIMean values of BDNF from patient
samples. |
Table II
Mean values of BDNF from patient
samples.
| Clinical/pathological
features | Sample (No.) | Mean | SE Mean | P-value |
|---|
| Normal | 68 | 212 | 171 | |
| Tumor | 88 | 2797 | 1003 | 0.013 |
| Paired N | 58 | 50 | 26 | |
| Paired T | 64 | 2224 | 887 | 0.017 |
| Differentation |
| Normal | 68 | 212 | 171 | |
| Diff 1 | 2 | 47.1 | 27 | 0.340 |
| Diff 2 | 50 | 1918 | 785 | 0.039 |
| Diff 3 | 14 | 7574 | 5023 | 0.170 |
| Diff 2&3 | 64 | 3155 | 1265 | 0.024 |
| T1234 |
| Normal | 68 | 212 | 171 | |
| T-1 | 2 | 1491 | 1491 | 0.550 |
| T2 | 9 | 9594 | 7107 | 0.220 |
| T3 | 37 | 2601 | 1307 | 0.078 |
| T4 | 18 | 914 | 499 | 0.200 |
| T2&3 | 46 | 3969 | 1740 | 0.037 |
| T3&4 | 55 | 2049 | 896 | 0.049 |
|
T2&3&4 | 64 | 3110 | 1266 | 0.027 |
| Normal | 68 | 212 | 171 | |
| No Invasion | 48 | 1770 | 931 | 0.110 |
| Invasive | 24 | 4989 | 2822 | 0.100 |
| Disease free | 32 | 4757 | 2406 | 0.069 |
| Incidence | 23 | 1874 | 1021 | 0.120 |
| Alive | 33 | 3728 | 2233 | 0.130 |
| Deceased | 22 | 3219 | 1559 | 0.069 |
RNA preparation and reverse transcription
PCR (RT-PCR)
Total cellular RNA was extracted from the cultured
cells using Total RNA Isolation reagent (ABgene, Epsom, UK). The
concentration of RNA was determined using an ultraviolet
spectrophotometer (WPA UV 1101; Biotech Photometer, Cambridge, UK).
cDNA was obtained by RT-PCR using a transcription kit (Sigma,
Poole, UK). The quality of DNA was verified using GAPDH primers.
The mRNA levels of BDNF were assessed using the BDNF primers. PCR
were run on a GeneAmp PCR System 2400 thermocycler (Perkin-Elmer,
Waltham, MA, USA). The PCR products were separated by 1% agarose
gel, stained with ethidium bromide and images were captured with a
digital camera mounted over a UV transluminator.
Quantitative PCR (qPCR)
The mRNA level of BDNF gene expression was
determined by the qPCR method using the prepared cDNA as the
template and BDNF primers. An additional primer sequence was added
to every qPCR system, known as the Z sequence (5′-ACT GAA CCT GAC
CGT ACA-3′), which is complementary to the universal Z probe
(Intergen Inc., Oxford, UK). The reaction was carried out in an
IcyclerIQ™ (Bio-Rad, Hemel Hemstead, UK) using 96-well plates.
GAPDH expression was used as an internal control. The reaction
conditions were as follows: 94°C for 7 min, 80 cycles of 94°C for
15 sec, 55°C for 35 sec (the data capture step) and 72°C for 20
sec. The levels of the transcripts were generated from an internal
standard that was simultaneously amplified with the samples.
Immunohistochemistry of clinical
samples
Cryostat sections of frozen tissues were cut at 6
μm, placed on Super Frost Plus slides (LSL UK, Rochdale, UK), air
dried and fixed in a 50:50 solution of alcohol:acetone. The
sections were then air dried again and stored at −20°C until used.
Immediately prior to immunostaining, the sections were washed in
buffered saline solution (BSS) for 5 min and treated with horse
serum (Sigma-Aldrich Co. Ltd., Gillingham, UK) for 20 min as a
blocking agent to non-specific binding. The sections were stained
using BDNF antibody (Santa Cruz Biotechnology, Inc., Santa Cruz,
CA, USA). Negative controls were used where necessary. The primary
antibody was used at 1:100 dilution for 60 min and the sections
were washed in buffer. The secondary biotinylated antibody at 1:100
dilution (Universal secondary, Vectastain Elite ABC; Vector
Laboratories Inc., Burlingame, CA, USA) was added (in horse
serum/buffer solution) for 30 min, followed by numerous washings.
Avidin/biotin complex (Vector Laboratories Ltd., Peterborough, UK)
was added for 30 min and followed by washes. Diaminobenzidine
(Sigma-Aldrich Co. Ltd.) was used as a chromogen to visualize the
antibody/antigen complex. Sections were counterstained with Mayer's
hematoxylin for 1 min, dehydrated, cleared and mounted in DPX.
Cell lines
The human colon cancer cell lines, Caco-2, HRT18 and
RKO were obtained from the European Collection of Animal Cell
Cultures (ECACC, Salisbury, England). Cells were maintained in
Dulbecco's modified Eagle's medium (DMEM) containing 10% fetal calf
serum, 100 U/ml penicillin and 100 μg/ml streptomycin (Gibco BRC,
Paisley, UK) at 37°C and 5% CO2.
Knockdown of BDNF expression using
ribozyme and screening of stable transfected cell line
Ribozymes targeted to human BDNF transcription
levels were designed based on the secondary structure of the gene
generated using the Zuker RNA mFold program (University of Albany,
New York, NY, USA). The ribozymes were accordingly synthesized and
then cloned into pEF6/V5-his-Topo T/A vector (Invitrogen, Paisley,
UK) and transfected into Caco-2 and HRT18 cells using an Easyjet
Plus electroporator (EquiBio, Kent, UK). Following selection with
culture medium containing 5 μg/ml blasticidin (Sigma-Aldrich Co.
Ltd.), the verified transfectants were cultured in maintenance
medium containing 0.5 μg/ml blasticidin. The primer sequences of
the BDNF ribozymes were as follows: 5′-CTG CAG TTG GCC TTT TGA TAC
AGG GAC CTT TTC AAG GAC TGT CTG ATG AGT CCG TGA GGA-3′ and 5′-ACT
AGT GCA GTG GAC ATG TCG GGC GGG ACG GTT TCG TCC TCA CGG ACT-3′.
Cell growth assay
Colon cancer cell growth rates were assessed using
an in vitro growth assay. Cells were planted in sextuplicate
into 96-well plates at a density of 2,000 cells per well. The
plates were then incubated for 24, 48, 72 and 120 h before being
fixed in 4% formaldehyde (v/v) and stained with 0.5% (w/v) crystal
violet. The crystal violet stain was then extracted using 10%
acetic acid (v/v) and cell density was determined by measuring the
absorbance of this solution at 540 nm using a Bio-Tek ELx800
multi-plate reader (Bio-Tek Instruments Inc., Winooski, VT,
USA).
Flow cytometric analysis of
apoptosis
All cells, including those floating in the culture
medium, were harvested following incubation. Cells were washed in
cold BSS and resuspended in 1X annexin V- binding buffer at a
density of 1×106 cells/ml after centrifugation for 8 min
at 13,000 × g. Fluorescein isothiocyanate (FITC)-annexin V (5 μl)
and 1 μl propidium iodide (PI) working solution (100 μg/ml;
Molecular Probes, Eugene, OR, USA) were added to 100 μl cell
suspension. After a 15-min incubation at room temperature, 400 μl
1X annexin V-binding buffer was added and mixed in gently, and the
samples were immediately kept on ice. The stained cells were
analyzed using a flow cytometer and FlowMax software (Partec UK
Ltd., Canterbury, UK).
Statistical analysis
Statistical analysis was performed using SPSS
software (SPSS, Inc., Chicago, IL, USA). P<0.05 was considered
to indicate a statistically significant result.
Results
Expression of BDNF in human colon
cancer
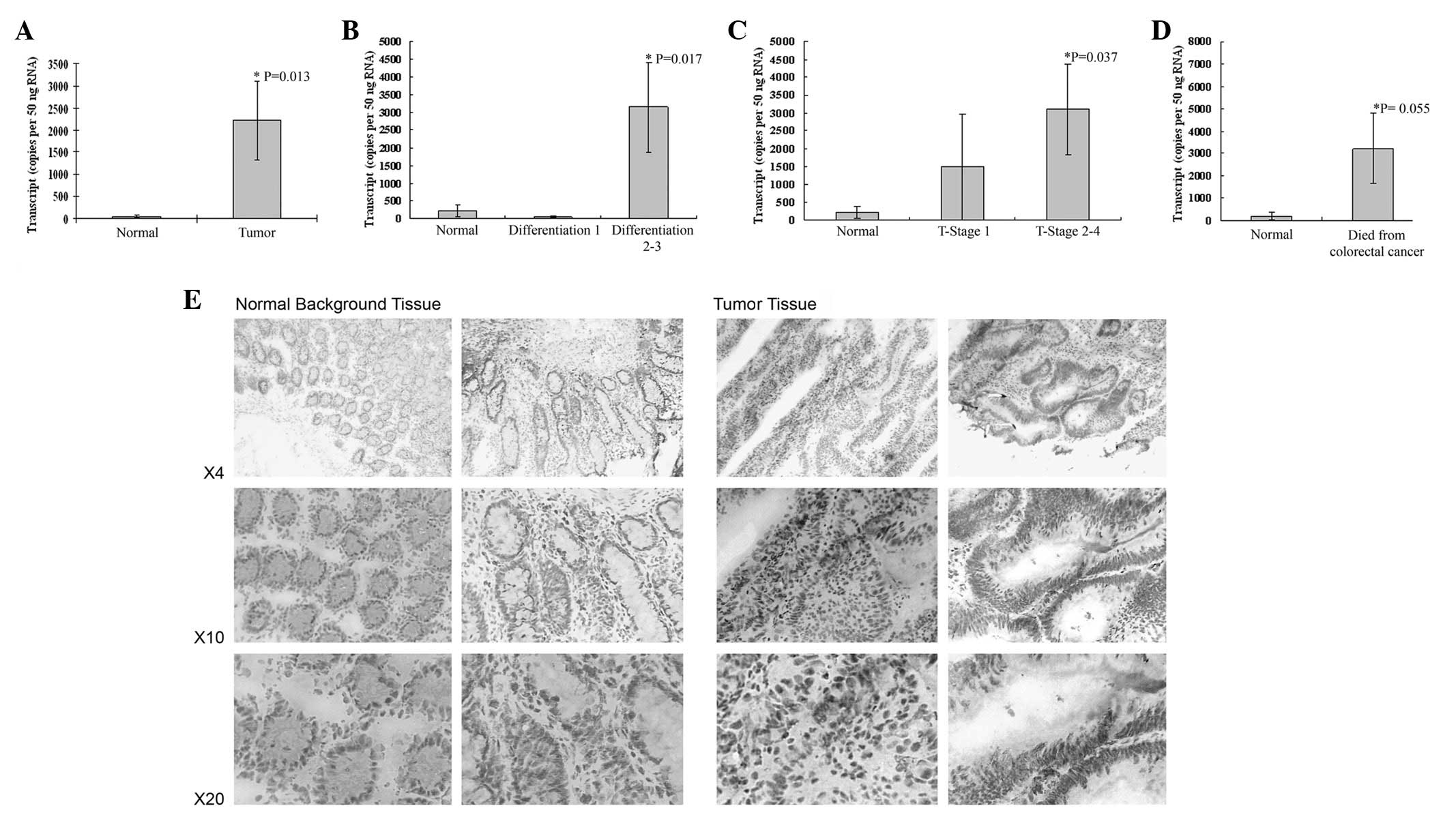
BDNF was observed to be expressed in colon cancer
tissues and normal tissues. BDNF mRNA expression levels were
detected using qPCR. Significantly higher mRNA levels were observed
in colon cancer tissues compared with normal tissues (P=0.017;
Fig. 1A). BDNF expression was
significantly increased with increasing differentiation of
colorectal tumors (well differentiated tumors versus
moderate/poorly differentiated, P=0.017; Fig. 1B) and also increased with
increasing T-staging (T-stage 1 versus 2 and 3, P=0.037; Fig. 1C). Patients who succumbed to
colorectal cancer also had elevated levels of BDNF, P=0.055, but
this did not reach significance (Fig.
1D). BDNF was also increased with Dukes staging [normal
(2312±171) versus Dukes A 314±195, P=0.031; normal versus Dukes B
and C (32761326), P=0.025]. Immunohistochemical staining showed
only a small increase in BDNF protein in tumor tissues, compared
with that in normal background colorectal tissues (Fig. 1E).
mRNA expression of BDNF in human colon
cancer cell lines
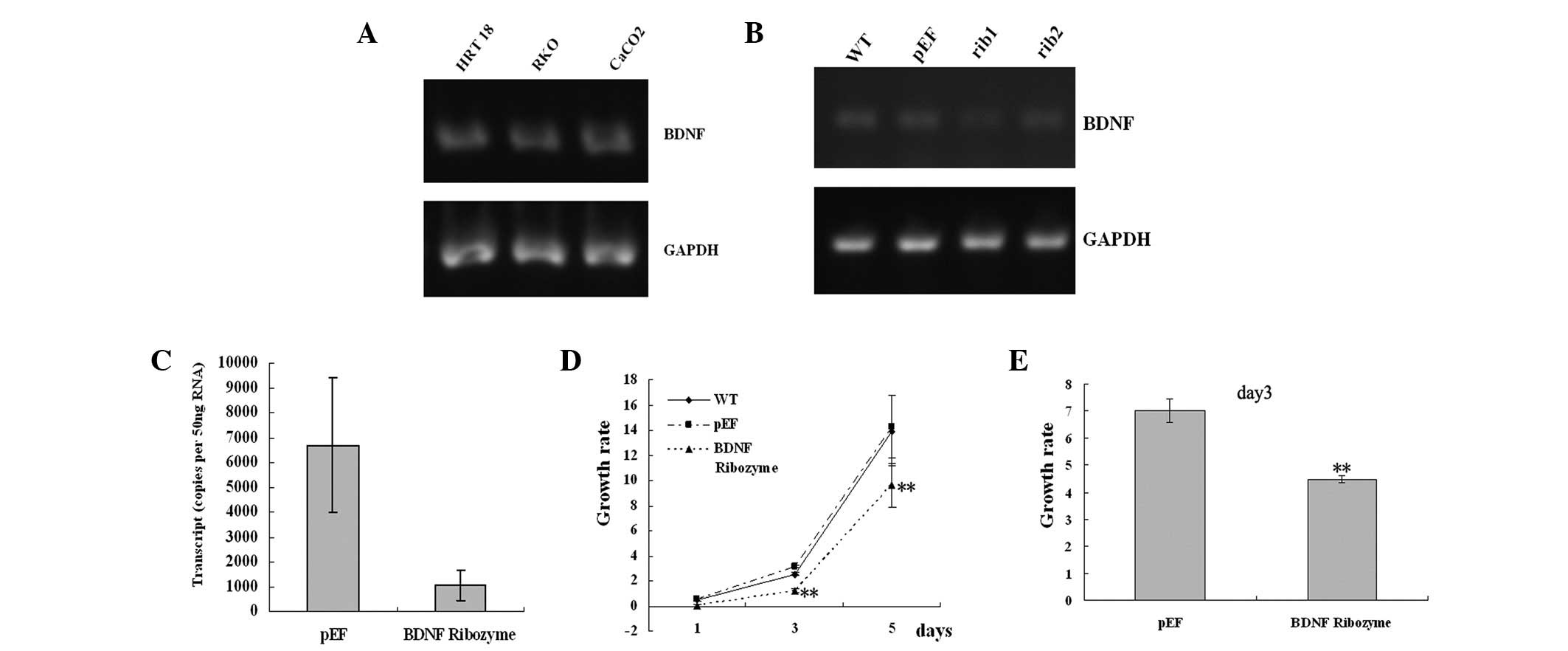
Human cancer cell lines Caco-2, HRT18 and RKO were
examined for the presence of BDNF using RT-PCR (Fig. 2A). BDNF was strongly expressed in
all three cell lines. Fetal kidney tissue was used as a positive
control. The negative control had no DNA template (data not
shown).
BDNF knockdown and establishment of
stable cell lines
The ribozymes targeting BDNF were cloned into
pEF6/V5-his-Topo T/A vector. Caco-2 and HRT18 wild type cells were
subjected to transfection using plasmids containing ribozymes
targeting BDNF or an empty vector control, respectively, followed
by the establishment of BDNF knockdown sub-lines and empty vector
(pEF) control cells. The expression of BDNF at the mRNA level was
reduced in BDNF knocked-down HRT18 and Caco-2 cells using RT-PCR
and qPCR (Fig. 2B and C). We then
characterized the effects of BDNF knockdown in these cells through
a series of in vitro studies.
Effects of BDNF knockdown on the growth
of human colon cancer cells
In the in vitro growth assay, knockdown of
BDNF in Caco-2 and HRT18 cells resulted in a reduction of cell
growth rate (growth rate in BDNF knocked-down Caco-2 cells by day 3
Rib=1.24±0.15, compared with 3.15±0.12 in pEF, P=0.0000; Fig. 2D). Loss of BDNF in HRT18 cells
resulted in reduction of cell growth rate (growth rate in BDNF
knocked-down HRT18 cells by day 3 Rib=4.47±0.13, compared with
7.00±0.43 in pEF, P=0.0001; Fig.
2E). These data demonstrate that BDNF may increase colon cancer
cell growth.
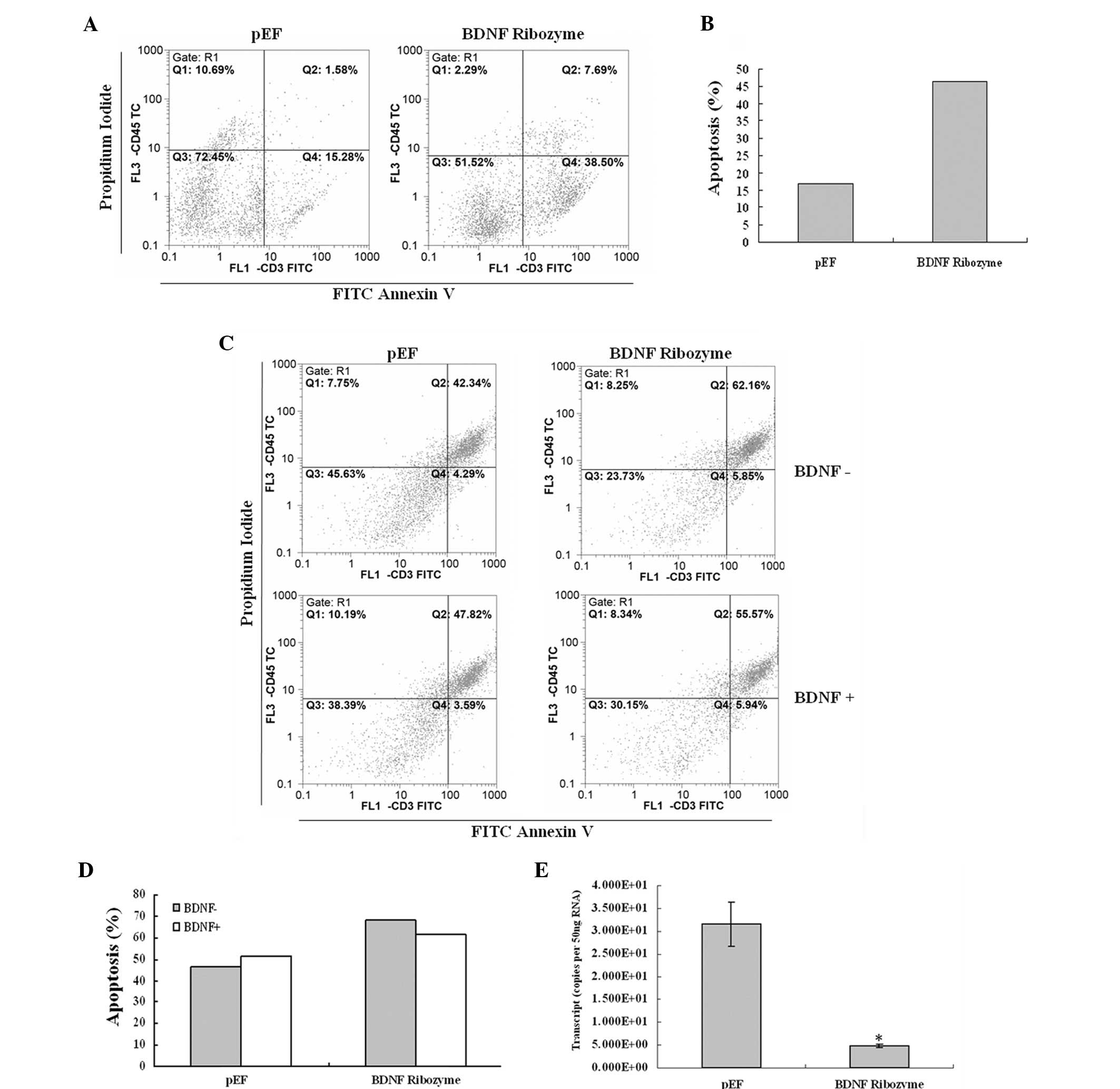
Effects of BDNF knockdown on cell
apoptosis
To investigate whether apoptosis is involved in the
effect of BDNF knockdown in Caco-2 and HRT18 cells, we determined
the proportion of apoptotic cells. As shown in Fig. 3A–D, there was an increase in cell
population towards apoptosis in the BDNF knocked-down Caco-2 and
HRT18 cells, which was 46.19% in the BDNF knocked-down Caco-2
cells, compared with 16.86% in the pEF control. There was also an
increase of apoptosis in the BDNF knocked-down HRT18 cells, which
was 68.01% in the BDNF knocked-down cells, compared with 46.63% in
the pEF control. These results suggest that BDNF decreases
apoptosis in these cells.
In addition, we confirmed whether or not this effect
was specific to BDNF knockdown by rescue experiments. BDNF protein
was added in the cell culture medium (50 ng/ml) and resulted in a
negated effect of BDNF knocked-down HRT18 cells compared with the
pEF controls (Fig. 3C and D).
Effects of BDNF knockdown on cellular
signal pathways
We screened the cells at the mRNA transcript level
for bcl-2 in BDNF knocked-down HRT18 cells using qPCR. The results
showed that the level of bcl-2 was reduced in BDNF knockdown HRT18
cells, indicating that BDNF stimulates the message for bcl-2.
(Fig. 3E).
Discussion
BDNF has been observed to be elevated in non-nervous
system solid tumors including colorectal cancer. However, the
impacts of BDNF transcription level on cellular biological function
and the molecular pathways induced by BDNF are unknown (15). In our patient cohort, we found BDNF
to have significantly elevated levels in tumors with poor prognosis
and, to the best of our knowledge, this is the first study of its
kind.
The expression levels of BDNF and TrkB mRNA have
been demonstrated to be higher in human cancer cell lines than in
normal tissues (16). Our results
also show that the mRNA expression level of BDNF in human colon
cancer is elevated. Therefore we utilized RNA knockdown experiments
to study the effects of BDNF expression on cellular function and
the possible molecular mechanisms. In the present study we obtained
stable knockdowns of BDNF in human colon cancer cell lines using
anti-BDNF ribozymes.
When BDNF was stably knocked down in Caco-2 and
HRT18 cell lines, the growth decreased compared with that of cells
transfected with the vector control, suggesting that reduced BDNF
gene expression inhibited cellular proliferation. These results
indicate that BDNF facilitates the proliferation of human colon
cancer cells.
The apoptosis experiments demonstrated that in the
Caco-2 and HRT18 cell lines in which BDNF was stably knocked down,
apoptosis increased compared with that of the cells transfected
with the vector control. In addition, we observed that when BDNF
was added the apoptosis increased in HRT18 cells in which BDNF was
stably knocked down. These results suggest that BDNF is a regulator
of apoptosis in these cells. We conclude that BDNF is able to
maintain colon cancer cell survival and proliferation.
The effect of BDNF on cellular biological functions
is induced mainly by its receptor TrkB. When BDNF binds to TrkB the
tyrosine kinase activity of the receptor is activated via
phosphorylation of tyrosine residues in the cytoplasmic region of
the receptor, which in turn induces cellular signaling (10,17).
The PI3K-AKT pathway is closely associated with cell
survival and PI3K plays a key role in anti-apoptotic survival and
proliferation (9,18,19).
BDNF activates the AKT pathway in order to maintain cell survival
(20). In this study, we also
investigated the expression of downstream molecules associated with
the AKT pathway. Bcl-2 as a member of the bcl-2 family, negatively
regulates cell death and acts as an anti-apoptosis factor. Bcl-2
was downregulated in the BDNF knocked-down HRT18 cells compared
with the level in the cells transfected with vector control.
Accordingly, BDNF induces the increased transcription of bcl-2 to
inhibit apoptosis and facilitate survival. BDNF downregulation
eliminated protection from apoptosis, likely via the BDNF-Akt-Bcl2
anti-apoptotic signaling pathway (19).
In conclusion, our study shows that BDNF facilitates
cell proliferation and inhibits cell apoptosis in human colon
cancer cells. Reduced BDNF expression induces changes in downstream
signaling molecules that are associated with cell survival and
apoptosis. BDNF is therefore a potential therapeutic target in
colon cancer and its effect in human colon cancer requires further
investigation.
Acknowledgements
The authors would like to thank Cancer Research
Wales, the Albert Hung Foundation and the Breast Cancer Hope
Foundation for support.
References
|
1
|
Jemal A, Siegel R, Ward E, Murray T, Xu J,
Smigal C and Thun MJ: Cancer statistics, 2006. CA Cancer J Clin.
56:106–130. 2006. View Article : Google Scholar
|
|
2
|
Zhang J, Dhaka IB, Zhao Z and Li L: Trends
in mortality from cancers of the breast, colon, prostate,
esophagus, and stomach in East Asia: role of nutrition transition.
Eur J Cancer Prev. 21:480–489. 2012. View Article : Google Scholar
|
|
3
|
Foretova L, Petrakova K, Palacova M,
Kalabova R, Svoboda M, Navratilova M, Schneiderova M, Bolcak K,
Krejci E, Drazan L, Mikova M, Hazova J, Vasickova P and Machackova
E: Genetic testing and prevention of hereditary cancer at the MMCI
- over 10 years of experience. Klin Onkol. 23:388–400.
2012.PubMed/NCBI
|
|
4
|
Glück S and Mamounas T: Improving outcomes
in early-stage breast cancer. Oncology (Williston Park). 24(Suppl
4): 1–15. 2010.
|
|
5
|
Brunetto de Farias C, Rosemberg DB, Heinen
TE, Koehler-Santos P, Abujamra AL, Kapczinski F, Brunetto AL,
Ashton-Prolla P, Meurer L, Reis Bogo M, Damin DC, Schwartsmann G
and Roesler R: BDNF/TrkB content and interaction with
gastrin-releasing peptide receptor blockade in colorectal cancer.
Oncology. 79:430–439. 2010.PubMed/NCBI
|
|
6
|
Barde YA, Edgar D and Thoenen H:
Purification of a new neurotrophic factor from mammalian brain.
EMBO J. 1:549–553. 1982.PubMed/NCBI
|
|
7
|
Numakawa T, Suzuki S, Kumamaru E, Adachi
N, Richards M and Kunugi H: BDNF function and intracellular
signaling in neurons. Histol Histopathol. 25:237–258.
2010.PubMed/NCBI
|
|
8
|
Huang YT, Lai PC, Wu CC, Hsu SH, Cheng CC,
Lan YF and Chiu TH: BDNF mediated TrkB activation is a survival
signal for transitional cell carcinoma cells. Int J Oncol.
36:1469–1476. 2010.PubMed/NCBI
|
|
9
|
Sun CY, Hu Y, Huang J, Chu ZB, Zhang L,
She XM and Chen L: Brain-derived neurotrophic factor induces
proliferation, migration, and VEGF secretion in human multiple
myeloma cells via activation of MEK-ERK and PI3K/AKT signaling.
Tumour Biol. 31:121–128. 2010. View Article : Google Scholar
|
|
10
|
Zhang L, Hu Y, Sun CY, Huang J and Chu ZB:
Brain-derived neurotrophic factor promotes the secretion of MMP-9
in human myeloma cell through modulation of nucleus factor-kappaB.
Zhonghua Xue Ye Xue Za Zhi. 29:243–246. 2008.(In Chinese).
|
|
11
|
Zhang S, Guo D, Luo W, Zhang Q, Zhang Y,
Li C, Lu Y, Cui Z and Qiu X: TrkB is highly expressed in NSCLC and
mediates BDNF-induced the activation of Pyk2 signaling and the
invasion of A549 cells. BMC Cancer. 10:432010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Akil H, Perraud A, Mélin C, Jauberteau MO
and Mathonnet M: Fine-tuning roles of endogenous brain-derived
neurotrophic factor, TrkB and sortilin in colorectal cancer cell
survival. PLoS One. 6:e250972011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Festuccia C, Gravina GL, Millimaggi D,
Muzi P, Speca S, Ricevuto E, Vicentini C and Bologna M: Uncoupling
of the epidermal growth factor receptor from downstream signal
transduction molecules guides the acquired resistance to gefitinib
in prostate cancer cells. Oncol Rep. 18:503–511. 2007.
|
|
14
|
Li Z, Zhang J, Liu Z, Woo CW and Thiele
CJ: Downregulation of Bim by brain-derived neurotrophic factor
activation of TrkB protects neuroblastoma cells from paclitaxel but
not etoposide or cisplatin-induced cell death. Cell Death Differ.
14:318–326. 2007. View Article : Google Scholar
|
|
15
|
Patani N, Jiang WG and Mokbel K:
Brain-derived neurotrophic factor expression predicts adverse
pathological & clinical outcomes in human breast cancer. Cancer
Cell Int. 11:232011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Guo D, Hou X, Zhang H, Sun W, Zhu L, Liang
J and Jiang X: More expressions of BDNF and TrkB in multiple
hepatocellular carcinoma and anti-BDNF or K252a induced apoptosis,
supressed invasion of HepG2 and HCCLM3 cells. J Exp Clin Cancer
Res. 30:972011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kawamura N, Kawamura K, Manabe M and
Tanaka T: Inhibition of brain-derived neurotrophic factor/tyrosine
kinase B signaling suppresses choriocarcinoma cell growth.
Endocrinology. 151:3006–3014. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li Z and Thiele CJ: Targeting Akt to
increase the sensitivity of neuroblastoma to chemotherapy: lessons
learned from the brain-derived neurotrophic factor/TrkB signal
transduction pathway. Expert Opin Ther Targets. 11:1611–1621. 2007.
View Article : Google Scholar
|
|
19
|
Sheikh AM, Malik M, Wen G, Chauhan A,
Chauhan V, Gong CX, Liu F, Brown WT and Li X: BDNF-Akt-Bcl2
antiapoptotic signaling pathway is compromised in the brain of
autistic subjects. J Neurosci Res. 88:2641–2647. 2010.PubMed/NCBI
|
|
20
|
Yu X, Liu L, Cai B, He Y and Wan X:
Suppression of anoikis by the neurotrophic receptor TrkB in human
ovarian cancer. Cancer Sci. 99:543–552. 2008. View Article : Google Scholar : PubMed/NCBI
|