Introduction
Recurrent miscarriage (RM) is a common gestational
complication that affects 2–5% of couples trying to conceive
(1,2), and previous successful live birth does
not preclude its development (3). RM
is considered to be a multifactorial disorder that may result from
either acquired or inherited factors (4). Several studies have been conducted to
examine and identify the molecular mechanism underlying the
pathophysiology of RM and yet 50–60% of the cases are of unknown
cause (4,5).
Although oxygen is essential for normal cell
function, it poses a risk by producing some toxic derivatives
through extensive metabolism (5).
Several studies have suggested that the oxidative stress (OS)
generated during angiogenesis and placental development might be
the cause of idiopathic RM (2,5–10). OS is known to result when there is an
increased production of reactive oxygen species (ROS) that cannot
be balanced by the produced antioxidants. However, ROS are
naturally produced during oxygen reduction and within limited
levels can act as essential regulators for several cell functions,
in addition to playing an important role in several reproductive
processes (11). Increased ROS
levels are common during placental development and are normally
balanced by higher levels of antioxidants (9).
Superoxide anions (SOA), hydrogen peroxide
(H2O2), and hydroxyl radicals (•OH) are the
most commonly generated ROS in an electron-rich environment, which
mainly occurs within the mitochondrial inner membrane and the
respiratory chain (6,12). The conversion of oxygen to SOA mainly
results after the leakage of 1–3% of electrons through respiratory
chain complex I and III (5,12,13).
Aerobic cells balance SOA by its dismutation into
H2O2 through antioxidant superoxide dismutase
(SOD) (6). Other antioxidants that
can convert SOA into H2O2 include cytosolic
copper (Cu) and zinc (Zn) dismutase and mitochondrial manganese
(Mn) SOD (7). Glutathione peroxidase
(GPx) and catalase (CAT) antioxidant enzymes can then transform
H2O2 into water. In addition, GPx can also
reduce lipid hydroperoxides into water by transferring the energy
of those reactive peroxides to reduced glutathione (GSH), and thus,
resulting in oxidised glutathione (GSSG). This can then be reduced
back to GSH by the enzyme glutathione reductase (GSR) via donated
electron by NADPH (7,14). However, excessive unbalanced ROS can
induce cytotoxicity by alteration in gene expression including
genes related to apoptosis (7).
Although, several studies have suggested that OS may
be a strong factor causing RM, there is a lack of consensus in the
literature. Therefore, the present study was conducted to gain
further knowledge regarding the impact of OS and the efficiency of
antioxidants in RM cases by examining free radicals and OS markers
in addition to measurements of antioxidant levels in placental
tissue and plasma from RM patients with reference to healthy
pregnant (HP) women. Moreover, these assays are paralleled by
profiling the differential expression level of selected
antioxidant, pro-inflammatory and apoptosis-related genes in the
placental tissues of RM cases in relation to HP women.
Patients and methods
All materials and solutions were purchased from
Sigma-Aldrich (Merck KGaA), unless otherwise specified.
Samples, collection and
preparation
The study included consenting patients who attended
the Department of Obstetrics and Gynaecology at the King Khalid
University Hospital (Riyadh, Kingdom of Saudi Arabia) from
September 2014 to January 2015, who signed the consent forms
provided. Ethics approval was obtained from the Ethics Review
Committee of the College of Applied Medical Sciences of King Saud
University (CAMS 27–34/35) and the study was conducted according to
the Declaration of Helsinki.
Placental tissue and blood plasma of idiopathic RM
women of at least 3 consecutive miscarriages (n=28), 35.8±5.3 years
of age, and of HP women (n=28), with no miscarriage history,
29.3±2.7 years of age, were examined. Blood plasma samples were
also collected from non-pregnant women (NP) (n=28), 28.6±4.91 years
of age. Patients with RM were excluded from the study if there were
risk factors, such as abnormal menstrual cycle (<21 and >35
days), genital infections, antiphospholipid syndrome (positive
anticardiolipin and/or b2 glycoprotein 1 antibodies), and uterus
anomalies (observed by ultrasound examination or
hysterosonography). No anticoagulant or immunological treatment was
applied during the pregnancy. Gestational age of RM patients at the
time of miscarriage was 12.6±2.8 weeks, with an average of 3.5±0.8
miscarriages.
Maternal blood and placental tissue samples were
collected at the time of delivery from HP women or at the time of
miscarriage from RM patients. Full depth placental tissue was
collected, in which the Decidua basalis and the chorionic
plate was trimmed off, leaving trophoblastic tissue. Tissue was
then washed in 0.1 M phosphate-buffered saline (PBS) and dissected
into 1.5 g pieces and placed into two Corning® cryogenic
vials (Corning, Inc.). One contained RNAlater® (Thermo
Fisher Scientific, Inc.) (4°C) for immediate RNA stabilization and
protection, and thus, reliable gene expression profiling, while the
other contained PBS for biochemical assays. All collected tubes
were kept at 4°C for 24 h. Subsequently, cryovials were immediately
snap-frozen in liquid nitrogen prior to storage at −80°C until
further use. Venous blood samples (6 ml) were collected from all
participants into cold BD Vacutainer™ Plastic Blood Collection
Tubes (BD Biosciences) with K2EDTA for the measurement
of biochemical OS markers (SOA, H2O2 and
lipid peroxides), activities of antioxidant enzymes (SOD, GPx, GSR
and CAT), in addition to the non-enzymatic antioxidants GSH, Zn,
selenium (Se), and Cu. All centrifugation steps were conducted at
room temperature. Plasma was obtained by centrifugation at 3,000 ×
g for 20 min and then transferred into the Eppendorf tubes within 1
h and stored at −80°C. For GSH and GSSG analysis, whole blood
aliquot samples (30 µl) were centrifuged and 33.3 µl of
5-sulphosalicylic acid (1 g/ml) were added for protein
precipitation and cellular disruption to release GSH. Samples were
then diluted with 936.7 µl sodium phosphate buffer (pH 7.5) and
then centrifuged for 5 min at 12,000 × g, and the supernatant was
kept at −80°C until the time of analysis.
Measurement of OS markers
H2O2
Levels of H2O2 were measured
as described previously (7).
Briefly, reaction mix (horseradish peroxidase dissolved in Kreb's
Ringer buffer 10 µg/ml, 100 µl; sodium phosphate reaction buffer 50
mM; pH 7.4) was added to 50 µl diluted samples and standards and
incubation followed for 30 min at room temperature. A total of 50
µl of 10 mM Amplex Red Reagent (ARR;
10-acetyl-3,7-dihydrophenoxazine; Thermo Fisher Scientific, Inc.)
was added to commence reaction, and fluorescence was measured at
590 nm. ARR reacts with H2O2 in the presence
of peroxide resulting in red fluorescent oxidation resorufin
products.
SOA
Samples (0.1 ml) were incubated for 5 min at 37°C
with 1 ml of PBS (2 g glucose, 2 g of fatty acid-free bovine serum
albumin/l) with and without 30 µg SOD following previous
publication (6,15), and were mixed with 0.1 ml reaction
solution of ferricytochrome-c (1.2 mM). Tube containing only buffer
and ferricytochrome-c was used as blank control. A
spectrophotometer, equipped with a thermostated cuvet, was used for
measuring absorbance at 550 nm. Results were converted to nM of
reduced ferricytochrome-c by using an absorptivity value of
1.96×104 l•mol−1. SOA levels were determined
by calculating the difference between the samples without SOD and
the samples with added SOD.
Lipid peroxidation (LPO)
The level of the end product of LPO was determined,
malondialdehyde (MDA), using thiobarbituric acid (TBA) which reacts
with MDA producing a fluorescence product that can be measured by
spectrophotometry (7,16,17).
Briefly, plasma (150 µl) or placental tissue supernatant (1 ml) was
mixed with 1 ml trichloroacetic acid (17.5%) and 1 ml TBA (0.6%),
followed by incubation in hot water bath (100°C) for 15 min, and
then left to cool. After that, 1 ml trichloroacetic acid (70%) was
added to the mixture, incubated for 20 min at room temperature, and
then centrifuged at 2,000 × g for 15 min. The supernatant was
decanted and the absorbance was measured at 535 nm. Applying
1.56×105 M−1•cm−1 as an extinction
coefficient, MDA levels were calculated.
Enzymatic assays for enzymatic
antioxidant measurements
Frozen tissue was thawed in ice, homogenized in 0.1
M potassium chloride buffer, and centrifuged at 10,000 × g, 4°C for
10 min. Supernatants were used for different assays.
Measurement of SOD activity
Serum samples (250 µl) were mixed with xanthine (25
µl, 1.142 mg•ml−¹), hydroxyl ammonium chloride (25 µl),
water (125 µl), and xanthine oxidase (75 µl, 0.1
U•ml−¹). The mixture was incubated at 25°C for 20 min,
and sulphonilic acid (0.5 ml, 3.3 mg•ml−¹) and
α-naphthylamine (0.5 ml, 1 ng•ml−¹) were added, and
further incubated at room temperature for 20 min. Absorbance was
measured at 530 nm.
Measurement of CAT activity
CAT activity was measured as previously described
(18), using
H2O2 as the substrate. The decomposition of
H2O2 was observed at 240 nm by measuring the
decrease in absorbance. The activity was expressed as µM/min/mg
protein using a coefficient of 0.0436
mM−1•mg−1.
Measurement of GPx activity
GPx activity was measured as described previously
(19). Briefly, assay solution (800
µl) of 50 mM Tris-HCl buffer (pH 7.6), 1 mM EDTA, 1 IU GSR, 0.25 mM
GSH and 0.2 mM NADPH were added to diluted plasma (1:10; 50 µl) or
placental tissue supernatant (50 µl). After 5 min, 100 µl of
H2O2 (15 mM) were added to the sample mix and
the absorbance change was examined at 340 nm for 30 min. GPx
activity was determined by calculating the enzyme catalysing the
oxidation of 1 µM or 1 nM GSH per minute.
Measurement of GSR activity
GSR activity was determined following a previously
described protocol (7). Briefly, 100
µl serum samples were added to 2 ml reaction mix (100 mM potassium
phosphate buffer pH 7.4, 50 µl of 80 mM EDTA, 100 µl 2 mM NADPH,
and 100 µl of 0.3 mM flavine adenine dinucleotide) and incubated
for 2 min followed by addition of 100 µl GSSG (7.5 mM). Using a
spectrophotometer, reaction was examined at 340 nm for 2 min. One
unit of GSR is equal to the amount of enzyme to reduce 1 µM of
NADPH/min at 25°C.
Non-enzymatic assays
Measurement of GSH and GSSG
Following a previous described protocol (20,21),
glutathione reductase-DTNB (5,5-dithiobis-2-nitrobenzoic acid) was
used to determine the GSH levels. Twenty-five microliters of
samples and standards (ranging from 20 to 80 µM) were incubated
separately for 3 min at 37°C with the reaction mix (150 µl of 100
mM sodium phosphate buffer pH 7.4, 50 µl of 8 mM EDTA, 50 µl DNTB
solution, and 100 µl of 2 mM NADPH). Then, 25 µl glutathione
reductase were added and absorbance was measured at 410 nm. GSSG
was measured similarly, but the reaction mix also contained
triethanolamine, to prevent pH increase and oxidation, and 2-vinyl
pyridine for GSH derivatization. GSSG standards ranged from 0 to 10
µM and were examined simultaneously.
Measurement of Se, Zn, Cu and Mn
antioxidants
Both plasma and placental tissue were freeze dried
at −45°C and ground into powder. Ground plasma (100 mg) and 20 mg
of ground placental tissue were used, in which samples were
digested with ultra-pure nitric acid (0.5 ml, 68%) and
H2O2 (0.2 ml, 35%). The product was diluted
using ultra-pure water and then measured for Se, Cu, Zn and Mn
using inductively coupled plasma mass spectrometry (ICP-MS) (HP
4500; Yokokawa Electric Co.), as described previously (22).
Gene expression profiling
Placental tissue samples previously stored in
RNAlater® at −80°C were allowed to thaw on ice, and were
subsequently used for total RNA extraction and relative gene
expression quantification using reverse transcription-quantitative
polymerase chain reaction (RT-qPCR). Approximately 100 mg of
placental tissues were homogenized in 1 ml TRIzol®
reagent (Invitrogen; Thermo Fisher Scientific, Inc.) by the use of
TissueLyser LT (Qiagen, Inc.), and total RNA was subsequently
extracted by standard procedure. Genomic DNA was then eliminated
and cDNA was synthesized from total RNA (1 µg) in a final reaction
volume of 20 µl using the QuantiTect Reverse Transcription kit
(QuantiTect®; Qiagen, Inc.), according to the
manufacturer's instructions. The RT reaction was carried out on
Veriti® 96-Well Thermal Cycler (Applied Biosystems) at
42°C for 45 min and then inactivated at 95°C for 15 min. The
resultant diluted cDNA (1:10, 5 µl) was then used to perform
RT-qPCR using the QuantiTect SYBR-Green PCR kit (Qiagen, Inc.). A
total of 100 nM of the following gene primers were used: CAT
(Hs_CAT_1_SG QuantiTect Primer Assay, QT00079674); GPx (Hs_GPx_1_SG
QuantiTect Primer Assay, QT00203392); GSR (Hs-GSR_1_SG QuantiTect
Primer Assay, QT00038325); SOD (Hs_SOS_1_SG QuantiTect Primer
Assay, QT01664327); tumor necrosis factor-α (TNF-α)
(Hs_TNF_3_SG QuantiTect Primer Assay, QT01079561); interleukin
(IL)-6 (Hs_IL6_1_SG QuantiTect Primer Assay, QT00083720); IL-8
(Hs_IL8_1_SG QuantiTect Primer Assay, QT00000322); and 18S
(Hs_RRN18S_1_SG QuantiTect Primer Assay, QT00199367), in a final
reaction volume of 25 µl, containing the cDNA sample (5 µl),
SYBR-Green PCR Master mix (12.5 µl), QuantiTect® Primer
Assay (10X, 2.5 µl), and RNase-free water (2.5 µl). A two-step
cycling reaction was conducted; following an initial polymerase
activation at 95°C for 10 min, samples were subjected to 40 cycles
of i) denaturation at 95°C for 15 sec, followed by ⅱ) annealing and
elongation at 60°C for 1 min. TaqMan® gene expression
assays (Applied Biosystems) were performed with the following gene
primers: tumor necrosis factor-related apoptosis-inducing ligand
(TRAIL) (Hs00921974_ml TaqMan® Gene Expression);
calcium binding protein A8 (S100A8) (Ha00374263_ml
TaqMan® Gene Expression); and TaqMan endogenous
housekeeping gene, hypoxanthine-guanine phosphoribosyltransferase
(HPRT1). The amplification program and PCR amplicon
specificity were performed and assessed as documented by us
previously (6). A three-step cycling
reaction was carried out; following an initial polymerase
activation at 95°C for 10 min, samples were subjected to 40 cycles
of ⅰ) denaturation at 95°C for 15 sec, followed by ⅱ) annealing at
55°C for 30 sec, and ⅲ) elongation at 72°C for 30 sec. Each tissue
sample was represented by two biological replicas and three
technical replicas, with the inclusion of a no-template control.
Raw data were analyzed using the Rotor-Gene Q software version 2.3
(Qiagen, Inc.) to calculate the threshold cycle (Cq) using the
second derivative maximum method. The relative gene expression
level (fold-change) was determined after normalization to the
expression levels of 18S and HPRT1 as a housekeeping
gene for SYBR-Green and TaqMan® assays, respectively,
with the 2−ΔΔCq method as previously described (6). The relative gene expression data were
subjected to Student's t-test in order to identify significant
differences between RM samples compared with uncomplicated HP
controls. Expression levels in terms of fold change were considered
statistically significant at P<0.05.
Statistical analysis
Statistical analysis was performed using the
computer-based package of Minitab software (v.13.1, 2001; Minitab
Ltd.). Sample analysis was run in duplicate for all investigated
parameters and results are presented as means ± SD. Values of the
activities and concentrations of individual parameters were
compared between different groups of the study subjects using
one-way analysis of variance (ANOVA) followed by the post-hoc
Tukey-HSD test for multiple comparisons. P<0.05 was considered
to indicate a statistically significant difference.
Results
Levels of OS markers in plasma and
placenta
Plasma H2O2 levels were
significantly higher (10.2±1.47 nmol/ml) in RM patients in relation
to both HP (8.11±1.14 nmol/ml; P<0.001) and NP (7.04±1.12
nmol/ml; P<0.0001) subjects. HP subjects, however, showed
moderately increased (P<0.05) plasma H2O2
levels compared with NP samples (Table
I). Placental tissue H2O2 generation
rates showed highly significantly elevated levels in RM patients
compared with HP women (3.38±0.46 vs. 2.41±0.35 nmol/min/mg
protein, respectively, P<0.0001; Table I). Similar increases of SOA (Table I) and MDA (Table I) were observed in both plasma and
placental tissue of RM patients compared with both HP (P<0.001
and P<0.0001, respectively) and NP (P<0.0001) samples. An
increase (P<0.05) of plasma SOA and MDA levels was observed in
HP in relation to NP subjects, as well.
 | Table I.Levels of H2O2,
SOA, and MDA, which is used as an index of LPO, in plasma and
placental tissue of NP, HP and RM women. |
Table I.
Levels of H2O2,
SOA, and MDA, which is used as an index of LPO, in plasma and
placental tissue of NP, HP and RM women.
|
| Subjects |
|---|
|
|
|
|---|
| Assayed
parameters | NP women
(n=28) | HP women
(n=28) | RM women
(n=28) |
|---|
| Plasma
H2O2 (nmol/ml) | 7.04±1.12 |
8.11±1.14a |
10.2±1.47b,c |
| Placental tissue
H2O2 (nmol/min/mg) | – | 2.41±0.35 |
3.38±0.46d |
| Plasma SOA
(nmol/ml) | 30.1±4.50 |
35.3±5.45a |
45.2±6.10b,c |
| Placental tissue
SOA (µmol/min/mg) | – | 4.67±0.62 |
5.93±0.78d |
| Plasma MDA
(nmol/ml) | 5.03±0.70 |
5.73±0.76a |
7.87±0.96b,c |
| Placental tissue
MDA (nmol/g wet weight) | – | 258±35.7 |
334±45.8d |
Levels of GPx, GSR, CAT and SOD
enzymatic antioxidants
All examined plasma enzymatic antioxidants showed
similar patterns of highly significant decreases in RM patients
compared with both HP (P<0.001) and NP (P<0.0001) samples
(Table II). However, the
antioxidant plasma levels of HP participants were slightly, however
significantly, decreased compared with those of NP samples
(P<0.05). Examination of GPx, GSR, CAT and SOD in placental
tissue of RM patients also revealed highly significant decreases
(P<0.0001) compared with the levels in HP placental tissues
(Table II).
 | Table II.GPx, GSR, CAT and SOD enzymatic
activities in plasma and placental tissue of NP, HP and RM
women. |
Table II.
GPx, GSR, CAT and SOD enzymatic
activities in plasma and placental tissue of NP, HP and RM
women.
|
| Subjects |
|---|
|
|
|
|---|
| Assayed
parameters | NP women
(n=28) | HP women
(n=28) | RM women
(n=28) |
|---|
| Plasma GPx
(nmol/min/ml) | 1.19±0.17 |
1.06±0.13a |
0.86±0.10b,c |
| Placental tissue
GPx (µmol/min/mg protein) | – | 0.36±0.04 |
0.28±0.04d |
| Plasma GSR
(nmol/min/ml) | 1.86±0.26 |
1.60±0.23a |
1.29±0.17b,c |
| Placental tissue
GSR (µmol/min/mg protein) | – | 0.61±0.08 |
0.48±0.06d |
| Plasma CAT
(nmol/min/ml) | 3.10±0.36 |
2.78±0.38a |
2.25±0.30b,c |
| Placental tissue
CAT (µmol/min/mg protein) | – | 0.91±0.12 |
0.70±0.10d |
| Plasma SOD
(nmol/min/ml) | 2.08±0.27 |
1.83±0.23a |
1.48±0.20b,c |
| Placental tissue
SOD (µmol/min/mg protein) | – | 1.88±0.20 |
1.52±0.19d |
Non-enzymatic antioxidants
Measurement of GSH and GSSG
GSH showed highly significant decreases in RM plasma
in relation to HP (P<0.001) and NP (P<0.001) subjects
(Table III). It was also observed
that HP subjects had moderately lower GSH levels (P<0.05)
compared with those in NP subjects. Similar findings were observed
in placental tissue samples of RM patients which had very
significantly decreased GSH levels compared with those of HP women.
However, GSSG plasma levels were very significantly higher in RM
patients compared with both HP (P<0.001) and NP (P<0.0001)
subjects, and also in HP compared with NP subjects (P<0.05;
Table III).
 | Table III.GSH, GSSG and GSH/GSSG ratio in
plasma and placental tissue of NP, HP and RM women. |
Table III.
GSH, GSSG and GSH/GSSG ratio in
plasma and placental tissue of NP, HP and RM women.
|
| Subjects |
|---|
|
|
|
|---|
| Assayed
parameters | NP women
(n=28) | HP women
(n=28) | RM women
(n=28) |
|---|
| Plasma GSH
(µmol/l) | 2.86±0.30 |
2.51±0.36a |
1.97±0.29b,c |
| Placental tissue
GSH (nmol/mg tissue) | – | 6.86±0.95 |
5.40±0.77d |
| Plasma GSSG
(µmol/l) | 0.049±0.007 |
0.056±0.008a |
0.071±0.010b,c |
| Placental tissue
GSSG (nmol/mg tissue) | – | 0.14±0.018 |
0.18±0.025d |
| Plasma
GSH/GSSG | 55.6±6.88 |
48.5±6.61a |
28.8±3.88b,c |
| Placental tissue
GSH/GSSG | – | 49.6±6.75 |
31.0±4.19d |
GSH/GSSG ratio
As GSH is considered an important scavenger for ROS,
its ratio with GSSG is used as an OS marker (23). The results (Table III) revealed that RM plasma samples
had very significantly lower GSH/GSSG ratios compared with those in
plasma of both HP (P<0.001) and NP (P<0.0001) subjects, and
HP plasma samples had significantly lower ratio than NP (P<0.05)
plasma. Similarly, GSH/GSSG ratios in placental tissue of RM
patients were highly significantly decreased when compared with
those found in HP women (P<0.0001).
Se, Cu, Zn and Mn
The examined non-enzymatic micronutrient
antioxidants (Table IV) showed very
similar patterns of highly significant decreases in plasma of RM
patients in relation to both HP (P<0.001) and NP (P<0.0001)
subjects. Data also indicated that plasma micronutrient levels were
moderately, however significantly, lower (P<0.05) in HP subjects
compared with the NP ones. Furthermore, the levels of Se, Cu, Zn
and Mn were very significantly decreased in placental samples of RM
patients compared with those of HP subjects (P<0.0001).
 | Table IV.Se, Cu, Zn and Mn levels in plasma
and placental tissue of NP, HP and RM women. |
Table IV.
Se, Cu, Zn and Mn levels in plasma
and placental tissue of NP, HP and RM women.
|
| Subjects |
|---|
|
|
|
|---|
| Assayed
parameters | NP women
(n=28) | HP women
(n=28) | RM women
(n=28) |
|---|
| Plasma Se
(µmol/l) | 1.09±0.14 |
0.95±0.13a |
0.78±0.09b,c |
| Placental tissue Se
(nmol/g) | – | 16.8±2.26 |
12.0±1.73d |
| Plasma Cu
(µmol/l) | 28.3±3.81 |
24.5±3.41a |
19.6±2.75b,c |
| Placental tissue Cu
(nmol/g) | – | 98.1±14.7 |
75.1±11.1d |
| Plasma Zn
(µmol/l) | 4.03±0.58 |
3.55±0.49a |
2.84±0.36b,c |
| Placental tissue Zn
(nmol/g) | – | 621±87.8 |
490±67.1d |
| Plasma Mn
(nmol/l) | 48.6±7.30 |
42.2±7.12a |
32.2±4.73b,c |
| Placental tissue Mn
(nmol/g) | – | 6.75±0.81 |
5.45±0.75d |
Gene expression profiling
Proinflammatory TNF-α and cytokines as
markers of OS and LPO
All pro-inflammatory cytokines and apoptosis-related
genes that were selected for the expression level examination
exhibited highly significant increases (P<0.001) in RM placenta
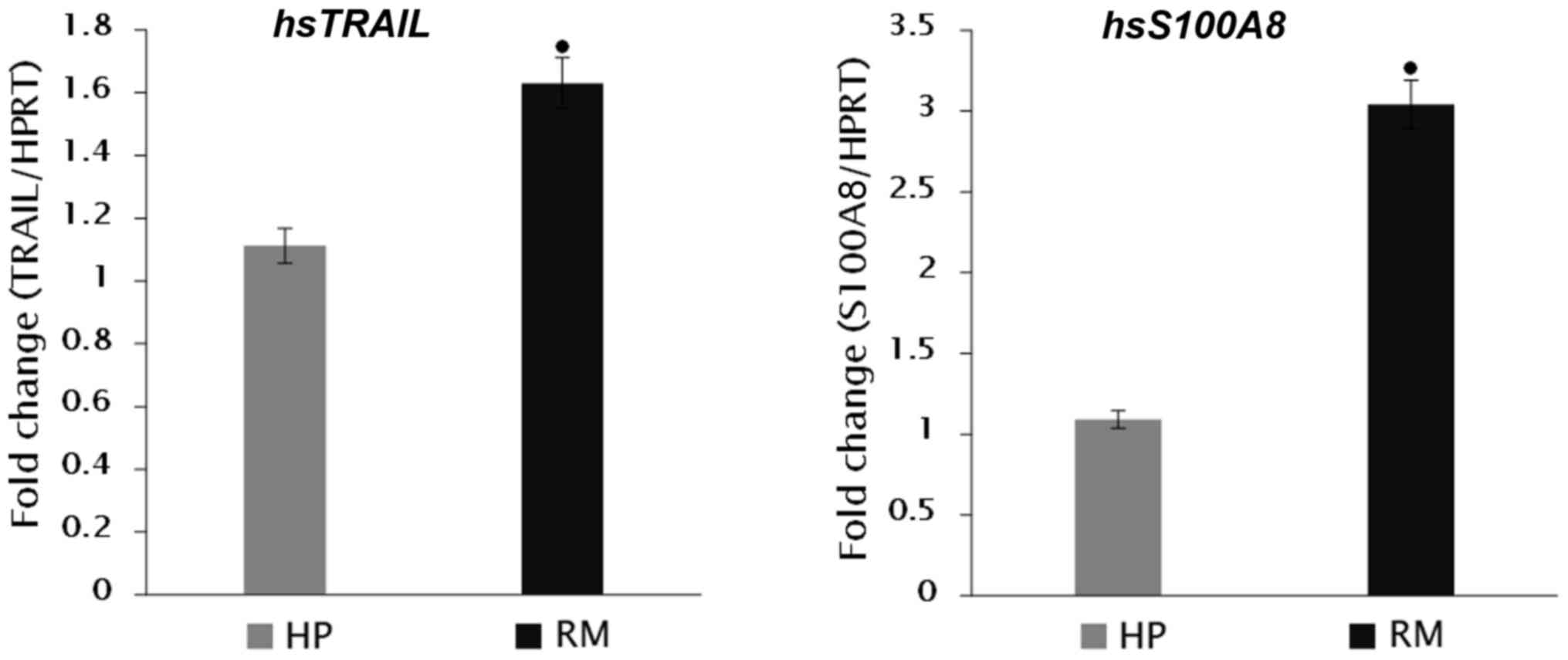
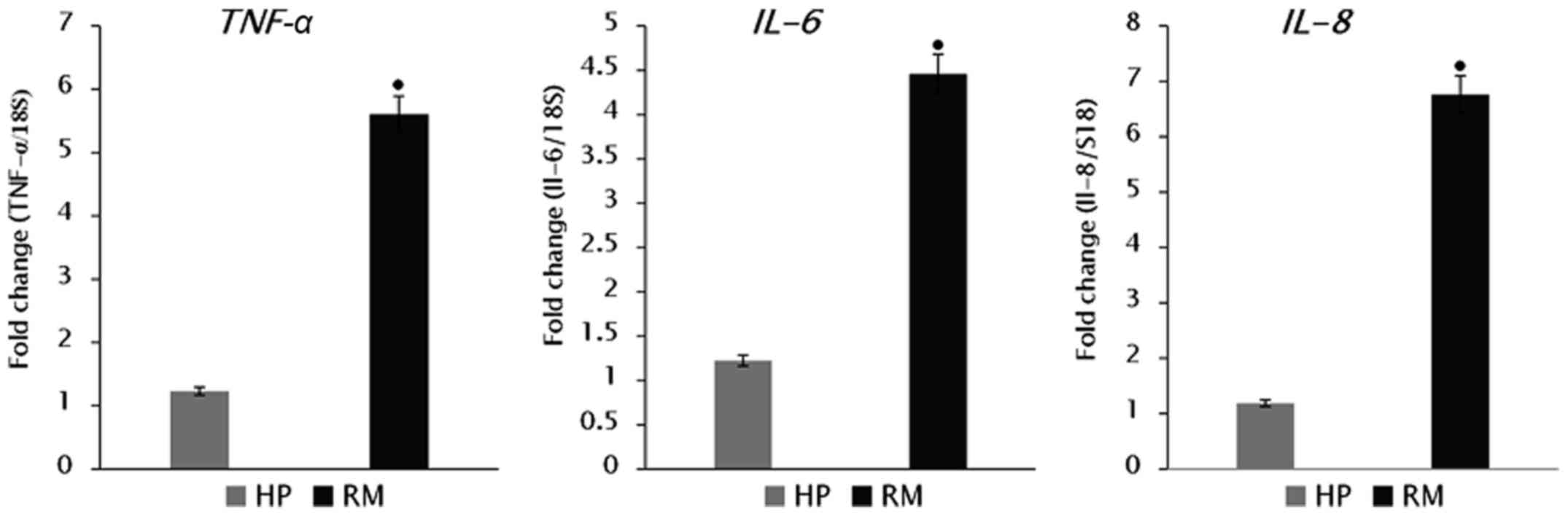
samples; TRAIL (1.63±0.27 fold-change), S100A8
(3.04±0.25 fold-change), TNF-α (5.61±0.26 fold-change),
IL-6 (4.46±0.348 fold-change), and IL-8 (6.76±0.428
fold-change), compared with HP women (Figs. 1 and 2).
Expression of antioxidant genes
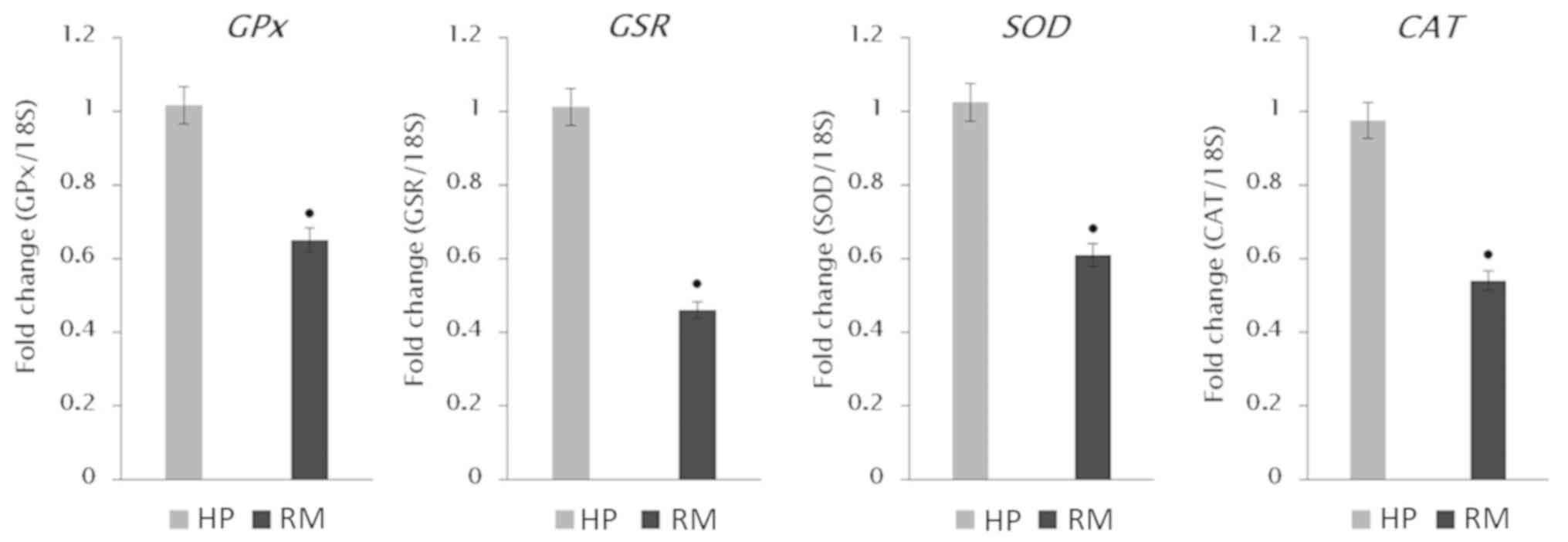
The mRNA expression levels (Fig. 3) showed highly significant decreases
(P<0.001) of all examined antioxidant genes; GPx
(fold-change 0.65±0.17), GSR (fold-change 0.46±0.09),
SOD (fold-change 0.61±0.15), and CAT (fold-change
0.55±0.087), compared with the expression levels observed in the HP
placental samples.
Discussion
The underlying molecular and subcellular mechanism
of RM pathogenesis is not fully understood, and thus, its
management is difficult and challenging (3). It was previously suggested that RM may
be a result of the action of ROS generated during the first
trimester of pregnancy. ROS are reactive chemical molecules
involved in cell signalling and homeostasis and can be increasingly
released in cell processes that involve high demand of oxygen
production or consumption (24),
such as placenta development. With all the evidence in the
literature suggesting the association of OS in RM patients
(2,5,10), there
is no consensus regarding its negative impact and the underlying
mechanism. Therefore, in addition to measuring levels of some OS
markers, the current study examined the levels of a comprehensive
list of key antioxidants in plasma and placental tissue of RM
patients in relation to those of HP and NP participants in order to
test the effect of OS. Moreover, the molecular basis of RM remains
unclear, thus the present study also attempted to quantify the gene
expression of some antioxidant enzymes and selected
pro-inflammatory and apoptosis-related genes in placental tissue of
RM patients compared with those of HP subjects.
LPO, resulting from ROS, can cause disruption in the
membrane lipid bilayer that may inactivate membrane-bound receptors
and enzymes, increasing the tissue permeability (25). Current results showed highly
significantly increased LPO levels in plasma and placental tissues
of RM patients compared with those of HP and NP women. High LPO, as
gestation progresses, has been previously reported in placenta and
umbilical cord (17). Similar
results have also been obtained by others reporting elevated MDA in
the serum of RM women and increase has also been observed in the
villous decidual tissues of women with early pregnancy loss
(26). This has also been supported
by other studies that showed increase of MDA levels in serum of
failed pregnancy (27) and in RM
women (28). The consensus between
current findings and previous studies supports the suggestion that
increased plasma and placental LPO is a high risk factor causing
oxidative cell damage that might result in miscarriage. SOA are the
most common oxygen-free radicals created from molecular oxygen by
an electron addition. SOA cannot penetrate the lipid membrane and
is mostly present within its production compartment (29). The present study demonstrated that
the levels of SOA and H2O2 generation rates
in plasma and placental tissue of RM patients are highly
significantly increased compared with those of HP and NP women, and
are moderately increased in plasma of HP women compared with those
of NP women. SOA generation within and out of the cell is an
initial step in ROS formation. Therefore, increased levels in
plasma or placental tissue will result in OS, and may have a direct
effect on causing RM. Dismutation of SOA and production of
H2O2 are catalysed by SOD, which is regarded
as the first line of defence. H2O2 is not a
free radical, however due to its high ability to penetrate
biological membranes is considered important by being an
intermediate molecule in forming more ROS radicals, such as
hydroxyl, which can cause increased damage to biological systems,
more than any other radical (30).
During normal pregnancy, the placental OS is
balanced by the quenching of ROS through the action of antioxidant
enzymes, including SOD, CAT, GPx and GSR, that act together with
non-enzymatic antioxidants, such as vitamin C and E, Se, and GSH.
This antioxidant control system of OS in normal pregnancy is
suggested to be a causative factor of RM when insufficiently
functioning. Thus, relating RM to OS may not only be due to
increased ROS generation in early gestation, but can also result
from insufficient levels of antioxidants required to combat the
excessive ROS, both of which have been demonstrated in RM subjects
of this study. It is likely that the significantly low GPx activity
and downregulation of its gene transcripts that was documented in
the plasma and placental tissue of RM patients in current study,
was due to the decreased levels of glutathione required for the
elimination of excessive ROS in RM women. Similar results have been
previously obtained showing that serum GPx activities are
significantly decreased in RM patients compared with NP women
(26,28), and in HP women compared with NP women
(30). Similarly, SOD activity
levels in the current study showed significant decreases in both
plasma and placental tissue of RM patients compared with those of
HP women. Moreover, the mRNA expression levels of SOD were found to
be significantly downregulated in trophoblast cells isolated from
RM placentas. This conforms with previous studies that have
reported similar significantly low levels of SOD in RM patients
compared with HP women (28,31,32).
This suggests that decreased levels of SOD enzyme in placental
tissue might be associated with increased generation of SOA, and
thus of OS, that may be involved in the pathogenesis of RM.
The control of H2O2 production
is the second enzymatic step that plays a vital role against ROS
propagation, and CAT and GPx catalyse the conversion of
H2O2 to H2O. Indeed, GPx is the
only enzyme able to quench H2O2 in
mitochondria, since CAT is not expressed there, and requires GSH to
convert H2O2 into H2O. In the
present study, CAT activity and its gene transcripts underwent
highly significant decreases and downregulation in plasma and
placental tissue of RM patients compared with those of HP and NP
women. Other studies have also reported significant decreases in
serum CAT activity in RM patients compared with HP women (28,32).
Thus, it is hypothesized that the decreases in CAT activity were
due to its consumption in protecting cells from the harmful effect
of H2O2. The present study also profiled
differential mRNA expression levels of GPx, GSR, SOD and
CAT genes, in placental tissue of RM patients compared with
those of HP women. The observed downregulation in the expression of
all investigated antioxidant enzyme levels might be linked to the
detrimental impact of OS on trophoblast cells, whereby leading to
ROS generation that may inactivate both de novo protein
synthesis, as well as gene transcription processes.
GSR and NADPH are important to maintain glutathione
in its active reduced state via the redox cycle activity. Under the
high OS conditions observed in the present study, GSH levels were
lowered, and subsequently the GSSG content was significantly higher
leading to a very significant drop in the GSH/GSSG ratio in RM
patients compared with HP and NP women. The GSH/GSSG ratio
represents the major cellular redox buffer, and therefore is a
representative indicator of the redox environment of the cell
(33). This observed reduction in
GSH/GSSG ratio should have been met with a proportional increase in
GSR activity in order to reduce the GSSG back to GSH, thereby
maintaining the natural balance of those two parameters. However,
the present results indicated significantly low GSR activity in
plasma and placental tissue of RM patients compared with those of
HP women, which is in agreement with previous studies (28,32,34).
This decrease in the enzyme activity might be a direct consequence
of OS damage caused by increased GSSG levels, leading to
downregulation of GSR gene expression levels presently
noted.
Pregnancy comes with physiological changes that can
cause reduced bioavailability of some dietary components in
addition to the increased demand for various nutrients and
micronutrients for developing the foetus (35). Se is known to be significantly low
during pregnancy and decreased levels in the blood of HP women have
been reported during delivery compared with those of NP women
(33,36), pre-eclamptic patients (37), as well as recurrent loss and pre-term
delivery patients (38) relative to
HP women. Results of the current study indicated a very significant
reduction in the plasma and placental tissue of RM patients
compared with HP subjects. Changes in Se homeostasis during
pregnancy are probably caused by increased oxygen demand in the
mother's body and developing foetus. A potential reason behind the
increased demand for Se during pregnancy may be the increased mass
of erythrocytes in the foetus (38).
It has been suggested that the loss of antioxidant capacity might
be attributed to low Se, leading to damage to biological membranes
and DNA (39), which may cause RM.
Cu, Zn and Mn are important cofactors for a number of antioxidant
enzymes, including Cu/Zn-SOD and Mn-SOD that may protect the
placenta from generating SOA and initiation of OS. In the present
study, the levels of Cu, Zn and Mn in plasma and placental tissues
of RM patients were significantly lower compared with those of HP
women. This is in agreement with previous studies that have
reported significant decreases in Cu and Zn levels in serum of
women with unexplained RM compared with normal HP women (26). Results of the present study also
indicated significantly decreased plasma Cu, Zn and Mn in HP
subjects compared with NP women. To this end, several studies have
reported that, whereas plasma Zn concentrations decline as the
pregnancy progresses (16,40,41), Cu
plasma levels increase before returning to normal NP values after
delivery (40,41). The increased levels with the
progression of pregnancy could be partly related to synthesis of
ceruloplasmin, a major Cu-binding protein, due to altered levels of
oestrogen (41).
This study also profiled the mRNA expression levels
of pro-inflammatory cytokines (TNF-α, IL-6 and IL-8),
S100A8 and TRAIL in RM samples compared with HP
women. TRAIL is also a TNF member that can induce apoptosis
via its apoptotic receptors (42).
Apoptosis plays an important role in normal human placental
development and any altered balance between proliferation and
apoptosis of villous trophoblast is associated with abnormal
pregnancies (43). Increased levels
of villous trophoblast apoptosis have been identified in several
placental pathologies including RM (44). TRAIL and its receptors appear to be
differentially expressed in villous placenta. Expression of TRAIL
and its two decoy receptors DcR1 and DcR2 are localized
predominantly in the syncytiotrophoblast, whereas cytotrophoblast
cells have been shown to express high levels of DR4 and DR5
(45) that trigger apoptotic
signalling via activation of the classic caspase-dependent ‘death’
pathway (46). It has been
previously shown (47) that
TRAIL and S100A8 are differentially expressed genes
in placental tissue in RM compared with normal pregnancy. The study
also suggested that TRAIL can be used as a potential predictive
biomarker in maternal serum for early pregnancy complications
(47). This supports our current
findings of highly significant expression levels of both
TRAIL and S100A8 in placental tissue of RM compared
with HP samples. Similar findings have been previously documented
(48) demonstrating that serum TRAIL
is significantly increased in patients of RM. This suggests that
TRAIL might have a direct effect on RM and could be used as a
predictive biomarker in maternal serum in women with history of
RM.
A number of cytokines have been reported essential
for the reproductive processes, and it has been suggested that a
lower index of T-helper1/T-helper2 (Th1/Th2) immune response
supports a physiological pregnancy (49). The increased production of Th1
cytokines, such as TNF-α and interferon-γ (IFN-γ), compared with
the Th2 cytokine has been linked to recurrent spontaneous abortions
(50). TNF-α is a cytokine that is
important in inflammation and has been strongly suggested to have a
role in cardiovascular disease pathogenesis. TNF-α is also found
during most inflammatory stages and can trigger the production of
the pro-inflammatory cytokine IL-6 (51). Proinflammatory cytokines have been
suggested as markers for OS and LPO (52).
It has been demonstrated that TNF-α acts in
synergism with other inflammatory cytokines and stimulates the
synthesis and release of prostaglandins inducing uterine
contractions and the onset of labour (53). It has also been proposed that TNF-α
plays an important role in apoptosis. Cytokine receptors, such as
tumor necrosis factor-receptor 1 (TNF-R1), have also been
associated with miscarriage. This is probably due to their role as
apoptotic mediators through pro-inflammatory cytokines, such as
TNF-α (54). TNF-α can inhibit the
proliferation of human trophoblast cells in vitro (55) and the administration of TNF-α and
INF-γ to normal pregnant mice has been shown to lead to abortion
(55). Our findings indicated
significant increases in the expression levels of TNF-α in
placental tissue of RM patients compared with those of HP women.
These results are supported by previous findings of the expression
of TNF-α in peripheral blood cells (56) and the similar high significant
increase in serum TNF-α in RM compared with HP women
(28,32). Taken together, these findings suggest
that OS and TNF-α have a potential detrimental effect on pregnancy
and high levels may play an important role in the pathogenesis of
RM.
Cytokines are also known to play an important role
in implantation and balance of locally produced pro-inflammatory,
and anti-inflammatory cytokines have been previously suggested to
be critical for a successful pregnancy (57). Thus, imbalanced cytokine production
may occur in early pregnancy loss. TNF-α and IL-6, as inflammatory
factors, may have immune-modulatory and anti-inflammatory effects
in addition to maintaining the maternal-foetal immune tolerance.
Their overexpression could induce several pathologies including RM
(58–61). The current results demonstrated that
the expression levels of TNF-α and IL-6 in placental
tissues of RM patients were significantly higher than those of HP
women. To this end, it has been reported that any disturbance in
the balance of TNF-α and IL-6 could eventually lead
to miscarriage (62,63). It has been previously shown that
TNF-α and IL-6 concentrations in murine trophoblasts with recurrent
spontaneous abortion are significantly higher than in control group
(63). This suggests that TNF-α and
IL-6 might play an important role in the embryonic development as
well. IL-8 is a pro-inflammatory cytokine that is often associated
with inflammation and its secretion is increased by OS, thereby
causing recruitment of inflammatory cells and inducing further
increase in OS mediators (64). The
present results showed upregulated mRNA expression of IL-8
gene in the placental tissue of RM women compared with HP samples.
It is thus proposed that the increased mRNA expression levels of
pro-inflammatory cytokines in placental tissue could be considered
as another causative factor of the pathogenesis of RM.
In conclusion, the current results confirm several
previous studies linking OS and RM. Antioxidant and pro-oxidant
status might be a useful tool in estimating the risk of OS and
associated diseases and they are becoming increasingly important.
This is because they may assist in designing strategies for the
prevention and management of OS. Such strategies include the use of
micronutrient supplements to maintain effective antioxidant defence
in women at risk of RM.
Acknowledgements
Not applicable.
Funding
This project was financially supported by King Saud
University, Vice Deanship of Research Chairs.
Availability of data and materials
The datasets used and/or analyzed during the
current study are available from the corresponding author on
reasonable request.
Authors contributions
MAMAS, YAAS and AFA conceived and designed the
study. MAMAS, MMA and AFA were responsible for the methodology.
MAMAS, FSA, HKG and YAAS were responsible for the data validation.
YAAS, HKG and MAMAS performed formal analysis. MAMAS, YAAS, MMA and
HKG were involved in the investigative aspects of the study. YAAS
and MMA provided resources. MAMAS and FSA wrote and prepared the
original draft. MAMAS wrote, reviewed and edited the manuscript.
YAAS was responsible for the project administration and funding
acquisition. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
Ethics approval was obtained from the Ethics Review
Committee of the College of Applied Medical Sciences of King Saud
University (CAMS 27–34/35; Riyadh, Kingdom of Saudi Arabia). Signed
consent forms were obtained from all subjects who participated in
this research.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Goodale LF, Hayrabedyan S, Todorova K,
Roussev R, Ramu S, Stamatkin C, Coulam CB, Barnea ER and Gilbert
RO: Pre implantation factor (PIF) protects cultured embryos against
oxidative stress: Relevance for recurrent pregnancy loss (RPL)
therapy. Oncotarget. 8:32419–32432. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Fortis MF, Fraga LR, Boquett JA, Kowalski
TW, Dutra CG, Gonçalves RO, Vianna FSL, Schüler-Faccini L and
Sanseverino MT: Angiogenesis and oxidative stress-related gene
variants in recurrent pregnancy loss. Reprod Fertil Dev.
30:498–506. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Tur-Torres MH, Garrido-Gimenez C and
Alijotas-Reig J: Genetics of recurrent miscarriage and fetal loss.
Best Pract Res Clin Obstet Gynaecol. 42:11–25. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Azani A, Hosseinzadeh A, Azadkhah R,
Zonouzi AA, Zonouzi AP, Aftabi Y, Khani H, Heidary L, Danaii S,
Bargahi N, et al: Association of endothelial nitric oxide synthase
gene variants (−786 T>C, intron 4 b/a VNTR and 894 G>T) with
idiopathic recurrent pregnancy loss: A case-control study with
haplotype and in silico analysis. Eur J Obstet Gynecol Reprod Biol.
215:93–100. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gupta S, Agarwal A, Banerjee J and Alvarez
JG: The role of oxidative stress in spontaneous abortion and
recurrent pregnancy loss: A systematic review. Obstet Gynecol Surv.
62:335–347; quiz 353–354. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ghneim HK, Al-Sheikh YA, Alshebly MM and
Aboul-Soud MA: Superoxide dismutase activity and gene expression
levels in Saudi women with recurrent miscarriage. Mol Med Rep.
13:2606–2612. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ghneim HK and Alshebly MM: Biochemical
markers of oxidative stress in Saudi women with recurrent
miscarriage. J Korean Med Sci. 31:98–105. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yiyenoğlu ÖB, Uğur MG, Özcan HÇ, Can G,
Öztürk E, Balat Ö and Erel Ö: Assessment of oxidative stress
markers in recurrent pregnancy loss: A prospective study. Arch
Gynecol Obstet. 289:1337–1340. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pereira AC and Martel F: Oxidative stress
in pregnancy and fertility pathologies. Cell Biol Toxicol.
30:301–312. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Park S, Lim W, Bazer FW and Song G:
Naringenin suppresses growth of human placental choriocarcinoma via
reactive oxygen species-mediated P38 and JNK MAPK pathways.
Phytomedicine. 50:238–246. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Menezo YJ, Silvestris E, Dale B and Elder
K: Oxidative stress and alterations in DNA methylation: Two sides
of the same coin in reproduction. Reprod Biomed Online. 33:668–683.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ishii T, Yasuda K, Miyazawa M, Mitsushita
J, Johnson TE, Hartman PS and Ishii N: Infertility and recurrent
miscarriage with complex II deficiency-dependent mitochondrial
oxidative stress in animal models. Mech Ageing Dev. 155:22–35.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Dröse S and Brandt U: Molecular mechanisms
of superoxide production by the mitochondrial respiratory chain.
In: Mitochondrial Oxidative PhosphorylationSpringer; Berlin: pp.
145–169. 2012
|
|
14
|
Couto N, Malys N, Gaskell SJ and Barber J:
Partition and turnover of glutathione reductase from
Saccharomyces cerevisiae: A proteomic approach. J Proteome
Res. 12:2885–2894. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Johnston RB Jr, Keele BB Jr, Misra HP,
Lehmeyer JE, Webb LS, Baehner RL and RaJagopalan KV: The role of
superoxide anion generation in phagocytic bactericidal activity.
Studies with normal and chronic granulomatous disease leukocytes. J
Clin Invest. 55:1357–1372. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ilhan N, Ilhan N and Simsek M: The changes
of trace elements, malondialdehyde levels and superoxide dismutase
activities in pregnancy with or without preeclampsia. Clin Biochem.
35:393–397. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Saleh S, Maraqa AD and Ali ME: Regional
distribution of superoxide dismutase activity in human placenta and
its correlation with lipid peroxidation. Jordan J Biol Sci.
3:125–132. 2010.
|
|
18
|
Aebi H: Catalase in vitro. Methods
Enzymol. 105:121–126. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ghneim HK and Al-Sheikh YA: Effect of
selenium supplementation on glutathione peroxidase and catalase
activities in senescent cultured human fibroblasts. Ann Nutr Metab.
59:127–138. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gherghel D, Griffiths HR, Hilton EJ,
Cunliffe IA and Hosking SL: Systemic reduction in glutathione
levels occurs in patients with primary open-angle glaucoma. Invest
Ophthalmol Vis Sci. 46:877–883. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gherghel D, Mroczkowska S and Qin L:
Reduction in blood glutathione levels occurs similarly in patients
with primary-open angle or normal tension glaucoma. Invest
Ophthalmol Vis Sci. 54:3333–3339. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Osada H, Watanabe Y, Nishimura Y, Yukawa
M, Seki K and Sekiya S: Profile of trace element concentrations in
the feto-placental unit in relation to fetal growth. Acta Obstet
Gynecol Scand. 81:931–937. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zitka O, Skalickova S, Gumulec J, Masarik
M, Adam V, Hubalek J, Trnkova L, Kruseova J, Eckschlager T and
Kizek R: Redox status expressed as GSH:GSSG ratio as a marker for
oxidative stress in paediatric tumour patients. Oncol Lett.
4:1247–1253. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kim J, Yun M, Kim EO, Jung DB, Won G, Kim
B, Jung JH and Kim SH: Decursin enhances TRAIL-induced apoptosis
through oxidative stress mediated-endoplasmic reticulum stress
signalling in non-small cell lung cancers. Br J Pharmacol.
173:1033–1044. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Girotti AW: Mechanisms of lipid
peroxidation. J Free Radic Biol Med. 1:87–95. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Talat T: The relationship between serum
copper, zinc, and glutathione peroxidase with malondialdehyde in
women with unexplained recurrent miscarriage. Kufa Med J. 12:29–37.
2009.
|
|
27
|
Sugino N, Nakata M, Kashida S, Karube A,
Takiguchi S and Kato H: Decreased superoxide dismutase expression
and increased concentrations of lipid peroxide and prostaglandin
F(2α) in the decidua of failed pregnancy. Mol Hum Reprod.
6:642–647. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
El-Far M, El-Sayed IH, El-Motwally A-G,
Hashem IA and Bakry N: Tumor necrosis factor-α and oxidant status
are essential participating factors in unexplained recurrent
spontaneous abortions. Clin Chem Lab Med. 45:879–883. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Aledo JC: Life-history constraints on the
mechanisms that control the rate of ROS production. Curr Genomics.
15:217–230. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Betteridge DJ: What is oxidative stress?
Metabolism. 49 (Suppl 1):3–8. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jenkins C, Wilson R, Roberts J, Miller H,
McKillop JH and Walker JJ: Antioxidants: Their role in pregnancy
and miscarriage. Antioxid Redox Signal. 2:623–628. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
El-Far M, El-Sayed IH, El-Motwally AE,
Hashem IA and Bakry N: Serum levels of TNF-α and antioxidant
enzymes and placental TNF-α expression in unexplained recurrent
spontaneous miscarriage. J Physiol Biochem. 65:175–181. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Schafer FQ and Buettner GR: Redox
environment of the cell as viewed through the redox state of the
glutathione disulfide/glutathione couple. Free Radic Biol Med.
30:1191–1212. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Vural P, Akgül C, Yildirim A and Canbaz M:
Antioxidant defence in recurrent abortion. Clin Chim Acta.
295:169–177. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Maggini S, Wintergerst ES, Beveridge S and
Hornig DH: Selected vitamins and trace elements support immune
function by strengthening epithelial barriers and cellular and
humoral immune responses. Br J Nutr. 98 (Suppl 1):S29–S35. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zachara BA, Dobrzyński W, Trafikowska U
and Szymański W: Blood selenium and glutathione peroxidases in
miscarriage. BJOG. 108:244–247. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Haque MM, Moghal MMR, Sarwar MS, Anonna
SN, Akter M, Karmakar P, Ahmed S, Sattar MA and Islam MS: Low serum
selenium concentration is associated with preeclampsia in pregnant
women from Bangladesh. J Trace Elem Med Biol. 33:21–25. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Rayman MP, Wijnen H, Vader H, Kooistra L
and Pop V: Maternal selenium status during early gestation and risk
for preterm birth. CMAJ. 183:549–555. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Barrington JW, Taylor M, Smith S and
Bowen-Simpkins P: Selenium and recurrent miscarriage. J Obstet
Gynaecol. 17:199–200. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Izquierdo Alvarez S, Castañón SG, Ruata
MLC, Aragüés EF, Terraz PB, Irazabal YG, González EG and Rodríguez
BG: Updating of normal levels of copper, zinc and selenium in serum
of pregnant women. J Trace Elem Med Biol. 21 (Suppl 1):49–52. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Liu J, Yang H, Shi H, Shen C, Zhou W, Dai
Q and Jiang Y: Blood copper, zinc, calcium, and magnesium levels
during different duration of pregnancy in Chinese. Biol Trace Elem
Res. 135:31–37. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wang S and El-Deiry WS: TRAIL and
apoptosis induction by TNF-family death receptors. Oncogene.
22:8628–8633. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Sharp AN, Heazell AE, Crocker IP and Mor
G: Placental apoptosis in health and disease. Am J Reprod Immunol.
64:159–169. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Choi HK, Choi BC, Lee SH, Kim JW, Cha KY
and Baek KH: Expression of angiogenesis- and apoptosis-related
genes in chorionic villi derived from recurrent pregnancy loss
patients. Mol Reprod Dev. 66:24–31. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Chen L, Liu X, Zhu Y, Cao Y, Sun L and Jin
B: Localization and variation of TRAIL and its receptors in human
placenta during gestation. Life Sci. 74:1479–1486. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Walczak H and Haas TL: Biochemical
analysis of the native trail death-inducing signaling complex.
Methods Mol Biol. 414:221–239. 2008.PubMed/NCBI
|
|
47
|
Rull K, Tomberg K, Kõks S, Männik J, Möls
M, Sirotkina M, Värv S and Laan M: Increased placental expression
and maternal serum levels of apoptosis-inducing TRAIL in recurrent
miscarriage. Placenta. 34:141–148. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Agostinis C, Bulla R, Tisato V, De Seta F,
Alberico S, Secchiero P and Zauli G: Soluble TRAIL is elevated in
recurrent miscarriage and inhibits the in vitro adhesion and
migration of HTR8 trophoblastic cells. Hum Reprod. 27:2941–2947.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Raghupathy R, Makhseed M, Azizieh F, Omu
A, Gupta M and Farhat R: Cytokine production by maternal
lymphocytes during normal human pregnancy and in unexplained
recurrent spontaneous abortion. Hum Reprod. 15:713–718. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Reid JG, Simpson NA, Walker RG, Economidou
O, Shillito J, Gooi HC, Duffy SR and Walker JJ: The carriage of
pro-inflammatory cytokine gene polymorphisms in recurrent pregnancy
loss. Am J Reprod Immunol. 45:35–40. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Pearson D and Shaw S: Proinflammatory
cytokine tumor necrosis factor-alpha increases with hostility and
aggression. Life Enhancement Mag. 7:2004, http://206.220.200.3/magazine/article/1002-proinflammatory-cytokine-tumor-necrosis-factor-alpha
|
|
52
|
Stentz FB, Umpierrez GE, Cuervo R and
Kitabchi AE: Proinflammatory cytokines, markers of cardiovascular
risks, oxidative stress, and lipid peroxidation in patients with
hyperglycemic crises. Diabetes. 53:2079–2086. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Haddad EK, Duclos AJ and Baines MG: Early
embryo loss is associated with local production of nitric oxide by
decidual mononuclear cells. J Exp Med. 182:1143–1151. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Yu XW, Yan CF, Jin H and Li X: Tumor
necrosis factor receptor 1 expression and early spontaneous
abortion. Int J Gynaecol Obstet. 88:44–48. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Chaouat G, Menu E, Clark DA, Dy M,
Minkowski M and Wegmann TG: Control of fetal survival in CBA ×
DBA/2 mice by lymphokine therapy. J Reprod Fertil. 89:447–458.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Daher S, Fonseca F, Ribeiro OG, Musatti CC
and Gerbase-DeLima M: Tumor necrosis factor during pregnancy and at
the onset of labor and spontaneous abortion. Eur J Obstet Gynecol
Reprod Biol. 83:77–79. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Choi YK and Kwak-Kim J: Cytokine gene
polymorphisms in recurrent spontaneous abortions: A comprehensive
review. Am J Reprod Immunol. 60:91–110. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Lin QD and Qiu LH: Pathogenesis,
diagnosis, and treatment of recurrent spontaneous abortion with
immune type. Front Med China. 4:275–279. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Teklenburg G, Salker M, Heijnen C, Macklon
NS and Brosens JJ: The molecular basis of recurrent pregnancy loss:
Impaired natural embryo selection. Mol Hum Reprod. 16:886–895.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Li D and Li J: Association of
miR-34a-3p/5p, miR-141-3p/5p, and miR-24 in decidual natural killer
cells with unexplained recurrent spontaneous abortion. Med Sci
Monit. 22:922–929. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Finan RR, Al-Irhayim Z, Mustafa FE,
Al-Zaman I, Mohammed FA, Al-Khateeb GM, Madan S, Issa AA and Almawi
WY: Tumor necrosis factor-α polymorphisms in women with idiopathic
recurrent miscarriage. J Reprod Immunol. 84:186–192. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Xiao SJ, Zhao AM and Bao SΜ: Relationship
between expression of cox-2, its signal transduction pathways and
autoimmune-type recurrent miscarriage. J Shanghai Jiaotong Univ
(Medical Science). 7:0072011.
|
|
63
|
Hua F, Li CH, Wang H and Xu HG:
Relationship between expression of COX-2, TNF-α, IL-6 and
autoimmune-type recurrent miscarriage. Asian Pac J Trop Med.
6:990–994. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Vlahopoulos S, Boldogh I, Casola A and
Brasier AR: Nuclear factor-kappaB-dependent induction of
interleukin-8 gene expression by tumor necrosis factor α: Evidence
for an antioxidant sensitive activating pathway distinct from
nuclear translocation. Blood. 94:1878–1889. 1999. View Article : Google Scholar : PubMed/NCBI
|