Introduction
The Gram-negative bacterium, Pseudomonas
aeruginosa, is a ubiquitous and versatile microorganism that is
able to survive in soil, marsh and marine habitats on plant and
animal tissue and on non-living surfaces (1). In a clinical context, P.
aeruginosa functions as an opportunistic pathogen and is a
leading cause of nosocomial infections, particularly chronic lung
infections in patients with cystic fibrosis (CF) or patients with
other immune deficiencies (2).
Infections caused by P. aeruginosa have become a worldwide
problem due to the increasing rates of morbidity and mortality, and
the expense associated with hospitalized patients (3).
P. aeruginosa is able to form a biofilm on
numerous types of surface, including the lung tissue of patients
with CF (4), and on abiotic
surfaces, including contact lenses and catheter lines (5,6). As a
result of this biofilm-forming ability, P. aeruginosa
infections are difficult to eliminate, particularly lung infections
in CF patients, since the bacteria are highly resistant to a range
of antimicrobial agents (7). Certain
antimicrobial agents, including azithromycin (AZM),
third-generation cephalosporins, carbapenems, monobactams,
colistin, tobramycin and quinolones, are effective against the
majority of P. aeruginosa strains (8). However, P. aeruginosa typically
develops induced resistance to these agents via a number of
mechanisms, including horizontal transfer or overexpression of
resistance genes, or gene mutations that target the treatment drug
(9). In addition, the ability of
P. aeruginosa to form a biofilm results in a high level of
antibiotic resistance and virulence in patients, since the biofilm
is able to protect the bacterium from the inhibitory effects of
antibiotics and the host immune system (10). Due to this capability to form a
biofilm, P. aeruginosa is a model organism in bacterial
biofilm research (11). A variety of
agents have been investigated with the aim of suppressing the
biofilm formation, including products derived from plants (12–14).
Sodium houttuyfonate [SH; chemical composition,
CH3(CH2)8COCH2CHOHSO3Na]
is a compound of sodium bisulfite and houttuynin. Houttuynin is the
primary constituent of the volatile oil produced by Houttuynia
cordata Thunb, a wild perennial herb used widely in traditional
Chinese medicine (15). SH is easily
dissolved in hot water and alkaline solutions, is slightly soluble
in water and ethanol, and is insoluble in chloroform and benzene
(16). In China, SH has been
clinically used as an antimicrobial agent for numerous years, and
has been reported to effectively inhibit Gram-positive bacterial
infections, including Staphylococcus aureus, Moraxella
catarrhalis, Haemophilus influenzae and Streptococcus
pneumoniae (17). Ye et
al reported that Gram-positive bacteria were more sensitive to
houttuyfonate homologs compared with Gram-negative bacteria, owing
to the interactional differences between SH and the cell membrane
(18). In addition, a previous study
of transcriptional and functional analysis demonstrated SH-mediated
inhibition by autolysis in S. aureus (19). However, there are few reports
characterizing the effects of SH on the inhibition of Gram-negative
bacteria, such as P. aeruginosa.
In the present study, the inhibitory activity of SH
against biofilm formation and alginate production on a clinical
strain of P. aeruginosa (AH16) was investigated in
vitro. Alterations in the cellular morphology of P.
aeruginosa following treatment with SH were observed using
scanning electron microscopy (SEM). Furthermore, reverse
transcription-quantitative polymerase chain reaction (RT-qPCR) was
employed to identify any changes in the expression levels of genes
associated with alginate biosynthesis as a result of the SH
treatment.
Materials and methods
Bacteria strain and materials
An AH16 P. aeruginosa strain was isolated
from a patient with chronic pneumonia in the First Affiliated
Hospital of Anhui University of Traditional Chinese Medicine
(Hefei, China). This study was approved by the ethics committee of
the First Affiliated hospital of Anhui University of Traditional
Chinese Medicine. The strain was found to exhibit higher virulence
and produce more biofilm when compared with the wild-type P.
aeruginosa. SH and AZM were obtained from the National
Institute for the Control of Pharmaceutical and Biological Products
(Beijing, China). Luria-Bertani (LB), Mueller-Hilton (MH) and
Tryptic Soy Broth (TSB) media were purchased from Beijing Aoboxing
Bio-Tech Co., Ltd. (Beijing, China), and crystal violet solution
was purchased from bioMérieux, Inc. (Craponne, France). Alginate
standards were purchased from Sigma-Aldrich (St. Louis, MO, USA),
and Taq polymerase and PCR primers were purchased from Takara Bio,
Inc. (Otsu, Japan).
The AH16 strain was inoculated into LB broth, and
grown in a constant-temperature shaker (GLY; Fuma, Shanghai, China)
at 220 rpm for 6 h at 37°C. Subsequently, the bacteria were
harvested using a GL-20G-II high-speed refrigerated centrifuge
(Fuma) at 1,630 × g for 10 min. The supernatant was discarded and
the precipitate was resuspended with phosphate-buffered saline
(PBS; pH 7.2), after which the samples were centrifuged again at
1,630 rpm for 10 min. The harvested cells were resuspended in PBS
(pH 7.2) and adjusted to 2×105 colony-forming units
(CFU)/ml using the growth curve method (20).
Evaluation of the effects of SH on
biofilm formation
Sauer et al (21) previously described five stages of
biofilm formation as follows: i) Reversible attachment (0–2 h); ii)
irreversible attachment (2 h); iii) maturation stage 1 (day 3); iv)
maturation stage 2 (day 6); and v) dispersion (day 9–12). On the
basis of these stages, days 1, 3 and 7 were selected as the three
time points for observing the growth of the biofilm and to detect
any suppression as a result of the drugs used. The minimum
inhibitory concentrations (MICs) of SH and AZM were determined to
be 512 and 16 µg/ml, respectively, using the microdilution method
(22).
Fresh stock solutions of SH and AZM were prepared in
MH broth and filtered through a 0.22-µm filter (EMD Millipore,
Billerica, MA, USA). Six treatment groups were analyzed, which were
treated with SH or AZM at a MIC of 0.5, 1 or 2 (Fig. 1). In a 96-well plate, 28 wells were
filled with TSB medium, of which four wells were used for each of
the six treatment groups, with four further wells used as the
control. Next, 200 µl P. aeruginosa suspension was added and
the plate was incubated at 37°C. After 24 h, the culture suspension
was discarded, and the sedimented bacteria were washed in 1 ml PBS.
Fresh TSB medium with the aforementioned concentrations of
antibiotics was added to the relevant wells. The control wells
contained TSB medium with no antibiotics. In accordance with the
method previously described by O'Toole (23), the medium was exchanged for fresh
medium with the appropriate antibiotics every other day. At the end
of days 1 (attachment), 3 (maturation stage 1) and 7 (maturation
stage 2), cold PBS (4°C) was used to wash the planktonic bacteria
in one well of each group, after which 200 µl crystal violet
solution (1%) was added to the wells and left for 20 min. The wells
were rinsed with deionized water until no crystal violet was
visible, following which the wells were dried and 95% alcohol was
added for destaining. Subsequently, the destained solution from
each well was transferred to a cuvette and diluted with 3 ml
alcohol (95%). The optical density (OD) of the solutions was
determined at 570 nm using a UV spectrophotometer (U-2000; Hitachi,
Ltd., Tokyo, Japan). The OD value of the negative control was set
as 100%, from which the growth of the biofilm was calculated for
each group.
Evaluation of the effects of SH on
alginate production
Alginate is the predominant constituent of the P.
aeruginosa biofilm, and functions as a barrier to protect the
bacteria from antibiotics and the humoral and cellular host defense
system (24,25). Therefore, the effects of SH on
alginate production in the P. aeruginosa biofilm were
evaluated.
The groups analyzed were the same as those described
previously (0.5x, 1x and 2x MIC groups each for SH and AZM). A
sterile coverglass was used as a carrier for each group. The
coverglasses were placed into the wells of a six-well culture
plate, and 2 ml TSB medium and 0.2 ml bacterial suspension
(2×105 CFU/ml) were added. The plates were incubated for
1 day at 37°C, after which the medium was discarded. For each well,
the coverglass was removed, and the planktonic bacteria on the
coverglass were washed out with sterile PBS. The coverglass was
subsequently returned to the well prior to the addition of fresh
medium. As aforementioned, the medium added to the antibiotic
groups contained the appropriate antibiotic, while the negative
control contained medium without antibiotics. All the described
steps were repeated every 24 h. At the end of days 1, 3 and 7 of
the antibiotic treatment, the coverglasses were removed and the
planktonic bacteria were rinsed with PBS. Subsequently, the
coverglasses were each placed into a test-tube containing 6 ml PBS
and 1.2 ml sulfuric acid and sodium borate compound which was
prepared by dissolving 2.52 g sulfuric in 1:l sulfuric acid. The
tubes were boiled for 5 min and then cooled to 4°C. Each coverglass
received 20 µl hydroxybiphenyl (1%) for colorization, after which
the coverglasses were sonicated for 30 min in an ultrasonic
cleaning bath (2800; Branson Ultrasonics, Danbury, CT, USA) and the
absorbance was measured at 570 nm using the Hitachi UV spectrometer
(26). The alginate production of
each strain, defined as the quantity of alginate (µg/ml) adhered to
the coverglass, was calculated using a standard curve of alginate
standards following the subtraction of the blank control.
SEM imaging of the biofilm
morphology
Preparation of the carrier coverslips was performed
as aforementioned, up to the point at which the planktonic bacteria
were rinsed with PBS. Subsequently, the biofilm was subjected to
silver staining (27), and cellular
morphology was examined under SEM (Sirion 200 field-emission; FEI
Company, Hillsboro, OR, USA) at 500 kV and x40,000
magnification.
RT-qPCR of the alginate biosynthesis
genes
RT-qPCR was performed to determine the expression
levels of genes associated with alginate biosynthesis. The majority
of the alginate biosynthesis genes are clustered in the algD
operon (28). Alginate production is
highly regulated, and AlgR is among one of the key regulators
(29,30). Therefore, the algD and
algR genes were selected for subjection to RT-qPCR in order
to identify whether SH was able to attenuate the expression levels
of alginate biosynthesis-associated genes. In addition, the
expression levels of the biofilm-associated genes, pilL and
rhlI, were evaluated.
Initially, 1 ml biofilm bacterial suspension
culture, treated with 1x or 2x MIC concentrations of SH, was
centrifuged at 10,800 × g for 1 min. The supernatant was discarded
and the pellet was resuspended in 1 ml TRIzol reagent (Invitrogen
Life Technologies, Carlsbad, CA, USA) at room temperature for 20
min. Each sample received 200 µl chloroform and was vortexed for 15
sec, followed by centrifugation at 4,200 × g at 4°C for 15 min. The
liquid layer of the mixture was transferred to a fresh tube
containing 480 µl isopropanol, after which the tube was vortexed
for 15 sec and centrifuged at 4,200 × g at 4°C for 15 min. The
total RNA was washed with 70% ethanol, and the tube was centrifuged
again at 4,200 × g and 4°C for 10 min. The liquid was discarded,
and the total RNA was dissolved in RNase-free water. Purified RNA
(2 µg) was reverse transcribed into cDNA using a one-step method
commercial kit (Takara Bio, Inc.). The primers were designed to
produce products of 180–240 bp, and the sequences are detailed in
Table I. The rpoD gene was
used as a housekeeping control. PCR assays were conducted using an
ABI Prism thermal cycler (Applied Biosystems Life Technologies,
Foster City, CA, USA) using the following program: Initial
denaturation for 5 min at 95°C, followed by 40 cycles of 95°C for
15 sec, 58°C for 10 sec and 72°C for 20 sec. In the experiment,
there were three independent biological replicates and two
technical replicates. The calculated threshold cycle (Ct) of each
gene was normalized against the Ct of the rpoD gene
amplified from the corresponding sample. Fold change was calculated
according to the 2−ΔΔCt method (31).
 | Table I.Primers used for the reverse
transcription-quantitative polymerase chain reaction of the
alginate biosynthesis genes. |
Table I.
Primers used for the reverse
transcription-quantitative polymerase chain reaction of the
alginate biosynthesis genes.
| Gene | Sequence 5′→3′ |
|---|
| algR | F:
AGACCGGCTACGGCTACA |
|
| R:
GCGTCGTGCTTCTTCAGTT |
| algD | F:
AGAAGTCCGAACGCCACA |
|
| R:
TCCAGCTCGCGGTAGAT |
| rpoD | F:
AGGCCGTGAGCAGGGAT |
|
| R:
GGTGGTGCGACCGATGT |
Statistical analysis
All data were analyzed by SPSS statistical software,
version 17.0 (SPSS, Inc., Chicago, IL, USA), and expressed as the
mean ± standard deviation. Difference between the various group
data were compared using Students T-test. P-values were calculated
by comparison of the data of drug treatment and control groups.
Experiments were repeated in quadruplicate, and the results are
expressed as the mean of the four replicates.
Results
Effects of SH on biofilm
formation
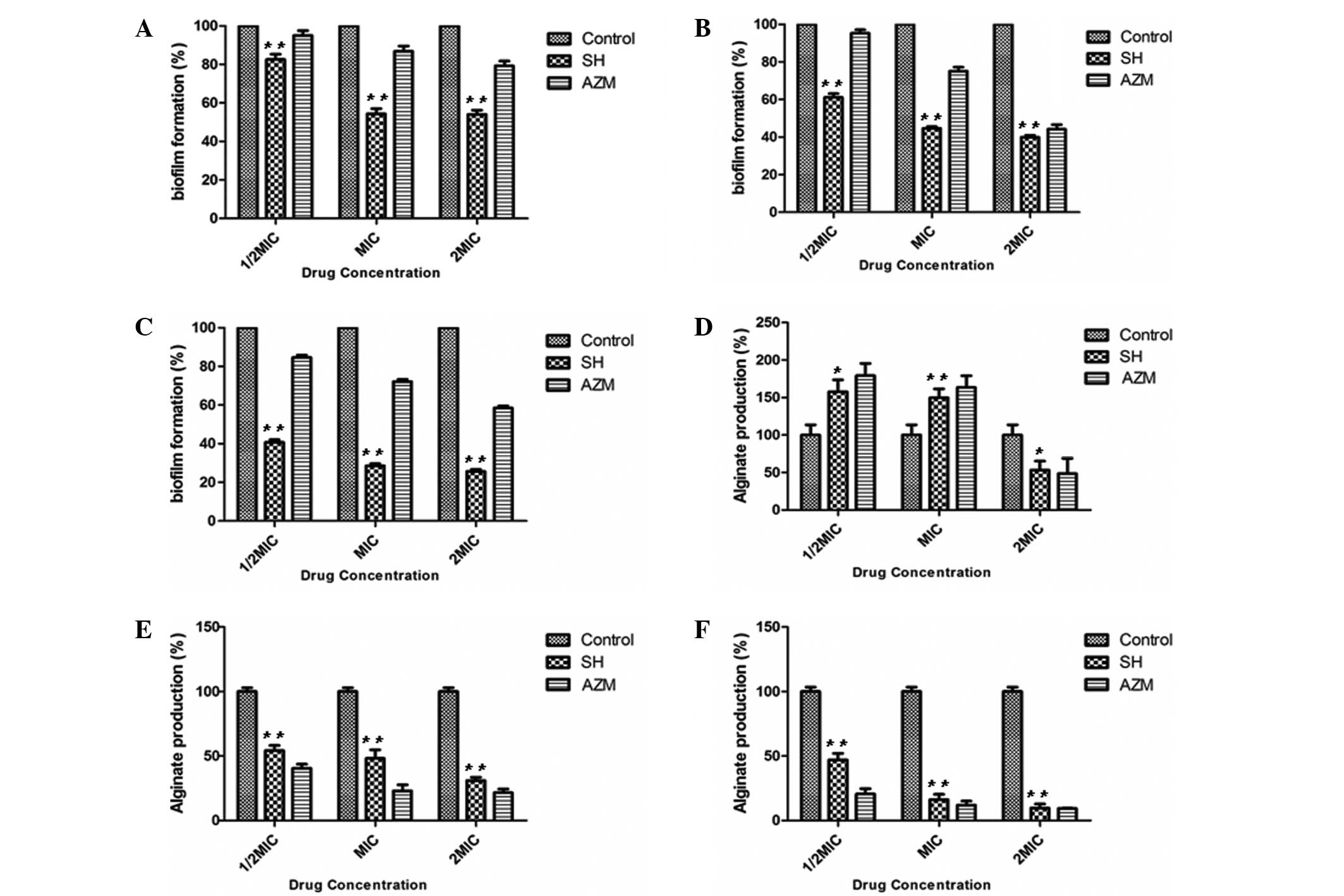
By day 1 of the antibiotic treatment, a significant
effect on biofilm growth was observed in the 0.5x, 1x and 2x MIC SH
treatment groups when compared with the control group (P<0.01;
Fig. 1A), with the 1x and 2x MIC
treatments producing a more marked effect compared with the 0.5x
MIC treatment. This effect continued throughout day 3 (Fig. 1B) and day 7 (Fig. 1C), with the higher concentrations
exhibiting the most notable effects. On day 7 (Fig. 1C), all the biofilms were reduced to
<50% of the control (P<0.01). In addition, SH was more
effective at repressing biofilm formation compared with AZM. These
results indicated that SH significantly inhibited biofilm formation
in P. aeruginosa (AH16) at various developmental stages.
Effects of SH on alginate
production
At day 1 of the antibiotic treatment (Fig. 1D), there was a statistically
significant (P<0.05) reduction in the alginate biofilm in the 2x
MIC SH group when compared with the control group; however, this
effect was not observed in the 0.5x or 1x MIC SH groups, which was
similar to the effects of AZM. By day 3 (Fig. 1E), the 0.5x, 1x and 2x MIC SH
treatment groups had induced significant reductions in alginate
production (P<0.01) in a dose-dependent manner. At day 7
(Fig. 1F), the inhibitory effects of
all three SH concentrations had produced reductions in the alginate
production of ≥50% compared with the control. Notably, the
inhibitory effects of SH were lower compared with AZM at 0.5x and
1x MIC, but were comparable to AZM at 2x MIC. These results
demonstrated that SH exerts an inhibitory effect on alginate
production in the P. aeruginosa AH16 clinical strain.
Biofilm morphology of P. aeruginosa
treated with SH
In the SEM images, the bacterial biofilm
morphologies were observed to clearly vary between the two groups.
In the control group (Fig. 2A), the
bacteria were completely covered by a thick mucous biofilm, and the
entire structure exhibited a mushroom shape with interlaced inner
pore channels towards the surface. By contrast, in the 1x MIC SH
treatment group (Fig. 2B), there was
no evident biofilm structure and the rod-shaped bacteria were
dispersed over the mucus. Thus, the morphology of the biofilm cells
indicated the inhibition of alginate production by SH.
Changes in the expression levels of
the alginate biosynthesis genes following SH treatment
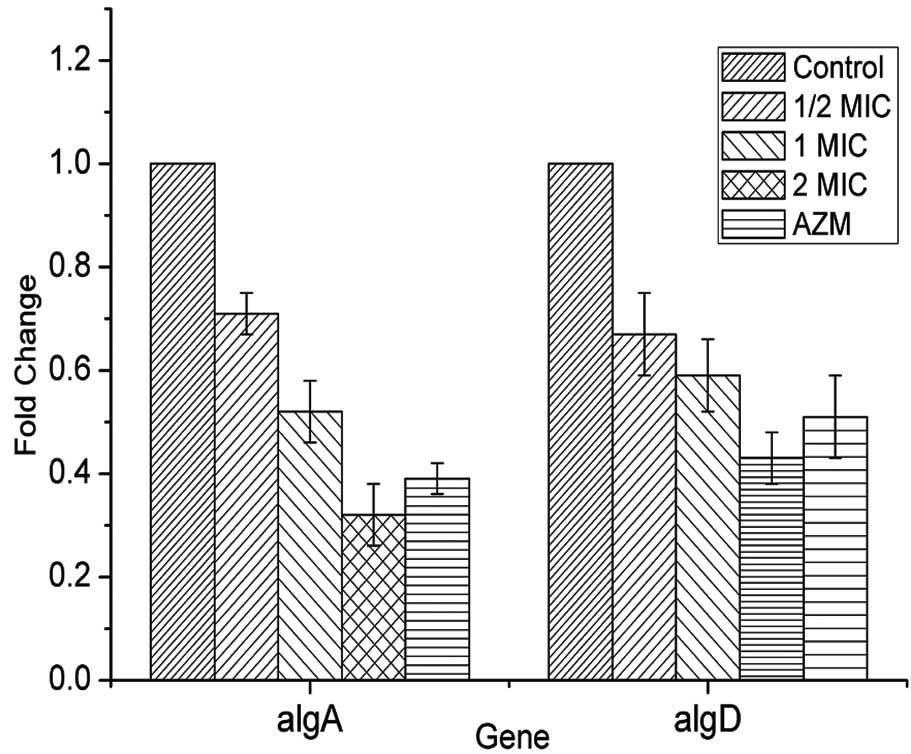
Expression levels of the algD and algR
genes were reduced in the SH treatment groups when compared with
the control group (Fig. 3). This
reduction was dose-dependent, with the expression level being more
notably reduced in the group treated with the highest concentration
of SH (2x MIC) compared with the medium concentration (1x MIC). By
contrast, the biofilm-associated genes, pilL and
rhlI, were not downregulated transcriptionally by SH (data
not shown). These results indicated that SH inhibits biofilm
formation in P. aeruginosa by repressing the expression of
genes associated with alginate biosynthesis.
Discussion
The production of a biofilm by a bacterial colony is
a key form of growth in environmental and clinical contexts. There
are three critical phases of biofilm development, namely adherence,
maturation and dispersion. Each of these stages involves
reinforcement by, or modulation of, the extracellular matrix
(32). The biofilm-forming ability
of P. aeruginosa is the primary factor enhancing the high
level of virulence and antibiotic resistance. The current study
investigated the inhibitory capacity of the plant-derived product,
SH, which has been reported to possess the ability to inhibit the
growth of clinical P. aeruginosa strains. SH was observed to
significantly repress biofilm formation in the attachment and
maturation stages of biofilm development. In addition, the
morphology of the biofilm was affected by SH treatment. Thus, the
results indicate that SH is able to inhibit the formation of
biofilms by a clinical strain of P. aeruginosa.
Overproduction of the exopolysaccharide alginate
provides P. aeruginosa with a selective advantage, and
facilitates survival in the lungs of patients with CF (33). The results of the present study
revealed that SH significantly inhibits alginate production at the
biofilm maturation stage (Fig.
1E–F); however, only the highest concentration of SH was able
to repress alginate production at the attachment stage (Fig. 1D), which is comparable to AZM.
Furthermore, the alginate biosynthesis genes, algD and
algR, were found to be downregulated as a result of the SH
treatment (Fig. 3). Thus, these
results indicate that SH inhibits biofilm formation in P.
aeruginosa by repressing alginate production. Expression of the
alginate machinery and biosynthetic enzymes are controlled by the
extracytoplasmic sigma factor (33).
However, further study is required to elucidate the mechanisms
underlying the SH-induced repression of alginate biosynthesis genes
in P. aeruginosa.
In conclusion, the present study indicated that SH
significantly inhibits biofilm formation in a clinical strain of
P. aeruginosa, and markedly reduced the expression of the
primary biofilm constituent, alginate, at various stages of growth.
Observations of cellular morphology demonstrated that SH alters the
biofilm structure of P. aeruginosa, while the results of the
RT-qPCR analysis indicated that SH may inhibit biofilm formation by
repressing the expression of alginate biosynthesis genes. Thus, the
present study provides novel insights into the effects of SH on
biofilm formation in the P. aeruginosa AH16 strain, and into
potential underlying mechanisms. However, further studies are
required to confirm the molecular mechanisms underlying the effects
of SH against biofilm formation in P. aeruginosa.
Acknowledgements
The authors thank Changfeng Zhang from the Clinical
Laboratory at the First Affiliated Hospital of Anhui University of
Traditional Chinese Medicine (Hefei, China) for providing the P.
aeruginosa clinical strain. This study was supported by a grant
from the National Natural Science Foundation of China (no.
81173629).
References
|
1
|
Hardalo C and Edberg SC: Pseudomonas
aeruginosa: Assessment of risk from drinking water. Crit Rev
Microbiol. 23:47–75. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bodey GP, Bolivar R, Fainstein V and
Jadeja L: Infections caused by Pseudomonas aeruginosa. Rev Infect
Dis. 5:279–313. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Rosenthal VD, Maki DG, Jamulitrat S, et
al: INICC Members: International Nosocomial Infection Control
Consortium (INICC) report, data summary for 2003–2008, issued June
2009. Am J Infect Control. 38:95–104. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
George AM, Jones PM and Middleton PG:
Cystic fibrosis infections: Treatment strategies and prospects.
FEMS Microbiol Lett. 300:153–164. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dutta D, Cole N and Willcox M: Factors
influencing bacterial adhesion to contact lenses. Mol Vis.
18:14–21. 2012.PubMed/NCBI
|
|
6
|
Mittal R, Aggarwal S, Sharma S, Chhibber S
and Harjai K: Urinary tract infections caused by Pseudomonas
aeruginosa: A minireview. J Infect Public Health. 2:101–111. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Breidenstein EB, de la Fuente-Núñez C and
Hancock RE: Pseudomonas aeruginosa: All roads lead to resistance.
Trends Microbiol. 19:419–426. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fothergill JL, Winstanley C and James CE:
Novel therapeutic strategies to counter Pseudomonas aeruginosa
infections. Expert Rev Anti Infect Ther. 10:219–235. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zavascki AP, Carvalhaes CG, Picao RC and
Gales AC: Multidrug-resistant Pseudomonas aeruginosa and
Acinetobacter baumannii: Resistance mechanisms and implications for
therapy. Expert Rev Anti Infect Ther. 8:71–93. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Høiby N, Ciofu O, Johansen HK, et al: The
clinical impact of bacterial biofilms. Int J Oral Sci. 3:55–65.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
O'Toole GA and Kolter R: Flagellar and
twitching motility are necessary for Pseudomonas aeruginosa biofilm
development. Mol Microbiol. 30:295–304. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Carneiro VA, Santos HS, Arruda FV,
Bandeira PN, et al: Casbane diterpene as a promising natural
antimicrobial agent against biofilm-associated infections.
Molecules. 16:190–201. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hengzhuang W, Wu H, Ciofu O, Song Z and
Høiby N: In vivo pharmacokinetics/pharmacodynamics of colistin and
imipenem in Pseudomonas aeruginosa biofilm infection. Antimicrob
Agents Chemother. 56:2683–2690. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cady NC, McKean KA, Behnke J, et al:
Inhibition of biofilm formation, quorum sensing and infection in
Pseudomonas aeruginosa by natural products-inspired organosulfur
compounds. PLoS One. 7:e384922012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang D, Yu Q, Eikstadt P, Hammond D, Feng
Y and Chen N: Studies on adjuvanticity of sodium houttuyfonate and
its mechanism. Int Immunopharmacol. 2:1411–1418. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yuan L, Wu J and Aluko RE: Size of the
aliphatic chain of sodium houttuyfonate analogs determines their
affinity for renin and angiotensin I converting enzyme. Int J Biol
Macromol. 41:274–280. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yu QH, Li S and Chu ZY: Studies on the
anti-inflammatory effect and mechanism of houttuyninum. Chin
Pharmacol Bull. 14:442–444. 1998.
|
|
18
|
Ye XL, Li XG, Yuan LJ, Ge LH, Zhang BS and
Zhou SB: Interaction of houttuyfonate homologues with the cell
membrane of Gram-positive and Gram-negative bacteria. Colloids and
Surfaces A: Physicochem Eng Aspects. 301:412–418. 2007. View Article : Google Scholar
|
|
19
|
Liu G, Xiang H, Tang X, et al:
Transcriptional and functional analysis shows sodium
houttuyfonate-mediated inhibition of autolysis in Staphylococcus
aureus. Molecules. 16:8848–8865. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Park S, Chibli H and Nadeau J:
Solubilization and bio-conjugation of quantum dots and bacterial
toxicity assays by growth curve and plate count. J Vis Exp.
65:e39692012.PubMed/NCBI
|
|
21
|
Sauer KC, Camper AK, Ehrlich GD, Costerton
JW and Davies DG: Pseudomonas aeruginosa displays multiple
phenotypes during development as a biofilm. J Bacteriol.
184:1140–1154. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Miller RA, Walker RD, Carson J, et al:
Standardization of a broth microdilution susceptibility testing
method to determine minimum inhibitory concentrations of aquatic
bacteria. Dis Aquat Organ. 64:211–222. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
O'Toole GA: Microtiter dish biofilm
formation assay. J Vis Exp. 30:24372011.
|
|
24
|
Costerton JW, Irvin RT and Cheng KJ: The
bacterial glycocalyx in nature and disease. Annu Rev Microbiol.
35:299–324. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Nivens DE, Ohman DE, Williams J and
Franklin MJ: Role of alginate and its O acetylation in formation of
Pseudomonas aeruginosa microcolonies and biofilms. J Bacteriol.
183:1047–1057. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shao J, Cheng H, Wang C and Wang Y: A
phytoanticipin derivative, sodium houttuyfonate, induces in vitro
synergistic effects with levofloxacin against biofilm formation by
Pseudomonas aeruginosa. Molecules. 17:11242–11254. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Joy DC and Pawley JB: High-resolution
scanning electron microscopy. Ultramicroscopy. 47:80–100. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gacesa P: Bacterial alginate biosynthesis
- Recent progress and future prospects. Microbiology.
144:1133–1143. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cody WL, Pritchett CL, Jones AK, et al:
Pseudomonas aeruginosa AlgR controls cyanide production in an
AlgZ-dependent manner. J Bacteriol. 191:2993–3002. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Morici LA, Carterson AJ, Wagner VE, et al:
Pseudomonas aeruginosa AlgR represses the Rhl quorum-sensing system
in a biofilm-specific manner. J Bacteriol. 189:7752–7764. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2−ΔΔCT Method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mann EE and Wozniak DJ: Pseudomonas
biofilm matrix composition and niche biology. FEMS Microbiol Rev.
36:893–916. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Damron FH and Goldberg JB: Proteolytic
regulation of alginate overproduction in Pseudomonas aeruginosa.
Mol Microbiol. 84:595–607. 2012. View Article : Google Scholar : PubMed/NCBI
|