Introduction
Spinal cord injury (SCI) poses a serious threat to
human health and typically results in incomplete or complete loss
of motor and sensory function (1).
The prevalence of SCI is relatively high. Notably, the prevalence
among the elderly (aged >60 years old) and the younger (aged
16–45 years old) have been estimated to be ~24 and 49%,
respectively, which are primarily due to traffic collisions
(2,3). Although various methods of treating SCI
are currently used, these treatments are not effective since SCIs
still result in substantial permanent morbidity and mortality
(2). Therefore, it is important to
develop a novel effective method of treating patients with SCI.
Apoptosis is a process of programmed cell death that
is increased under pathological conditions (2). Apoptosis inhibits nerve function
following SCI and this is the primary factor that causes secondary
injury of the spinal cord (4).
Previous studies have suggested that the mitochondrial and death
receptor signaling pathways are the two primary signaling pathways
that induce apoptosis (5–7). Furthermore, it has been demonstrated
that endoplasmic reticulum stress (ERS) induces apoptosis (8). The endoplasmic reticulum (ER) is an
important organelle in eukaryotic cells that regulates calcium ions
and the processing and synthesis of proteins (9). When ER function is altered during
pathological conditions, the ER will respond accordingly with an
ERS reaction (10,11). Subsequently, the unfolded protein
response (UPR), which is an adaptive response that predominantly
functions to maintain normal ER function, is induced (12). There are three processes associated
with UPR, which include the involvement of activating transcription
factor (ATF) 6, serine/threonine-protein kinase/endoribonuclease
inositol-requiring enzyme (IRE)1 and RNA-activated protein
kinase-like ER kinase (PERK) (13).
Under normal physiological conditions, these three proteins bind to
an intra-ER chaperone, glucose-regulated protein (GRP)78, which is
maintained in an inactive state in the ER membrane (13). However, in response to stress, GRP78
dissociates from the ER membrane to bind to misfolded proteins,
thereby initiating the signal transduction processes to
re-establish ER homeostasis (14).
Although the UPR typically acts to maintain homeostasis in cells by
removing misfolded proteins, elevated and sustained ER stress
induces cell death (15). Therefore,
therapeutic strategies targeting ER stress and its downstream
apoptosis signaling pathways may be used to treat patients with
SCI.
Methotrexate (MTX), which is characterized by
anti-inflammatory and immunosuppressive functions, is a first-line
prescription agent used in the treatment of rheumatoid arthritis
(RA) (16,17). Due to its safety and tolerability,
low doses of MTX has been used as a long-term therapy regimen for
patients with RA (18). However, to
the best of our knowledge, there have been no studies investigating
whether MTX improves traumatic SCI by inhibiting ERS. Therefore,
the present study aimed to explore the use of low-dose MTX in the
treatment of a rat model of SCI as a means to uncover a potential
inhibition mechanism of ERS.
Materials and methods
Establishment of a rat SCI model
A total of 72 specific-pathogen free male Sprague
Dawley rats (weight, 220–250 g, 6–8 weeks old) were purchased from
Shanghai Silaike Experimental Animal Limited Liability Company
(Shanghai, China). Rats were maintained in the animal experimental
center of Hongqi Hospital Affiliated to Mudanjiang Medical
University and four animals were housed per each cage with a 12-h
light/dark cycle. Room temperature was maintained at 23±1°C,
humidity was maintained at ~60% and all rats had free access to
food and water. Rats were randomly divided into three groups: A
sham, an SCI and an MTX+SCI group. MTX (Sigma-Aldrich; Merck KGaA,
Darmstadt, Germany) stock solutions were prepared in 0.1 mol/l NaOH
at 0.5 mg/ml and diluted 1:10 in 0.1 mol/l phosphate-buffered
saline (PBS) prior to use; the pH of the solution was adjusted to
7.4–7.6. For the sham group and SCI rats, no other treatments were
performed. For the MTX+SCI group, a total of 24 h following SCI,
rats were treated with 50 mg/kg MTX. A total of 8 rats were in the
sham group, 32 rats were in the SCI group and 32 rats were in
MTX+SCI group. Rats in the SCI and MTX+SCI groups were further
divided into 1, 3, 7 and 14 day groups (n=8, each group) according
to the different time points following injury.
The rat SCI model was established as previously
described (19). Rats in the sham
surgery group underwent all aspects of the surgery except for
contusion thoracic injury. Following 8 h fasting, rats were
anesthetized via intraperitoneal injection with 10% chloral hydrate
(400 mg/kg), according to a previous study (20). Subsequently, rats were placed in a
prone position and an incision (~2.5 cm in length) was made in the
middle of the back. Skin was cut layer by layer and the T8 to T10
vertebral plates were exposed. Total laminectomy was performed for
the T9 vertebral plate to expose the spinal dura mater. T8 and T10
spinous processes were fixated using forceps. A Kirschner wire (10
g) was inserted into the catheter that was inserted into the aorta
with a weight, which fell freely from a 25-mm height. Following
this, a semicircular slice (4×2 mm) made from thin plastic was hit,
and the wire was immediately removed, resulting in incomplete
injury of the rat spinal cord. The incision was sutured layer by
layer. Following the strike, rats exhibiting a tail-wagging reflex,
retraction flutter in the lower limbs and body, and flaccid
paralysis in the lower limbs in an awake state represented
successful model construction. For MTX treatment, MTX was delivered
intrathecally once per day (50 mg/kg) after SCI for 3 days and a
dose of 50 mg/kg MTX was selected based on the results of a
previous study, which indicated that the median lethal dose of MTX
in rats was 2,288.5 mg/kg (21). No
changes in motion or mental state were observed in rats from the
SCI, SCI+MTX-treated or the sham group in the present study.
Therefore, the dose of MTX selected was considered to be safe. The
present study was approved by the Animal Ethics Committee of the
Hongqi Hospital Affiliated to Mudanjiang Medical University
(Mudanjiang, China).
Basso, Beattie and Bresnahan (BBB)
scoring
Behavioral scoring was performed at different time
points (n=8, each time point) following SCI surgery. The recovery
of hind limb motor function was observed and scored using the BBB
scoring system (22). All behavioral
observations were performed at the same time (8:00 p.m.) to avoid
variations in the movement of animals between day and night.
Furthermore, an inclined plane test was performed. Behavior was
evaluated days 1, 3, 5, 7 and 14 days postoperatively using the
modified Rivlin's method (23),
which tested the ability of animals to balance on elevated wooden
beams. A simple device was constructed containing a moveable plate
with an adjustable angle of 0–90°. The rat's head was placed faced
forward, and the angle of inclination between the inclined plane
and the horizontal plane was increased gradually, until the rats
were unable to maintain a constant position (45°) for 5 sec. The
angle was considered to be the critical value and then
recorded.
Cell culture
PC12 cells were purchased from the Chinese Academy
of Medical Sciences (Beijing, China). Cells were cultured in
high-glucose Dulbecco's modified Eagle's medium/F12 (GE Healthcare
Life Sciences, Logan, UT, USA) supplemented with 10% fetal bovine
serum (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA,
USA) and 100 U/ml penicillin and 100 U/ml streptomycin in
25-cm2 culture flasks at 37°C in a humidified atmosphere
containing 5% CO2.
MTT colorimetric assay
Triglycerides (TG, Sigma-Aldrich; Merck KGaA) were
used to establish the in vitro ERS cell model. To
investigate the influence of TG on PC12 cell viability, PC12 cells
were seeded in 96-well tissue culture plates at a density of
5×104 cells per well in DMEM medium. When the confluence
reached 70%, 1, 2.5, 5 and 10 µM TG was added to each well and the
cells were incubated at 37°C for 48 h. Cell viability was examined
with MTT assay kits (Sigma-Aldrich; Merck KGaA). The blue formazan
products in the cells were dissolved in dimethyl sulfoxide (DMSO,
Sigma-Aldrich; Merck KGaA) and spectrophotometrically measured at a
wavelength of 550 nm. All experiments were performed in
triplicate.
Drug treatment
In brief, 106 PC12 cells were seeded into
the 6-well plates for 24 h at 37°C. To establish the in
vitro ERS cell model, 5 µM TG were dissolved in DMSO and
applied to PC12 cells for 24 h. Cells in the normal control (NC)
group was treated with DMSO for 24 h. Subsequently, PC12 cells were
treated with 100 ng/ml MTX dissolved in PBS for 48 h and
subsequently administered to the cells.
Western blotting
Rats were anesthetized with intraperitoneal
injection of 400 mg/kg 10% chloral hydrate (20) and subjected to perfusion with
fixatives. Spinal cords were stripped bluntly through the incision
on the back of the rat, resulting in a single 1-cm spinal cord
segment with the damaged part at the center. The spinal cords were
then processed into homogenized tissue using a homogenizer and then
the homogenates were centrifuged at 10,000 × g for 15 min at 4°C.
Following this, spinal cord tissues or PC12 cells were treated with
radioimmunoprecipitation assay buffer (Beijing Solarbio Science
& Technology Co., Ltd., Beijing, China) containing 1% (v/v)
phenylmethylsulfonyl fluoride (Beijing Solarbio Science &
Technology Co., Ltd.), 0.3% (v/v) protease inhibitor
(Sigma-Aldrich; Merck KGaA) and 0.1% (v/v) phosphorylated
proteinase inhibitor (Sigma-Aldrich; Merck KGaA). A BCA protein
assay kit (Pierce; Thermo Fisher Scientific, Inc.) was used to
determine the protein concentration. Subsequently, supernatants
were extracted from the lysates following centrifugation at 11,000
× g at 4°C for 15 min. Equal amounts of protein (15 µg/lane) were
separated using 10% SDS-PAGE at 300 mA for 2 h and transferred onto
a polyvinylidene fluoride membrane, as previously reported
(10). Nonspecific binding was
blocked using 8% (w/v) milk in Tris-buffered saline with Tween-20
(TBS-T) for 2 h at room temperature. The following primary
antibodies were used: β-actin (cat. no. 4970), cleaved-caspase-3
(cat. no. 9664), GRP78 (cat. no. 3177), CHOP (cat. no. 5554),
caspase-12 (cat. no. 2202), ATF6 (cat. no. 65880), IRE1α (cat. no.
3294) and eukaryotic initiation factor 2 (eIF2)α (cat. no. 5324;
all 1:1,000 dilution; Cell Signaling Technology, Inc., Danvers, MA,
USA). Following several washes with Tris-buffered saline with
Tween-20, the membranes were incubated with horseradish
peroxidase-conjugated goat anti-rabbit IgG (1:5,000; ZB-2306;
Zhongshan Gold Bridge Biological Technology Co., Beijing, China)
for 2 h at room temperature and then washed with TBS-T. Proteins
were detected using enhanced chemiluminescence
RapidStep™ ECL, according to the manufacturer's protocol
(cat. no. 345818; Merck KGaA). ImageJ 1.8.0 (National Institutes of
Health, Bethesda, MD, USA) was applied to quantify the relative
protein levels. GAPDH was used as an internal control.
Apoptosis assay
To determine the effects of TG or MTX on the
apoptosis of PC12 cells, PC cells (50–60% confluence) were treated
with 5 µM TG for 24 h at 37°C. Once they reached 50–60% confluence,
PC12 cells were incubated with or without 100 ng/ml MTX at 37°C for
48 h. Subsequently, cells were washed with 1X PBS three times and
an Annexin V-fluorescein (FITC)-propidium iodide (PI) Apoptosis kit
(Beyotime Institution of Biotechnology, Shanghai, China) according
to the manufacturer's protocol. Cells were analyzed by FC500 flow
cytometry instrument equipped with CXP software (Beckman Coulter,
Bethesda, MA, USA).
Terminal
deoxynucleotidyl-transferase-mediated dUTP nick end (TUNEL)
In the SCI model group, apoptotic cells in the
spinal cord were examined after 7 days of SCI, while the number of
apoptotic cells in the MTX group were evaluated after 7 days
post-administration of MTX. Four-µm-thick spinal cord tissues were
acquired following fixation, dehydration, paraffin embedding and
serial sectioning. Nuclear fragmentation was detected using TUNEL
staining with an In Situ Cell Death Detection kit (Roche
Diagnostics, Indianapolis, IN, USA) according to the supplier's
instructions. The transfected cells were fixed using 4%
paraformaldehyde for 30 min, followed by incubation with TUNEL
buffer for 1 h at 37°C. After rinsing with PBS, the number of
TUNEL-positive apoptotic cells and the total number of cells in
five different random high-power fields were counted using a
microscope (Olympus Corporation, Tokyo, Japan) at a magnification
of 400. The percentage of apoptotic cells was calculated as the
ratio of the number of TUNEL-positive cells to the total number of
cells.
Statistical analysis
Data were expressed as the mean ± standard error of
the mean, as indicated. Each experiment was performed in
triplicate. Multiple comparisons were performed using one-way
analysis of variance followed by Tukey's multiple comparison test.
P<0.05 was considered to indicate a statistically significant
difference. The data were analyzed using SPSS software, version
13.0 (SPSS, Inc., Chicago, IL, USA).
Results
MTX inhibits TG-induced PC12 cell
apoptosis
PC12 cells are cancer cells that have been used as a
cell model for the study of SCI (24–26) and
were therefore selected for use in the present study. PC12 cells
were treated with TG to establish an in vitro ERS cell
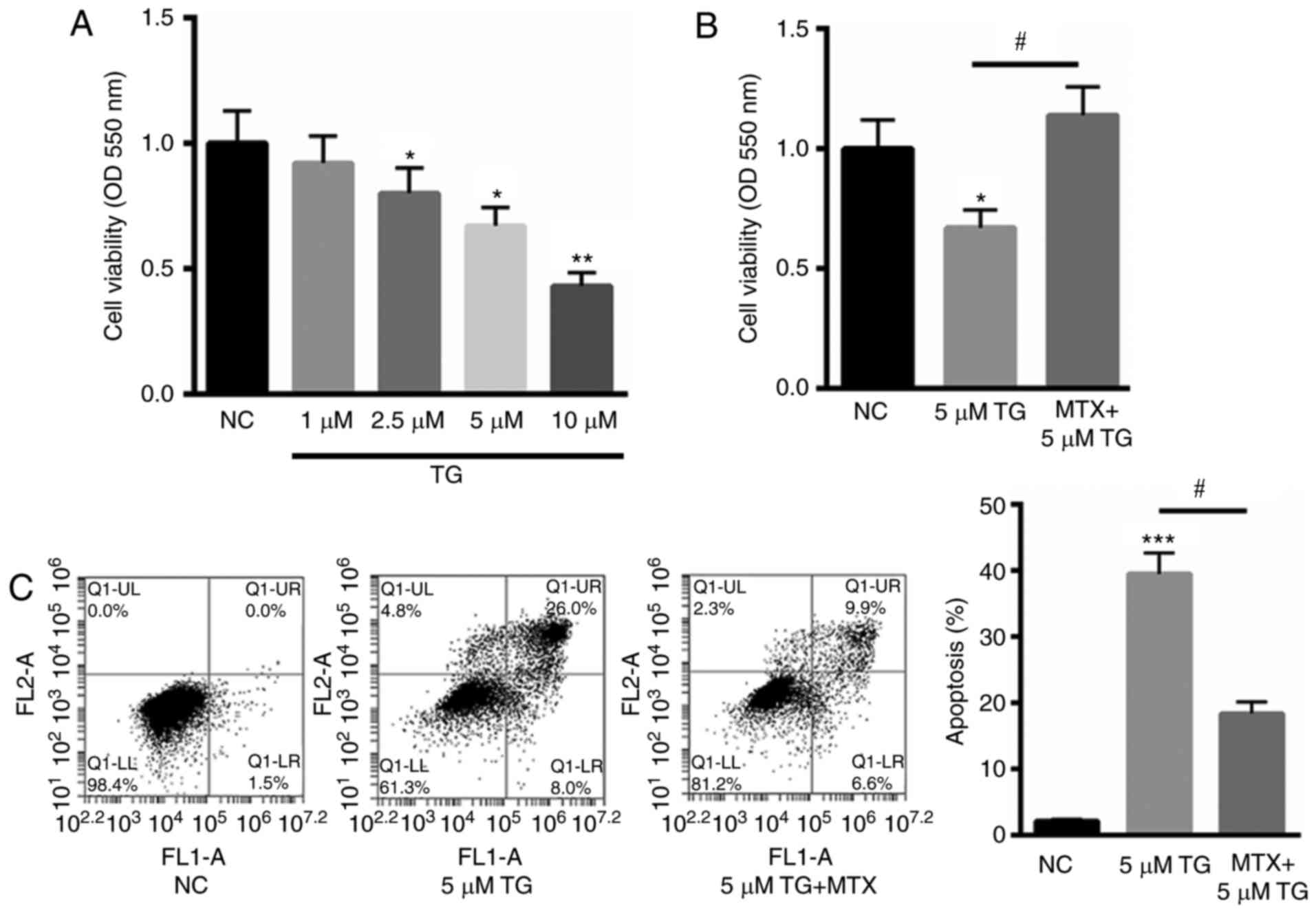
model. As indicated in Fig. 1A,
treatment with 1, 2.5, 5 and 10 µM TG decreased cell viability to
92, 80, 67 and 43%, in a dose-dependent manner. As 10 µM TG reduced
>50% cell viability, for subsequent experiments, 5 µM TG was
selected to stimulate PC12 cells. Subsequently, PC12 cells were
stimulated with TG and assessed in the presence or absence of 100
ng/ml MTX. The results indicated that MTX significantly reversed
the TG-induced reduction in cell viability (P<0.05; Fig. 1B). Furthermore, the protective role
of MTX in TG-induced PC12 cell apoptosis was assessed using Annexin
V-FITC staining. Compared with the normal control (NC), 5 µM TG
significantly increased cell apoptosis (P<0.001). However,
treatment with MTX significantly reduced this effect (P<0.05;
Fig. 1C). These results indicate
that MTX may inhibit TG-induced apoptosis and may be involved in
ERS.
MTX inhibits TG-induced ERS in PC12
cells
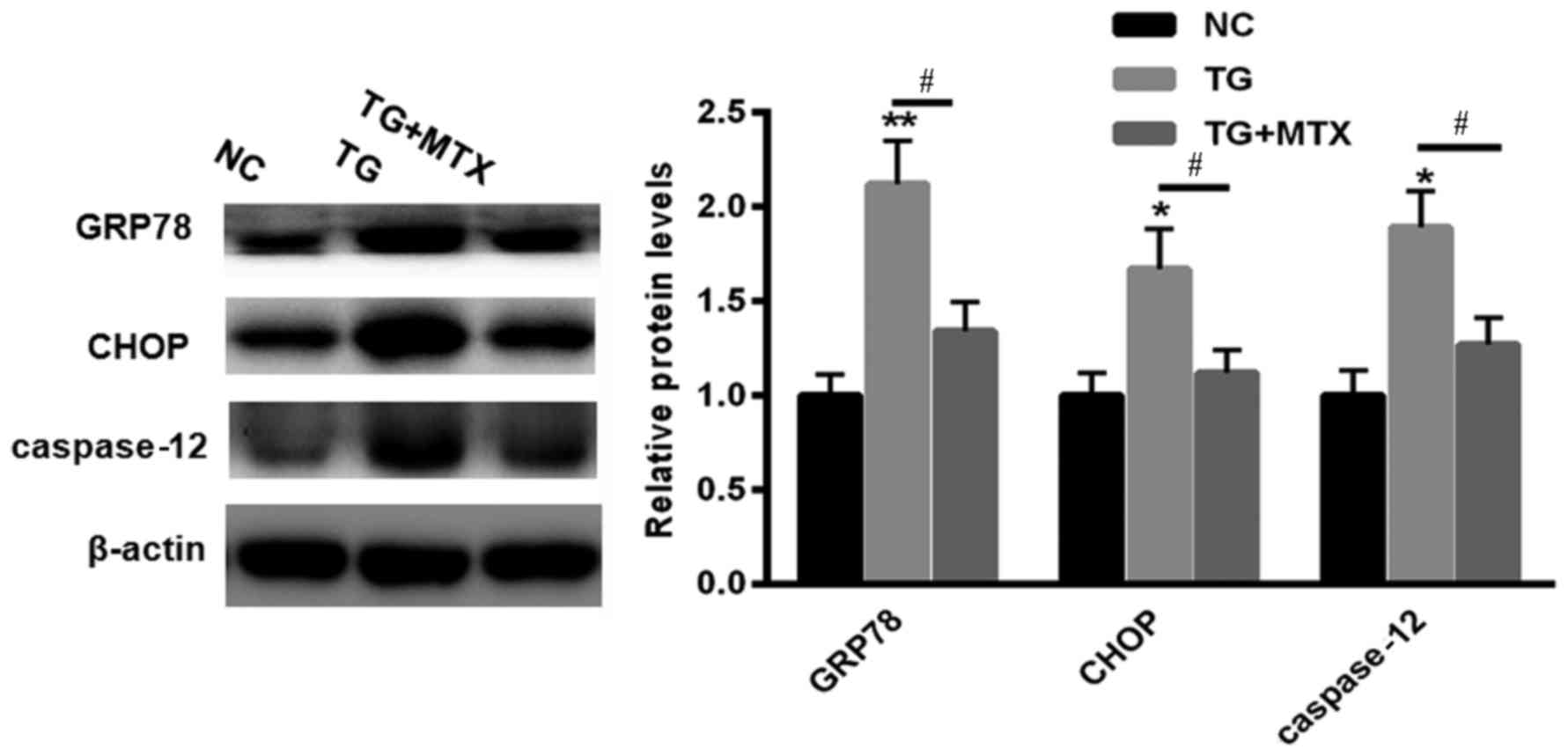
To explore the role of MTX in TG-induced ERS, levels
of GRP78, CHOP and caspase-12 expression in PC12 cells incubated
with 5 µM TG in the presence or absence of MTX were assessed using
western blotting. Levels of GRP78, CHOP and caspase-12 expression
were all significantly increased compared with the NC group
following TG treatment (P<0.01 or P<0.05; Fig. 2). However, following administration
of MTX, levels of GRP78, CHOP and caspase-12 expression were all
significantly decreased compared with the PC12 cells that received
TG treatment alone (all P<0.05; Fig.
2). These results suggest that MTX may inhibit the ERS induced
by TG in PC12 cells.
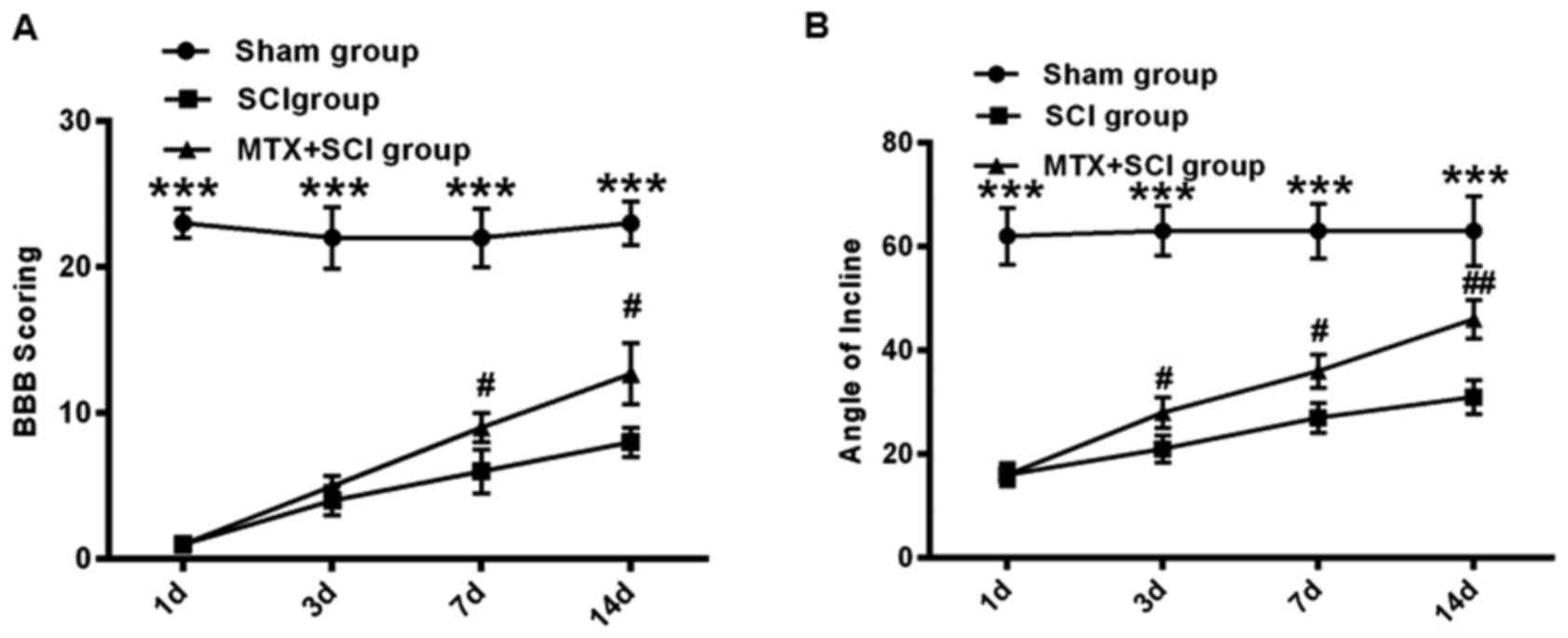
MTX improves motor behavioral function
in rats with SCI
The effect of MTX on ERS was further assessed in the
SCI rat model. Following SCI, the hind legs of rats were paralyzed
and the rats exhibited limited movement. To assess the hind limb
function recovery following MTX administration, the rats in the
sham, SCI and MTX+SCI groups were observed and their limb function
was scored. Using the BBB scoring system, hind limb function
recovery was assessed in rats 1, 3, 7 and 14 days following SCI.
Compared with the sham group, severe paralysis was identified in
all rats that had undergone SCI (P<0.001; Fig. 3A). However, on days 7 and 14,
movement capacity was significantly improved in the MTX+SCI group
compared with the SCI group (P<0.05; Fig. 3A). After 14 days, the BBB score was
increased by 12.7±2.1 in the MTX+SCI group compared with that in
the SCI group (P<0.05; Fig. 3A).
Additionally, as determined by inclined plane test, the hind limb
strength of rats in the sham group was strong. Rats in the sham
group were able to stand on a 65° sloped plate for ≥5 sec. The hind
limb strength of rats in the SCI group was significantly weaker,
and they were unable to stand on the sloped plate for ≥5 sec
(P<0.001). However, rats from the MTX-SCI group exhibited
significantly strengthened hind limb force compared with the SCI
group 3 and 7 days after SCI initiation (P<0.05), and this
improvement was continued 14 days after SCI induction (P<0.01;
Fig. 3B).
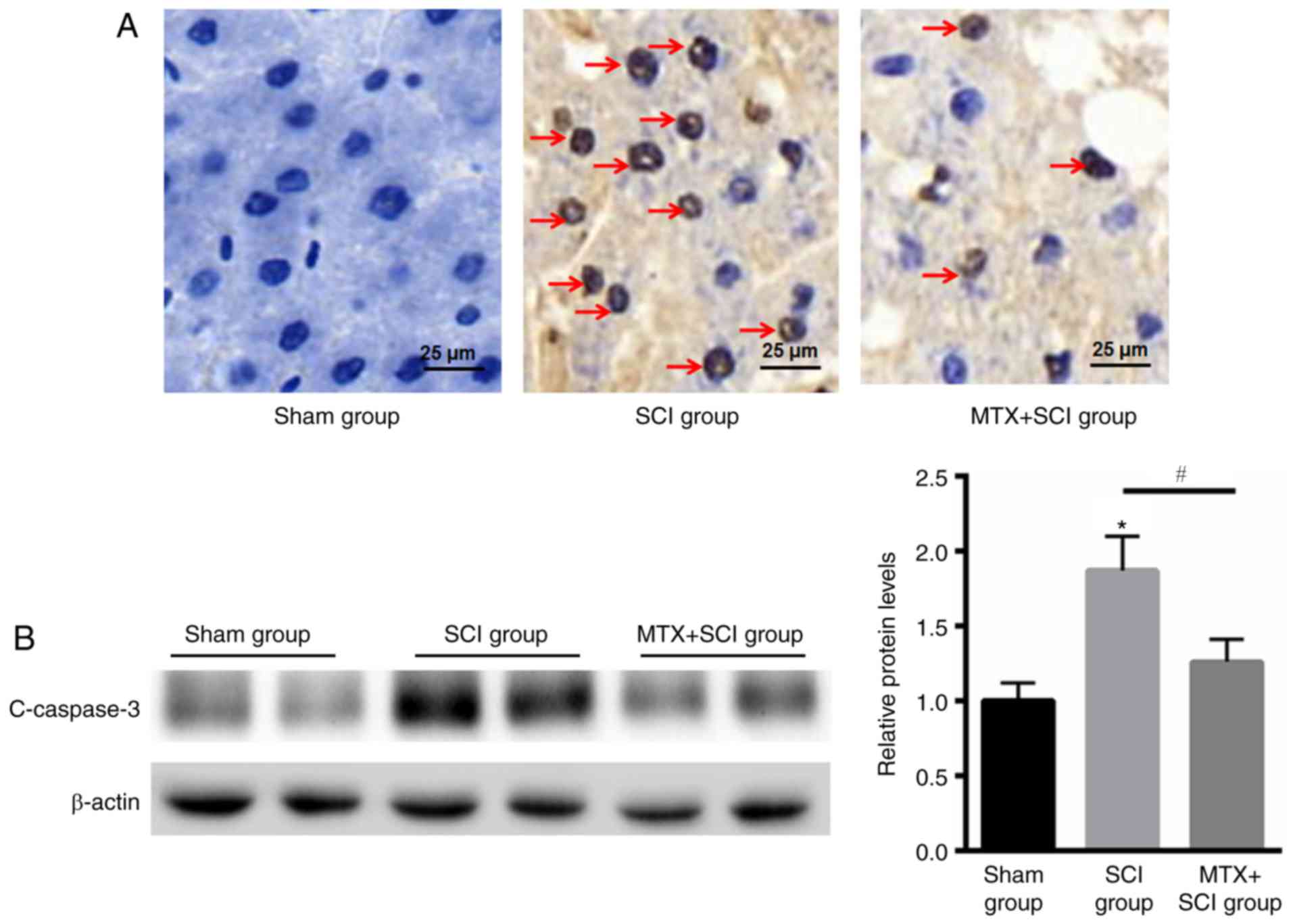
MTX reduces apoptosis in rats with
SCIs
TUNEL staining indicated that there were no
apoptotic cells present in rats from the sham group. In the SCI
model group apoptotic cells were identified after 7 days, but the
number of apoptotic cells was decreased in the MTX group after an
additional 7 days (Fig. 4A). The
caspase family serves an important role in mediating apoptosis.
Caspase-3 is a key molecule that functions in various signaling
pathways associated with apoptotic signaling (27). To determine the anti-apoptotic effect
of MTX in rat spinal cord nerve cells, levels of cleaved caspase-3
expression were assessed using western blotting. The results
indicated that levels of cleaved caspase-3 expression were
significantly increased in rats that had undergone SCI compared
with the sham group; however, MTX treatment significantly
attenuated this effect at day 7 (P<0.05; Fig. 4B). These data suggested that MTX may
protect rats from SCI by suppressing apoptosis.
MTX inhibits the expression of GRP78
in cells in the region of SCI
To detect whether SCI induces ERS, levels of ATF6,
IRE1α, eIF2α and GRP78 expression were assed using western
blotting. Compared with sham group, levels of ATF6, IRE1α, eIF2α
and GRP78 expression were significantly increased following SCI
(P<0.05, P<0.01 and P<0.001; Fig. 5A). This suggests that SCI may induce
ERS. Notably, on day 7, levels of ATF6, IRE1α, eIF2α and GRP78
expression were significantly decreased in the MTX+SCI group
compared with the SCI group on day 7 days (each band represented
one rat; P<0.05, P<0.01 and P<0.001; Fig. 5B). These data indicate that MTX may
protect against ERS.
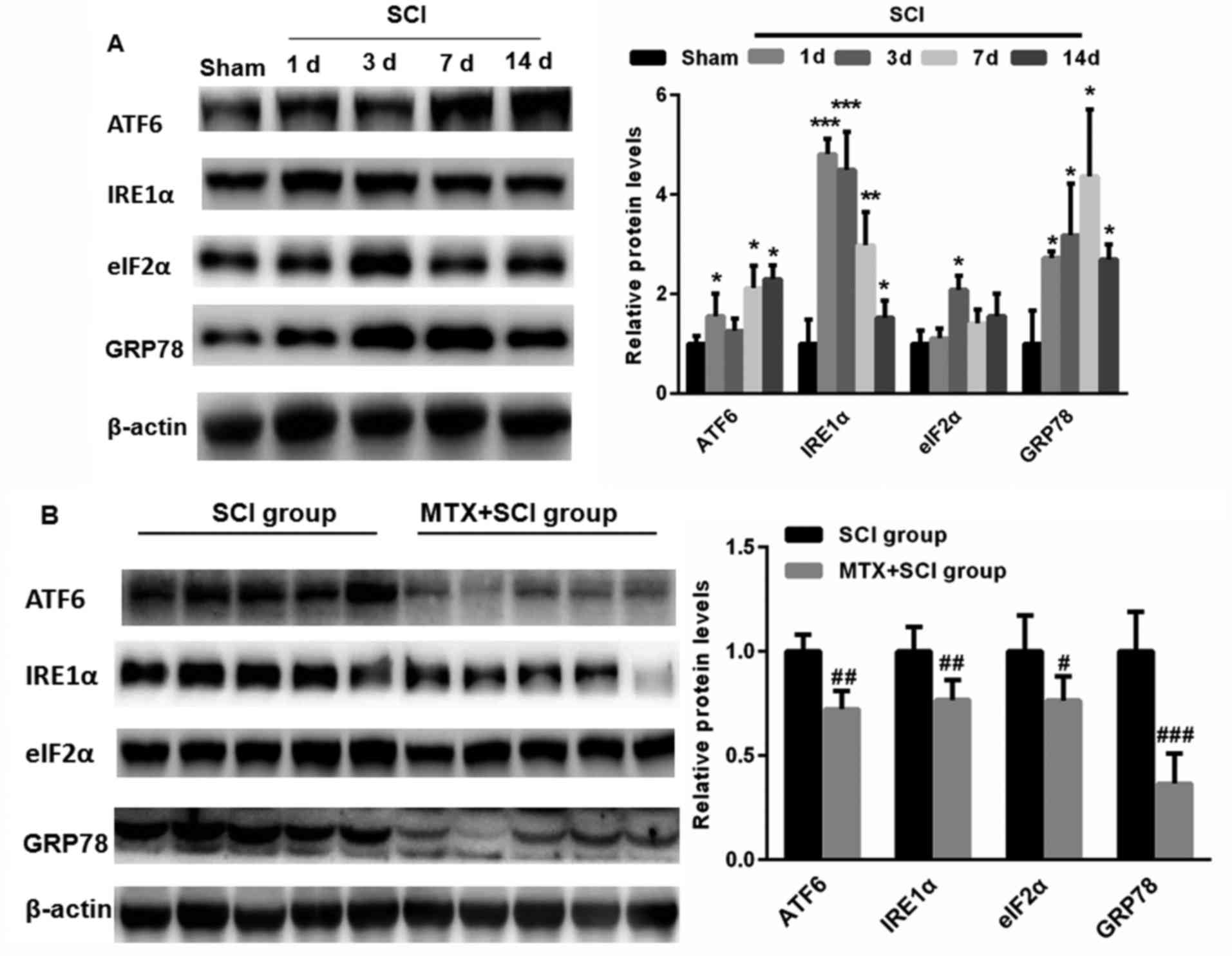 | Figure 5.MTX inhibited the expression of
endoplasmic reticulum stress-associated proteins in the region of
SCI. (A) Compared with the sham group, levels of ATF6, IRE1α, eIF2α
and GRP78 expression were significantly increased in rats following
SCI. (B) Following treatment with MTX, levels of ATF6, IRE1α, eIF2α
and GRP78 expression were significantly decreased compared with the
SCI group (n=8 rats per group). Data are presented as the mean ±
standard error of the mean. *P<0.05, **P<0.01, ***P<0.001
vs. Sham; #P<0.05, ##P<0.01,
###P<0.001 vs. SCI. SCI, spinal cord injury; MTX,
methotrexate; ATF6, activating transcription factor 6; IRE1α,
serine/threonine-protein kinase/endoribonuclease inositol-requiring
enzyme 1 α; eIF2α, eukaryotic initiation factor 2 α; GRP78,
glucose-regulated protein 78; c-caspase-3, cleaved caspase-3. |
Discussion
MTX is the most commonly used disease-modifying
anti-rheumatic agent and is also effective at treating patients
with breast cancer (28,29). Previous studies have demonstrated
that MTX induces greater cytotoxicity and growth inhibition in
leukemia and other malignant cell types compared with other
treatments (30,31). It has been demonstrated that
sustained ERS is a major contributor to MTX-induced cell death, as
indicated by the induction of several unfolded protein response
markers, which ultimately induce apoptosis (32). Furthermore, MTX prevents epidural
fibrosis via the ERS signaling pathway (33). The present study aimed to evaluate
the possible protective effects and the potential mechanisms of
low-dose MTX in rats with SCI and in PC12 cells.
In the present study, an in vitro cell model
of ERS was established. PC12 cells were treated with 1, 2.5, 5 and
10 µM TG, and MTT assays demonstrated that TG decreased PC12 cell
viability in a dose-dependent manner. Additionally, flow cytometric
analysis indicated that TG significantly increased the apoptosis of
PC12 cells. It has been suggested that, during the early stages of
ERS, upregulation of GRP78 may aid cell survival by removing
misfolded proteins; however, prolonged elevation of ERS levels may
induce cell death (34). In addition
to the activation of GRP78 during sustained ERS, the activation of
CHOP, p53 upregulated modulator of apoptosis and caspase-12 is
induced, thereby further promoting cell death (12,35). In
accordance with previous studies, the results of the present study
indicated that levels of the ERS-associated proteins GRP78, CHOP
and caspase-12 were significantly increased in PC12 cells following
treatment with TG. An increase in cell apoptosis and the expression
of ERS-associated proteins was identified in a rat model of SCI.
The results of the present study imply that ERS may be involved in
this process.
Apoptosis is a type of programmed cell death that
differs from necrosis (36). This
process serves an important role in SCI, particularly during
secondary injury. By blocking apoptosis, cells may survive and
retain more nerve function (37).
Neuronal apoptosis following SCI is an important pathological
process and often leads to secondary injury in SCI (38). It has been reported that excessive
apoptosis may result in decreased functioning of the spinal cord
anterior horn and the spinal nerve fibers (38).
The ERS response regulates the homeostasis of
organisms by modulating protein degradation and protein folding
(39). Under normal physiological
conditions, the ER chaperone GRP78 combines with PERK, ATF6 and
IRE1, and is maintained in its inactive state (40). When the UPR is activated, unfolded
proteins accumulate in the ER, which then trigger the ERS-induced
cell apoptosis (41). GRP78,
CHOP/GADDl53 and caspase-12 are ERS-specific transcription factors
that are not activated in either the death receptor or the
mitochondrial signaling pathways (42). In the present study, the motor
behavioral function of rats with SCI was significantly improved
following MTX treatment on day 7. Furthermore, the present study
revealed that decreased neuronal cell apoptosis was accompanied by
significantly reduced levels of GRP78, CHOP and caspase-12
expression, indicating that the neuroprotective effects of MTX may
be associated with the inhibition of ERS-induced apoptosis.
In conclusion, the present study revealed that MTX
treatment increased neuronal survival in the region of SCI and
improved the functional recovery of rats with acute SCI. According
to the results from the present in vivo and in vitro
experiments, it was suggested that the protective effect of MTX in
neuronal cells may be mediated by the inhibition of ERS-induced
apoptosis. These data suggest that MTX is suitable for the
treatment of SCI as it is able to reduce ERS-induced apoptosis.
However, further studies are required to determine the most
effective application of MTX in the treatment of SCI.
Acknowledgements
Not applicable.
Funding
This study was supported by a grant from Doctoral
Fund of Mudanjiang Medical University (grant no.
MDJU-20170234).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
FR performed the experiments and analyzed the data.
WT performed the experiments. XG and KL performed part of the
animal experiments. JW designed the whole experiments, analyzed the
data and gave final approval of the version to be published.
Ethics approval and consent to
participate
The present study was approved by the Animal Ethics
Committee of the Hongqi Hospital Affiliated to Mudanjiang Medical
University (Mudanjiang, China).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Motiei-Langroudi R and Sadeghian H:
Traumatic spinal cord injury: Long-term motor, sensory, and urinary
outcomes. Asian Spine J. 11:412–418. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Shank CD, Walters BC and Hadley MN:
Management of acute traumatic spinal cord injuries. Handb Clin
Neurol. 140:275–298. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen Y, Tang Y, Allen V and DeVivo MJ:
Aging and spinal cord injury: External causes of injury and
implications for prevention. Top Spinal Cord Inj Rehabil.
21:218–226. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lee JY, Maeng S, Kang SR, Choi HY, Oh TH,
Ju BG and Yune TY: Valproic acid protects motor neuron death by
inhibiting oxidative stress and endoplasmic reticulum
stress-mediated cytochrome C release after spinal cord injury. J
Neurotrauma. 31:582–594. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu X, Yang J, Li Z, Liang F, Wang Y, Su Q
and Li C: Hyperbaric oxygen treatment protects against spinal cord
injury by inhibiting endoplasmic reticulum stress in rats. Spine
(Phila Pa 1976). 40:E1276–E1283. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Matsuyama D, Watanabe M, Suyama K, Kuroiwa
M and Mochida J: Endoplasmic reticulum stress response in the rat
contusive spinal cord injury model-susceptibility in specific cell
types. Spinal Cord. 52:9–16. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ohri SS, Maddie MA, Zhang Y, Shields CB,
Hetman M and Whittemore SR: Deletion of the pro-apoptotic
endoplasmic reticulum stress response effector CHOP does not result
in improved locomotor function after severe contusive spinal cord
injury. J Neurotrauma. 29:579–588. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang ZL, Zhou ZG, Chen Y, Li XT and Sun
YS: Support vector machines model of computed tomography for
assessing lymph node metastasis in esophageal cancer with
neoadjuvant chemotherapy. J Comput Assist Tomogr. 41:455–460. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ohri SS, Maddie MA, Zhao Y, Qiu MS, Hetman
M and Whittemore SR: Attenuating the endoplasmic reticulum stress
response improves functional recovery after spinal cord injury.
Glia. 59:1489–1502. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ohri SS, Mullins A, Hetman M and
Whittemore SR: Inhibition of GADD34, the stress-inducible
regulatory subunit of the endoplasmic reticulum stress response,
does not enhance functional recovery after spinal cord injury. PLoS
One. 9:e1097032014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Penas C, Guzmán MS, Verdú E, Forés J,
Navarro X and Casas C: Spinal cord injury induces endoplasmic
reticulum stress with different cell-type dependent response. J
Neurochem. 102:1242–1255. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tka Hadj Ayed K, Boussaid Mahfoudh A,
Zaouali MA, Kammoun R, Bejaoui M, Ghoul Mazgar S, Catafau Rosello J
and Ben Abdennebi H: Melatonin modulates endoplasmic reticulum
stress and Akt/GSK3-beta signaling pathway in a rat model of renal
warm ischemia reperfusion. Anal Cell Pathol (Amst).
2015:6351722015.PubMed/NCBI
|
|
13
|
Halliday M and Mallucci GR: Review:
Modulating the unfolded protein response to prevent
neurodegeneration and enhance memory. Neuropathol Appl Neurobiol.
41:414–427. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ben Mosbah I, Alfany-Fernández I, Martel
C, Zaouali MA, Bintanel-Morcillo M, Rimola A, Rodés J, Brenner C,
Roselló-Catafau J and Peralta C: Endoplasmic reticulum stress
inhibition protects steatotic and non-steatotic livers in partial
hepatectomy under ischemia-reperfusion. Cell Death Dis. 1:e522010.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yang X, Shao H, Liu W, Gu W, Shu X, Mo Y,
Chen X, Zhang Q and Jiang M: Endoplasmic reticulum stress and
oxidative stress are involved in ZnO nanoparticle-induced
hepatotoxicity. Toxicol Lett. 234:40–49. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Curtis JR, Wright GC, Strand V, Davis CS,
Hitraya E and Sasso EH: Reanalysis of the multi-biomarker disease
activity score for assessing disease activity in the abatacept
versus adalimumab comparison in biologic-naive rheumatoid arthritis
subjects with background methotrexate study: Comment on the Article
by Fleischmann et al. Arthritis Rheumatol. 69:863–865. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Fleischmann R, Mease PJ, Schwartzman S,
Hwang LJ, Soma K, Connell CA, Takiya L and Bananis E: Efficacy of
tofacitinib in patients with rheumatoid arthritis stratified by
background methotrexate dose group. Clin Rheumatol. 36:15–24. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ito Y, Hozumi K, Okada Y and Kurimoto S:
Adalimumab with methotrexate in treatment-naive Japanese patients
with rheumatoid arthritis at risk of progressive structural joint
damage: A postmarketing observational study. Rheumatol Ther.
4:151–166. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen H, Li J, Liang S, Lin B, Peng Q, Zhao
P, Cui J and Rao Y: Effect of hypoxia-inducible factor-1/vascular
endothelial growth factor signaling pathway on spinal cord injury
in rats. Exp Ther Med. 13:861–866. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Vachon P, Faubert S, Blais D, Comtois A
and Bienvenu JG: A pathophysiological study of abdominal organs
following intraperitoneal injections of chloral hydrate in rats:
comparison between two anaesthesia protocols. Lab Anim. 34:84–90.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Nakase Y, Hagiwara A, Kin S, Fukuda K, Ito
T, Takagi T, Fujiyama J, Sakakura C, Otsuji E and Yamagishi H:
Intratumoral administration of methotrexate bound to activated
carbon particles: Antitumor effectiveness against human colon
carcinoma xenografts and acute toxicity in mice. J Pharmacol Exp
Ther. 311:382–387. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Thuret S, Moon LD and Gage FH: Therapeutic
interventions after spinal cord injury. Nat Rev Neurosci.
7:628–643. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rivlin AS and Tator CH: Objective clinical
assessment of motor function after experimental spinal cord injury
in the rat. J Neurosurg. 47:577–581. 1977. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Qiao Y, Peng C, Li J, Wu D and Wang X:
Spinal cord ischemia-reperfusion causes damage of neurocyte by
inhibiting RAP2C. Neurol Res. 39:877–884. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tian R and Shi R: Dimercaprol is an
acrolein scavenger that mitigates acrolein-mediated PC-12 cells
toxicity and reduces acrolein in rat following spinal cord injury.
J Neurochem. 141:708–720. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lee DH, Seubert S, Huhn K, Brecht L,
Rötger C, Waschbisch A, Schlachetzki J, Klausmeyer A, Melms A,
Wiese S, et al: Fingolimod effects in neuroinflammation: Regulation
of astroglial glutamate transporters? PLoS One. 12:e01715522017.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xia Y, Xia H, Chen D, Liao Z and Yan Y:
Mechanisms of autophagy and apoptosis mediated by JAK2 signaling
pathway after spinal cord injury of rats. Exp Ther Med.
14:1589–1593. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Park JH, Im SA, Byun JM, Kim KH, Kim JS,
Choi IS, Kim HJ, Lee KH, Kim TY, Han SW, et al: Cyclophosphamide,
methotrexate and 5-fluorouracil as palliative treatment for heavily
pretreated patients with metastatic breast cancer: A multicenter
retrospective analysis. J Breast Cancer. 20:347–355. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Fekry B, Esmaeilniakooshkghazi A, Krupenko
SA and Krupenko NI: Ceramide synthase 6 is a novel target of
methotrexate mediating its antiproliferative effect in a
p53-dependent manner. PLoS One. 11:e01466182016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Giordano L, Akinyede O, Bhatt N, Dighe D
and Iqbal A: Methotrexate-induced neurotoxicity in hispanic
adolescents with high-risk acute leukemia-a case series. J Adolesc
Young Adult Oncol. 6:494–498. 2017.PubMed/NCBI
|
|
31
|
Bohme D, Krieghoff J and Beck-Sickinger
AG: Double methotrexate-modified neuropeptide Y analogues express
increased toxicity and overcome drug resistance in breast cancer
cells. J Med Chem. 59:3409–3417. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kuznetsov JN, Leclerc GJ, Leclerc GM and
Barredo JC: AMPK and Akt determine apoptotic cell death following
perturbations of one-carbon metabolism by regulating ER stress in
acute lymphoblastic leukemia. Mol Cancer Ther. 10:437–447. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chen H, Yan L, Wang J, Sun Y, Li X, Zhao
S, Wang D, Zhu G and Liang Y: Methotrexate prevents epidural
fibrosis through endoplasmic reticulum stress signalling pathway.
Eur J Pharmacol. 796:131–138. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sharma S, Sarkar J, Haldar C and Sinha S:
Melatonin reverses fas, E2F-1 and endoplasmic reticulum stress
mediated apoptosis and dysregulation of autophagy induced by the
herbicide atrazine in murine splenocytes. PLoS One. 9:e1086022014.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Tuñón MJ, San-Miguel B, Crespo I, Laliena
A, Vallejo D, Álvarez M, Prieto J and González-Gallego J: Melatonin
treatment reduces endoplasmic reticulum stress and modulates the
unfolded protein response in rabbits with lethal fulminant
hepatitis of viral origin. J Pineal Res. 55:221–228. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sui T, Ge DW, Yang L, Tang J, Cao XJ and
Ge YB: Mitomycin C induces apoptosis in human epidural scar
fibroblasts after surgical decompression for spinal cord injury.
Neural Regen Res. 12:644–653. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
He Z, Zhou Y, Huang Y, Wang Q, Zheng B,
Zhang H, Li J, Liu Y, Wu F, Zhang X, et al: Dl-3-n-butylphthalide
improves functional recovery in rats with spinal cord injury by
inhibiting endoplasmic reticulum stress-induced apoptosis. Am J
Transl Res. 9:1075–1087. 2017.PubMed/NCBI
|
|
38
|
Pei JP, Fan LH, Nan K, Li J, Dang XQ and
Wang KZ: HSYA alleviates secondary neuronal death through
attenuating oxidative stress, inflammatory response, and neural
apoptosis in SD rat spinal cord compression injury. J
Neuroinflammation. 14:972017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang C, Shi D, Song X, Chen Y, Wang L and
Zhang X: Calpain inhibitor attenuates ER stress-induced apoptosis
in injured spinal cord after bone mesenchymal stem cells
transplantation. Neurochem Int. 97:15–25. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Xue Q, Li C, Chen J, Guo H, Li D and Wu X:
The protective effect of the endoplasmic reticulum stress-related
factors BiP/GRP78 and CHOP/Gadd153 on noise-induced hearing loss in
guinea pigs. Noise Health. 18:247–255. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhu SP, Wang ZG, Zhao YZ, Wu J, Shi HX, Ye
LB, Wu FZ, Cheng Y, Zhang HY, He S, et al: Gelatin nanostructured
lipid carriers incorporating nerve growth factor inhibit
endoplasmic reticulum stress-induced apoptosis and improve recovery
in spinal cord injury. Mol Neurobiol. 53:4375–4386. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Feng J, Chen X and Sun X, Wang F and Sun
X: Expression of endoplasmic reticulum stress markers GRP78 and
CHOP induced by oxidative stress in blue light-mediated damage of
A2E-containing retinal pigment epithelium cells. Ophthalmic Res.
52:224–233. 2014. View Article : Google Scholar : PubMed/NCBI
|