Introduction
Type 2 diabetes is an endocrine and metabolic
disease characterized by a disturbance of the blood glucose and
lipid metabolism. The cause of this disease is pancreatic β-cell
dysfunction and insulin resistance (1,2).
Patients with newly diagnosed type 2 diabetes have impaired insulin
secretion and insulin resistance. The lack of insulin secretion is
mainly due to the inhibition of islet function caused by
hyperglycemia (3). Hyperglycemia
induces and exacerbates pancreatic β-cell damage and insulin
resistance. Impaired secretion of β-cells in pancreatic islets
results in oxidative stress and subsequent apoptosis. Under
high-glucose conditions, the oxidative stress in pancreatic β-cells
increases when a series of cytokines and reactive oxygen species
(ROS) are released, triggering the activation of death signaling
pathways in islet cells (4).
However, early islet β-cell dysfunction is partially reversible and
early intensive treatment may control blood glucose levels to
protect islet β-cells to achieve the goal of improving the
prognosis of patients with type 2 diabetes (5,6).
Programs for intensive treatment commonly include the use of
insulin pumps for continuous subcutaneous infusion of insulin and
short-acting pre-prandial insulin may be added as subcutaneous
intermediate-acting or long-term insulin as an injection prior to
bedtime.
Insulin pump therapy [continuous subcutaneous
insulin infusion (CSII)] activates the physiological secretion
pattern of insulin in patients with newly detected type 2 diabetes,
while not affecting oral hypoglycemic agents, and which may rapidly
and efficiently regulate blood glucose to the anticipated level in
order to relieve high glycotoxicity and enhance the islet function
of islet β-cells.
The present study examined and compared the effect
of two different short-term intensive treatment schedules of
insulin pump and numerous subcutaneous insulin injections on blood
lipids, blood glucose and islet β-cell secretion in patients with
newly diagnosed type 2 diabetes mellitus, and explored the possible
mechanisms.
Materials and methods
General data
A total of 100 patients with newly diagnosed type 2
diabetes mellitus hospitalized at Daqing Oilfield General Hospital
(Daqing, China) between January 2016 to December 2017 were selected
for the present study. Patients who met the diagnostic criteria for
diabetes of the World Health Organization from 1999 (7), fasting plasma glucose >11.1 mmol/l
and/or glycosylated hemoglobin (HbA1c) ≥9.0%, never received any
oral hypoglycemics, lipid-regulating drugs or insulin treatment,
glutamic acid decarboxylase antibody, were negative for islet cell
antibody and urinary protein, had no acute complications and no
diabetic retinopathy on fundus examination were included in the
present study. The exclusion criteria were as follows: Severe
heart, liver or renal insufficiency or severe systemic
complications, accompanied by endocrine or immune system diseases.
Patients were divided into a control group and an experimental
group according to the random number table method, with 50 patients
in each group. In the control group, there were 28 males and 22
females aged between 40 to 57, with an average age of 48.31±7.83
years and a mean body mass index (BMI) of 27.20±2.67
kg/m2; while the experimental group comprised 27 males
and 23 females aged between 39 to 58 with an average age of
48.53±8.63 years and the average BMI was 27.31±2.95
kg/m2. There was no statistically significant difference
between the two groups in terms of age, sex and other general data,
indicating comparability between the two groups.
Methods
Patients in the control group were administered a
subcutaneous injection of insulin 15 min prior to meals and insulin
was given subcutaneously prior to bedtime. The specific dosage was
calculated according to the patients' BMI. The daily starting dose
was 0.4–0.6 IU/kg, based on blood glucose levels. The dosage was
adjusted according to the monitoring results, with continuous
treatment for 2 weeks. The patients in the study group were treated
with a Misting MTI-PII insulin pump (Milestone). The pumps were
located on the subcutaneous tissue of both sides of the umbilicus,
and insulin aspart was used. The specific dosage was calculated
according to the patients' BMI and the daily starting dose was
0.4–0.6 IU/kg. Of the daily insulin dose, 50% was administered by
continuous subcutaneous injection, whilst the remaining insulin was
divided into three equal portions pumped prior to meals with the
dose adjusted according to blood glucose monitoring results, with
continuous treatment for two weeks. The calculation of the total
amount of insulin, the amount of basal and the amount of
pre-loading insulin prior to meals was calculated for each
patient.
Observational indicators
The patients' fasting venous blood was collected in
the morning and the associated indicators were examined and
compared prior to- and following 2 weeks of treatment. The blood
lipid indicators of total cholesterol (TC), triglycerides (TG),
low-density lipoprotein-cholesterol (LDL-C) and high-density
lipoprotein-cholesterol (HDL-C) were measured with an automatic
blood biochemical analyzer (SMT-100; Perlong Medical Equipment Co.,
Ltd.). The general blood glucose parameters, including fasting
plasma glucose (FPG), 2-h post-prandial blood glucose (2 h PG,
HbA1c level, fasting C-peptide (CP) and post-prandial 2 h
C-peptides (2hC-P) were detected. The insulin function index
[homeostasis model assessment of insulin resistance (HOMA-IR)] and
insulin secretion index [HOMA of β-cell function (HOMA-β)] of islet
cell function were calculated by using the following formula:
HOMA-IR=FPGxFast Insulin/22.5, HOMA-β=20×Fast Insulin/(FPG-3.5).
Oxidative stress markers, including malondialdehyde (MDA) and
superoxide dismutase (SOD) levels, were detected by ELISA (cat. no.
ab188392; Abcam).
Isolation of peripheral blood
mononuclear cells (PBMCs)
Venous blood (12 ml) was collected from the patients
early in the morning. PBMCs were isolated by density gradient
centrifugation in a Ficoll lymphocyte separation solution within 2
h as follows: Anti-coagulation agent was mixed with an equal volume
of PBS, and an equal volume of lymphocyte separation solution was
slowly added, followed by horizontal centrifugation at 12,000 × g
for 25 min at 37°C. The cells in the buffy coat were aspirated and
washed twice with PBS, and the PBMCs were isolated.
Dichloro-dihydro-fluorescein diacetate
(DCFH-DA) assay
The ROS levels in PBMCs were detected by using a
Reactive Oxygen Species ROS Detection Kit (Cell Biolabs, Inc.). The
experimental procedure was according to the manufacturer's
protocol. The mean fluorescence intensity (MFI) of samples was
measured using a flow cytometer with an excitation wavelength of
488 nm and an emission wavelength of 525 nm.
Detection of the mRNA level of heme
oxygenase 1 (HO-1) and Nrf2 nuclear factor erythroid 2-related
factor 2 (Nrf2)
The total RNA of PBMCs was extracted by using TRIzol
and the transcription levels of HO-1 and Nrf2 were detected by
reverse transcription (RT)-quantitative PCR. The specific primers
were as follows: HO-1 forward, 5′-ATGGCCTCCCTGTACCACATc-3′ and
reverse, 5′-TGTTGCGCTCAATCTCCTCCT-3′; Nrf2 forward,
5′-TGAGGTTTCTTCGGCTACGTT-3′ and reverse,
5′-CTTCTGTCAGTTTGGCTTCTGG-3′; GAPDH forward,
5′-ACCAGCCTCAAGATCATCAGC-3′ and reverse,
5′-TGCTAAGCAGTTGGTGGTGC-3′. GAPDH was used as an internal
reference. The complementary DNA was synthesized using the
PrimeScript RT reagent kit (RR037A; Takara Bio, Inc.) according to
the manufacturer's protocol. qPCR was was performed using SYBRGreen
(Thermo Fisher Scientific, Inc.) and the Roche Light Cycler 480 II
(Roche Diagnostics Ltd.). The PCR conditions were as follows:
Denaturation at 95°C for 5 min, followed by 40 cycles of 95°C for
30 sec, 58°C for 30 sec and 72°C for 30 sec. The quantification
cycle (Cq) value was obtained from the PCR amplification curve and
relative quantification was performed using the 2−∆∆Cq
method (8).
Statistical methods
SPSS 17.0 statistical software (SPSS, Inc.) was used
for statistical analysis of the data. The count data are presented
as frequencies and percentages, while measurement data with a
normal distribution are presented as the mean ± standard deviation
analyzed by Shapiro-Wilk test. One-way analysis of variance with
Tukey's post-hoc test was performed to analyze differences among
multiple groups. P<0.05 was considered to indicate statistical
significance.
Results
Short-term intensive insulin pump
therapy improves blood lipid levels
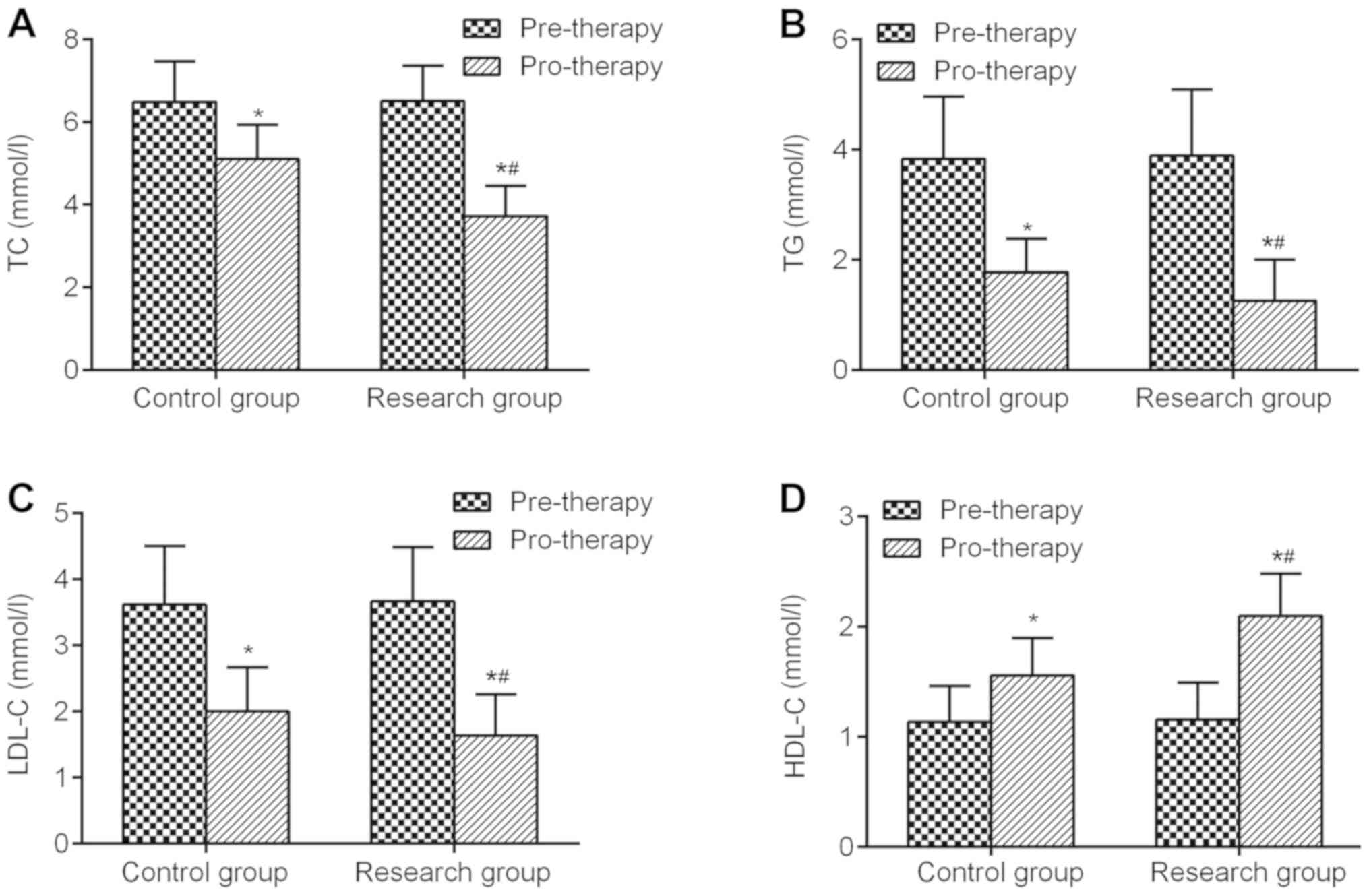
As blood lipid levels, serum TC, TG, LDL-C and HDL-C
level were measured in the two groups. Prior to insulin treatment,
the blood lipid levels of TC, TG, LDL-C and HDL-C were similar
between the two groups, with no statistically significant
difference (Fig. 1). The blood lipid
levels of all of the patients after intensive treatment were
significantly improved. The levels of TC, TG, and LDL-C was
significantly lowered in the experimental group compared with those
in the control group (Fig. 1A-C),
and the level of HDL-C was higher than that in the control group
(Fig. 1D).
Short-term intensive insulin pump
therapy improves glycemic index in patients
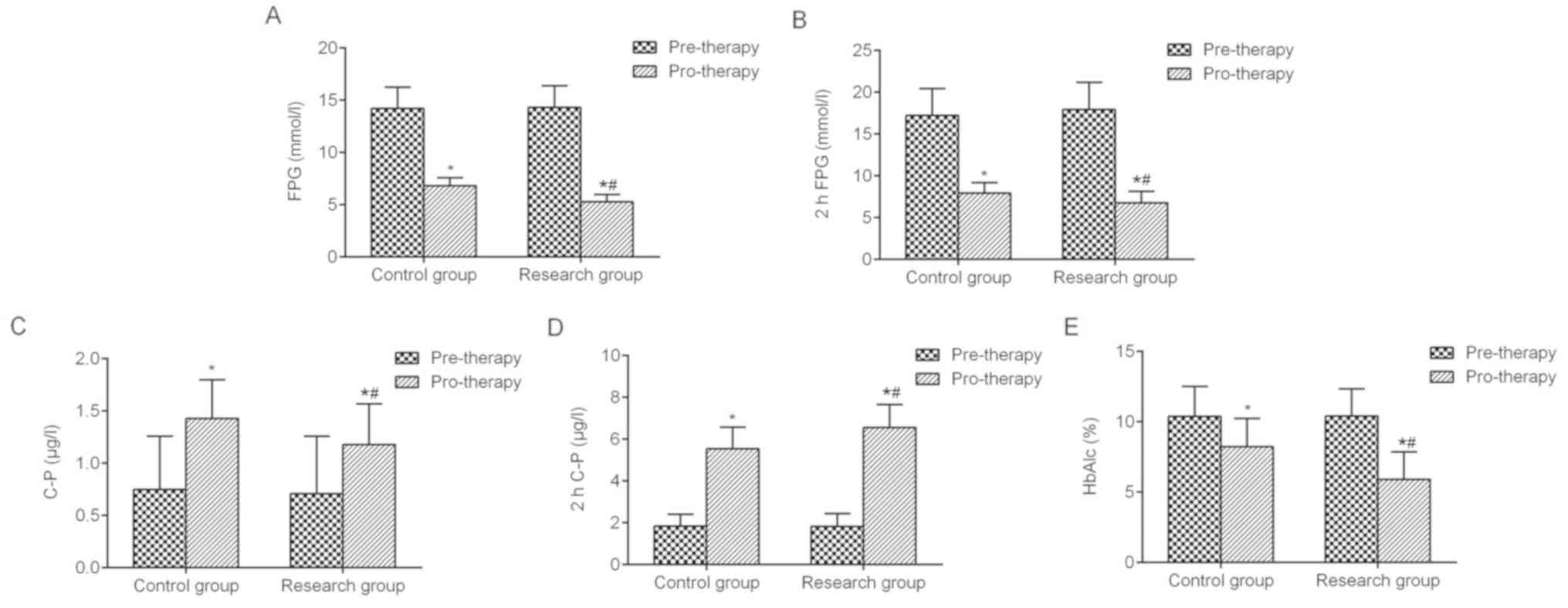
In order to detect blood glucose levels in the two
groups, FPG, 2hPG, C-P, 2hC-P and HbA1c were measured. Prior to
treatment, the serum FPG, 2hPG, CP, 2hC-P and HbA1c levels were
similar between the two groups with no statistically significant
difference (Fig. 2). Of note, the
blood glucose levels in the two groups were significantly improved
after intensive treatment compared with pre-therapy. The levels of
FPG, 2hPG and HbA1c in the experimental group were lower compared
with the control group (Fig. 2A, B and
E), while the levels of CP and 2hC-P were higher compared with
the pre-therapy group (Fig. 2C and
D). Furthermore, the results of pro-therapy between control
group and experimental groups was of remarkable significance.
Short-term intensive insulin pump
therapy improves the function of islet β-cells
To detect the function of islet β-cells in the two
groups, the HOMA-β and HOMA-IR indices were determined. Prior to
treatment, there was no significant difference between the two
groups regarding their HOMA-β and HOMA-IR. After intensive
treatment, the function of the islet β-cells was significantly
improved in each group and the HOMA-β in the experimental group was
significantly higher than that in the control group, while the
HOMA-IR was lower than that in the control group (Table I).
 | Table I.Short-term intensive insulin pump
therapy significantly improves the function of islet β-cells in
newly diagnosed type 2 diabetics. |
Table I.
Short-term intensive insulin pump
therapy significantly improves the function of islet β-cells in
newly diagnosed type 2 diabetics.
| Group/time-point | HOMA-β | HOMA-IR |
|---|
| Control (n=50) |
|
|
|
Pre-therapy | 14.73
(6.53–22.67) | 3.45 (1.87–6.03) |
|
Pro-therapy | 46.53
(30.79–62.43)a | 2.34
(1.09–3.55)a |
| Experimental group
(n=50) |
|
|
|
Pre-therapy | 14.85
(6.81–23.07) | 3.48 (1.92–5.86) |
|
Pro-therapy | 57.57
(32.57–82.63)a,b | 1.36
(0.71–2.15)a,b |
Short-term intensive insulin pump
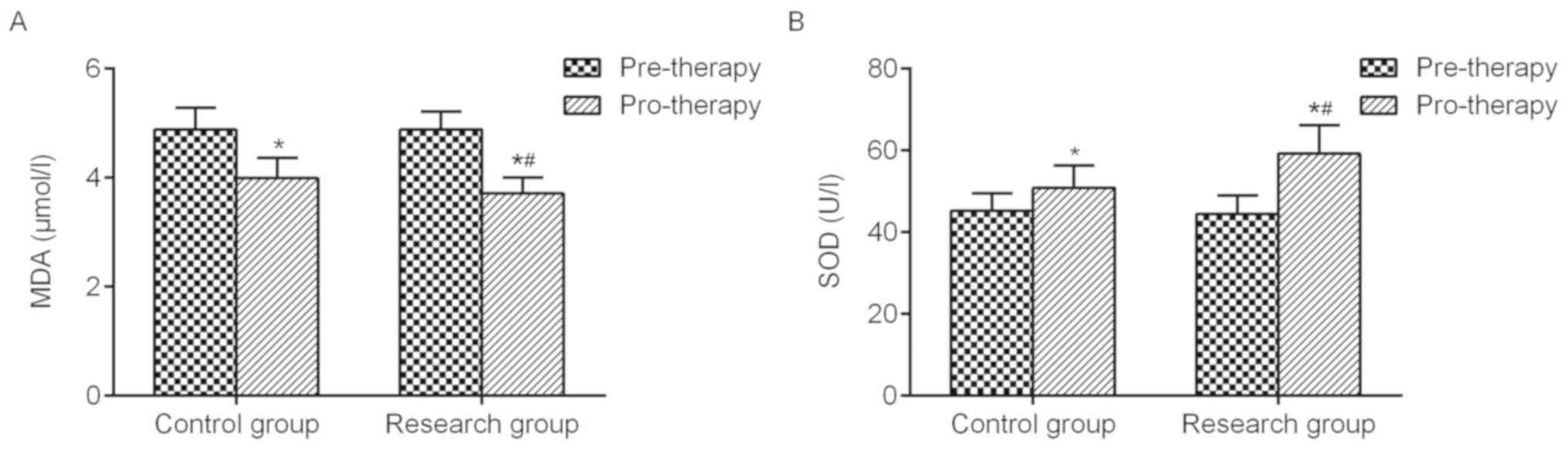
therapy improves oxidative stress
In order to detect redox indicators, the serum MDA
and SOD levels were determined in the two groups. Prior to
treatment, serum MDA and SOD levels were similar between the two
groups and no significant differences were observed. After
intensive treatment, the redox parameters of the two groups were
significantly improved and the serum MDA levels in the experimental
group were significantly lower than those of the control group. The
serum SOD levels in the experimental group were significantly
higher than those in the control group, as presented in Fig. 3A and B. Moreover, the results of
pro-therapy between control group and experimental groups was of
remarkable significance.
Short-term intensive insulin pump
therapy decreases the level of ROS in PBMCs
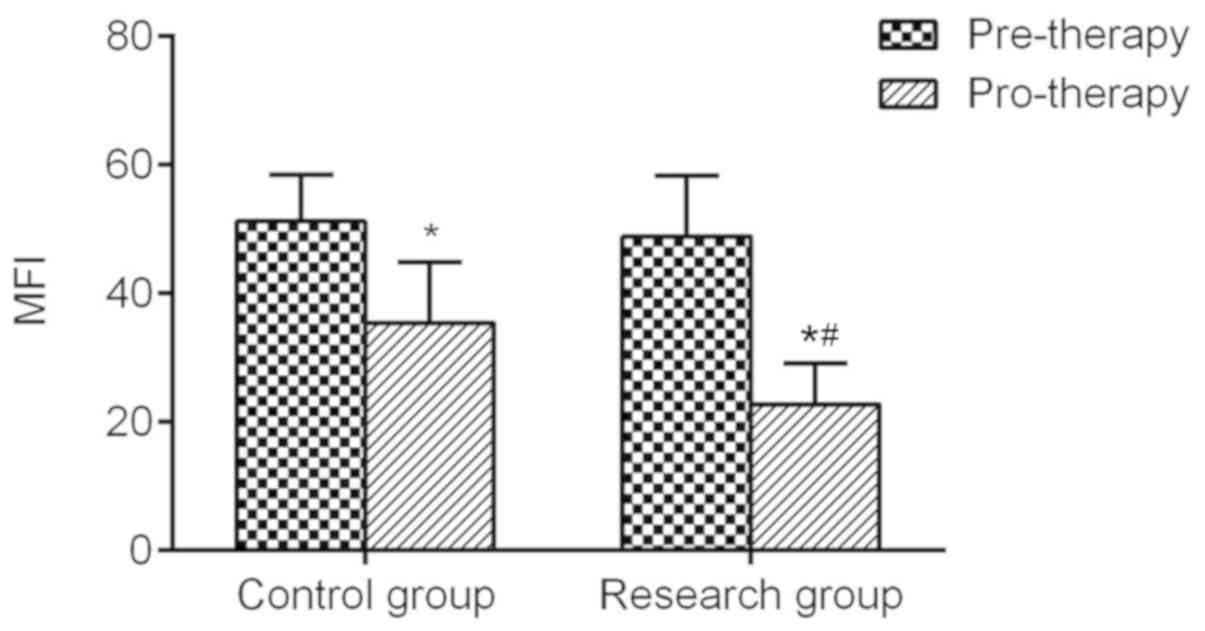
To detect the ROS levels, the DCFH-DA assay was
performed using the isolated PBMCs. Prior to treatment, the ROS
levels in PBMCs were similar between the two groups, and there was
no significant difference. After intensive treatment, the ROS
levels in the two groups were significantly decreased and the ROS
levels in the experimental group were significantly lower than
those in the control group (Fig. 4).
The expression level of MFI pro-therapy in experimental group was
significantly lower than that in control group
Short-term intensive insulin pump
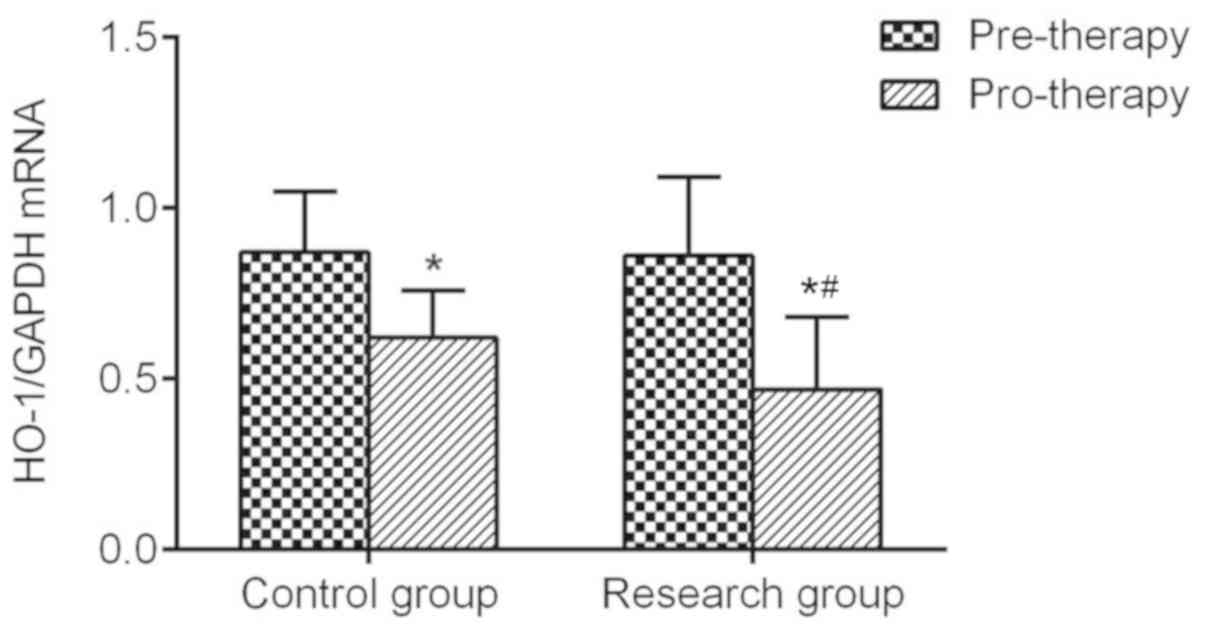
therapy decreases the mRNA levels of HO-1
There was no significant difference in the mRNA
levels of HO-1 between the two groups prior to treatment; however,
after intensive treatment, the transcription levels of HO-1 were
significantly decreased in each group, and HO-1 was significantly
decreased in the experimental group compared with that in the
control group (Fig. 5). Compared
with control group, the expression of HO-1 pro-therapy in
experiment group was significantly decreased.
Short-term intensive insulin pump
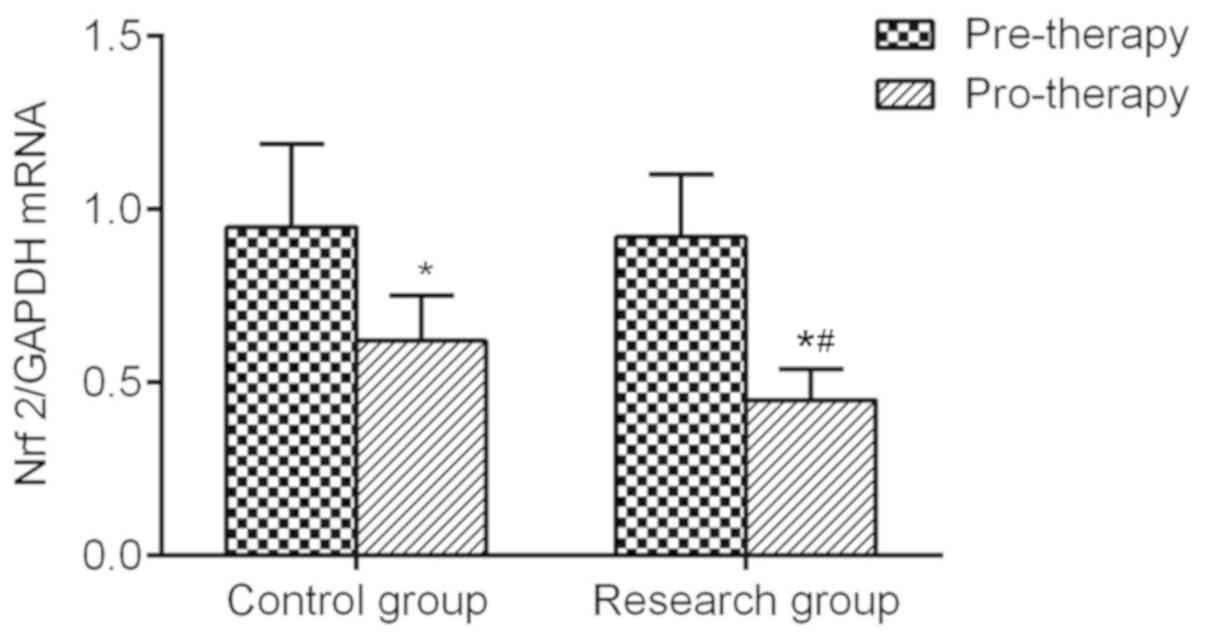
therapy decreases the mRNA levels of Nrf2
The expression of HO-1 is regulated by the upstream
transcription factor Nrf2, so the transcriptional level of Nrf2 was
also detected in the present study. Prior to treatment, the
transcription levels of PBMCs were similar between the two groups,
without exhibiting any statistically significant difference. After
intensive treatment, the mRNA levels of Nrf2 in the two groups were
significantly decreased. Furthermore, the mRNA levels of Nrf2 in
the experimental group were significantly reduced when compared
with those in the control group (Fig.
6). The expression of Nrf2 pro-therapy in experimental group
was significantly downregulated in comparison with control
group.
Discussion
Type 2 diabetes is a slowly progressing systemic
disease with an unclear cause. It is generally thought that defects
in insulin resistance and secretion have an important role in the
development of type 2 diabetes. Insulin secretion defects are
associated with islet β-cell dysfunction (9). Numerous factors may lead to a decrease
in islet β-cell function, of which hyperglycemia is the most
important and persistent cause (10). Elevated blood glucose may reduce the
expression of insulin-associated genes, promote apoptosis of islet
β-cells that may lead to a decrease in insulin secretion and
increase blood glucose, thereby forming a vicious circle and
ultimately leading to the loss of islet β-cell function (11). Studies have indicated that certain
toxic effects of hyperglycemia on islet β-cells are reversible
after maintaining short-term normal glucose levels (12–14).
Therefore, only strict control of blood glucose is able to prevent
the toxic effects of hyperglycemia on the function of islet
β-cells. Short-term intensive usage of insulin may rapidly control
blood glucose levels to reach the normal range. If insulin is
supplemented in an approximate physiological manner, the occurrence
of hyperinsulinemia may also be avoided, which may, in turn, alter
the clinical course of diabetes. At the same time, lipo-toxicity
caused by abnormal blood lipids also enhances the decline in the
function of islet β-cells and exacerbates deficiencies in insulin
action. TG may impair pro-proinsulin biosynthesis and damage the
oxidation process of glucose in the islets, as well as increasing
islet β-cell apoptosis (15).
Insulin sensitivity decreased with increasing amounts of TG in the
muscle.
The insulin pump mimics the pulsatile release of
insulin during sustained basal secretion and feeding, making
insulin release more physiologically compatible. The present study
indicated that the short-term intensive treatment of newly
diagnosed type 2 diabetes using insulin pump rapidly and
efficiently controlled the blood glucose levels, improved blood
lipids and increased insulin sensitivity and islet β-cell
function.
Oxidative stress is a persistent imbalance between
free radical production and oxidative defense, which results in
tissue damage when the body suffers from various harmful stimuli.
Numerous studies have indicated that the gradual decline of islet
β-cell function in type 2 diabetic patients is linked to oxidative
stress caused by various factors, including high glucose and high
fat (16,17). Among them, MDA is the final breakdown
product of lipid peroxidation, which reflects the lipid
peroxidation rate or intensity of tissue cells (18). The concentration of MDA reflects the
extent of lipid peroxidation and cell damage. In the present study,
the serum level of MDA was significantly decreased in patients with
newly diagnosed type 2 diabetes after short-term intensive
treatment, while the level of SOD was significantly increased. On
the other hand, increased ROS is also an important factor of
oxidative stress. In the present study, CSII significantly reduced
ROS levels in PBMCs from patients newly diagnosed with type 2
diabetes, which was consistent with the study by Zhao et al
(19). However, the underlying
molecular mechanisms remains to be fully elucidated.
The anti-oxidant system also has important roles in
the development of diabetes and its complications (20). HOs constitute one of the important
anti-oxidant systems in the body. HO-1 reduces the sensitivity of
tissue and oxidative stress to reduce cell damage and apoptosis.
Nrf2 is a leucine zipper protein of the ‘CNC’ family that binds to
the DNA sequence of the anti-oxidation response element in the
nucleus that regulates the expression of a series of downstream
phase II detoxification enzymes and anti-oxidant genes, including
HO-1. In the absence of any stimulation, Nrf2 binds to the
cytosolic chaperone protein Keap1 to depress its function, which is
resolved upon its release into the cytoplasm. Under oxidative
stress conditions, Nrf2 is separated from Keap1 and translocates
into the nucleus, where it binds to nuclear anti-oxidant response
elements and initiates the expression of a series of phase II
detoxification enzymes and anti-oxidant genes, including HO-1
(21). The present study indicated
that the transcriptional level of HO-1 and its transcriptional
regulatory protein Nrf2 decreased with the intensive treatment of
short-term insulin pumping in patients newly diagnosed with
diabetes, and their transcription level was positively paralleled
with direct or indirect indicators reflecting the body's oxidative
stress, which in turn maintained a balance of oxidation and
anti-oxidant levels in the body. In particular, after a short-term
insulin pump treatment, the redox index in diabetic patients was
significantly improved.
Of note, the present study had certain limitations.
Only the short-term benefits of intensive insulin treatment were
assessed without long-term observations. Furthermore, the study was
performed at one institution and was limited due to the small
sample.
In conclusion, short-term intensive treatment with
an insulin pump significantly improves blood lipids and glucose
levels in patients with newly diagnosed type 2 diabetes. The
function of islet β-cells was partially restored with involvement
of the oxidative stress mechanism. Compared with the traditional
intensive treatment with insulin injections, the effect of
short-term intensive therapy with insulin pump is more effective
and useful for clinical application.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HTL conceived and designed the study. YG was
responsible for the collection and analysis of the data. HTL and YG
interpreted the data and drafted the manuscript. All authors read
and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Daqing Oilfield General Hospital (Daqing, China).
Written informed consent was obtained from all patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Urakami T, Suzuki J, Mugishima H, Amemiya
S, Sugihara S, Kawamura T, Kikuchi T, Sasaki N, Matsuura N and
Kitagawa T: Screening and treatment of childhood type 1 and type 2
diabetes mellitus in Japan. Pediatr Endocrinol Rev. 10 (Suppl
1):S51–S61. 2012.
|
|
2
|
Halban PA, Polonsky KS, Bowden DW, Hawkins
MA, Ling C, Mather KJ, Powers AC, Rhodes CJ, Sussel L and Weir GC:
β-cell failure in type 2 diabetes: Postulated mechanisms and
prospects for prevention and treatment. J Clin Endocrinol Metab.
99:1983–1992. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mu J, Woods J, Zhou YP, Roy RS, Li Z,
Zycband E, Feng Y, Zhu L, Li C, Howard AD, et al: Chronic
inhibition of dipeptidyl peptidase-4 with a sitagliptin analog
preserves pancreatic beta-cell mass and function in a rodent model
of type 2 diabetes. Diabetes. 55:1695–1704. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Robertson RP, Harmon J, Tran PO, Tanaka Y
and Takahashi H: Glucose toxicity in beta-cells: Type 2 diabetes,
good radicals gone bad, and the glutathione connection. Diabetes.
52:581–587. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Retnakaran R: Novel strategies for
inducing glycemic remission during the honeymoon phase of type 2
diabetes. Can J Diabetes. 39:(Suppl). S142–S147. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Marchetti P, Lupi R, Del Guerra S,
Bugliani M, D'Aleo V, Occhipinti M, Boggi U, Marselli L and Masini
M: Goals of treatment for type 2 diabetes beta-cell preservation
for glycemic control. Diabetes Care. 32 (Suppl 2):S178–S183. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gabir MM, Hanson RL, Dabelea D, Imperatore
G, Roumain J, Bennett PH and Knowler WC: Plasma glucose and
prediction of microvascular disease and mortality: Evaluation of
1997 American Diabetes Association and 1999 World Health
Organization criteria for diagnosis of diabetes. Diabetes Care.
23:1113–1118. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chiu KC, Chu A, Go VL and Saad MF:
Hypovitaminosis D is associated with insulin resistance and beta
cell dysfunction. Am J Clin Nutr. 79:820–825. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Meier JJ and Bonadonna RC: Role of reduced
β-cell mass versus impaired β-cell function in the pathogenesis of
type 2 diabetes. Diabetes Care. 36 (Suppl 2):S113–S119. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rorsman P and Ashcroft FM: Pancreatic
β-cell electrical activity and insulin secretion: Of mice and men.
Physiol Rev. 98:117–214. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Robertson RP: Beta-Cell deterioration
during diabetes: What's in the gun? Trends Endocrinol Metab.
20:388–393. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
White MG, Shaw JA and Taylor R: Type 2
diabetes: The pathologic basis of reversible β-Cell dysfunction.
Diabetes Care. 39:2080–2088. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Steven S and Taylor R: Restoring
normoglycaemia by use of a very low calorie diet in long- and
short-duration type 2 diabetes. Diabet Med. 32:1149–1155. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Robertson RP, Harmon J, Tran PO and
Poitout V: Beta-cell glucose toxicity, lipotoxicity, and chronic
oxidative stress in type 2 diabetes. Diabetes. 53 (Suppl
1):S119–S124. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jung KY, Kim KM and Lim S: Therapeutic
approaches for preserving or restoring pancreatic β-Cell function
and mass. Diabetes Metab J. 38:426–436. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mizukami H, Takahashi K, Inaba W, Osonoi
S, Kamata K, Tsuboi K and Yagihashi S: Age-associated changes of
islet endocrine cells and the effects of body mass index in
Japanese. J Diabetes Investig. 5:38–47. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dragun Z, Filipović Marijić V, Krasnići N,
Ramani S, Valić D, Rebok K, Kostov V, Jordanova M and Erk M:
Malondialdehyde concentrations in the intestine and gills of Vardar
chub (Squalius vardarensis Karaman) as indicator of lipid
peroxidation. Environ Sci Pollut Res Int. 24:16917–16926. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhao QB, Wang HF, Sun CF, Ma AQ and Cui
CC: Effect of short-term intensive treatment with insulin pump on
beta cell function and the mechanism of oxidative stress in newly
diagnosed type 2 diabetic patients. Nan Fang Yi Ke Da Xue Xue Bao.
27:1878–1879. 2007.(In Chinese). PubMed/NCBI
|
|
20
|
Bao W, Song F, Li X, Rong S, Yang W, Zhang
M, Yao P, Hao L, Yang N, Hu FB and Liu L: Plasma Heme Oxygenase-1
concentration is elevated in individuals with type 2 diabetes
mellitus. PLoS One. 5:e123712010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Fukushima-Uesaka H, Saito Y, Maekawa K,
Kamatani N, Kajio H, Kuzuya N, Noda M, Yasuda K and Sawada J:
Genetic variations and haplotype structures of transcriptional
factor Nrf2 and its cytosolic reservoir protein Keap1 in Japanese.
Drug Metab Pharmacokinet. 22:212–219. 2007. View Article : Google Scholar : PubMed/NCBI
|