Introduction
Cushing's disease is the most common etiology of
endogenous hypercortisolism, accounting for ~70% of cases (1,2).
Patients with this disease suffer from excess adrenocorticotropic
hormone (ACTH) secretion from the pituitary adenoma cells.
Cardiovascular diseases, refractory diabetes mellitus, as well as
infections are the major causes of mortality. During
endocrinological remission, the risk of death from cardiovascular
events may still be higher than that in the general population
(1,2).
In 1969, Hardy developed the endoscopic
transsphenoidal surgery (ETS) for pituitary ACTH adenoma with the
assistance of micro-neurosurgical techniques. Since then, the
transsphenoidal approach has been regarded as the optimal method
for the treatment of pituitary ACTH adenoma (3–5). The
ever-growing clinical implementation of ETS resulted in encouraging
outcomes (remission rates of ~80%) (6–9).
However, >80% of ACTH pituitary adenomas are microadenomas, and
for those, magnetic resonance imaging (MRI) may only provide normal
scans in 50% of the cases examined (10). Recently, the application of ETS in
Qilu Hospital of Shandong University (Jinan, China) for the
treatment of functional pituitary adenomas has been used to
routinely inactivate peritumoral tissue by cauterization if a clear
border of lesion cannot be identified. To the best of our
knowledge, the current study is the first to demonstrate the
aforementioned method being used to inactivate peritumoral tissue.
The present study aimed to review and compare endocrinological
remission, recurrence and post-operative complications in patients
who underwent inactivation and those who did not.
Material and methods
General information
The medical records of 79 consecutive patients with
Cushing's disease who were treated at Qilu Hospital (Jinan, China)
between January 2010 and June 2016 were reviewed. The cohort
comprised 58 women and 21 men, and the age of the subjects ranged
between 33 and 61 years, with a median age of 44 years. A total of
67 cases exhibited truncal obesity, 35 cases had purple striae, 9
cases had visual field defects, 61 cases presented with
hypertension and 47 cases with diabetes. A total of 18 cases had
transient ischemic attacks (defined as at least one attack within 5
years), 7 female patients had irregular menstruation or amenorrhea
and two male patients exhibited defective secondary sexual
characteristics. None of the patients included received any medical
therapy, including ketoconazole, prior to surgery.
Pre-operative endocrine
evaluation
All of the 79 patients exhibited elevated serum
cortisol, ranging from 22.1 to 43.5 µg/dl with an average of 27.9
µg/dl. The serum ACTH level ranged between 26.2 and 59.1 pg/ml,
with an average level was 37.2 pg/ml. A total of 16 patients were
not responsive in high-dose dexamethasone suppression testing, and
13 patients without visible tumors on routine enhanced MRI
underwent bilateral inferior petrosal sinus sampling (BIPSS). BIPSS
confirmed the diagnosis of Cushing's syndrome with IPS based on a
ratio of ACTH (IPS/peripheral) of >2.0. These cases underwent
surgery. The erratic availability of corticotropin-releasing
hormone (CRH) in China prevented the application of CRH stimulation
tests to the patients. With the exception of four cases with
slightly increased prolactin (PRL<100 ng/ml), other
pre-operative pituitary hormones were in the normal range among the
remaining patients of the cohort.
Neuroimaging
All patients underwent sphenoid sinus computed
tomography three-dimensional reconstruction prior to surgery. The
results revealed 66 cases of the sellar type and 13 cases of the
presellar type. With the exception of two patients who had metal
implants due to a previous surgery, enhanced MRI examination was
performed in all patients pre-operatively. If the information
provided by the routine enhanced MRI was unsatisfactory, pituitary
dynamic MRI scanning was performed. Neuroimaging revealed 11 cases
of pituitary hyperplasia, 51 cases of microadenomas (2 cases of
microadenoma were confirmed by operation without MRI examination)
and 14 cases of macroadenoma.
Surgical treatment
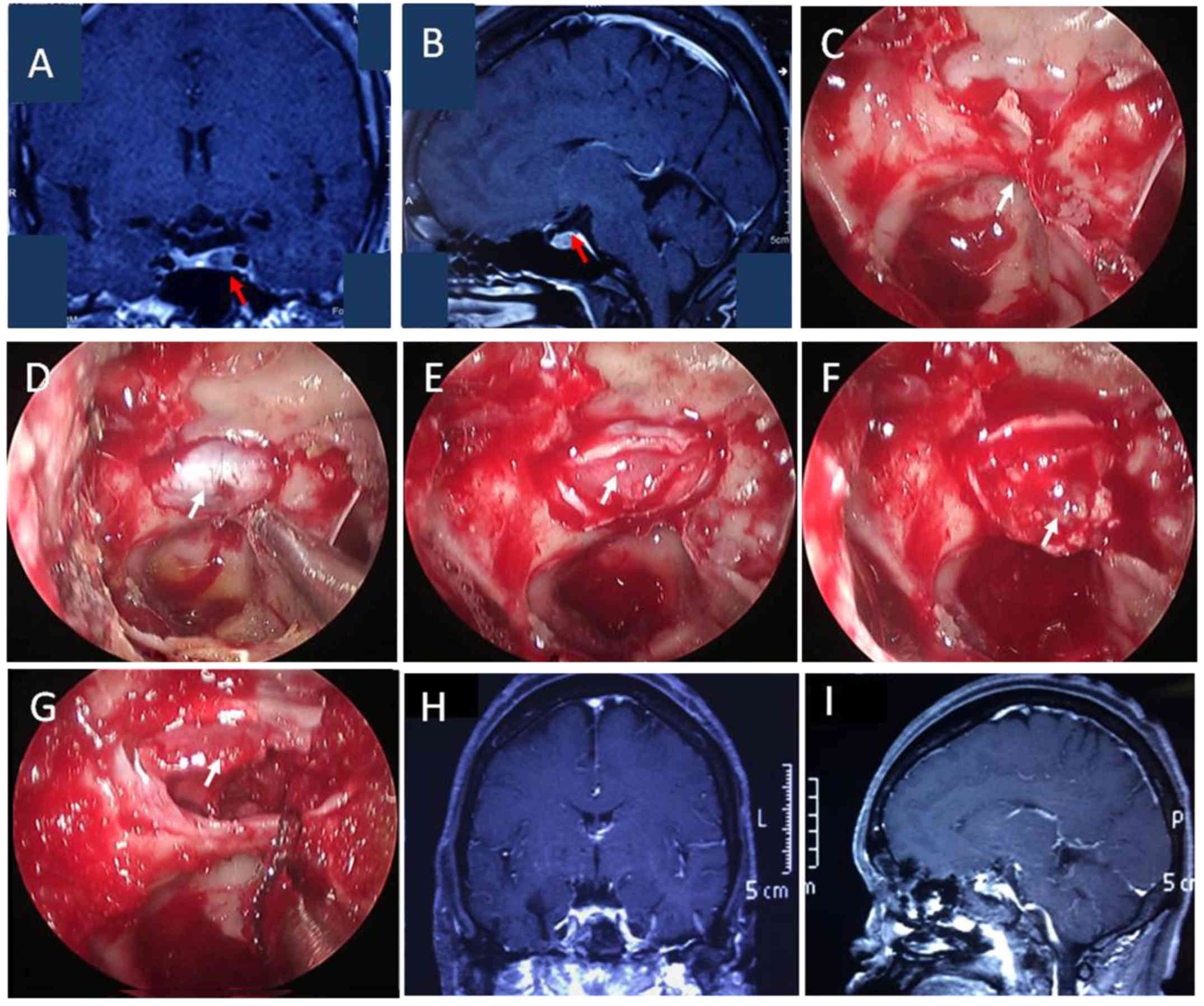
All of the included patients underwent ETS, as
exemplified in the illustrative case presented in Fig. 1. Rigid 0° and 30° endoscopes with an
external diameter of 4.5 mm (Aesculap; B. Braun, Melsungen,
Germany) were used. Once the pseudocapsule was identified in part
of the tumors, complete tumor resection was achieved along that
plane, as exemplified in the illustrative case presented in
Fig. 2. However, larger tumors
without an identifiable pseudocapsule were resected in a piecemeal
fashion with the assistance of tumor forceps, angled curette and
suction. If the remaining pituitary gland had a diameter of ≥8 mm,
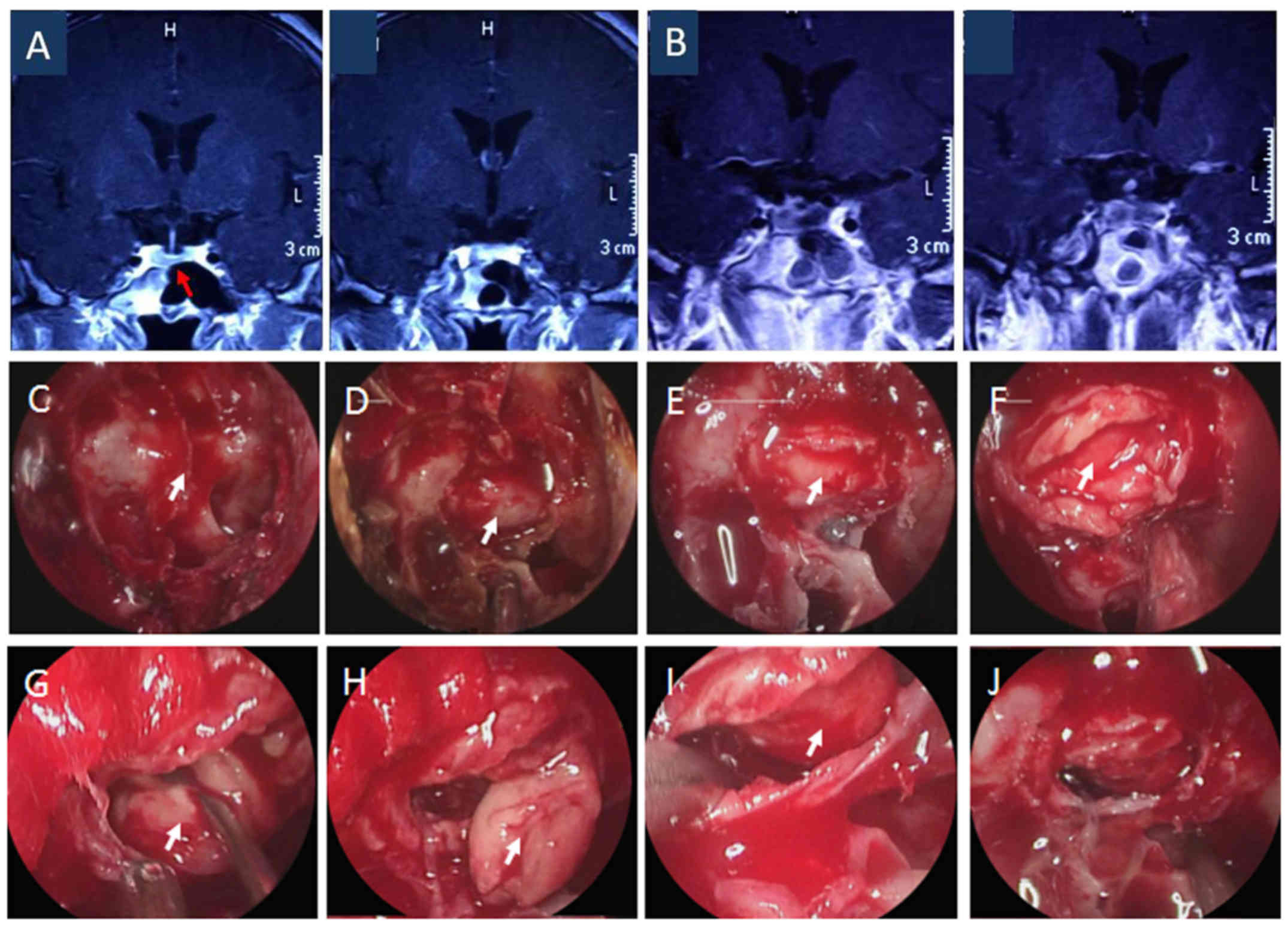
the peritumoral tissue was inactivated (11,12).
Careful inactivation of the tissue was applied through extensive
cauterization following the detection of color changes (from
off-white to orange), which indicated the presence of peritumoral
tissue. With regard to pituitary microadenomas or lesions that were
not identifiable by pre-operative imaging, selective exploration of
the pituitary was performed according to the pre-operative imaging
results and IPSS. During this process, off-white tumor tissues were
always identified and removed from the sella. A total of 24
patients in the study group experienced different degrees of
rupture of the arachnoid membrane around the diaphragm sellae.
Intra-operatively, autologous fat was harvested in order to fill
the intrasellar region and reconstruct the sellar floor.
Hematoxylin and eosin stains, and comprehensive immunohistochemical
analysis (13) on several
parameters, such as ACTH, were performed for all specimens.
Post-operative endocrine
evaluation
Normal fasting plasma cortisol and ACTH levels
within the first week following surgery were considered to indicate
early endocrinal remission. If signs and symptoms of
hypopituitarism were present, replacement therapy was provided. In
contrast to hypopituitarism, recurrence was defined as an early
remission followed by recurrent hypercortisolism. In addition to
these conditions, post-operative complications, including diabetes
insipidus, brain infarction and cerebrospinal fluid rhinorrhea were
recorded. Persistent rhinorrhea was defined as a long-lasting
rhinorrhea for >7 days that was not improved following
conservative treatment. Persistent hypopituitarism and/or diabetes
insipidus were defined based on the requirement for replacement
therapy in the first month of the follow-up period. All of the
patients were evaluated at 1, 3, 9 and 12 months post-operatively,
and every 12 months thereafter. Pituitary function and enhanced MRI
were also evaluated in the follow-up period.
Statistical analysis
The incidence of endocrinological remission,
recurrence and post-operative complications among the patients who
underwent inactivation and those without inactivation was compared
by Fisher's exact test. All statistical analyses were conducted
using GraphPad (version 6.0; GraphPad Software, Inc., La Jolla, CA,
USA). P<0.05 indicated that the difference between groups was
statistically significant.
Results
Surgical outcomes
Early endocrinological remission was achieved in 71
cases, while 8 cases exhibited no apparent improvement (Table I). In patients with pre-operative
visual impairment, various degrees of visual improvement were
noted. In addition, serum PRL returned to normal levels. At 3
months after the operation, the enhanced pituitary MRI scans
indicated the following: Gross total resection was achieved in 73
cases and partial resection in 6 cases. Pathological examination
indicated pituitary adenoma in 75 cases and normal pituitary tissue
in 4 cases, while immunohistochemical analysis revealed positive
ACTH staining.
 | Table I.Clinicopathological characteristics
and examination results of the patients (n=79). |
Table I.
Clinicopathological characteristics
and examination results of the patients (n=79).
| Parameter | Value |
|---|
| Age (years) | 44 (33–61) |
| Sex |
|
| Male | 21 (26.6%) |
|
Female | 58 (73.4%) |
| Pre-op cortisol
levels (µg/dl) | 33
(22.1–43.5)a |
| Pre-op ACTH levels
(pg/ml) | 41
(26.2–59.1)b |
| High-dose
dexamethasone test |
|
| Suppressed | 48 (60.8%) |
|
Unsuppressed | 16 (20.2%) |
| Bilateral inferior
petrosal sinus sampling | 31 (39.2%) |
| No obvious lesion on
MRI | 13 (16.4%) |
| Complications with
MRI scan | 2 (2.5%) |
| (Metallic
implants) |
|
| Pre-op
comorbidities |
|
|
Hypertension | 61 (77.2%) |
| Diabetes
mellitus | 47 (59.4%) |
| Transient ischemic
attackc | 18 (22.8%) |
| Pituitary
hyperplasia | 11 (13.9%) |
| Tumor size (cm) |
|
|
<1 | 52 (65.8%) |
| ≥1 | 14 (17,7%) |
| Intra-op rupture of
diaphragma sellae and arachnoid membrane | 24 (30.4%) |
| Extent of tumor
resection |
|
|
Total | 73 (92.4%) |
|
Partial | 6 (7.6%) |
|
Inactivation of peritumoral
tissue by cauterization | 35 (44.3%) |
| Pathological
examination |
|
| Pituitary
adenoma | 75 (94.9%) |
| Normal
pituitary tissue | 4 (5.1%) |
| Early endocrinal
remissiond |
|
|
Achieved | 71 (89.9%) |
|
Stable | 8 (10.1%) |
| Stable
disease after partial resection | 6 (7.6%) |
| Recurrence at
follow-upe | 5 (6.3%) |
| Post-op
complications | 18 (24.1%) |
|
Hypopituitarism | 7 (8.9%) |
|
Rhinorrhea | 4 (5.1%) |
| Brainstem
infarction | 1 (1.3%) |
| Diabetes
insipidus | 9 (11.4%) |
Post-operative complications
Post-operative complications occurred in 18
patients, of whom 7 patients developed persistent symptoms of
hypopituitarism, 4 presented with cerebrospinal rhinorrhea, 1 with
brain stem infarction and 9 with transient diabetes insipidus
(Table II). Three patients
simultaneously developed persistent hypopituitarism and transient
diabetes insipidus. Regarding complications, no significant
differences were identified between patients who underwent
inactivation and those who did not. A total of 2 out of the 4
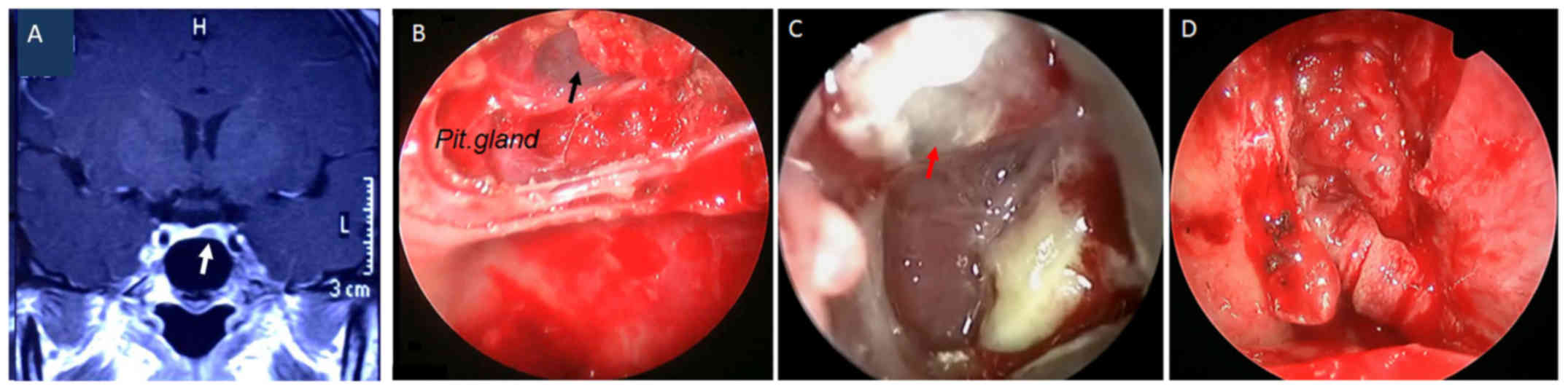
patients with post-operative cerebrospinal fluid rhinorrhea were
cured by re-operation, as exemplified in the illustrative case
presented in Fig. 3, and the other
two were successfully managed by conservative therapy. No cases of
confirmed intracranial infection or death associated with the
surgical procedures were encountered.
 | Table II.Association between the cauterization
of peritumoral tissue and outcomes/complications. |
Table II.
Association between the cauterization
of peritumoral tissue and outcomes/complications.
| Item | Inactivation (n=35)
(%) | No inactivation
(n=44) (%) | P-value |
|---|
| Outcome |
|
|
|
| Early
remissiona | 30 (38.0) | 41 (51.9) | 0.2744 |
|
Recurrenceb | 2 (2.5) | 3 (3.8) | 0.9157 |
| Complications |
|
|
|
| Diabetes
insipidus | 6 (7.6) | 3 (3.8) | 0.1743 |
|
Persistent
hypopituitarism | 5 (6.3) | 2 (2.5) | 0.2314 |
|
Concomitant diabetes insipidus
and persistent hypopituitarism | 2 (2.5) | 1 (1.3) | 0.4266 |
|
Intraoperative rupture of
diaphragma sellae and arachnoid membrane | 13 (16.5) | 11 (13.9) | 0.3255 |
|
Rhinorrhea | 2 (2.5) | 2 (2.5) | 1.0000 |
Follow-up
The mean interval of the follow-up was 26 months
with a range of 11–62 months. At the time that the follow-up was
terminated, all of the 19 patients with post-operative
complications were in remission. A total of 4 out of the 7 patients
with hypopituitarism who had undergone surgery withdrew from
replacement therapy. All of the patients without early remission
(n=8) and all the cases of recurrence (n=5) achieved
endocrinological remission following radiotherapy. The enhanced
pituitary MRI that was performed during the follow-up period in the
6 patients with partial resection indicated no enlargement of the
residual tumor.
Discussion
Despite the presence of classical clinical symptoms
and signs of Cushing's disease, the high-dose dexamethasone
suppression test may be negative in certain cases, and the enhanced
pituitary MRI scan may fail to indicate the presence of
ACTH-secreting adenomas (4,5). Under such circumstances, the BIPSS
should be considered (3,5). The pathological results of the present
study confirmed the efficiency of this diagnostic strategy.
Pituitary surgery with the assistance of endoscopic technology is
the preferred option among the treatments for Cushing's disease, as
it allows for more accurate visualization of the operative field
and reduces blind spots for the surgeon, so as to avoid injury to
normal structures (8,14). Hence, the endoscope is able to lay a
technical foundation for minimally invasive microneurosurgery
within the concept of ‘minimal trauma, minimal risk and the
greatest degree of lesion resection’ (15–17).
With the improvement of the neuroimaging techniques and endocrine
examinations, the pre-operative diagnostic rate of Cushing's
disease has significantly improved, and pre-operative pituitary MRI
scans (including plain, enhanced and dynamic enhanced scan) are
able to visualize ~80% of micro-adenomas (18). Despite the possibility of omission,
the tumor tissue was successfully localized during exploration in
94.9% of the patients (75/79), even in the 9 of the 13 cases with a
negative dynamic MRI scan. The method introduced by Cebula et
al (11) was used to explore and
localize the adenoma when lesions were not identified on the MRI
scans. According to the experience of the authors of the current
study, after resecting a macroadenoma that did not have a clear
border, patients with a remaining pituitary gland of <8 mm had a
tendency to go into remission. Hence, it was concluded that those
patients require inactivation of peritumoral tissue in order to
achieve the highest possible post-operative endocrinal remission
rate. Since the location and/or an apparent border of a tumor
cannot always be detected, this method was developed to maximize
the probability of a curative operation. In our opinion, ETS for
Cushing's disease should be performed in high-volume neurosurgical
centers with experienced and trained surgeons to reconstruct the
sellar floor. By using this surgical method, the tumors may be
accessed and endoscopy may be used in order to facilitate sellar
floor reconstruction. When using ETS, the authors of the current
study worried that cauterization may cause a higher rate of
cerebrospinal fluid rhinorrhea. In the present study, a total of 24
patients experienced various degrees of rupture of the diaphragm
sellae and arachnoid membrane, while the reconstruction of the
sellar floor with autologous fat and hemostatic materials was
largely satisfactory. The results suggested that if surgeons
operate meticulously, these ruptures would not lead to major
problems, such as cerebrospinal fluid rhinorrhea.
In the early stage of the application of peritumoral
activation, two cases of persistent post-operative cerebrospinal
fluid rhinorrhea were encountered that were successfully treated by
re-operation. To reduce the possibility of post-operative
cerebrospinal fluid rhinorrhea, the technical protocol that was
followed during the resection of the pituitary ACTH adenoma
included the following points: i) Maintenance of a clean surgical
field to avoid blind curettage of the tumors. The risk of bleeding
during the operation, the major sources of which are intercavernous
sinuses, was managed by elevating the upper body to a 20–30° angle
in order to ensure that the head was in a higher position than the
heart, thereby reducing venous bleeding. ii) Extrocapsular tumor
resections were applied in order to remove pituitary ACTH tumors
with pseudocapsule and reduce damage to the normal pituitary
tissues, diaphragma sellae and arachnoid membranes. During
inactivation of the peritumoral tissue, care was taken to
distinguish the pituitary tissue that closely adheres to the
diaphragma sellae. Post-operative cerebrospinal fluid rhinorrhea
was thereby reduced. iii) Once intra-operative cerebrospinal fluid
rhinorrhea occurred, the reconstruction of the diaphragma sellae
with fat and/or other autologous tissues was immediately performed
at the end of surgery. Sterilization and draping of an alternative
abdominal incision prior to ETS is our common practice and may
reduce the duration of the surgery. iv) Following surgery, the
patient was maintained in an elevated head position, and any
postoperative discomforts, including constipation and prostatic
hyperplasia, were treated. Furthermore, patients were instructed to
avoid sneezing and coughing by force. Post-operative lumbar
drainage and antibiotic treatment were applied in cases with
high-volume intra-operative cerebrospinal fluid rhinorrhea. In
cases of unsuccessful conservative therapy and persistence of
rhinorrhea for >7 days, re-operation should be considered.
The characteristic manifestation of pituitary ACTH
adenoma is Cushing's syndrome. Hypercortisolemia may hinder the
body's ability to fight off infection (18). Infections of the central nervous and
respiratory systems are particularly prone to occur in these
patients (19). Hypertension may
lead to instability of vital signs during the surgery, which may
cause additional intra-operative bleeding and intracranial hematoma
following the operation. High levels of blood glucose prior to and
following the surgery may also increase the risk of infection and
impair wound healing. In the present study, one case presented with
post-operative brain stem infarction, possibly due to a poor
condition of the vasculature and surgical stress. Although the
patient gradually recovered and returned to work, this case
exemplifies the importance of peri-operative management, including
fluid balance (as patients can be affected by post-operative
diabetes insipidus), blood pressure and glucose control. The
multidisciplinary team involved in treating the patients of in the
current study was dedicated to the management of the surgery of the
functional pituitary adenoma. At the beginning, the majority of
patients enrolled in the present study were admitted to the
Department of Endocrinology of Qilu Hospital of Shandong University
(Jinan, China). All of the patients were subjected to endocrinal
examinations and comprehensive assessments were performed by
endocrinologists. Subsequently, the expert team, consisting of
experienced neurosurgeons, endocrinologists, anesthesiologists,
radiologists and pathologists, reviewed the medical records of each
of the patients and derived an individualized protocol. In terms of
remission, it should be kept in mind that the outcomes across
various studies are different depending on the criteria used to
define remission and the timing of evaluation. A recent surgical
series revealed remission rates of 65–85% and recurrence rates of
10–35% (1). With regard to the
patients with recurrent post-operative Cushing's syndrome,
radiotherapy was able to attain endocrinological remission.
In conclusion, although limited by its retrospective
nature, the present study included a relatively large population of
79 patients with Cushing's disease. The overall post-operative
remission rate was 83.5% (66/79), which is comparable to that of
other studies (1,2). Overall, compared with conventional ETS
procedures, the data suggest that inactivation of peritumoral
tissue is a comparable method in the treatment of intricate
Cushing's disease in terms of efficacy and safety. However, the
present results require further verification by large prospective
studies.
Acknowledgements
The authors would like to thank Dr Hongwei Qin
(Department of Cell, Development, and Integrative Biology,
University of Alabama at Birmingham, Birmingham, AL, USA) for
revising this manuscript.
Funding
This work was supported by the National Natural
Science Foundation of China (grant no. 81201986) and the foundation
for outstanding young and middle-aged scientists of Shandong
province (grant no. BS2013YY018).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
SC and FLin were major contributors in revising the
manuscript as well as analyzing and interpreting the data. SX was
the major surgeon performing ETS. XZ made substantial contributions
to acquisition of the data and the preparation of the manuscript.
MD and FLiu performed the pre-operative endocrinal examinations and
assessment. XL and XM made substantial contributions to the
conception and design and supervised the study. All authors read
and approved the final manuscript.
Ethics approval and consent to
participate
This study was approved by the Ethics Committee of
Qilu Hospital (Jinan, China) and written informed consent was
obtained from all patients.
Patient consent for publication
Written informed consent and/or oral consent was
obtained from all included patients and/or their relatives for
publication of associated images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Lonser RR, Nieman L and Oldfield EH:
Cushing's disease: Pathobiology, diagnosis, and management. J
Neurosurg. 126:404–417. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Molitch ME: Diagnosis and treatment of
pituitary adenomas: A review. JAMA. 317:516–524. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Esposito V, Santoro A, Minniti G, Salvati
M, Innocenzi G, Lanzetta G and Cantore G: Transsphenoidal
adenomectomy for GH-, PRL- and ACTH-secreting pituitary tumours:
Outcome analysis in a series of 125 patients. Neurol Sci.
25:251–256. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Hardy J: Transphenoidal microsurgery of
the normal and pathological pituitary. Clin Neurosurg. 16:185–217.
1969. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mehta GU, Lonser RR and Oldfield EH: The
history of pituitary surgery for Cushing disease. J Neurosurg.
116:261–268. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dehdashti AR, Ganna A, Karabatsou K and
Gentili F: Pure endoscopic endonasal approach for pituitary
adenomas: Early surgical results in 200 patients and comparison
with previous microsurgical series. Neurosurgery. 62:1006–1015.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Frank G, Pasquini E, Farneti G, Mazzatenta
D, Sciarretta V, Grasso V and Faustini Fustini M: The endoscopic
versus the traditional approach in pituitary surgery.
Neuroendocrinology. 83:240–248. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gondim JA, Schops M, de Almeida JP, de
Albuquerque LA, Gomes E, Ferraz T and Barroso FA: Endoscopic
endonasal transsphenoidal surgery: Surgical results of 228
pituitary adenomas treated in a pituitary center. Pituitary.
13:68–77. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kabil MS, Eby JB and Shahinian HK: Fully
endoscopic endonasal vs. transseptal transsphenoidal pituitary
surgery. Minim Invasive Neurosurg. 48:348–354. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Pivonello R, Isidori AM, De Martino MC,
Newell-Price J, Biller BM and Colao A: Complications of Cushing's
syndrome: State of the art. Lancet Diabetes Endocrinol. 4:611–629.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cebula H, Baussart B, Villa C, Assié G,
Boulin A, Foubert L, Aldea S, Bennis S, Bernier M, Proust F and
Gaillard S: Efficacy of endoscopic endonasal transsphenoidal
surgery for Cushing's disease in 230 patients with positive and
negative MRI. Acta Neurochir (Wien). 159:1227–1236. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Starke RM, Reames DL, Chen CJ, Laws ER and
Jane JA Jr: Endoscopic transsphenoidal surgery for cushing disease:
Techniques, outcomes, and predictors of remission. Neurosurgery.
72:240–247. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lopes MBS: The 2017 World Health
Organization classification of tumors of the pituitary gland: A
summary. Acta Neuropathol. 134:521–535. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Goudakos JK, Markou KD and Georgalas C:
Endoscopic versus microscopic trans-sphenoidal pituitary surgery: A
systematic review and meta-analysis. Clin Otolaryngol. 36:212–220.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ammirati M, Wei L and Ciric I: Short-term
outcome of endoscopic versus microscopic pituitary adenoma surgery:
A systematic review and meta-analysis. J Neurol Neurosurg
Psychiatry. 84:843–849. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tabaee A, Anand VK, Barrón Y, Hiltzik DH,
Brown SM, Kacker A, Mazumdar M and Schwartz TH: Endoscopic
pituitary surgery: A systematic review and meta-analysis. J
Neurosurg. 111:545–554. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sarkar S, Rajaratnam S, Chacko G, Mani S,
Hesargatta AS and Chacko AG: Pure endoscopic transsphenoidal
surgery for functional pituitary adenomas: Outcomes with Cushing's
disease. Acta Neurochir (Wien). 158:77–86. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hofstetter CP, Shin BJ, Mubita L, Huang C,
Anand VK, Boockvar JA and Schwartz TH: Endoscopic endonasal
transsphenoidal surgery for functional pituitary adenomas.
Neurosurg Focus. 30:E102011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Choe JH, Lee KS, Jeun SS, Cho JH and Hong
YK: Endocrine outcome of endoscopic endonasal transsphenoidal
surgery in functioning pituitary adenomas. J Korean Neurosurg Soc.
44:151–155. 2008. View Article : Google Scholar : PubMed/NCBI
|