Introduction
The pituitary gland is located below the center of
the brain and over the sella on the cerebral surface of the body of
the sphenoid. The sphenoid contains two sinuses, which open into
the roof of the nasal cavity via the apertures on the posterior
wall of the sphenoethmoidal recess directly above the turbinates.
Since only thin layers of bone separate the sphenoid sinuses from
the nasal cavities below and the sella turcica above,
transsphenoidal surgery (TSS) is the first choice option for the
removal of pituitary lesions rather than the transcranial
approach.
The transsphenoidal approach has evolved
considerably since it was first successfully performed by Schloffer
in 1907 (1). Since then, TSS has
been performed on numerous patients via different methods and the
surgical routes are well formulated. However, even with the aid of
fluoroscopy, the development of this technique was hampered by poor
illumination and visualization of the surgical field. In 1967,
Hardy first introduced the operating microscope to TSS, which laid
a cornerstone foundation for the development of modern TSS
(2). Since then, minimally
invasive transsphenoidal surgical approaches to the sella turcica
have undergone significant changes from sublabial transseptal,
transnasal, to pure endonasal approaches. TSS has now become the
standard approach for the surgical removal of pituitary adenoma
(3,4). Compared with the transcranial
approach, TSS does not require skin incision and external
craniotomy, thereby offering the advantages of fewer complications,
less discomfort and quicker recovery (5). Despite the increasing popularity of
endoscopic techniques in recent years, microscopic TSS remains the
mainstay of surgical treatment for pituitary lesions as it offers
stereoscopic vision of the sella, excellent coaxial illumination
and the capability for neurosurgeons to use traditional
neurosurgical instruments (6).
Moreover, access may be somewhat narrower in the absence of a nasal
speculum, with some likening the endoscopic technique to ‘operating
with chopsticks’ (7).
However, the surgical path of TSS is extremely deep
and narrow, and the view is usually blocked by crucial
neurovascular structures. In addition, the close proximity of the
sphenoid sinus to the carotid artery and the optic canal, plus the
high levels of variation between the anatomical structures of the
sphenoid sinus and sellar floor, make the approach even more
difficult, hence the success of the treatment greatly relies on the
experience of the surgeon and the familiarity with anatomical
landmarks through the surgical route. To date, the methods and
techniques of TSS adopted by different surgeons with respect to
surgical guidance and important landmarks vary significantly. Our
knowledge regarding the anatomical structures relevant to TSS is
mainly based on postmortem or imaging studies (8–13).
However, the actual view at the surgical level under the microscope
is different in real-world scenarios. However, in some patients
with complex sellar anatomy, non-pneumatized sphenoid sinuses, or
those undergoing reoperation, the typical appearance of the sella
turcica and its relationship with the tuberculum sellae and clivus
may be less conspicuous and identification of the midline is often
more challenging, substantially increasing the risk of the surgery
(14). Although advances in
intraoperative neuronavigation have improved the accuracy
associated with transsphenoidal and related extended endonasal
skull base surgery over the last decade, they by no means obviate
the requirement for knowledge of the relevant surgical anatomy. A
practical anatomical study of the landmarks relevant to TSS is
therefore warranted. We conducted a study based on 148 cases of
endonasal TSS to delineate the important anatomical landmarks
relevant to the three major regions across the procedure. We
believe that these landmarks will provide useful guidance in
clinical practice.
Materials and methods
General data
This study retrospectively reviewed 148 surgical
records of single-nostril endonasal TSS for sellar lesions
performed in our department in the period between May 2002 and
February 2008. The patients included 78 males and 70 females with a
mean age of 39.2 years (range, 12–78 years). Preoperative magnetic
resonance imaging (MRI) was performed for all patients to assess
variations of the sphenoid bone, sphenoid sinus and sellar floor.
Postoperative histopathological examination confirmed the diagnosis
of pituitary lesions. The study was approved by the ethics
committee of Fuzhou General Hospital (Fuzhou, China). Written
informed consent was obtained from all participants.
Surgical procedure
Patients lay in the supine position with the head
extended by 20°. Surgeons were positioned directly behind the
patient’s head. The microscope was orientated perpendicularly to
the surface of the surgical floor first and then later adjusted
towards the mucosal aperture of the sphenoid sinus. All surgeries
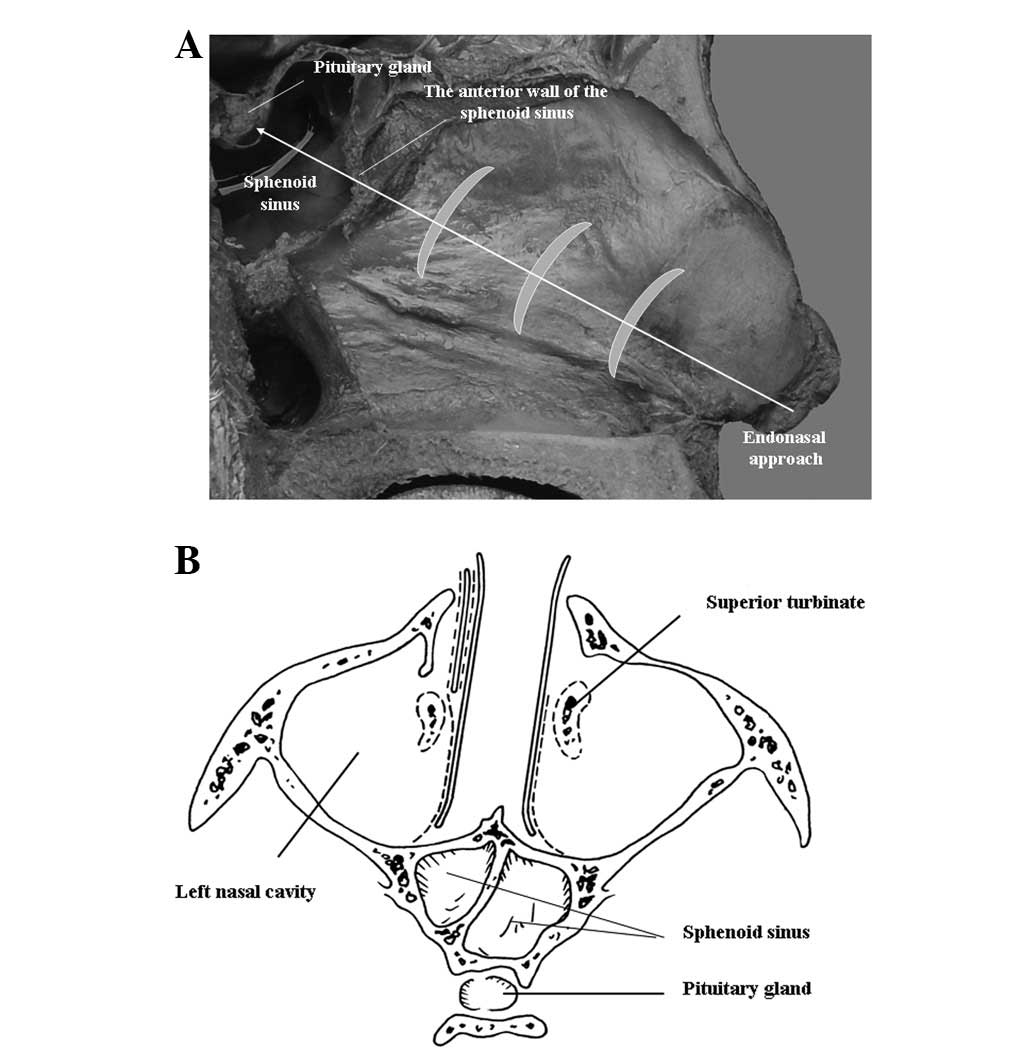
were performed via a unilateral endonasal transsphenoidal approach
(Fig. 1). This route approached
the roof of the nasal cavity and the anterior wall of the sphenoid
sinus and then entered the sphenoid sinuses by anterior
sphenoidotomy followed by entry through the top of the sphenoid
bone into the sella turcica. The method was modified three times
with different positions of mucosal incision to obtain the optimal
surgical view.
Method A
An endoscope was used in 26 patients to examine and
identify the nasal structures and the mucosal aperture of the
sphenoid sinus. Under an operating microscope, the sphenoid sinus
was approached either by expanding the aperture or by incising the
ipsilateral mucoperiosteum at the posterior third of the nasal
septum, fracturing the vomer and separating the bilateral
mucoperiosteum to finally expose the anterior wall of the sphenoid
sinus, followed by an anterior sphenoidotomy. The sphenoid septum
was then excised, the orientation of the sellar floor was
determined and the bony sellar floor and dura were opened to
approach the pituitary gland and lesion. After removing the
pituitary lesion, the dural defect of the sellar floor was closed
with a small piece of autologous muscle harvested from the thigh
and coated with fibrin glue. In a few difficult cases,
neuronavigation was employed to guide access to the sella
turcica.
Method B
In another 63 patients, the surgical procedure was
similar to method A. However, the mucoperiosteal incision was made
on the posterior nasal septum (∼0.5–1.5 cm from the anterior wall
of the sphenoid sinus) and then the perpendicular plate of the
ethmoid bone was fractured and pushed to the opposite side before
performing an anterior sphenoidotomy.
Method C
In the final 59 patients, the mucoperiosteal
incision was made at the osseocartilaginous junction of the nasal
septum (∼3 cm from the naris). The cartilaginous nasal septum was
pushed to the opposite side and the perpendicular plate of the
ethmoid bone was excised to expose the anterior wall of the
sphenoid sinus, followed by an anterior sphenoidotomy. The rest of
the procedure was identical to the previous two methods.
Anatomical assessment
The structure of the nasal cavity and sphenoid
sinus, position of the apertures of the sphenoid sinus and relevant
arteries and the morphological characteristics of the anterior wall
of the sphenoid sinus and sellar bulge were observed and recorded.
Nasal structures and anatomical anomalies that would affect the
surgical approach were photographed and recorded.
Results
General outcomes
Comparing the three surgical methods, the approach
with the mucoperiosteal incision made at the osseocartilaginous
junction of the nasal septum provided a greater surgical view
compared with the other two methods. The most common pituitary
lesion was pituitary macroadenoma, occurring in 70.9% of patients
(Table I). There were two cases of
meningitis but no optic nerve or carotid artery injuries. The
procedure-related complications included cerebrospinal fluid (CSF)
leak (3.4%), mild subarachnoid hemorrhage (2.7%), nasal bleeding
(6.8%) and mild nostril injuries (12.2%).
 | Table IDiagnosis of pituitary lesions. |
Table I
Diagnosis of pituitary lesions.
| Pituitary
lesions | Number of cases |
|---|
| Pituitary
adenoma | 136 |
| Knosp-Steiner
classification for parasellar extension | |
| 0–II | 122 |
| III | 24 |
| IV | 2 |
| Classification by
size | |
| Microadenoma (≤10
mm) | 10 |
| Macroadenoma
(10–40 mm) | 105 |
| Giant adenoma
(≥40 mm) | 21 |
| Dural invasion on
the sellar floor | 13 |
| Rathke cysts | 4 |
| Sphenoid
mucocele | 2 |
| Pituitary
abscess | 2 |
| Empty sella | 2 |
| Meningioma | 1 |
| Metastatic tumor | 1 |
| Pituitary
hyperplasia | 1 |
Structure and anatomical landmarks
located in the nasal cavities
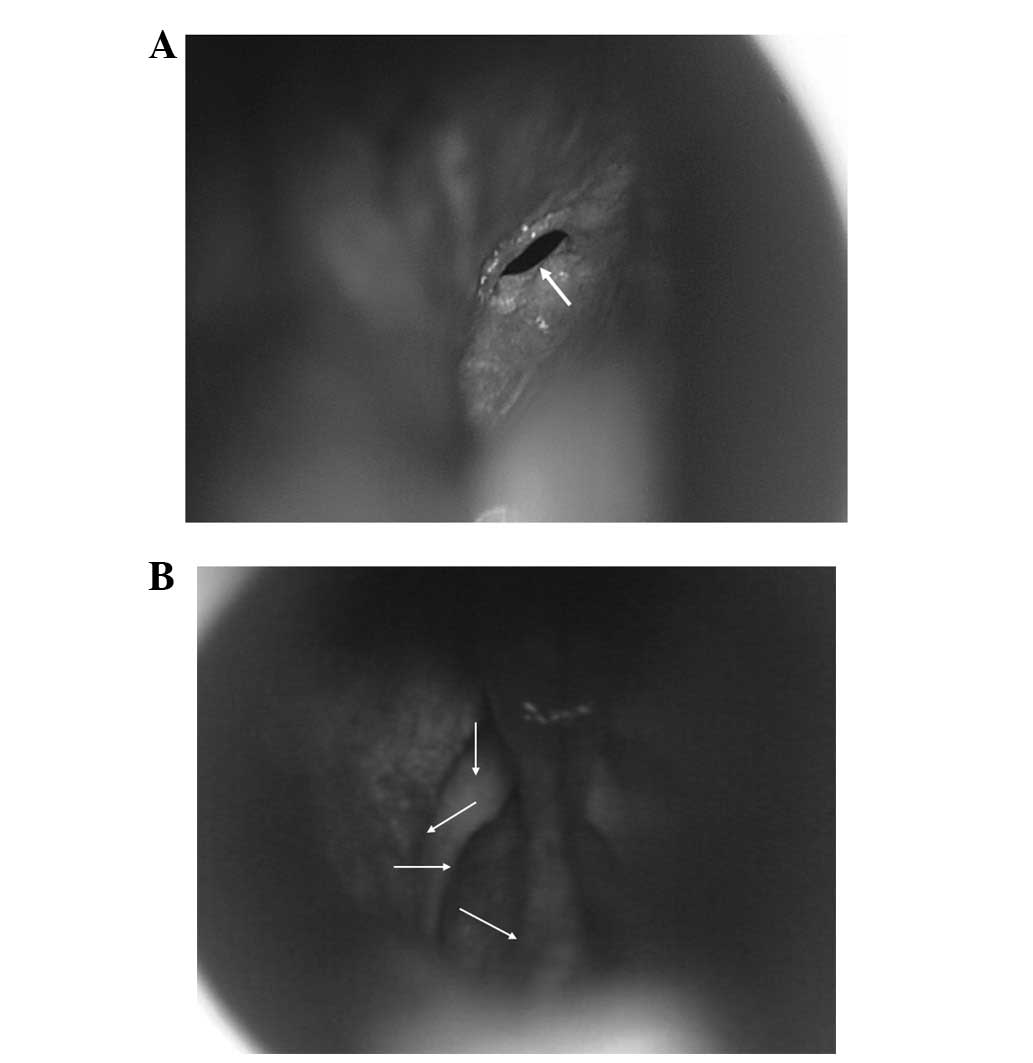
The most important landmark in the nasal cavity is
the mucosal aperture of the sphenoid sinus, which could be observed
under the microscope in 79 patients (53.4%) after pushing the
middle turbinate laterally (Fig.
2A). However, in the other 69 patients (46.6%), fracturing of
the middle and superior turbinates was required. In addition, there
was usually a blunt longitudinal bulge on the posterior nasal
septum towards the sphenoid crest. The mucosal aperture was
observed lateral to the end of this bulge (Fig. 2B). Once the aperture position was
confirmed, the objective mirror of the microscope was fixed towards
it. The sphenoid sinus together with the anteroinferior wall of the
sella could then be easily approached from this direction.
Anatomical landmarks after dissection of
the nasal septal mucoperiosteum
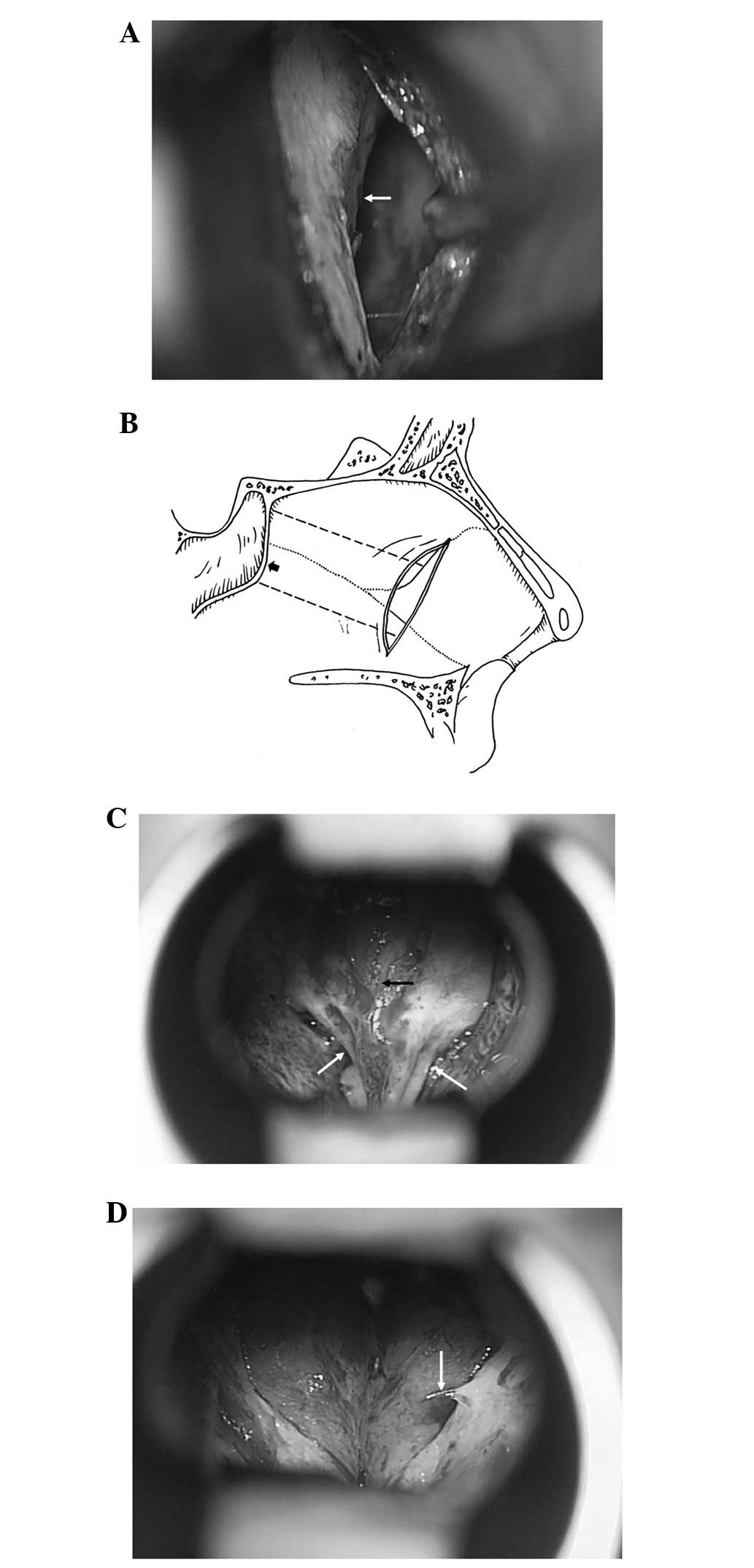
In this region, the first landmark noted was the
osseocartilaginous junction of the nasal septum, which could be
observed after the septal mucoperiosteum was opened 3 cm from the
naris (Fig. 3A). As illustrated
(Fig. 3B), the anterior wall of
the sphenoid sinus was situated at the end of the osseous septum,
approximately perpendicular to the surgical view with the superior
aspect tilting slightly posteriorly and the midline (the sphenoid
crest) protruding anteriorly. This resembled a protruding bow under
the microscope. The top of the bow was the perpendicular plate of
the ethmoid bone, which was narrow and deeply tilted. The bottom of
the bow was broad and shallow, bulged at the midline like a bird
beak and two osseous apertures were located on both sides of the
bulge (Fig. 3C). The ‘bow sign’ of
the sphenoid bone is the most important landmark in this region,
which indicated the correct direction of the approach. If the
surgical route was directed slightly above the bulge, it would
enter the ethmoid sinus. If the surgical route was directed below
the bulge, it would not approach the sellar floor appropriately.
The position of the ‘bird beak’ between the two apertures is the
best place to enter the sphenoid sinus. In addition, there were
usually 1–3 small nutrient arteries, 0.1–0.2 mm in diameter,
arising from the superior branch of the posterior septal artery on
each side. These arteries traveled inferomedially along the
subperiosteum into the sphenoid bone below the osseous aperture of
the sphenoid sinus. We found that one of these arteries
consistently appeared at 3–7 mm inferior to the osseous aperture,
which could serve as a surrogate marker for locating the aperture
(Fig. 3D). Caution should be taken
when separating the mucoperiosteum in this area in order to avoid
damaging the blood vessels.
Anatomical landmarks within the sphenoid
sinus
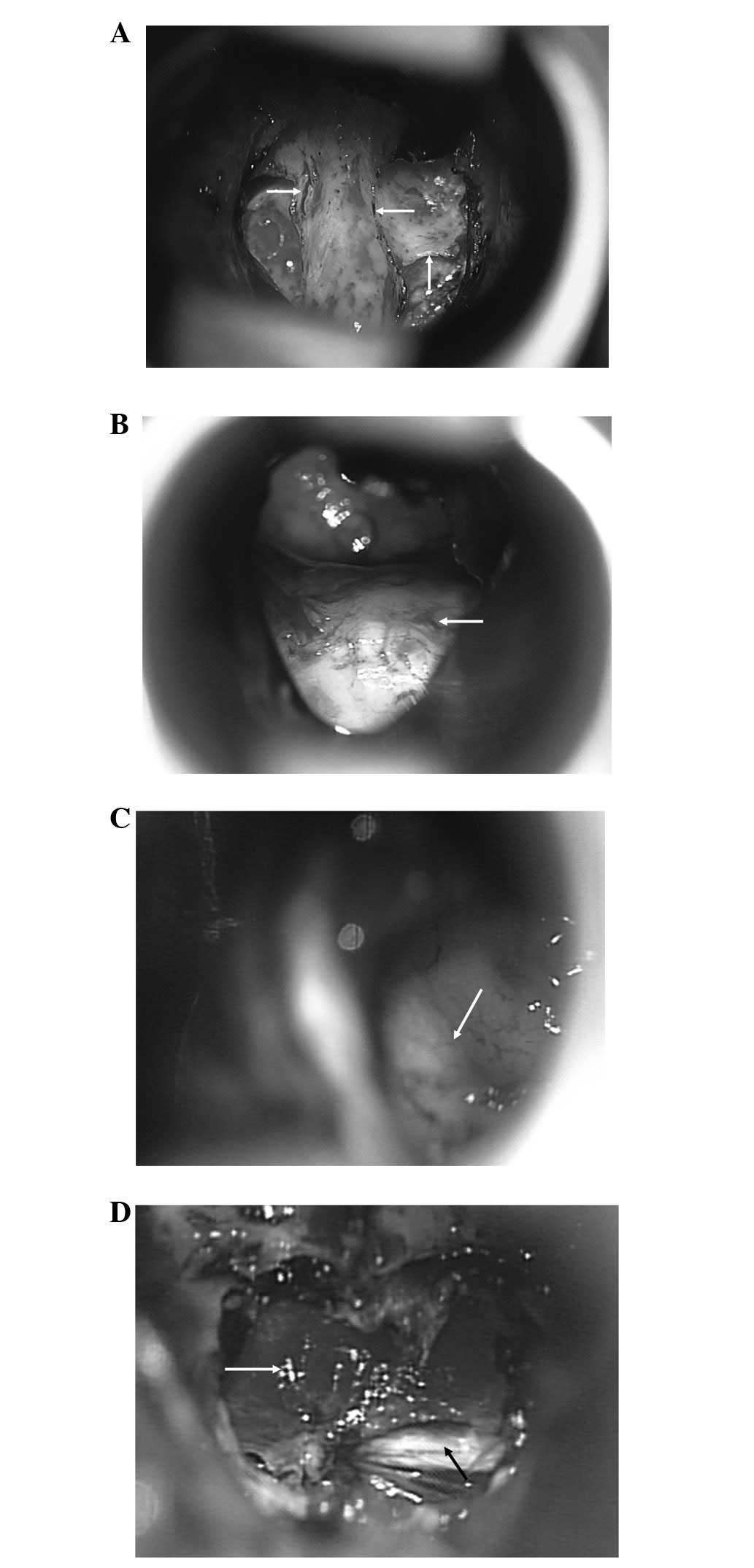
The position, thickness, deviation and degree of
development of the sphenoid septum varied greatly, which should be
identified with the aid of preoperative MRI. In our study, most
patients had only one sphenoid septum but multiple sphenoid septa
were observed in a few cases (Fig.
4A). The important landmark in the sphenoid sinus is the bulge
of the sellar floor (Fig. 4B),
which is beneath the pituitary fossa. Its morphology varied in
accordance with the degree of the sphenoid sinus pneumatization and
the development of the pituitary fossa. It may also present as an
increased inferior and lateral convexity or destruction in patients
with pituitary macroadenomas. We were able to define this bulge in
most patients. When this bulge was ill-defined, the observation of
carotid prominence would be useful as it was always located in the
parasellar space and could be observed after tilting the microscope
slightly to the left or right (Fig.
4C). In the present study, successful access to the sella fossa
was achieved in all our patients. Neuronavigation was only used in
a few difficult cases during the early periods of our practice.
Differentiation of the pituitary gland
and dura
The outcome of TSS is related to the proper removal
of lesions and the protection of normal pituitary gland tissue. As
shown in Fig. 4D, normal pituitary
gland tissue was orange-colored and tough, whereas pituitary
nervous tissue was pale and soft. It is also important to
distinguish the type of dura to confirm whether the surgical
approach is appropriate or not. The differences in appearance and
texture between the dura of the anterior wall of the pituitary
turcica and that of the tuberculum sella could be identified. The
former was smooth and thin without any pattern, usually orange,
light blue, light yellow or white in color. The latter was white
and thick with a horizontal streaky pattern and larger collagen
fiber bundles, which were located beneath the anterior lamina
terminalis cistern, inter-hemispheric cistern and gyrus rectus.
Discussion
In the present study, we delineated important
anatomical landmarks for endonasal TSS, including the mucosal
aperture of the sphenoid sinus, a blunt longitudinal prominence on
the posterior nasal septum, the osseocartilaginous junction of the
nasal septum, the ‘bow sign’ of the anterior wall of the sphenoid
sinus, the osseous aperture and its relationship with nutrient
arteries, the bulge of the sellar floor and the carotid
protuberance. These landmarks outline a clear route to the sella
turcica providing the optimal view and causing less tissue damage.
Based on these landmarks, we successfully accessed the sella
turcica and dissected pituitary lesions in all patients without any
assistance from intraoperative CT scan and fluoroscopic
navigation.
Several postmortem and imaging studies attempting to
illustrate anatomical landmarks for TSS have been conducted
previously (6). Using cadaveric
heads and 10 skulls, Campero et al produced a spheno-sellar
point and a spheno-nostril line to guide the head positioning for
TSS (8). However, the clinical
applicability of such types of measurement is limited due to the
small number of subjects and the difficultly in evaluating the
procedure as a result of limited standard verification, despite
being attempted in 102 surgical procedures. Other studies have
tried to disclose the variations of human skulls which may affect
the transsphenoidal approach. Campero et al studied dry
skulls and found that the location of apertures varied greatly
(9). Tatreau et al
(15) reported that the periform
aperture and pneumatization to the planum and sella changed with
age in pediatric patients, but not in adult patients. Hamid et
al (10) used CT and MRI scans
to study variations of the sphenoid sinus in 296 patients with
pituitary lesions. The authors found that the degree of
pneumatization of the sphenoid sinus varied greatly but the
appearance of the sellar bulge was prominent, which appeared in 75%
of patients. In the present study, we did not find a great
variation in the position of aperture. If this is the case, we may
still use other landmarks, such as the blunt longitudinal
prominence on the posterior nasal septum and the nutrient arteries,
to determine its position to guide surgical direction. The
appearance of these landmarks, especially the position of nutrient
arteries, was prominent. We also identified the sellar bulge in
most patients, which is consistent with the study by Hamid et
al (10). In the cases of
ill-defined sellar bulges, the identification of the carotid
protuberance will be useful to decide the position of the sella.
Within all these anatomical landmarks, the ‘bow sign’ on the
anterior wall of the sphenoid sinus is the most important and has
thus far never been reported in previous studies. The ‘bow sign’
was consistent in the majority of patients, thus it is useful to
guide the surgical direction towards the sella turcica regardless
of the variation in the location of apertures. The combination of
all these landmarks can therefore minimize the risk of anatomic
disorientation so as to avoid major complications in our patients.
In addition, we made the mucoperiosteal incision in three distinct
positions of the nasal septum and found that the approach from the
incision at the osseocartilaginous junction provided the optimal
surgical view and direction. This incision was also recommended by
Marquardt et al (16).
Over the last 10 years, endoscopic techniques have
seen a marked development and led to a trend in transsphenoidal
surgical approaches. However, the preference of endoscopic TSS or
microscopic TSS depends on the technological refinements and
economical restraints. Although an endoscopic approach permits a
better view in the sphenoid sinus and the parasellar region
(17), it cannot magnify the area
being viewed and solve the narrowness of the anatomical spaces and
the consequent limited view before sphenoidotomy, which is an
advantage of the microscopic approach (16). It also has difficulty in
intracranial hemorrhage control and the closure of the dural and
osseous defects following tumor dissection, subsequently increasing
the risk of postoperative CSF leak, meningitis, etc. (18). Goudakos et al (19) reviewed all studies from 1952 to
2010 regarding endoscopic TSS and microscopic TSS and demonstrated
that the two techniques provided similar rates of complete tumor
excision and remission rates. Endoscopic surgery was associated
with fewer complications related to surgical technique. However,
another study identified no statistically significant differences
between the two approaches (20).
It should be noted that these studies did not take the surgical
experience or concomitant use of intraoperative imaging modalities
into account. Nevertheless, we did not underestimate the value of
the endoscopic approach even though most surgeries were performed
under the microscope. The endoscope was used to aid the
establishment of surgical techniques in the first 26 patients of
the present study. In fact, the endoscopic approach for pituitary
lesions involves exactly the same surgical steps as the microscopic
approach. As described by Jho et al (21,22),
it opens exactly the same anatomical tissue layers and provides an
identical wide exposure of the concerned structures as a
microscopic approach. Therefore, the anatomical landmarks that were
summarized from our experience with microscopic TSS can be also
applied to endoscopic approaches.
Although the aforementioned anatomical landmarks are
useful to guide the surgical procedure, neuronavigation techniques
are invaluable in evaluating the variation of sphenoid sinus and
the sellar region, especially in those with residual or recurrent
masses in the setting of previous TSS, which may inevitably alter
the normal anatomical structure of the skull base (23). In the present study, we used
neuronavigation to distinguish the sellar structure in the few
patients we experienced difficulties in identifying the sellar
floor in the early period of the present study. Therefore,
neuronavigation techniques and anatomical landmarks are
complementary to each other. The combination of the two may improve
surgical outcomes.
Since we mainly focused on endonasal TSS, we did not
compare our findings with those using another surgical approach. We
believe, however, that the anatomical landmarks for endonasal TSS
are also applicable to other approaches.
Locating the sphenoid sinus aperture is the gold
standard to direct the surgical route of TSS. The ‘bow sign’ and
the sellar bulge are critical landmarks for the accurate entry into
the sphenoid sinus and sella fossa. These landmarks outline a clear
route to the sella turcica with the optimal view causing less
tissue damage. The application of these landmarks will aid the
reduction of complications and improvement of outcomes of TSS.
Acknowledgements
The authors would like to thank Dr
Heping Zheng of the Clinical Anatomy Research Center in Fuzhou
General Hospital for his technical assistance.
References
|
1
|
Schmidt RF, Choudhry OJ, Takkellapati R,
Eloy JA, Couldwell WT and Liu JK: Hermann Schloffer and the origin
of transsphenoidal pituitary surgery. Neurosurg Focus. 33:e52012.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ciric IS and Zhao J: Transsphenoidal
microsurgery: past, present and future. Expert Rev Anticancer Ther.
6(Suppl 9): S75–S78. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cavallo LM, Messina A, Cappabianca P,
Esposito F, de Divitiis E, Gardner P and Tschabitscher M:
Endoscopic endonasal surgery of the midline skull base: anatomical
study and clinical considerations. Neurosurg Focus.
19:e22005.PubMed/NCBI
|
|
4
|
Liu JK, Das K, Weiss MH, Laws ER Jr and
Couldwell WT: The history and evolution of transsphenoidal surgery.
J Neurosurg. 95:1083–1096. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ceylan S, Koc K and Anik I: Endoscopic
endonasal transsphenoidal approach for pituitary adenomas invading
the cavernous sinus. J Neurosurg. 112:99–107. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Powell M and Gnanalingham KK: Endoscopic
trans-sphenoidal pituitary surgery: is it here to stay? Br J
Neurosurg. 21:315–317. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jho HD: Endoscopic transsphenoidal tumor
surgery. Operative Tech Neurosurg. 5:218–225. 2002. View Article : Google Scholar
|
|
8
|
Campero A, Socolovsky M, Torino R, Martins
C, Yasuda A and Rhoton AL Jr: Anatomical landmarks for positioning
the head in preparation for the transsphenoidal approach: the
spheno-sellar point. Br J Neurosurg. 23:282–286. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Campero A, Emmerich J, Socolovsky M,
Martins C, Yasuda A, Agustin Campero A and Rhoton A Jr:
Microsurgical anatomy of the sphenoid ostia. J Clin Neurosci.
17:1298–1300. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hamid O, El Fiky L, Hassan O, Kotb A and
El Fiky S: Anatomic variations of the sphenoid sinus and their
impact on transsphenoid pituitary surgery. Skull Base. 18:9–15.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cavallo LM, de Divitiis O, Aydin S,
Messina A, Esposito F, Iaconetta G, Talat K, Cappabianca P and
Tschabitscher M: Extended endoscopic endonasal transsphenoidal
approach to the suprasellar area: anatomic considerations - part 1.
Neurosurgery. 61(3 Suppl): 24–34. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zada G, Agarwalla PK, Mukundan S Jr, Dunn
I, Golby AJ and Laws ER Jr: The neurosurgical anatomy of the
sphenoid sinus and sellar floor in endoscopic transsphenoidal
surgery. J Neurosurg. 114:1319–1330. 2011.
|
|
13
|
Wang J, Bidari S, Inoue K, Yang H and
Rhoton A Jr: Extensions of the sphenoid sinus: a new
classification. Neurosurgery. 66:797–816. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cavallo LM, de Divitiis O, Aydin S,
Messina A, Esposito F, Iaconetta G, Talat K, Cappabianca P and
Tschabitscher M: Extended endoscopic endonasal transsphenoidal
phenoidal approach to the suprasellar area: anatomic considerations
- part 1. Neurosurgery. 62(6 Suppl 3): 1202–1212. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tatreau JR, Patel MR, Shah RN, McKinney
KA, Wheless SA, Senior BA, Ewend MG, Germanwala AV, Ebert CS Jr and
Zanation AM: Anatomical considerations for endoscopic endonasal
skull base surgery in pediatric patients. Laryngoscope.
120:1730–1737. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Marquardt G, Yahya H, Hermann E and
Seifert V: Direct transnasal approach for pituitary surgery.
Neurosurg Rev. 27:83–88. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Spencer WR, Das K, Nwagu C, Wenk E,
Schaefer SD, Moscatello A and Couldwell WT: Approaches to the
sellar and parasellar region: anatomic comparison of the microscope
versus endoscope. Laryngoscope. 109:791–794. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ceylan S, Koc K and Anik I: Extended
endoscopic approaches for midline skull-base lesions. Neurosurg
Rev. 32:309–319. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Goudakos JK, Markou KD and Georgalas C:
Endoscopic versus microscopic trans-sphenoidal pituitary surgery: a
systematic review and meta-analysis. Clin Otolaryngol. 36:212–220.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Smith SJ, Eralil G, Woon K, Sama A, Dow G
and Robertson I: Light at the end of the tunnel: the learning curve
associated with endoscopic transsphenoidal skull base surgery.
Skull Base. 20:69–74. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jho HD: Endoscopic microscopic
transpedicular thoracic discectomy. Technical note. J Neurosurg.
87:125–129. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jho HD, Carrau RL, Ko Y and Daly MA:
Endoscopic pituitary surgery: an early experience. Surg Neurol.
47:213–223. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jagannathan J, Prevedello DM, Ayer VS,
Dumont AS, Jane JA Jr and Laws ER: Computer-assisted frameless
stereotaxy in transsphenoidal surgery at a single institution:
review of 176 cases. Neurosurg Focus. 20:e92006. View Article : Google Scholar : PubMed/NCBI
|