Introduction
It has previously been demonstrated that treatment
of rats with diethylnitroseamine (DENA) induced moderate and
well-differentiated forms of hepatocellular carcinoma (HCC) that
resembled human HCC (1) HCC is the
second most common cause of cancer-associated mortality in males
and the sixth among females (2). In
previous studies, a diet rich in dietary antioxidants and
phytochemicals was shown to decrease the risk of HCC (3–5).
Therefore, identifying potential chemopreventive agents in the
human diet, and their underlying molecular mechanisms, has been
considered the best strategy for protecting against human
hepatocarcinogenesis (3). Numerous
investigators have attempted to use naturally occurring compounds
for preventing or inhibiting carcinogenesis (6–10).
Furthermore, various natural bioactive compounds have been shown to
exert anti-cancer effects, including reversing the epigenetic
mechanisms underlying cancer, reducing tumorigenesis and
metastasis, and targeting cancer stem cells (11).
Previous studies have demonstrated that taurine,
which is an amino-acid derivative, exhibits a protective effect
against oxidation-induced cellular stress by acting as a free
radical scavenger in various cells and tissues (12–15). In
addition, taurine has been shown to exert hepatoprotective effects,
including inhibiting the accumulation of extracellular matrix
components in an experimental model of liver fibrosis (16,17).
Furthermore, it was shown to protect against genotoxicity induced
by anti-neoplastic drugs in somatic and germ tissues, and may be of
therapeutic potential in alleviating the risk of secondary tumors
following chemotherapy (18,19). Taurine also reduced lipid
peroxidation and the levels of proinflammatory cytokines in a rat
model of acute pancreatitis by increasing the levels of superoxide
dismutase and glutathione peroxidase (20). In addition, taurine has been shown to
exert an anti-cancer effect by inducing the apoptosis of cancer
cells, although the underlying molecular mechanism of this effect
is not well understood (21).
In a previous study, p53-upregulated modulator of
apoptosis (PUMA) was shown to respond to taurine treatment in human
colorectal cancer cells, which may be useful in the targeted
therapy of p53-deficient colorectal cancer (22). Furthermore, it has been demonstrated
that silencing the PUMA gene with specific small interfering RNA
(siRNA) was able to significantly reduce the ability of taurine to
inhibit the proliferation and induce the apoptosis of human HCC
HepG2 cells (23).
Curcumin (diferuloyl methane), which is a yellow
curry pigment obtained from turmeric extract (Curcuma
longa), has been shown to be a potent anti-inflammatory,
anti-carcinogenic and anti-oxidative agent in a number of
pre-clinical trials (24–27). In addition, curcumin was demonstrated
to exert chemopreventive effects against DENA-induced murine
hepatocarcinogenesis (28). The
inhibition of tumor formation by curcumin has been attributed to
its anti-initiation and anti-promotional effects. The former effect
may result from its ability to inhibit the formation of DNA damage
(29–31), whereas the latter may be mediated via
anti-proliferation effects or induction of apoptosis in the
initiated cells (32–35).
Curcumin has previously been shown to be a potent
immunomodulatory agent that can modulate the activation of T-cells,
B-cells, macrophages, neutrophils, natural killer cells and
dendritic cells (36). Furthermore,
curcumin was shown to downregulate the expression of various
proinflammatory cytokines, including tumor necrosis factor-α
(TNF-α), interleukin (IL)-1, IL-2, IL-6, IL-8 and IL-12, by
inactivation of the transcription factor nuclear factor-κB
(37).
In another study, curcumin and taurine alone or in
combination inhibited the growth of leukemic cells propagated in
vitro, and increased the level of interferon-γ (IFN-γ) in the
conditioned media of the cells (38). Furthermore, curcumin and taurine
treatment resulted in a significant increase in CD25+
mononuclear leukocytes in healthy control and cirrhotic patients,
and when propagated in vitro, although the increase was not
significant in HCC patients (39).
The present study aimed to investigate the
protective effects of a combination of taurine and curcumin in a
rat model of DENA-induced hepatocarcinogenesis.
Materials and methods
Animals
A total of 100 male albino rats, weighing 80–100 g
and aged 6–8 weeks, were obtained from the Institute of
Ophthalmology (Giza, Egypt). The rats were housed five per cage
under optimal conditions of temperature (27–30°C) and humidity
(40–70%), and were fed a standard pellet diet and water. The diet
consisted of ~20% protein, 5% fiber, 3.5% fat and 6.5% ash, as well
as vitamin mixtures. Feed and water were available ad
libitum throughout the experimental period. Ethical approval
for the present study was granted by the Institutional Animal Care
and Use Committee of Cairo University (Cairo, Egypt).
Experimental procedure
A suspension of curcumin in saline was prepared at a
concentration of 400 mg/40 ml, after which 2.5 ml curcumin
suspension was orally administered to a rat (100 mg/kg body
weight). Taurine in saline was prepared at a concentration of 1,000
mg/100 ml, after which 1 ml taurine solution was administered
orally to a rat (100 mg/kg body weight).
Rats were divided into eight groups, as follows: i)
Control group (n=10), in which the rats were treated with daily
intraperitoneal injection of saline parallel for 8 weeks; ii)
DENA-injected group (n=13), in which the rats were
intraperitoneally injected with DENA (100 mg/kg body weight;
Sigma-Aldrich, St. Louis, MO, USA) twice a week for 8 weeks; iii)
taurine plus curcumin treated group (n=10), in which the rats were
injected into the stomach with taurine (500 mg/kg body weight) and
curcumin (100 mg/kg body weight) daily, with a 1 h interval between
the two injections, for 8 weeks; iv) the taurine, curcumin and DENA
treated group (n=14), in which the rats were treated with the same
dose of taurine and curcumin for one week prior to DENA injection,
and received continued treatment [1 ml curcumin (25 mg/100 g body
weight) and 2.5 ml taurine (10 mg/100 g body weight] until the day
of sacrifice; v) the curcumin-treated group (n=10), in which the
rats were injected into the stomach with curcumin (100 mg/kg body
weight) daily for 8 weeks; vi) the curcumin plus DENA treated group
(n=12), in which the rats were treated with the same dose of
curcumin for one week prior to DENA injection, and received
continued treatment until the day of sacrifice; vii) the
taurine-treated group (n=10), in which the rats were injected into
the stomach with taurine (500 mg/kg body weight) for 8 weeks; and
viii) the taurine plus DENA treated group (n=12), in which the rats
were treated with the same dose of taurine for one week prior to
DENA injection, and received continued treatment until the day of
sacrifice.
Histopathological analysis
After 8 weeks of treatment, the rats were sacrificed
via an overdose of 50 mg/ml ketamine (40 mg/kg; Sigmatech
Pharmaceutical Industries, Cairo, Egypt) and sacrificed to obtain
liver tissue samples for the detection of HCC. Liver tissue was
fixed in 10% normal saline and buffered neutral formalin for 7
days, after which the tissues were washed and dehydrated in
ascending grades of ethyl alcohol, cleared using benzene and
embedded in paraffin for 1.5 h in the oven at 55°C. Serial sections
(5 µm) were cut using a microtome and stained with hematoxylin and
eosin, as described previously (40). The tissue sections were then analyzed
under a light microscope.
Analysis of liver tumor markers
Serum levels of α-fetoprotein (AFP) were analyzed
using an ELISA kit (ShARa/AFP/FITC; Nordic-MUbio, Susteren, The
Netherlands). Serum levels of α-L-fucosidase (AFU) were determined
using a colorimetric method, according to a previous study
(41). Serum levels of the cytokines
IL-2 (MBS494288) and IFN-γ (RIF00) were determined using ELISA kits
from MyBioSource, Inc. (San Diego, CA, USA) and R&D Systems,
Inc. (Minneapolis, MN, USA), respectively.
Electron microscope analysis
The rat livers were removed immediately following
sacrifice, and the liver tissue was fixed in 4% formaldehyde and 1%
gluteraldehyde in 0.1 M phosphate buffer (pH 7.4) to prepare for
electron microscopy analysis, according to a previous study
(42). The liver tissue sections
were then analyzed under an electron microscope.
Statistical analysis
Data are presented as the mean ± standard deviation.
Differences among groups and differences between different time
points were analyzed using one-way analysis of variance, followed
by Newman-Keuls method, where appropriate. P<0.05 was considered
to indicate a statistically significant difference.
Results
Biochemical analysis
The serum levels of AFP and AFU were significantly
elevated in the DENA-injected group, as compared with the control
group (P<0.001). Conversely, the serum levels of AFP and AFU
were significantly decreased in the groups treated with curcumin
and/or taurine (P<0.001). However, no significant differences
were detected in the serum levels of these markers among the
curcumin alone, taurine alone and curcumin plus taurine groups
(P>0.05; Table I).
 | Table I.Serum levels of AFP and AFU in rats
injected with DENA and treated with curcumin and taurine. |
Table I.
Serum levels of AFP and AFU in rats
injected with DENA and treated with curcumin and taurine.
| Group | Rats (n) | AFP (ng/ml) | AFU (U/l) |
|---|
| Control | 10 |
1.96±0.67 |
251.9±57.44 |
| DENA | 13 | 18.01±1.26 | 669.54±41.08 |
| Curcumin | 10 |
0.9±0.43 | 299.46±36.79 |
| Curcumin +
DENA | 12 | 12.28±0.74 |
312.4±39.93 |
| Taurine | 10 |
1.32±0.27 | 295.28±62.67 |
| Taurine + DENA | 12 |
7.84±0.69 | 296.55±41.66 |
| Taurine +
curcumin | 10 |
1.53±0.47 | 175.54±30.66 |
| Taurine + curcumin
+ DENA | 14 |
7.95±0.43 | 271.05±36.99 |
| F-value |
|
|
|
|
Control |
| 0.8 | 1.5 |
|
Treatments |
|
32.94a |
23.18a |
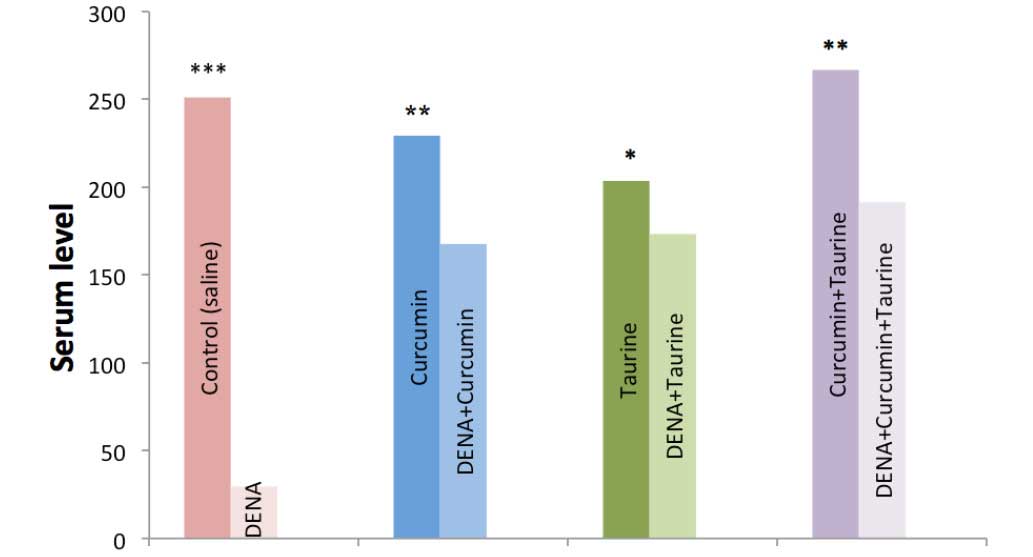
Serum levels of IL-2 were significantly decreased in
the DENA-injected group, as compared with the control group
(P<0.001). Conversely, the serum levels of IL-2 were
significantly elevated in the rats treated with curcumin and/or
taurine. Furthermore, the present results demonstrated there were
no significant differences in the serum levels of IL-2 among the
curcumin alone, taurine alone and curcumin plus taurine groups
(Table II and Fig. 1).
 | Table II.Serum levels of IL-2 and IFN-γ in
rats injected with DENA and treated with curcumin and taurine. |
Table II.
Serum levels of IL-2 and IFN-γ in
rats injected with DENA and treated with curcumin and taurine.
| Group | Rats (n) | IL-2 (pg/ml) | IFN-γ (pg/ml) |
|---|
| Control | 10 |
251.15±14.39 |
161.59±10.28 |
| DENA | 13 |
29.56±2.9 |
16.54±0.91 |
| Curcumin | 10 |
229.35±9.87 |
209.17±34.36 |
| Curcumin +
DENA | 12 |
167.66±13.56 | 109.44±4.74 |
| Taurine | 10 | 203.61±9.2 | 181.22±5.83 |
| Taurine + DENA | 12 |
173.34±5.63 | 102.73±4.72 |
| Taurine +
curcumin | 10 |
266.81±12.68 | 171.32±8.01 |
| Taurine + curcumin
+ DENA | 14 |
191.43±4.69 | 122.83±4.89 |
| F-value |
|
|
|
|
Control |
| 1.489 | 1.15 |
|
Treatments |
| 83.44a | 130.92a |
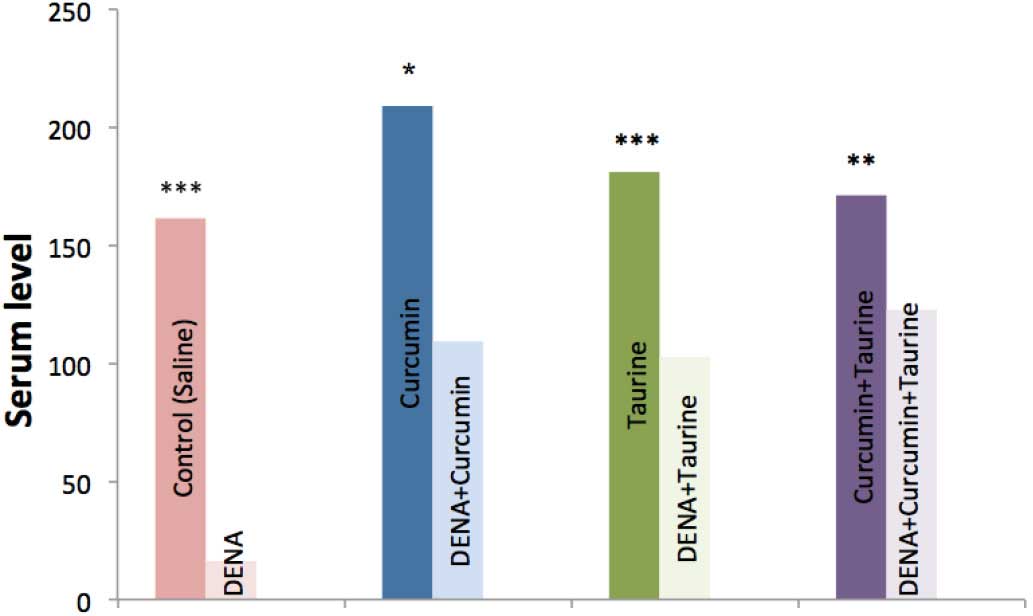
Serum levels of IFN-γ were significantly decreased
in the DENA-injected group, as compared with the control group
(P<0.001), whereas, the serums level of this cytokine were
significantly elevated in the curcumin and/or taurine treated
groups (P<0.001). In addition, there were no significant
differences in the serum levels of IFN-γ among the curcumin alone,
taurine alone and curcumin plus taurine groups (P>0.05; Table II and Fig. 2).
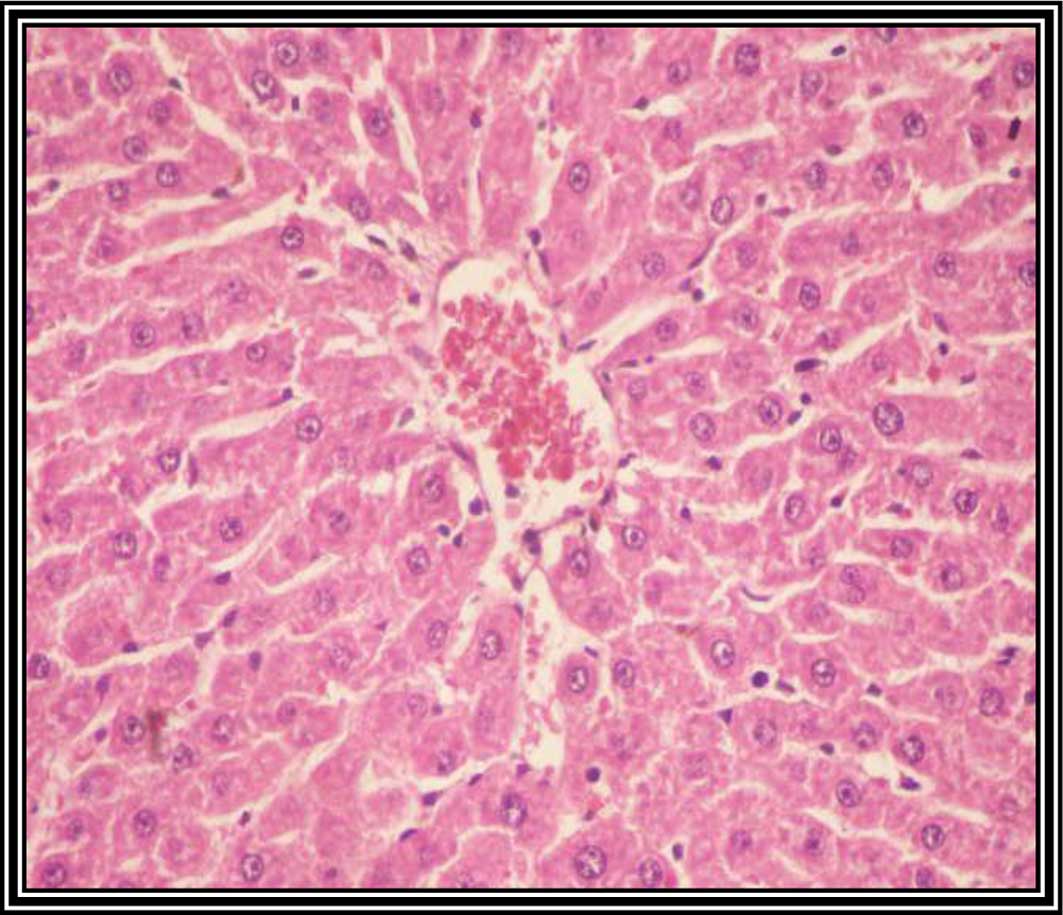
Pathological observations
Fig. 3 shows a liver
tissue section from a control group rat. It shows a normal liver
tissue pattern with normal hepatocytes. A histological observation
of liver tissue sections from DENA-injected rats at 4 weeks
post-injection showed only mild dysplastic changes, with no obvious
malignant changes (not shown). However, at 6 weeks following DENA
injection, the tissue sections of the liver showed dysplastic
changes, including disorganized hepatic cords (not shown), and
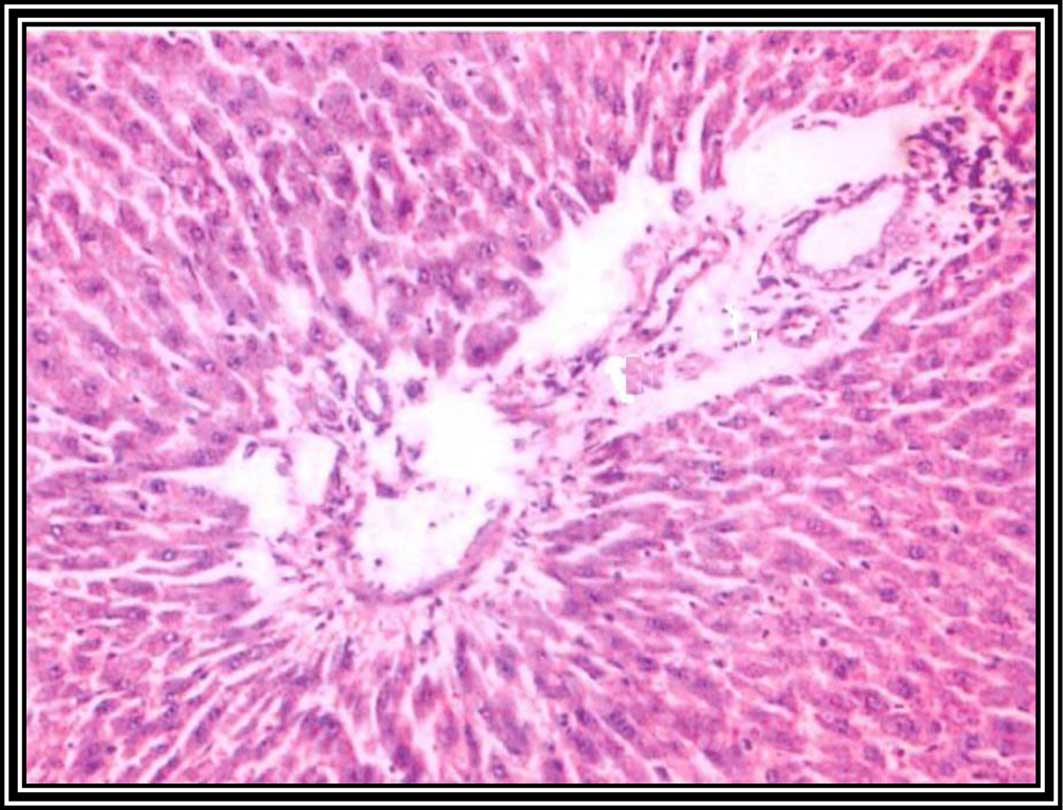
liver sections at 8 weeks following DENA injection showed malignant
changes associated with HCC grade 1, including nuclear
hyperchromasia, irregular anaplasia and pleomorphism, with
scattered mitotic figures (Fig.
4).
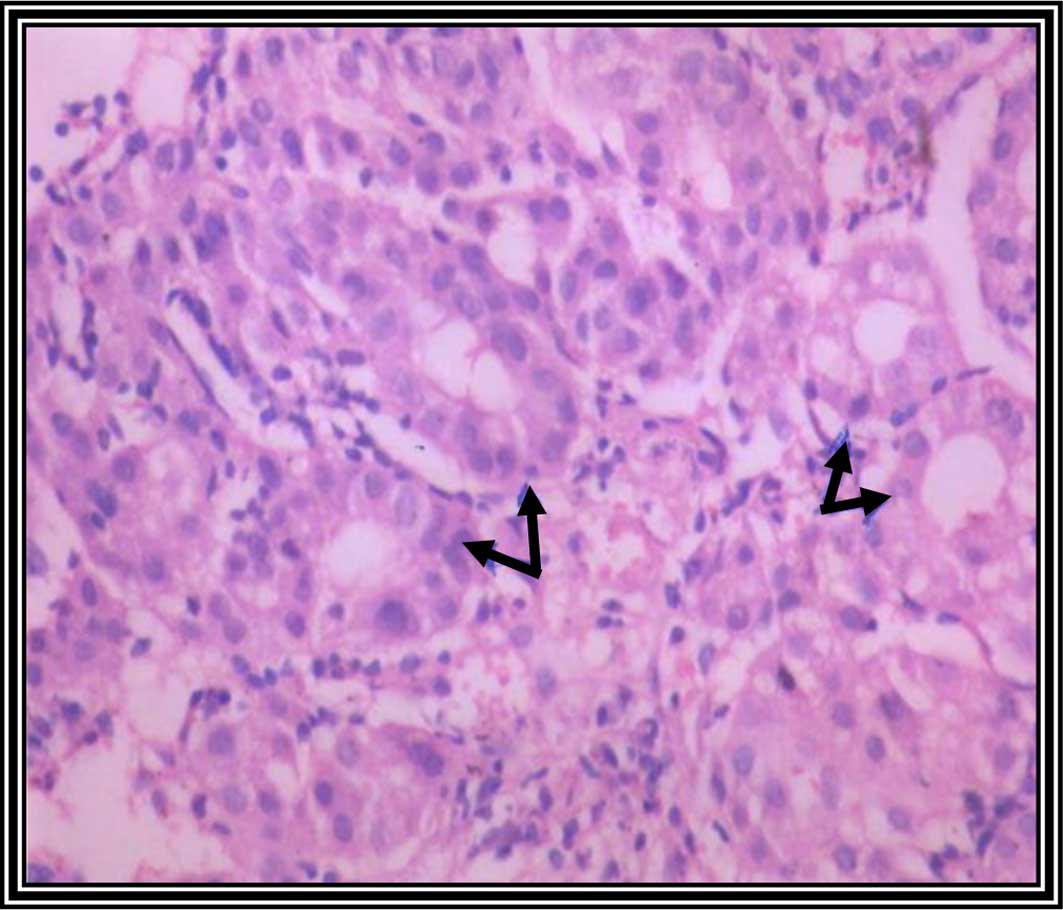
Liver tissue sections from the rats in the curcumin
plus taurine group showed normal liver architecture, with
well-preserved hepatocytes (Fig. 5).
In addition, the architecture and morphology of the rat liver
tissue section from the curcumin plus taurine group were similar to
the control group. These results suggest that curcumin and taurine
do not have toxic effects on the animal liver.
Electron microscope observations
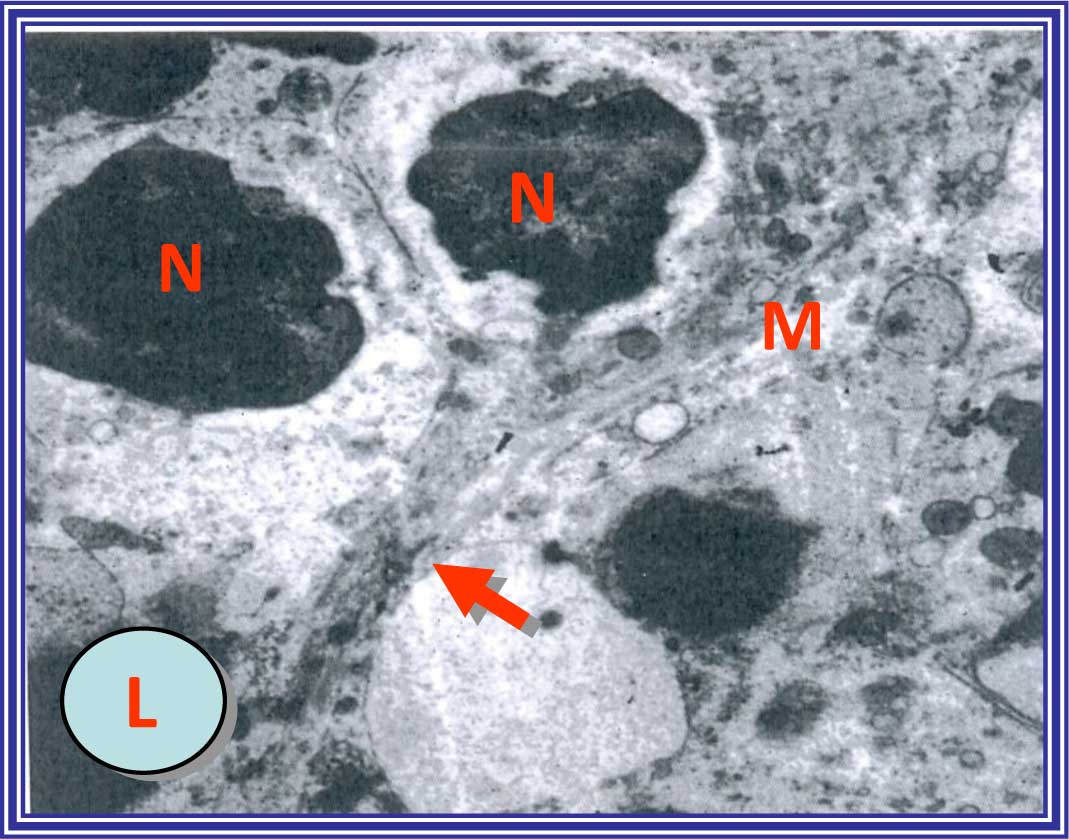
In the normal control rats, the hepatocytes showed
markedly swollen rough endoplasmic reticulum (RER), with few
ribosomes attached to their cisternae (Fig. 6). The liver sections of the
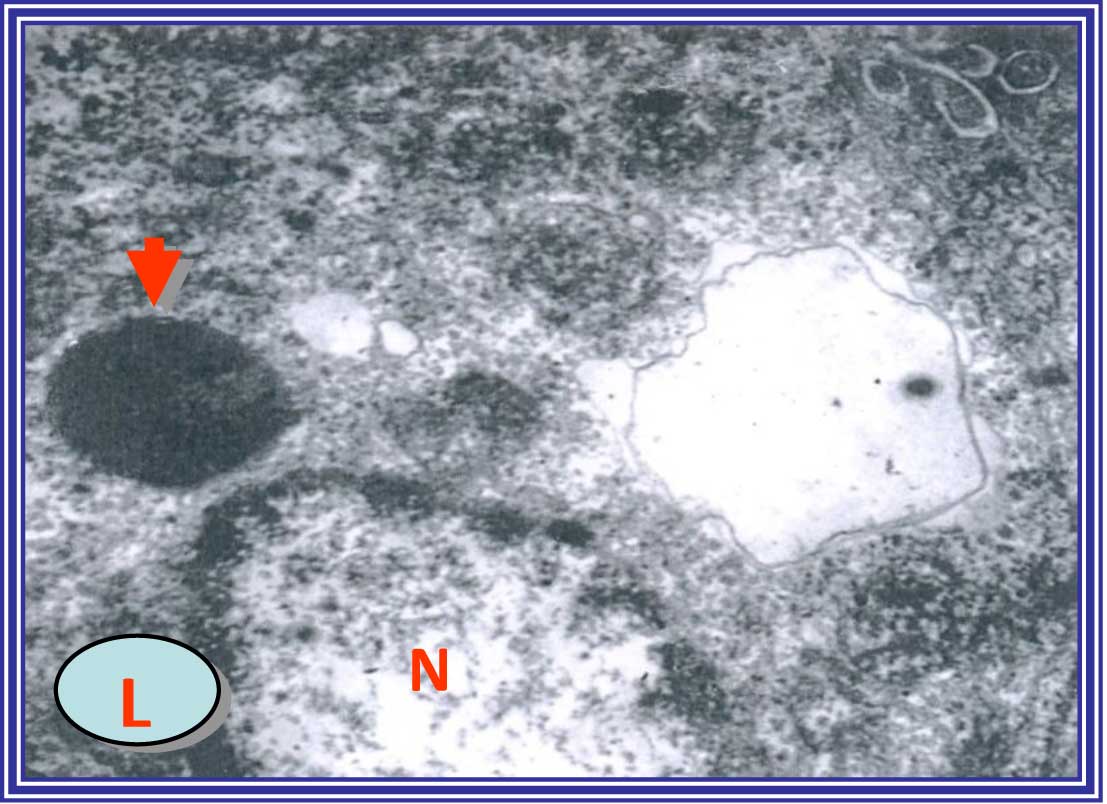
DENA-treated group showed degenerative cytoplasmic vacuolization,
disrupted organelles and malignant changes of the nucleus. In
addition, the mitochondria and ER cisternae appeared irregular, and
infiltration of inflammatory cells, including macrophages and
lymphocytes, were observed in the DENA-treated rat livers (Fig. 7).
In the curcumin-treated group, the number of damaged
hepatocytes was unchanged, as compared with the DENA-treated group,
although the extent of their internal damage was decreased.
Microvilli of the hepatocytes continued to project into the
expanded intercellular spaces; however, recovery of the fine
structures of the cellular organelles was evident (data not shown).
In the DENA-injected group, hepatic necrosis and infiltration of
inflammatory cells, particularly into the pericentral region, were
observed. However, in the taurine-treated group, these biological
changes were diminished and the development of hepatic fibrosis in
the pericentral region was inhibited (data not shown).
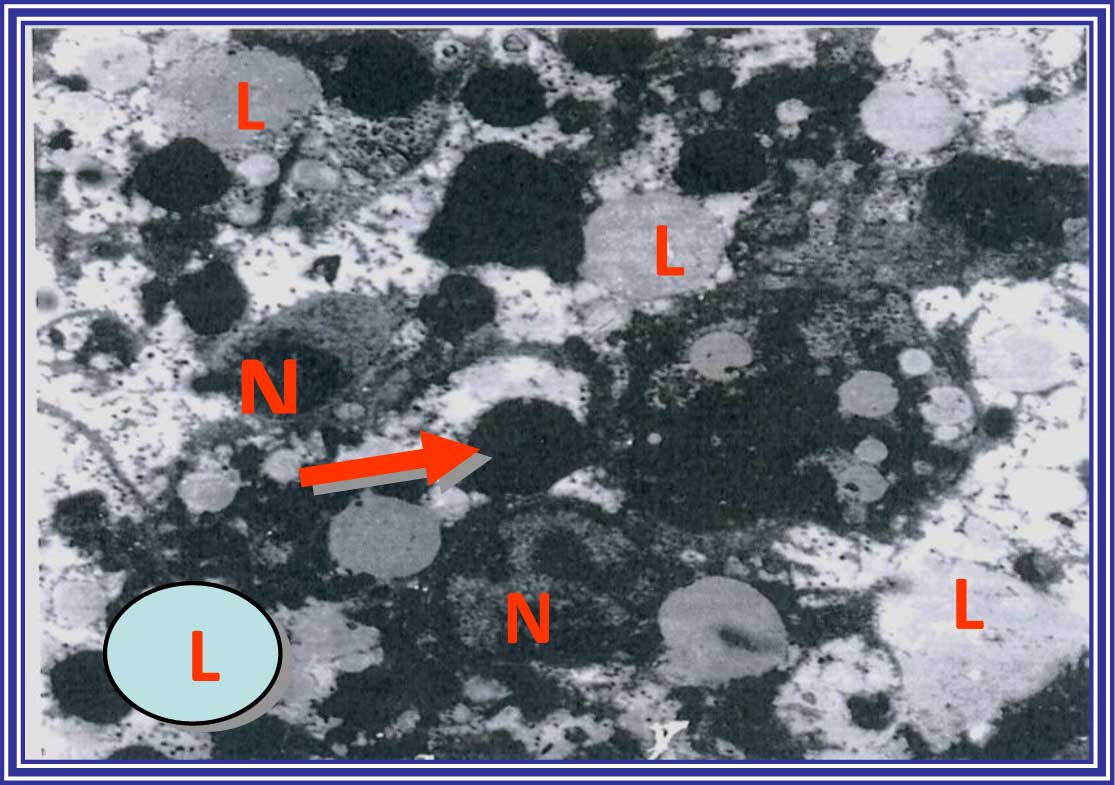
In the curcumin- plus taurine-treated group, the
hepatocytes appeared enlarged and pale, and some exhibited mitotic
activity. Furthermore, these cells showed marked dilatation of the
RER, indicating some loss of their ribosomes. The periorbital
hepatocytes remained unaffected and appeared intact and almost
normal. In addition, the nuclei, mitochondria and other organelles
were more regular in the curcumin plus taurine group, as compared
with the DENA-injected group (Fig.
8).
Discussion
The present study aimed to investigate the
protective effects of taurine and curcumin, used alone or in
combination, against hepatocarcinogenesis in a DENA-induced rat
model of HCC. In the present study, serum levels of AFP and AFU
were significantly elevated in the DENA-injected rats, which was
consistent with the results of previous studies that demonstrated
an association between AFP and AFU serum levels in rats with
DENA-induced HCC (43,44).
Conversely, the serum levels of AFP and AFU were
significantly decreased in the curcumin-treated rats, thus
suggesting a potential protective role of this natural agent
against hepatocarcinogenesis. Previous studies have reported
anti-inflammatory, antioxidant and anti-cancer activities for
curcumin (45–49). The serum levels of AFP and AFU were
similarly decreased in the taurine-treated rats, as compared with
the DENA-injected rats. These results suggested that taurine may
also have a protective effect against hepatocarcinogenesis. Taurine
has previously been shown to attenuate or prevent hepatic damage
(necrosis and fibrosis) induced by carbon tetrachloride (CCL4)
(50–53). Furthermore, in the present study, the
serum levels of AFP and AFU were significantly downregulated in the
curcumin- plus taurine-treated group, as compared with the
DENA-injected group.
The mechanism of action of these two natural
compounds is based on the secretion of various cytokines that
function via the following pathway: Stimulation of antitumor immune
responses, followed by induction of tumor cell apoptosis,
inhibition of the uncontrolled proliferation of cancer cells and
suppression of angiogenesis (54,55). In
the present study, the serum levels of IL-2 and IFN-γ were
significantly decreased in the DENA-treated group, as compared with
the control group. These results were consistent with a previous
study, in which the production of various cytokines, including
IL-2, IL-4, IL-5, IFN-γ and TNF-α, was shown to be suppressed in
cancer patients (56).
The present study demonstrated that the pathogenic
alterations induced by DENA could be inhibited by curcumin
treatment. This finding may have been due to the immunomodulatory
effect of curcumin, since previous studies have reported that
curcumin is a safe and useful immunomodulator (57–60). In
the present study, the serum levels of IL-2 and IFN-γ were
significantly elevated in the taurine-treated group; thus
suggesting that taurine may also exert an immunoregulatory role.
This finding is consistent with a previous study, in which taurine
was shown to alter the activity of the immune system to protect
hepatocytes against membrane disintegration during rat
hepatocarcinogenesis (61).
Potential immunoregulatory properties for taurine
have been suggested, which are based on the modulation of key
proinflammatory cells, in particular neutrophils and T lymphocytes
(62). Furthermore, another study
using a taurine derivative (taurine chloramines) demonstrated that
taurine was able to regulate the synthesis of proinflammatory
cytokines, thus suggesting that taurine may have a role in the
initiation and propagation of the immune response (63). In addition, a previous study reported
that the levels of IL-2 and IFN-γ were markedly elevated following
treatment with curcumin and taurine, and were approximately equal
to the control untreated levels (64).
In the present study, a histopathological analysis
of liver tissue sections from the DENA-treated group at 4 weeks
following DENA injection showed mild dysplastic changes and
inflammatory cell infiltration. At 6 weeks, the DENA-injected rat
liver tissues showed dysplastic changes, including disorganized
hepatic tissue. These results were consistent with a previous
study, in which hepatocarcinogenesis was shown to be induced in
rats at 6 weeks following injection with DENA and weekly
subcutaneous injection of CCL4 (65). In the present study, DENA injection
for 8 weeks resulted in malignant changes of the liver tissues,
including disorganized and irregularly arranged hepatocytes.
Furthermore, electron microscopy confirmed the histopathological
changes and showed the presence of HCC in the DENA-treated group,
as indicated by mitochondrial accumulation, RER stacks and nuclear
malignant changes. These results were consistent with a previous
study, which demonstrated that normal hepatocytes had fewer
mitochondria as compared with HCC cells (66). In addition, HCC cells were shown to
contain a greater abundance of, and more widely dispersed, RER.
The present study demonstrated that curcumin
effectively inhibited DENA-induced hepatic damage in a rat model of
HCC. A histopathological analysis showed moderate improvements in
the hepatocellular structure of curcumin-treated rats, as well as
the absence of nuclear changes and malignancy. However, some
apoptotic changes, such as cell membrane lysis, were observed,
which may have been due to the anti-cancer effect of curcumin.
These results were in agreement with a previous study that reported
direct anti-carcinogenic activities for curcumin (67). Similarly, previous studies
demonstrated that curcumin was able to inhibit the proliferation of
various cancer cells in culture and prevent carcinogenesis in
rodents (29,47,68). In
the present study, electron microscopy detected numerous
mitochondria, condensed chromatin, small nucleoli and stacks of RER
in the hepatocytes of the curcumin-treated rats. These results were
consistent with a previous study (69), in which numerous hallmarks of
apoptosis, including DNA laddering, chromatin condensation,
fragmentation and apoptosis, were observed in curcumin-treated
human hepatoblastoma cells.
The present study demonstrated a protective effect
for taurine against hepatic damage, as shown by the well-preserved
hepatocytes in lung tissue sections from taurine-treated rats. This
finding may be due to one or more of the biological roles that have
previously been reported for taurine, including antioxidant (free
radical scavenging), membrane stabilizing, and immune system
potentiation activities (70).
Furthermore, these results were supported by a previous study that
reported that taurine was a powerful prophylactic agent against
hepatocarcinogenesis in rats (71).
The electron microscopic observations in the present study
suggested that taurine preserves the morphology of major organelles
in hepatocytes and delays the development of fibrosis, which was
consistent with Tasci et al (72).
In the present study, combined treatment with
curcumin and taurine prior to DENA injection resulted in
well-preserved hepatocytes and reorganization of venous congestion
in the central veins. These findings suggested that the combination
of taurine and curcumin exerted a protective effect against
hepatocarcinogenesis, which may be due to the anti-cancer activity
of curcumin (29), and the
anti-neoplastic effect of taurine and its derivatives via
inhibition of cell proliferation, induction of tumor cell apoptosis
and suppression of angiogenesis (73–77).
In conclusion, the present study demonstrated that
combined use of curcumin and taurine exerted a protective effect
against hepatocarcinogenesis in a rat model of DENA-induced HCC.
Further studies should assess the efficacy of using curcumin and
taurine as a potential prophylactic combination therapy against
human hepatocarcinogenesis in high-risk groups that are exposed to
chemical carcinogens or viral infections, such as hepatitis B or
C.
Acknowledgements
The authors would like to thank Mr. Ahmed
EL-Husseiny, a research assistant at the Department of Cancer
Biology at the National Cancer Institute, Cairo University, for his
efforts in supporting the publication of this work, and the staff
at the animal care facility for their support in taking care of the
animals during the experimental procedures.
References
|
1
|
Di Stefano G, Fiume L, Bolondi L, Lanza M,
Pariali M and Chieco P: Enhanced uptake of lactosaminated human
albumin by rat hepatocarcinomas: Implications for an improved
chemotherapy of primary liver tumors. Liver Int. 25:854–860. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mann CD, Neal CP, Garcea G, Manson MM,
Dennison AR and Berry DP: Phytochemicals as potential
chemopreventive and chemotherapeutic agents in
hepatocarcinogenesis. Eur J Cancer Prev. 18:13–25. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Glauert HP, Calfee-Mason K, Stemm DN,
Tharappel JC and Spear BT: Dietary antioxidants in the prevention
of hepatocarcinogenesis: a review. Mol Nutr Food Res. 54:875–896.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lim DY, Jeong Y, Tyner AL and Park JH:
Induction of cell cycle arrest and apoptosis in HT-29 human colon
cancer cells by the dietary compound luteolin. Am J Physiol
Gastrointest Liver Physiol. 292:G66–G75. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cheng YL, Chang WL, Lee SC, Liu YG, Chen
CJ, Lin SZ, Tsai NM, Yu DS, Yen CY and Harn HJ: Acetone extract of
Angelica sinensis inhibits proliferation of human cancer cells via
inducing cell cycle arrest and apoptosis. Life Sci. 75:1579–1594.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Baliga MS, Meleth S and Katiyar SK: Growth
inhibitory and antimetastatic effect of green tea polyphenols on
metastasis-specific mouse mammary carcinoma 4T1 cells in vitro and
in vivo systems. Clin Cancer Res. 11:1918–1927. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Michaud DS, Feskanich D, Rimm EB, Colditz
GA, Speizer FE, Willett WC and Giovannucci E: Intake of specific
carotenoids and risk of lung cancer in 2 prospective US cohorts. Am
J Clin Nutr. 72:990–997. 2000.PubMed/NCBI
|
|
9
|
Kim ND, Mehta R, Yu W, Neeman I, Livney T,
Amichay A, Poirier D, Nicholls P, Kirby A, Jiang W and Mansel R:
Chemopreventive and adjuvant therapeutic potential of pomegranate
(Punica granatum) for human breast cancer. Breast Cancer Res Treat.
71:203–217. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yang SF, Yang WE, Chang HR, Chu SC and
Hsieh YS: Luteolin induces apoptosis in oral squamous cancer cells.
J Dental Res. 87:401–406. 2008. View Article : Google Scholar
|
|
11
|
Pistollato F, Giampieri F and Battino M:
The use of plant-derived bioactive compounds to target cancer stem
cells and modulate tumor microenvironment. Food Chem Toxicol.
75:58–70. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Schuller-Levis G, Mehta PD, Rudelli R and
Sturman J: Immunologic consequences of taurine deficiency in cats.
J Leukoc Biol. 47:321–331. 1990.PubMed/NCBI
|
|
13
|
Lake N, Wright ED and Lapp WS: Effects of
taurine deficiency on immune function in mice. Adv Exp Med Biol.
315:241–243. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bustamante J, Lobo MV, Alonso FJ, Mukala
NT, Giné E, Solís JM, Tamarit-Rodriguez J and Del Río R Martín: An
osmotic-sensitive taurine pool is localized in rat pancreatic islet
cells containing glucagon and somatostatin. Am J Physiol Endocrinol
Metab. 281:E1275–E1285. 2001.PubMed/NCBI
|
|
15
|
Miyazaki T, Karube M, Matsuzaki Y, Ikegami
T, Doy M, Tanaka N and Bouscarel B: Taurine inhibits oxidative
damage and prevents fibrosis in carbon tetrachloride-induced
hepatic fibrosis. J Hepatol. 43:117–125. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tasci I, Mas MR, Vural SA, Deveci S,
Comert B, Alcigir G, Mas N, Akay C, Bozdayi M, Yurdaydim C, et al:
Pegylated interferon-alpha plus taurine in treatment of rat liver
fibrosis. World J Gastroenterol. 13:32372007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tasci I, Mas N, Mas MR, Tuncer M and
Comert B: Ultrastructural changes in hepatocytes after taurine
treatment in CCl4 induced liver injury. World J Gastroenterol.
14:48972008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cetiner M, Sener G, Sehirli AO,
Ekşioğlu-Demiralp E, Ercan F, Sirvanci S, Gedik N, Akpulat S,
Tecimer T and Yeğen BC: Taurine protects against
methotrexate-induced toxicity and inhibits leukocyte death. Toxicol
Appl Pharmacol. 209:39–50. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Alam SS, Hafiz NA and Abd El-Rahim AH:
Protective role of taurine against genotoxic damage in mice treated
with methotrexate and tamoxfine. Environ Toxicol Pharmacol.
31:143–152. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Akay C, Yaman H, Oztosun M, Cakir E,
Yildirim AO, Eyi YE, Agilli M, Akgul EO, Aydin I, Kaldirim U, et
al: The protective effects of taurine on experimental acute
pancreatitis in a rat model. Hum Exp Toxicol. 32:522–529. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tu S, Zhang X, Luo D, Liu Z, Yang X, Wan
H, Yu L, Li H and Wan F: Effect of taurine on the proliferation and
apoptosis of human hepatocellular carcinoma HepG2 cells. Exp Ther
Med. 10:193–200. 2015.PubMed/NCBI
|
|
22
|
Zhang X, Tu S, Wang Y, Xu B and Wan F:
Mechanism of taurine-induced apoptosis in human colon cancer cells.
Acta Biochim Biophys Sin (Shanghai). 46:261–272. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tu S, Zhang X, Luo D, Liu Z, Yang X, Wan
H, Yu L, Li H and Wan F: Effect of taurine on the proliferation and
apoptosis of human hepatocellular carcinoma HepG2 cells. Exp Ther
Med. 10:193–200. 2015.PubMed/NCBI
|
|
24
|
Punithavathi D, Venkatesan N and Babu M:
Curcumin inhibition of bleomycin-induced pulmonary fibrosis in
rats. Br J Pharmacol. 131:169–172. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kim KA, Lee WJ, Lim HK, Park CM, Park CK,
Cha IH and Seol HY: Small hepatocellular carcinoma:
Ultrasonographic findings and histopathologic correlation. Clin
Imaging. 27:340–345. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Duvoix A, Blasius R, Delhalle S,
Schnekenburger M, Morceau F, Henry E, Dicato M and Diederich M:
Chemopreventive and therapeutic effects of curcumin. Cancer Lett.
223:181–190. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gulcubuk A, Altunatmaz K, Sonmez K,
Haktanir-Yatkin D, Uzun H, Gurel A and Aydin S: Effects of curcumin
on tumour necrosis factor-alpha and interleukin-6 in the late phase
of experimental acute pancreatitis. J Vet Med A Physiol Pathol Clin
Med. 53:49–54. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chuang SE, Kuo ML, Hsu CH, Chen CR, Lin
JK, Lai GM, Hsieh CY and Cheng AL: Curcumin-containing diet
inhibits diethylnitrosamine-induced murine hepatocarcinogenesis.
Carcinogenesis. 21:331–335. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Huang MT, Newmark HL and Frenkel K:
Inhibitory effects of curcumin on tumorigenesis in mice. J Cell
Biochem Suppl. 27:26–34. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Subramanian M, Sreejayan, Rao MN,
Devasagayam TP and Singh BB: Diminution of singlet oxygen-induced
DNA damage by curcmin and related antioxidants. Mutat Res.
311:249–255. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Parshad R, Sanford KK, Price FM, Steele
VE, Tarone RE, Kelloff GJ and Boone CW: Protective action of plant
polyphenols on radiation-induced chromatid breaks in cultured human
cells. Anticancer Res. 18:3263–3266. 1998.PubMed/NCBI
|
|
32
|
Samaha HS, Kelloff GJ, Steele V, Rao CV
and Reddy BS: Modulation of apoptosis by sulindac, curcumin,
phenylethyl-3-methylcaffeate, and 6-phenylhexyl isothiocyanate:
Apoptotic index as a biomarker in colon cancer chemoprevention and
promotion. Cancer Res. 57:1301–1305. 1997.PubMed/NCBI
|
|
33
|
Sikora E, Bielak-Zmijewska A, Piwocka K,
Skierski J and Radziszewska E: Inhibition of proliferation and
apoptosis of human and rat T lymphocytes by curcumin, a curry
pigment. Biochem Pharmacol. 54:899–907. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chen HW and Huang HC: Effect of curcumin
on cell cycle progression and apoptosis in vascular smooth muscle
cells. Br J Pharmacol. 124:1029–1040. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Jee SH, Shen SC, Tseng CR, Chiu HC and Kuo
ML: Curcumin induces a p53-dependent apoptosis in human basal cell
carcinoma cells. J Invest Dermatol. 111:656–661. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jagetia GC and Aggarwal BB: “Spicing up”
of the immune system by curcumin. J Clin Immunol. 27:19–35. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Bruck R, Ashkenazi M, Weiss S, Goldiner I,
Shapiro H, Aeed H, Genina O, Helpern Z and Pines M: Prevention of
liver cirrhosis in rats by curcumin. Liver Int. 27:373–383. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
El-Houseini ME, Refaei MO, Amin AI and
Abol-Ftouh MA: Potential role of curcumin and taurine combination
therapy on human myeloid leukemic cells propagated in vitro. Leuk
Lymphoma. 54:2281–2287. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Abdeen SH, El-Houseini ME, El-Sherbiny M,
Tabashy R and Salah A: Ex vivo assessment of the protective effect
of curcumin and taurine against human hepatocarcinogenesis. J Basic
Appl Zool. 66:180–187. 2013. View Article : Google Scholar
|
|
40
|
Leventhal, Ruth and Russell F Cheadle:
Medical parasitology: A Self-Instructional Text. FA Davis.
2011.
|
|
41
|
El-Houseini, ME, El-Sherbiny M, Awad, ME,
El-Din Awad M, Amer MA, El Din AH and Hussein TD: Serum
alpha-L-fucosidase enzyme activity as a marker for hepatocellular
carcinoma: Comparison with AFP using ROC analysis. J Egypt Natl
Cancer Inst. 13:227–283. 2001.
|
|
42
|
Bravman JC and Sinclair R: The preparation
of cross-section specimens for transmission electron microscopy. J
Electron Microsc Tech. 1:53–61. 1984. View Article : Google Scholar
|
|
43
|
Liu Y, Daley S, Evdokimova VN, Zdobinski
DD, Potter DM and Butterfield LH: Hierarchy of alpha fetoprotein
(AFP)-specific T cell responses in subjects with AFP-positive
hepatocellular cancer. J Immunol. 177:712–721. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Butterfield LH, Ribas A, Potter DM and
Economou JS: Spontaneous and vaccine induced AFP-specific T cell
phenotypes in subjects with AFP-positive hepatocellular cancer.
Cancer Immunol Immunother. 56:1931–1943. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Guo LY, Cai XF, Lee JJ, Kang SS, Shin EM,
Zhou HY, Jung JW and Kim YS: Comparison of suppressive effects of
demethoxycurcumin and bisdemethoxycurcumin on expressions of
inflammatory mediators in vitro and in vivo. Arch Pharm Res.
31:490–496. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Sandur SK, Pandey MK, Sung B, Ahn KS,
Murakami A, Sethi G, Limtrakul P, Badmaev V and Aggarwal BB:
Curcumin, demethoxycurcumin, bisdemethoxycurcumin,
tetrahydrocurcumin and turmerones differentially regulate
anti-inflammatory and anti-proliferative responses through a
ROS-independent mechanism. Carcinogenesis. 28:1765–1773. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Suryanarayana P, Satyanarayana A,
Balakrishna N, Kumar PU and Reddy GB: Effect of turmeric and
curcumin on oxidative stress and antioxidant enzymes in
streptozotocin-induced diabetic rat. Med Sci Monit. 13:BR286–BR292.
2007.PubMed/NCBI
|
|
48
|
Kunnumakkara AB, Diagaradjane P, Guha S,
Deorukhkar A, Shentu S, Aggarwal BB and Krishnan S: Curcumin
Sensitizes human colorectal cancer xenografts in nude mice to
gamma-radiation by targeting nuclear factor-kappaB-regulated gene
products. Clin Cancer Res. 14:2128–2136. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Lin J and Chen A: Activation of peroxisome
proliferator-activated receptor-gamma by curcumin blocks the
signaling pathways for PDGF and EGF in hepatic stellate cells. Lab
Invest. 88:529–540. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Waterfield CJ, Turton JA, Scales MD and
Timbrell JA: Taurine, a possible urinary marker of liver damage: A
study of taurine excretion in carbon tetrachloride-treated rats.
Arch Toxicol. 65:548–555. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Chen Y, Li S and Zhang X: Taurine inhibits
deposition of extracellular matrix in experimental liver fibrosis
in rats. Zhong Hua Gan Zang Bing Za Zhi. 7:165–167. 1999.(In
Chinese).
|
|
52
|
Wu QD, Wang JH, Fennessy F, Redmond HP and
Bouchier-Hayes D: Taurine prevents high-glucose-induced human
vascular endothelial cell apoptosis. Am J Physiol. 277:C1229–C1238.
1999.PubMed/NCBI
|
|
53
|
Dinçer S, Ozenirler S, Oz E, Akyol G and
Ozoğul C: The protective effect of taurine pretreatment on carbon
tetrachloride-induced hepatic damage-a light and electron
microscopic study. Amino Acids. 22:417–426. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Boujendar S, Arany E, Hill D, Remacle C
and Reusens B: Taurine supplementation of a low protein diet fed to
rat dams normalizes the vascularization of the fetal endocrine
pancreas. J Nutr. 133:2820–2825. 2003.PubMed/NCBI
|
|
55
|
Yoysungnoen P, Wirachwong P, Bhattarakosol
P, Niimi H and Patumraj S: Effects of curcumin on tumor
angiogenesis and biomarkers, COX-2 and VEGF, in hepatocellular
carcinoma cell-implanted nude mice. Clinical Hemorheol Microcirc.
109–115. 2006.
|
|
56
|
Tomova R, Pomakov J, Jacobs JJ, Adjarov D,
Popova S, Altankova I, Den Otter W and Krastev Z: Changes in
cytokine profile during local IL-2 therapy in cancer patients.
Anticancer Res. 26:2037–2047. 2006.PubMed/NCBI
|
|
57
|
Antony S, Kuttan R and Kuttan G:
Immunomodulatory activity of curcumin. Immunol Invest. 28:291–303.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Jagetia GC and Aggarwal BB: ‘Spicing up’
of the immune system by curcumin. J Clin Immunol. 27:19–35. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Yadav VS, Mishra KP, Singh DP, Mehrotra S
and Singh VK: Immunomodulatory effects of curcumin. Immunopharmacol
Immunotoxicol. 27:485–497. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Aggarwal BB, Surh YJ and Shishodia S: The
Molecular Targets and Therapeutic Uses of Curcumin in Health and
Disease. Springer Science+Business Media; 2007, View Article : Google Scholar
|
|
61
|
You JS and Chang KJ: Taurine protects the
liver against lipid peroxidation and membrane disintegration during
rat hepatocarcinogenesis. Adv Exp Med Biol. 442:105–112. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Redmond HP, Stapleton PP, Neary P and
Bouchier-Hayes D: Immunonutrition: The role of taurine. Nutrition.
14:599–604. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Kim H, Jeon H, Kong H, Yang Y, Choi B, Kim
YM, Neckers L and Jung Y: A molecular mechanism for the
anti-inflammatory effect of taurine-conjugated 5-aminosalicylic
acid in inflamed colon. Mol Pharmacol. 69:1405–1412. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Chorazy M, Kontny E, Marcinkiewicz J and
Maśliński W: Taurine chloramine modulates cytokine production by
human peripheral blood mononuclear cells. Amino Acids. 23:407–413.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
aziz MT Abdel, El Asmar MF, Atta HM,
Mahfouz S, Fouad HH, Roshdy NK, Rashed LA, Sabry D, Hassouna AA and
Taha FM: Efficacy of mesenchymal stem cells in suppression of
hepatocarcinorigenesis in rats: Possible role of Wnt signaling. J
Exp Clin Cancer Res. 30:492011. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Couch JA: Light and electron microscopic
comparisons of normal hepatocytes and neoplastic hepatocytes of
well-differentiated hepatocellular carcinomas in a teleost fish.
Dis Aquat Org. 16:1–14. 1993. View Article : Google Scholar
|
|
67
|
Aggarwal S, Ichikawa H, Takada Y, Sandur
SK, Shishodia S and Aggarwal BB: Curcumin (diferuloylmethane)
down-regulates expression of cell proliferation and antiapoptotic
and metastatic gene products through suppression of IkappaBalpha
kinase and Akt activation. Mol Pharmacol. 69:195–206.
2006.PubMed/NCBI
|
|
68
|
He SM, Li CG, Liu JP, Chan E, Duan W and
Zhou SF: Disposition pathways and pharmacokinetics of herbal
medicines in humans. Curr Med Chem. 17:4072–4113. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Aggarwal BB, Kumar A and Bharti AC:
Anticancer potential of curcumin: Preclinical and clinical studies.
Anticancer Res. 23:363–398. 2003.PubMed/NCBI
|
|
70
|
Koyama I, Nakamura T, Ogasawara M, Nemoto
M and Yoshida T: The protective effect of taurine on the
biomembrane against damage produced by the oxygen radical. Adv Exp
Med Biol. 315:355–359. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
El-Agamy DS: Comparative effects of
curcumin and resveratrol on aflatoxin B(1)-induced liver injury in
rats. Arch Toxicol. 84:389–396. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Tasci I, Mas N, Mas MR, Tuncer M and
Comert B: Ultrastructural changes in hepatocytes after taurine
treatment in CCl4 induced liver injury. World J Gastroenterol.
14:4897–4902. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
McCourt M, Wang JH, Sookhai S and Redmond
HP: Taurolidine inhibits tumor cell growth in vitro and in vivo.
Ann Surg Oncol. 7:685–691. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Calabresi P, Goulette FA and Darnowski JW:
Taurolidine: Cytotoxic and mechanistic evaluation of a novel
antineoplastic agent. Cancer Res. 61:6816–6821. 2001.PubMed/NCBI
|
|
75
|
Stendel R, Stoltenburg-Didinger G, Al
Keikh CL, Wattrodt M and Brock M: The effect of taurolidine on
brain tumor cells. Anticancer Res. 22:809–814. 2002.PubMed/NCBI
|
|
76
|
Petrovic L, Schlegel KA, Ries J, Park J,
Diebel E, Schultze-Mosgau S and Wiltfang J: In vitro effect of
taurolidine on squamous cell carcinoma in the oral cavity. Mund
Kiefer Gesichtschir. 7:102–107. 2003.(In German). View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Rodak R, Kubota H, Ishihara H, Eugster HP,
Könü D, Möhler H, Yonekawa Y and Frei K: Induction of reactive
oxygen intermediates-dependent programmed cell death in human
malignant ex vivo glioma cells and inhibition of the vascular
endothelial growth factor production by taurolidine. J Neurosurg.
102:1055–1068. 2005. View Article : Google Scholar : PubMed/NCBI
|