Introduction
Glycyrrhizin, a naturally occurring licorice
flavonoid glycoside extracted from licorice (Glycyrrhiza
uralensis Fisch) root has been reported to have multiple
functions (1–3); it can scavenge free radicals, have
anti-bacterial, anti-inflammatory, antiviral and anti-ulcer effects
and protect cytochrome enzymes as an antioxidant (1–6).
Studies have shown that glycyrrhizin can regulate the secretion of
various cytokines and the functioning of immunomodulatory effects
in the central nervous system (6,7).
Glycyrrhizin can also improve intracerebral ischemic injury
(6,8,9).
During 20 years of anti-inflammatory application for the treatment
of liver diseases, glycyrrhizin has not generated any toxicity or
side-effects (10–12). However, the effects of glycyrrhizin
on functional recovery following peripheral nerve injury are not
well understood.
The p75 neurotrophin receptor (p75NTR), one of the
nerve signaling proteins, plays a significant role in neuronal
survival, apoptosis and axonal growth in the peripheral and central
nervous systems. p75NTR, as an apoptosis-inducing receptor, has
been demonstrated to induce cell death in developing peripheral
sympathetic neurons and early retinal neurons (13–17).
Küst et al found that p75NTR is also associated with the
inflammatory response of autoimmune encephalomyelitis in C57BL/6
mice (18). The present study
explored the reparative effects of glycyrrhizin on sciatic nerve
injury and the p75NTR-associated immune inflammatory response in a
BALB/c mouse model.
Materials and methods
Reagents and animals
Glycyrrhizin (IUPAC name,
[3β,18α]-30-hydroxy-11,30-dioxoolean-12-en-3-yl
2-O-β-D-glucopyranuronosyl-β-D-glucopyranosiduronic acid; CAS
number, 1405-86-3; molecular formula,
C42H62O16; molecular weight,
822.93 Da; purity, 98%) was purchased from Xi’an Tianxingjian
Natural Bio-products Group (Xi’an, Shaanxi, China), and its
structure is shown in Fig. 1. In
total, 160 healthy adult male BALB/c mice (weighing 25±2 g) were
housed and fed in Jilin University School of Medicine [Experimental
Animal Center, Changchun, China; License No. SCXK (Kyrgyzstan)
2007-0001], with a normal diet, access to water ad libitum
and room temperature conditions. The animal experiments were
approved by Jilin University Ethics Committee.
Modeling and treatment
The sciatic nerve injury model was created as
previously described (19,20), with certain modifications. Briefly,
all mice underwent intraperitoneal injection of 3% ketamine using
100 mg/kg weight for anesthesia. The two cm-longitudinal incisions
were aseptically made on the unilateral rear thigh. The sciatic
nerve cords were completely interrupted at 0.5 cm below the ischial
tuberosity and immediately underwent microsurgical anastomosis
using 11-0 microsutures (Sharpoint™ MicroSutures, Angiotech
Pharmaceuticals, Inc., Vancouver, BC, Canada). Finally, the muscle
and skin were sutured. Mice were randomized into four groups of 40
mice each and treated with different concentrations of glycyrrhizin
for four different time periods. Glycyrrhizin was dissolved in 0.9%
NaCl solution and administered intragastrically, daily for 14
continuous days. The final high-, mid- and low-doses were 20, 10
and 5 g/kg weight/day, respectively. An NaCl solution (0.9%) of an
equivalent volume served as the control. Mice were sacrificed one,
two, four and eight weeks after injury.
Electroneurophysiological testing
Affected sciatic nerves underwent
electroneurophysiological testing using an electromyograph and
evoked potential machine (Keypoint®, Medtronic A/C Inc.,
Skovlunde, Denmark) prior to the mice being sacrificed, as
previously described (20), with
certain modifications. Briefly, the sciatic nerves were exposed at
24°C, then pin electrodes were pierced into the musculi soleus for
wave recording (M point) and grounding electrodes were placed on
the tails. A single current stimulus (10 mA) was employed to
stimulate proximal anastomotic ischial tuberosity (P point) and the
distal sciatic nerve bifurcation point (D point), respectively,
using a parallel stimulating electrode with a fixed space of 3.3 mm
between two tips. Motor nerve conductive velocity (MNCV) equaled
the distance between the P and D points divided by the difference
value of potential latency.
Preparation of spinal cord specimens
The affected spinal cord segments, L4–L6, connected
to injured sciatic nerves, were dissected, dissociated and removed
as previously described (19,20).
Tissues were stored in liquid nitrogen until use in quantitative
polymerase chain reaction (qPCR) and western blotting analysis.
Alternatively, tissues were soaked in 10% neutral buffered formalin
for fixation for 72 h, then dehydrated by gradient alcohols and
embedded in paraffin for use.
Western blotting and qPCR analysis
Western blotting procedures were routinely
performed, as previously described (20,21),
but the goat anti-rat p75NTR polyclonal antibodies (Santa Cruz
Biotechnology, Inc., Dallas, TX, USA) were diluted 100 times with
phosphate-buffered saline containing 1% bovine serum albumin
(Beyotime Inc., Nanjing, China). In the qPCR experiments, the total
RNA was extracted from the tissues using the TRIzol (Invitrogen,
Carlsbad, CA, USA) reagent method. cDNA was synthesized by reverse
transcription using total RNA templates. p75NTR mRNA was detected
by qPCR using cDNA templates. Reduced glyceraldehyde-phosphate
dehydrogenase (GAPDH) served as a housekeeping gene in each
reaction system. The relative p75NTR/GAPDH mRNA levels were
determined. The reaction conditions were 35 cycles at 95°C for 30
sec, 57°C for 60 sec and 72°C for 60 sec. p75NTR and GAPDH primers
were designed based on the NCBI Genbank sequences using Beacon
Designer 7 software (Premier Biosoft Inc., Palo Alto, CA, USA) and
synthesized by Sangon Bioengineering, Inc. (Shanghai, China). The
p75NTR primer sequences were: Sense, 5′-AGTGGCGGATATGGTGAC-3′;
antisense, 5′-GAGCAATAGACAGGAATGAGG-3′; and probe, 5′-TCC
TGACTCCGTTGCTGCTCCCGA-3′. The GAPDH primer sequences were: Sense,
5′-AATGTGTCCGTCGTGGAT CTG-3′; antisense,
5′-CAACCTGGTCCTCAGTGTAGC-3′; and probe,
5′-CGTGCCGCCTGGAGAAACCTGCC-3′.
Histological examination
Paraffin-embedded tissues were sectioned into
transverse slices of 3 μm in thickness. Slices were stained using
combined Marsland, Glees and Erikson’s silver stain (Marsland’s)
and Luxol fast blue (LFB) staining methods, as previously described
(21,22). Nerve fibers were stained black and
myelin was stained blue (23). In
each slice, five microscopic fields of ×400 magnification were
randomly selected and their images captured. The diameter and
number of myelinated fibers was calculated using image analysis
software Image-Pro® Plus 6.0 (Media Cybernetics Inc.,
Silver Spring, MD, USA).
Immunofluorescence staining
Paraffin-embedded tissues were sectioned into slices
of 3 μm in thickness. Indirect immunofluorescence staining was
routinely performed to detect expression in astrocytes. Glial
fibrillary acidic protein (GFAP) was stained blue and
neuron-specific enolase (NSE) was stained green. Goat anti-rat GFAP
and NSE monoclonal antibodies (Santa Cruz Biotechnology, Inc.) were
diluted 1,000 times. The fluorescence intensity of five random
fields was measured using a confocal laser scanning microscope
(FV1200CLSM; Olympus Inc., Beijing, China). The excitation
wavelengths were 488 nm and 561 nm for blue and green fluorescence,
respectively.
Statistical analysis
Data are presented as the mean ± standard deviation.
The SPSS 17.0 statistical package (SPSS, Inc., Chicago, IL, USA)
was used for the statistical analysis. Student’s t-test was used
for comparisons between groups. P<0.05 was used to indicate a
statistically significant difference.
Results
Electrophysiological testing
The functional recovery of the sciatic nerves was
determined by the action potential amplitude and MNCV (Table I). The amplitude and MNCV of the
high- and mid-dose groups at one, two, four and eight weeks after
injury were significantly higher than for the low-dose and control
groups (P<0.05). There was no statistically significant
difference between the high- and mid-dose groups or between the
low-dose and control groups. Administration of glycyrrhizin at 10
and 20 mg/kg/day accelerated the functional recovery of the injured
sciatic nerves in the mice.
 | Table INeuroelectrophysiological potential
amplitude (mV) and MNCV (m/sec) of each dose group at each
time-point following injury (n=10). |
Table I
Neuroelectrophysiological potential
amplitude (mV) and MNCV (m/sec) of each dose group at each
time-point following injury (n=10).
| Glycyrrhizin,
mg/kg/day | Action potential
amplitude, mV | MNCV, m/sec |
|---|
|
|
|---|
| 1 week | 2 weeks | 4 weeks | 8 weeks | 1 week | 2 weeks | 4 weeks | 8 weeks |
|---|
| 20 (High) | 2.32±0.21a | 4.64±0.17a | 26.13±0.16a | 26.44±0.68a | 20.8±0.31a | 41.9±0.59a | 64.8±1.73a | 68.8±1.34a |
| 10 (Mid) | 2.29±0.16a | 4.39±0.19a | 21.87±0.26a | 23.11±0.33a | 19.6±0.38a | 35.5±0.72a | 55.7±1.33a | 60.4±0.61a |
| 5 (Low) | 1.40±0.13 | 2.66±0.07 | 17.56±0.32 | 19.76±0.36 | 13.0±0.19 | 30.6±0.84 | 52.0±0.66 | 55.6±0.43 |
| 0 (Control) | 1.31±0.10 | 2.19±0.15 | 13.29±0.06 | 15.77±0.68 | 10.4±0.17 | 27.6±0.48 | 49.1±0.34 | 46.9±2.17 |
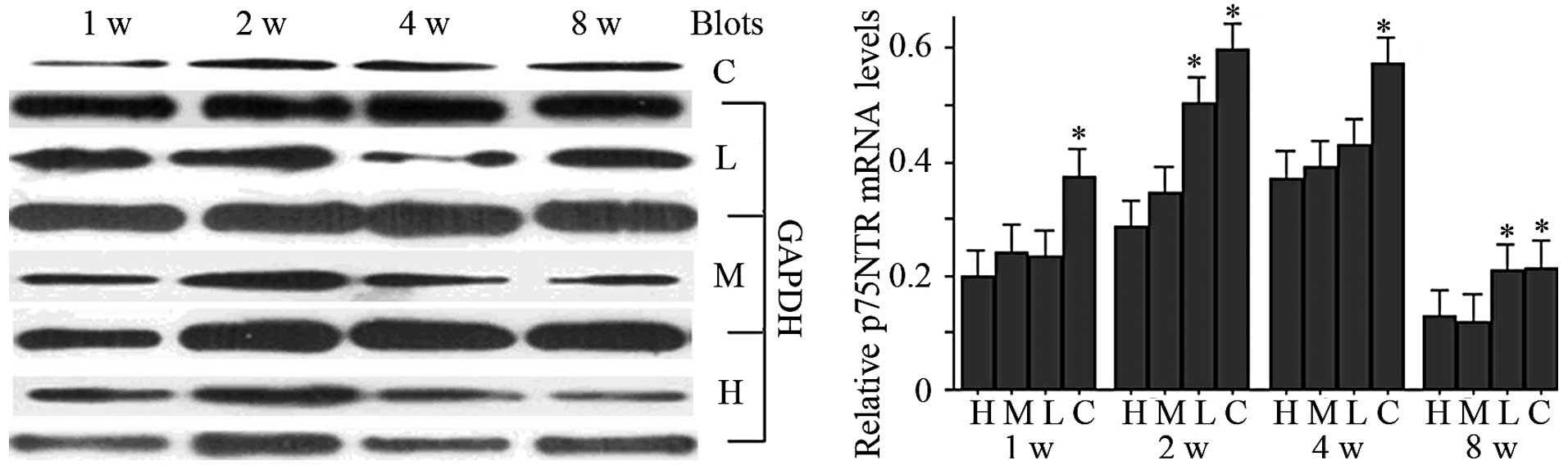
Western blotting and qPCR analysis
The western blotting analysis of the results is
shown in Fig. 2, and the blot
grayscale ratio in Table II.
Levels of p75NTR protein in each group increased markedly between
one and two weeks after injury, and then decreased markedly between
two and eight weeks. At each time-point, each glycyrrhizin
treatment group had significantly reduced p75NTR protein expression
versus the control group (P<0.05 for each). Meanwhile, the high-
and mid-dose groups at one and four weeks showed lower levels of
p75NTR protein versus the low-dose groups (P<0.05 for each), and
the high-dose group expressed significantly reduced levels versus
the low-dose group at two weeks (P<0.05). The qPCR analysis of
the results is shown in Fig. 2.
Overall, the levels of p75NTR mRNA in each group increased
following injury and then decreased. The levels of p75NTR mRNA in
the low-dose and control groups increased between one and four
weeks and then decreased between four and eight weeks. While a
decrease in p75NTR mRNA expression in the high- and mid-dose groups
occurred early between two and eight weeks, p75NTR mRNA expression
in the high- and mid-dose groups was significantly lower versus the
control (P<0.05) at each time-point. Application of
glycyrrhizin, particularly using doses of 20 and 10 mg/kg/day,
markedly inhibited the expression of p75NTR at the protein and mRNA
levels.
 | Table IIRelative grayscale of p75NTR/GAPDH
blots (n=5) of each dose group at each time-point following
injury. |
Table II
Relative grayscale of p75NTR/GAPDH
blots (n=5) of each dose group at each time-point following
injury.
| Relative
grayscale |
|---|
|
|
|---|
| Glycyrrhizin,
mg/kg/day | 1 week | 2 weeks | 4 weeks | 8 weeks |
|---|
| 20 (High) | 0.512±0.020a | 1.088±0.022a | 0.697±0.032a | 0.233±0.025b |
| 10 (Mid) | 0.599±0.026a | 1.207±0.022b | 0.758±0.033a | 0.232±0.019b |
| 5 (Low) | 0.874±0.021b | 1.333±0.022b | 1.111±0.020b | 0.288±0.030b |
| 0 (Control) | 1.311±0.035 | 1.735±0.24 | 1.432±0.013 | 0.523±0.004 |
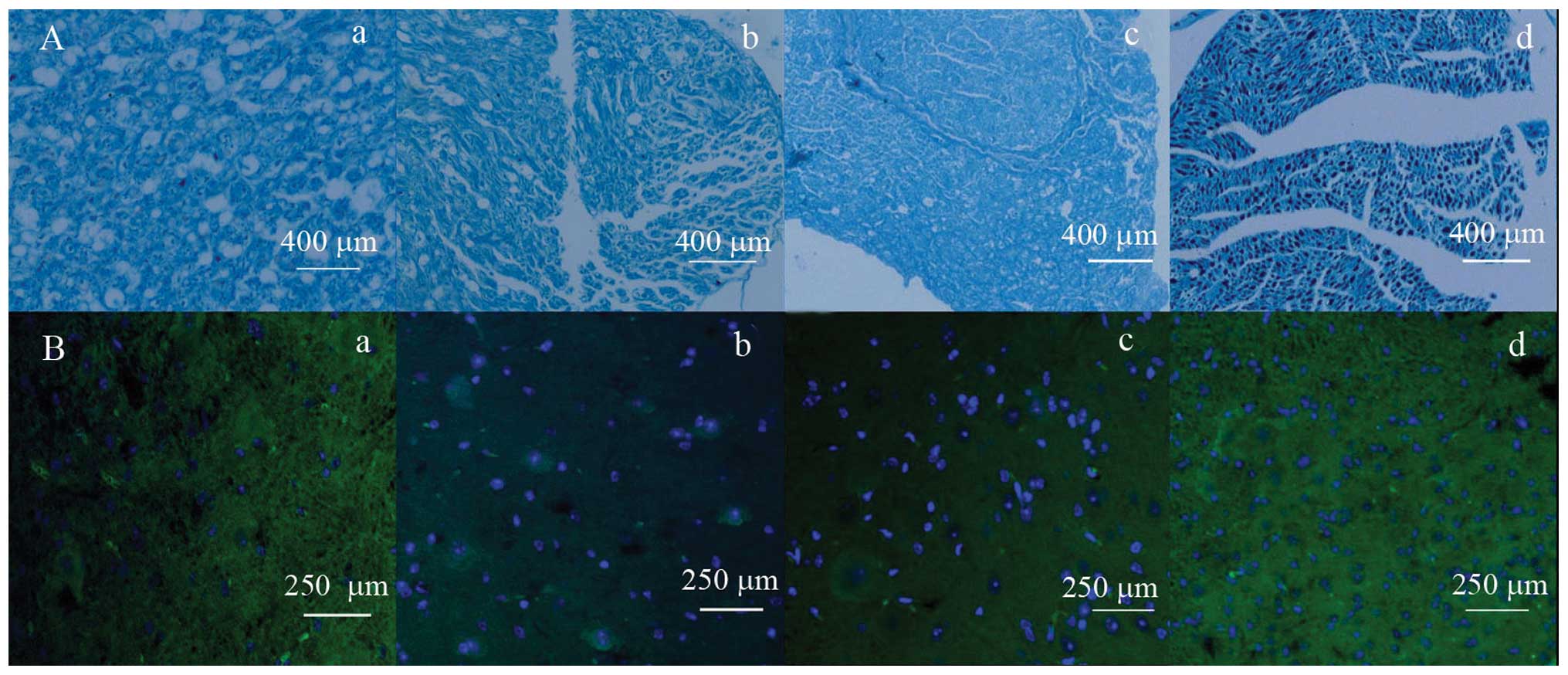
Histological staining
Nerve fibers and myelin were dually stained using
combined Marsland’s and LFB staining methods to detect nerve
regeneration and myelinization at eight weeks after injury. Nerve
fibers were stained black and myelin was stained blue (Fig. 3A). Myelin sheaths in the high-
(Fig. 3Aa) and mid-dose groups
(Fig. 3Ab) were regularly shaped,
having uniform thickness and possessing clear borders or little
fibrous hyperplasia. Myelin sheaths in the low-dose group were of
irregular shape and thickness, and possessed clear borders and
visible fibrous hyperplasia (Fig.
3Ac). Myelin sheaths in the control group were extremely
irregularly shaped with marked fibrous hyperplasia (Fig. 3Ad). Based on these dual stained
images, the number of myelinated fibers in five random microscopic
fields and their average diameters were analyzed using image
analysis software Image-Pro® Plus 6.0 (Table III). The number and diameter of
myelinated fibers in the high- and mid-dose groups at each
time-point were significantly higher than in the low-dose and
control groups, respectively (P<0.05). There was no
statistically significant difference between the high- and mid-dose
groups or between the low-dose and control groups. Using 10 and 20
mg/kg doses of glycyrrhizin per day accelerated nerve
regeneration.
 | Table IIIMyelinated fiber status and
immunofluorescence intensity eight weeks after injury (n=5). |
Table III
Myelinated fiber status and
immunofluorescence intensity eight weeks after injury (n=5).
| Glycyrrhizin,
mg/kg/day | Myelinated fiber
count, n/mm2 | Myelinated fiber
diameter, μm | GFAP fluorescence
intensity (a.u.) | NSE fluorescence
intensity (a.u.) |
|---|
| 20 (High) | 76±3a | 2.46±0.30a |
133.21±24.18a |
466.26±23.12a |
| 10 (Middle) | 70±1a | 2.30±0.17a |
222.88±21.32b |
298.48±32.77b |
| 5 (Low) | 55±3 | 1.96±0.31 |
246.39±15.88b |
288.02±30.15b |
| 0 (Control) | 49±1 | 1.56±0.25 | 299.74±17.31 | 178.71±15.13 |
Immunofluorescence staining
The expression of GFAP and NSE in astrocytes was
detected by indirect immunofluorescence staining at eight weeks
after injury. GFAP staining appeared as blue dots and NSE stains
appeared as an abundance of green in the cytoplasm (Fig. 3B). Overall, GFAP and NSE were
expressed in each of the four groups. The high-dose group (Fig. 3Ba) was less expression of GFAP, as
indicated by fewer blue dots and more expression of NSE, as
indicated by more green dots in cytoplasm, than the mid-dose
(Fig. 3Bb) and low-dose (Fig. 3Bc) groups. The mid-dose and
low-dose groups were less expression of GFAP and more NSE in the
cytoplasm than the controls (Fig.
3Bd). In the control group, certain specimens were observed to
form glial scars. Fluorescence densities in each group are shown in
Table III. Each glycyrrhizin
treatment group had less blue fluorescence and more green
fluorescence than the controls (P<0.05 for each). The high-dose
group had less blue fluorescence and more green fluorescence than
the low-dose and control groups (P<0.05 for each). The
application of glycyrrhizin increased NSE and reduced GFAP
expression in the astrocytes in order to improve sciatic nerve
repair.
Discussion
In the present study, 10 and 20 mg/kg doses of
glycyrrhizin were shown to promote the functional recovery of
sciatic nerves. This was verified by measuring the potential
amplitude and MNCV in mice. Dual staining of the affected spinal
cord segments, L4–L6, connected to injured sciatic nerves, was
performed to examine the structural evidence that 10 and 20 mg/kg
doses of glycyrrhizin can elevate the number and diameter of
myelinated fibers, accelerating nerve regeneration. Subsequently,
the GFAP- and NSE-marked fluorescence in astrocytes was employed to
assess the nerve damage-associated molecules. Upregulation of GFAP
in astrocytes partially marks the formation of glial scars, showing
that astrocytes interact with fibrous tissues to reconstruct the
glial layers around the central injury core (24,25).
NSE, a homodimer, is a biomarker for mature neurons and
neuroendocrine cells (26).
High-dose glycyrrhizin results in increased NSE and reduced GFAP
expression in astrocytes, indicating sciatic nerve repair.
Nerve repair and regeneration following injury
involves a complex biological process, including an inflammatory
response, an adhesion reaction, a synergistic effect of the
extracellular matrix, the regulatory function of neurotrophic
factors on the regeneration, synthesis and release of a
neurotransmitter, the formation and extension of a growth cone and
final neuronal regeneration (9,14).
Following nerve injury, the local excessive immune response leads
to expression of inflammatory response genes, forming a local glial
scar. This scar can directly affect the recovery of nerve
conduction. Reducing the scar can provide a favorable external
environment for the repair and regeneration of nerves.
In the present study, glycyrrhizin, particularly at
doses of 10 and 20 mg/kg, markedly inhibited the expression of
p75NTR at the protein and mRNA levels throughout the eight weeks of
the experiment. This downregulation of p75NTR synchronizes the
functional recovery of the sciatic nerve, structural myelination
and neuronal regeneration and the improvement of molecular markers.
Considering p75NTR is an apoptosis-inducing receptor and is
associated with the inflammatory response of autoimmune
encephalomyelitis (13–18), the downregulation of p75NTR may
likely result in the reduction of the local inflammatory response,
lessening the glial scar and recovering nerve conduction function.
Simultaneously, downregulation of p75NTR may attenuate the
apoptosis-inducing receptor function of p75NTR, promoting neuronal
regeneration and the expression of marker molecules. Therefore,
sciatic nerve regeneration is associated with the downregulation of
p75NTR.
In conclusion, 10 and 20 mg/kg doses of glycyrrhizin
per day are effective in promoting sciatic nerve regeneration and
functional repair, and such regeneration or repair is associated
with the downregulation of p75NTR.
References
|
1
|
Cinatl J, Morgenstern B, Bauer G, Chandra
P, Rabenau H and Doerr HW: Glycyrrhizin, an active component of
liquorice roots, and replication of SARS-associated coronavirus.
Lancet. 361:2045–2046. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Dhiman RK and Chawla YK: Herbal medicines
for liver diseases. Dig Dis Sci. 50:1807–1812. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kim SC, Byun SH, Yang CH, Kim CY, Kim JW
and Kim SG: Cytoprotective effects of Glycyrrhizae radix extract
and its active component liquiritigenin against cadmium-induced
toxicity (effects on bad translocation and cytochrome c-mediated
PARP cleavage). Toxicology. 197:239–251. 2004. View Article : Google Scholar
|
|
4
|
Jo EH, Kim SH, Ra JC, Kim SR, Cho SD, Jung
JW, et al: Chemopreventive properties of the ethanol extract of
Chinese licorice (Glycyrrhiza uralensis) root: induction of
apoptosis and G1 cell cycle arrest in MCF-7 human breast cancer
cells. Cancer Lett. 230:239–247. 2005.PubMed/NCBI
|
|
5
|
Yuan H, Ji WS, Wu KX, Jiao JX, Sun LH and
Feng YT: Anti-inflammatory effect of Diammonium Glycyrrhizinate in
a rat model of ulcerative colitis. World J Gastroenterol.
12:4578–4581. 2006.PubMed/NCBI
|
|
6
|
Kim SW, Jin Y, Shin JH, Kim ID, Lee HK,
Park S, Han PL and Lee JK: Glycyrrhizic acid affords robust
neuroprotection in the postischemic brain via anti-inflammatory
effect by inhibiting HMGB1 phosphorylation and secretion. Neurobiol
Dis. 46:147–156. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gong G, Yuan LB, Hu L, Wu W, Yin L, Hou
JL, Liu YH and Zhou LS: Glycyrrhizin attenuates rat ischemic spinal
cord injury by suppressing inflammatory cytokines and HMGB1. Acta
Pharmacol Sin. 33:11–18. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hwang IK, Lim SS, Choi KH, Yoo KY, Shin
HK, Kim EJ, et al: Neuroprotective effects of roasted licorice, not
raw form, on neuronal injury in gerbil hippocampus after transient
forebrain ischemia. Acta Pharmacol Sin. 27:959–965. 2006.
View Article : Google Scholar
|
|
9
|
Ohnishi M, Katsuki H, Fukutomi C,
Takahashi M, Motomura M, Fukunaga M, et al: HMGB1 inhibitor
glycyrrhizin attenuates intracerebral hemorrhage-induced injury in
rats. Neuropharmacology. 61:975–980. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ogiku M, Kono H, Hara M, Tsuchiya M and
Fujii H: Glycyrrhizin prevents liver injury by inhibition of
high-mobility group box 1 production by Kupffer cells after
ischemia-reperfusion in rats. J Pharmcol Exp Ther. 339:93–98. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
van Rossum TG, Vulto AG, Hop WC and Schalm
SW: Glycyrrhizin-induced reduction of ALT in European patients with
chronic hepatitis C. Am J Gastroenterol. 96:2432–2437.
2001.PubMed/NCBI
|
|
12
|
Kumada H: Long-term treatment of chronic
hepatitis C with glycyrrhizin [stronger neo-minophagen C (SNMC)]
for preventing liver cirrhosis and hepatocellular carcinoma.
Oncology. 62(Suppl 1): 94–100. 2002.
|
|
13
|
Majdan M, Lachance C, Gloster A, Aloyz R,
Zeindler C, Bamji S, et al: Transgenic mice expressing the
intracellular domain of the p75 neurotrophin receptor undergo
neuronal apoptosis. J Neurosci. 17:6988–6998. 1997.PubMed/NCBI
|
|
14
|
Frade JM and Barde YA: Genetic evidence
for cell death mediated by nerve growth factor and the neurotrophin
receptor p75 in the developing mouse retina and spinal cord.
Development. 126:683–690. 1999.PubMed/NCBI
|
|
15
|
Harada C, Harada T, Nakamura K, Sakai Y,
Tanaka K and Parada LF: Effect of p75NTR on the regulation of
naturally occurring cell death and retinal ganglion cell number in
the mouse eye. Dev Biol. 290:57–65. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jansen P, Giehl K, Nyengaard JR, Teng K,
Lioubinski O, Sjoegaard SS, et al: Roles for the pro-neurotrophin
receptor sortilin in neuronal development, aging and brain injury.
Nat Neurosci. 10:1449–1457. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Paul CE, Vereker E, Dickson KM and Barker
PA: A pro-apoptotic fragment of the p75 neurotrophin receptor is
expressed in p75NTRExonIV null mice. J Neurosci. 24:1917–1923.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Küst B, Mantingh-Otter I, Boddeke E and
Copray S: Deficient p75 low-affinity neurotrophin receptor
expression does alter the composition of cellular infiltrate in
experimental autoimmune encephalomyelitis in C57BL/6 mice. J
Neuroimmunol. 174:92–100. 2006.
|
|
19
|
Du JS, Zhao Q, Zhang YL, Wang Y and Ma M:
7,8-dihydroxycoumarin may promote sciatic nerve regeneration by
suppressing NF-κB expression in mice. Mol Med Rep. 8:1525–1530.
2013.PubMed/NCBI
|
|
20
|
Cao J, Jiang YW, Sun Y, Zhang XZ, Li LS
and Man Y-H: Immunosuppression of NF-κB by intragastric brazilein
in motor neuron of spinal cord connected with injured sciatic nerve
in mouse. Biomed Res (India). 23:199–206. 2012.
|
|
21
|
Marsland TA, Glees P and Erikson LB:
Modification of the Glees silver impregnation for paraffin
sections. J Neuropathol Exp Neurol. 13:587–591. 1954. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang XH and Chen JJ: The mechanism of
astragaloside IV promoting sciatic nerve regeneration. Neural Regen
Res. 8:2256–2265. 2013.PubMed/NCBI
|
|
23
|
Timperley WR: ACP Best Practice No 158.
Neuropathology J Clin Pathol. 53:255–265. 2000.PubMed/NCBI
|
|
24
|
Bunge MB, Bunge RP and Ris H:
Ultrastructural study of remyelination in an experimental lesion in
adult cat spinal cord. J Biophys Biochem Cytol. 10:67–94. 1961.
View Article : Google Scholar
|
|
25
|
Smith ME and Eng LF: Glial fibrillary
acidic protein in chronic relapsing experimental allergic
encephalomyelitis in SJL/J mice. J Neurosci Res. 18:203–208. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yokobori S, Hosein K, Burks S, Sharma I,
Gajavelli S and Bullock R: Biomarkers for the clinical differential
diagnosis in traumatic brain injury - systematic review. CNS
Neurosci Ther. 19:556–565. 2013. View Article : Google Scholar
|