Introduction
In the natural reproductive cycle, the luteal phase
is the result of intermittent stimulation of the corpus luteum by
pituitary luteinizing hormone (LH), which is different from
assisted reproductive technology (ART) cycles. Luteal-phase
deficiency is common during the follicular stimulation phase of the
menstrual cycle (1) and leads to a
decreased embryo implantation rate, a lower pregnancy rate and an
increased miscarriage rate (2).
Controlled ovarian stimulation is usually involved in in
vitro fertilization (IVF)/intracytoplasmic sperm injection
(ICSI)/embryo transfer (ET) technology, causing the development of
multiple follicles and the formation of multiple embryos to
increase the clinical pregnancy rate (CPR) and live birth rate. The
development of multiple follicles leads to abnormally high levels
of estrogen and multiple corpora lutea are formed after aspiration,
which maintain the abnormally high production of steroids (3,4). In a
natural pregnancy, LH is continuously produced after the ovulation
surge until human chorionic gonadotropin (hCG) is secreted by the
proliferating trophoblast. In addition, the high steroid levels
exert a negative feedback on the pituitary gland, thus prematurely
inhibiting the production of LH during ART cycles (4). By contrast, aspiration of granulosa
cells may interfere with the production of progesterone (P)
(5), leading to a reduction in the
luteal phase to cause a condition known as premature luteolysis
(3).
To overcome this issue, pharmacological support,
including combinations of estradiol, P and hCG, have been
frequently applied to directly or indirectly increase the low
levels of P (6). Recently,
gonadotropin-releasing hormone (GnRH) agonist has been used for
luteal-phase support (LPS) (7,8). A
possible explanation for the effect of GnRH agonist is that it
extends LH production, thus preventing the occurrence of premature
luteolysis (3). However, it was
observed that a single dose GnRH agonist at the time of
implantation improved the pregnancy rate in recipients after
artificial endometrial preparation, inducing the downregulation of
GnRH followed by a decrease in the levels of estrogen and P
(8). In addition, GnRH agonist may
directly influence the quality of the early embryo for the
recipients without corpus luteum (4,8),
although a direct effect on the endometrium cannot be excluded. A
previous in vitro study provided similar results, since GnRH
agonist receptor was indicated to be broadly expressed in the human
morula and at the blastocyst stage (9). The development of porcine and murine
pre-implantation embryos is enhanced when incubated with GnRH
agonist and diminished when incubated with GnRH antagonist
(10,11).
In total, four previous systematic reviews concluded
that there may be benefits from the addition of single-dose GnRH
agonist to improve luteal support (3,12–14), and
it was suggested that this treatment is relatively safe and
effective (15). However, it is
required to perform a comprehensive and unbiased systematic review
analyzing intensive studies and recent results (16). The aim of the present meta-analysis
was to identify, analyze and summarize evidence from randomized
controlled trials (RCTs) and examine the effects of single-dose
GnRH agonist for luteal support in females undergoing IVF/ICSI
cycles.
Materials and methods
Eligibility criteria
All published and ongoing RCTs assessing the
administration effect of single-dose GnRH agonist during the luteal
phase on IVF/ICSI outcomes were included in the present
meta-analysis. Trials including egg donation and frozen embryo
transfer cycles were excluded. Studies with multiple-dose GnRH
agonist treatments in the luteal phase were excluded due to the
lack of safety assessment and the large difference in GnRH agonist
application protocols.
Grouping
In total, four groups were considered: i)
Single-dose GnRH agonist administered as LPS at 5/6 days after
IVF/ICSI procedures (GnRHa group); ii) regular support, where
progesterone/estradiol/hCG was used for LPS (control group); iii)
GnRH agonist was used to suppress premature LH surge (GnRH-a
group); and iv) GnRH antagonist was used to suppress premature LH
surge (GnRH-A group). In the GnRH-a group, a single GnRH agonist
(Decapeptyl, 1.25–3.75 mg) was administered on day 24–26 of the
cycle. After 2 weeks, Gn (Gonadotropin) are used to stimulate
follicular development. In GnRH-A group, GnRH antagonist (Ganirelix
acetate) was given 0.25 mg/d from day 5 or 6 of Gn administration
until hCG day.
Outcome measures
The outcome measures used for the present
meta-analysis were as follows: i) Ongoing pregnancy or live birth
rate per transfer; ii) CPR per transfer; iii) multiple pregnancy
rate; and iv) clinical abortion rate. Data in which the live birth
rate was recorded was generally preferred. However, when live birth
was not reported, data from an ongoing pregnancy (intrauterine live
fetus with a gestational age ≥12 weeks) were used as a measure for
live birth, since the difference between live birth and ongoing
pregnancy rates are limited and <1% of pregnancies result in
stillbirth (17,18). When studies reported on clinical
pregnancy and ongoing pregnancy without miscarriage rates, the
number of clinical abortions was considered as being equal to the
difference between the number of clinical pregnancies and ongoing
pregnancies.
Search strategy
Published studies were searched in the following
electronic databases: i) China National Knowledge Infrastructure
(CNKI), ii) Wanfang database, iii) Chinese Biomedicine Literature
Database (CBM), iv) Pubmed, v) EMBASE and vi) Cochrane Controlled
Trials Register. The entries analyzed were published prior to June
1st, 2018. There was no language restriction. The following terms
were used, adjusting for each database as necessary: ‘Fertilization
in vitro’ OR ‘in vitro fertilization’ OR in
vitro fertilizations’ OR ‘test-tube fertilization’ OR
‘fertilization, test-tube’ OR ‘fertilizations, test-tube’ OR ‘test
tube fertilization’ OR ‘test-tube fertilizations’ OR
‘fertilizations in vitro’ OR ‘test-tube babies’ OR ‘babies,
test-tube’ OR ‘baby, test-tube’ OR ‘test tube babies’ OR
‘intracytoplasmic sperm injection’ OR ‘IVF’ OR ‘ICSI’ AND ‘luteal
support’ OR ‘luteal phase support’ AND ‘gonadotropin-releasing
hormone agonist’ OR ‘GnRH-a’ OR ‘GnRHa’ AND ‘randomized controlled
trial’ OR ‘randomized’.
Study selection and search
results
The two researchers (MLS and CLL) independently
screened the studies as follows: i) Duplicated articles were
removed using NoteExpress software (version 3.2; Aegean Software
Corp.); ii) two researchers read the titles and abstracts and
manually removed the articles that did not meet the inclusion
criteria of the present study; ii) the articles were further
screened by reading the full text and excluded or included
according to the inclusion and exclusion criteria of the present
study. When inconsistent results were obtained, the issues were
resolved by discussing with relevant experts in the field. Missing
data were obtained from the authors of the respective studies
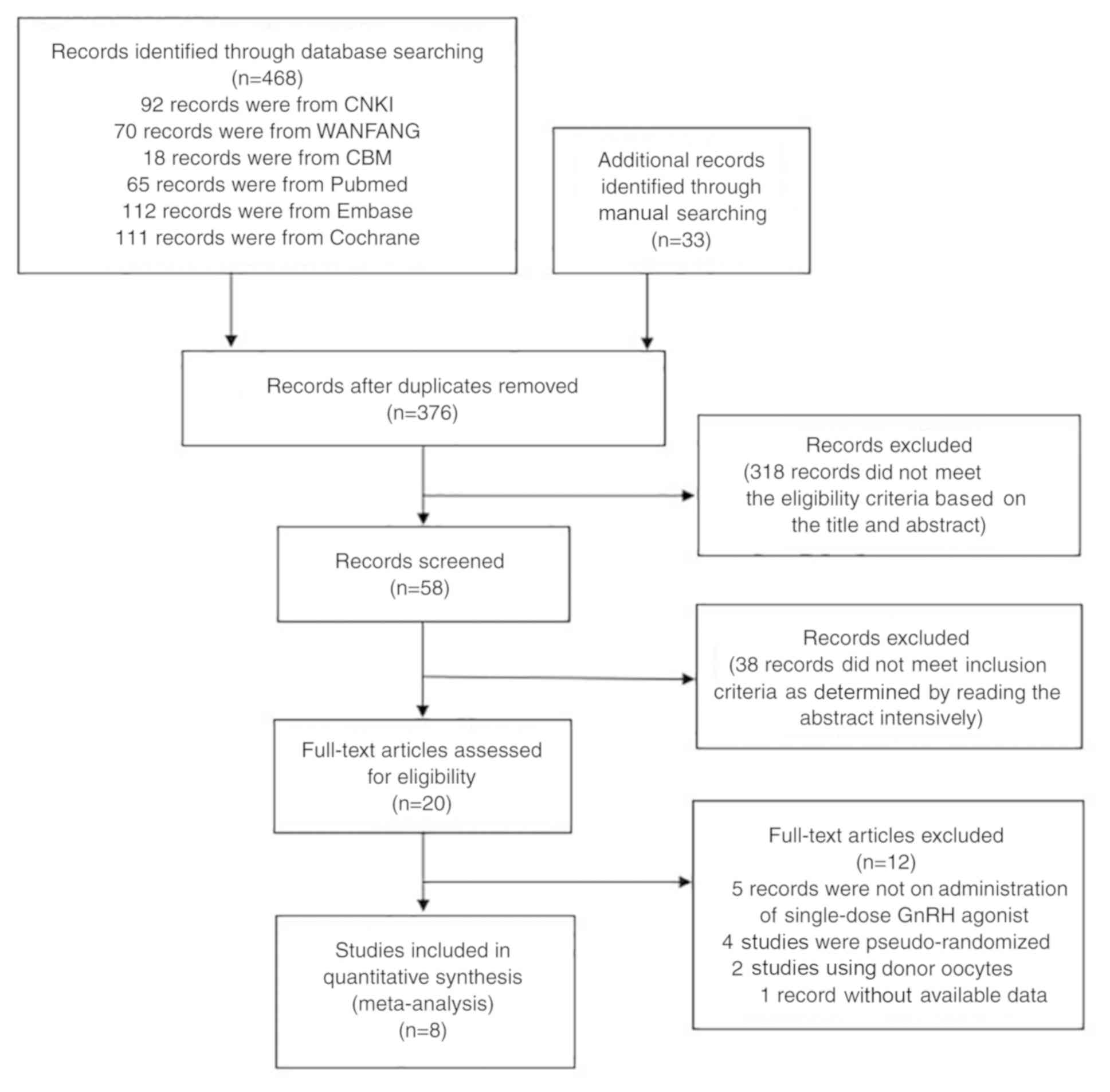
whenever possible. In total, 468 records were retrieved in the
initial electronic search: i) 92 records were from CNKI; ii) 70
records were from WANFANG; iii) 18 records were from CBM; iv) 65
records were from Pubmed; v) 112 records were from Embase; and vi)
111 records were from Cochrane. A total of 33 additional records
were retrieved through manual search of potentially eligible
studies and relevant reviews. A total of 501 records were assessed
for eligibility. After removal of duplicate articles, 376 documents
were retained and after reading the titles and abstracts, 318
records were removed. In addition, 38 articles that did not meet
the inclusion criteria were excluded. Finally, among the 20
potentially relevant studies were obtained and a total of 8 studies
fulfilled the inclusion criteria (19–26). A
flowchart depicting the selection process is provided in Fig. 1.
Data extraction
In three studies (20,22,24), the
long GnRH-a ovarian treatment protocol starting in the mid-luteal
phase of the preceding cycle was used, in four studies (21,23,25,26), the
GnRH-A multi low-dose ovarian treatment protocol was applied and in
one study (19), both ovarian
treatment protocol types were used. The trials published by Tesarik
et al (8) used both the
GnRH-a and the GnRH-A treatment protocol. In all studies, IVF or
ICSI were performed. The characteristics of the eight studies
identified, including the eligibility criteria, are provided in
Table I.
 | Table I.Major characteristics of randomized
controlled trials on the administration GnRH agonist in the luteal
phase. |
Table I.
Major characteristics of randomized
controlled trials on the administration GnRH agonist in the luteal
phase.
| Author (year) | Funding source | Randomization
method/allocation concealment | Protocol
stimulation | Luteal phase study
group GnRH-a | Control group | Others medicines
(all patients) | Fertilization | Day of embryo
transfer | Embryos transfered
(n) | (Refs.) |
|---|
| Tesarik (2006) | Not reported | Embryo transfer day
Computer-generated list Opaque envelopes Blinding | GnRH-a long
protocol/GnRH- ant multiple doses + r-FSH/HMG | GnRH-a long
protocol (n=150) GnRH-A protocol (n=150) Single injection Dose: 0.1
mg triptorelin Day 6 after ICSI | GnRH-a long
protocol (n=150) GnRH-A protocol Placebo (n=150) | E2
valerate (4 mg) + vaginal micronized progesterone (400 mg) + r-HCG
(single dose) | ICSI | 3 | 1–3 | (19) |
| Ata (2008) | Not reported | Embryo transfer day
Computer-generated list Opaque envelopes Blinding | GnRH-a long
protocol + r-FSH | Single injection
(n=285) Dose: 0.1 mg triptorelin Day 6 after ICSI | Placebo
(n=285) | Vaginal
progesterone gel 90 mg | ICSI | 3 | 1–3 | (20) |
| Isik (2009) | Not reported | Embryo transfer day
Computer-generated list Blinding | GnRH-A multiple
doses + r-FSH/HMG | Single injection
(n=74) Dose: 0.5 mg leuprolide Day 6 after ICSI | No placebo
(n=80) | Vaginal micronized
progesterone (600 mg) + HCG (single dose) | ICSI | 3 | 1–5 | (21) |
| Razieh (2009) | Same
institution | Drawing piece of
paper from a bag | GnRH-a long
protocol + r-FSH | Single injection
(n=90) Dose: 0.1 mg triptorelin Day 5/6 after ICSI | Placebo (n=90) | Vaginal micronized
progesterone (800 mg) | ICSI | 2 or 3 | 2 or 3 | (22) |
| Ata and Urman
(2010) | Not reported | Embryo transfer day
Computer-generated list Opaque envelopes Blinding | GnRH-A multiple
doses + r-FSH | Single injection
(n=38) Dose: 0.1 mg triptorelin Day 6 after ICSI | Placebo (n=52) | Vaginal
progesterone gel 90 mg | ICSI | 3 | 1–3 | (23) |
| Yildiz (2014) | Not reported | Computer-generated
list | GnRH-a long
protocol + r-FSH | Single injection
(n=100) Dose: 1 mg leuprolide Day 6 after ICSI | No placebo
(n=100) | 17βE2 (4 mg) +
Vaginal micronized progesterone (600 mg) | ICSI | 3 | 1–3 | (24) |
| ZSa (2015) | Not reported | OCP administration
day Computer-generated list | GnRH-ant multiple
doses + r-FSH/HMG | Single injection
(n=43) Dose: 0.1 mg triptorelin Day 6 after ICSI | No placebo
(n=40) | Vaginal
progesterone (800 mg) | ICSI | 3 | 1–3 | (25) |
| BAb (2017) | Same
institution | Embryo transfer day
Computer-generated list Blinding | GnRH-ant multiple
doses + r-FSH | Single injection
(n=165) Dose: 0.1 mg triptorelin Day 6 after OPU | No placebo
(n=163) | E2 (4 mg) + Vaginal
micronized progesterone (600 mg) + r-HCG (single dose) | IVF/ICSI | 2 or 3 | 1–3 | (26) |
Risk of bias within trials
The two researchers (MLS and CLL) evaluated the
methodological quality of the studies included according to the RCT
bias risk assessment tool recommended by the Cochrane systematic
review guidelines (16). The
evaluation included the following: i) Selection bias (random
sequence generation and allocation concealment); ii) performance
bias (blinding of participants and personnel); iii) detection bias
(blinding of outcome assessment); iv) attrition bias (incomplete
outcome data); v) reporting bias (selective reporting); and vi)
other bias. In total, nine trials from eight studies were rated as
‘low risk’, ‘unknown risk’ or ‘high risk’. In the case of
disagreement, the two researchers discussed or resolved the issue
by discussion with a third researcher (RH).
Statistical analysis
The results for each of the studies eligible for the
present meta-analysis were expressed as the risk ratio (RR) and the
precision of estimates were evaluated using the 95% CI. The results
of the previous studies were combined for the present meta-analysis
using the DerSimonian and Laird method and a random-effects model
was applied. The heterogeneity analysis was performed using the
I2 test. When P≤0.05 and I2≥50%, the studies
were considered heterogeneous; 0≤I2≤25% indicated low
heterogeneity, 25%<I2≤50% indicated moderate
heterogeneity and 50%<I2≤75% indicated high
heterogeneity. All results were combined for the meta-analysis
using Revman 5.3 software (Cochrane Collaboration). In addition,
sensitivity analysis and subgroup analysis were performed for the
GnRH-a and GnRH-A groups according to a pituitary downregulation
protocol, where the levels of FSH and LH are reduced. Sensitivity
analysis was performed using a method that removed one document at
a time to assess the impact of a single study on the results of the
present meta-analysis. A re-analysis was performed after excluding
the study identified as the source of heterogeneity.
Results
Ongoing pregnancy or live birth rate
per transfer
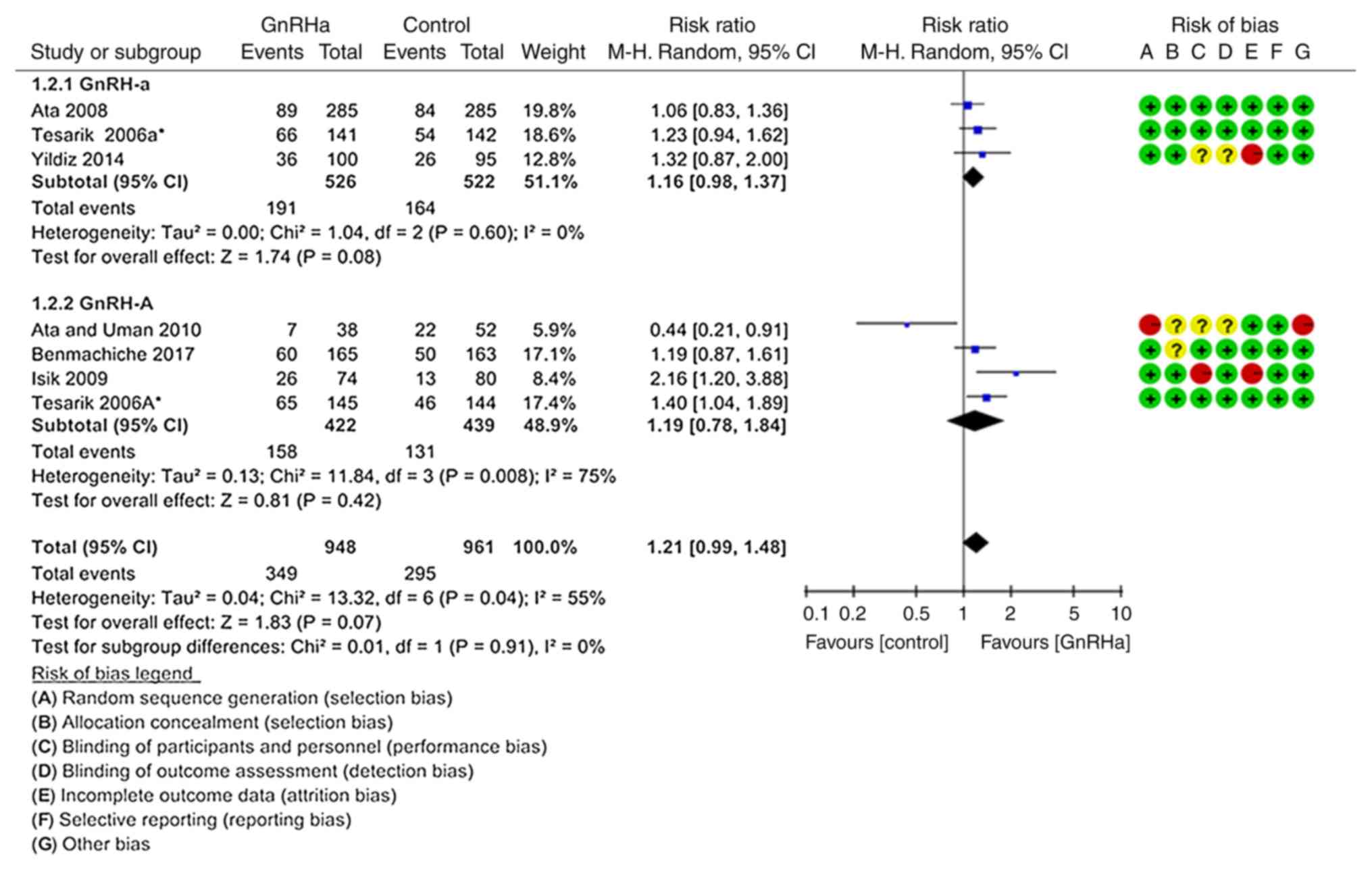
In total, seven records from six studies were
included to determine the effect of the treatments on ongoing
pregnancy or live birth rate per transfer (Fig. 2) (19–21,23,24,26). The
pooled ongoing pregnancy or live birth rate per transfer was not
significantly different between the GnRHa group (36.81%, 349/948)
and the control group (30.70%, 295/961; P=0.070; RR=1.21; 95%
CI=0.99–1.48). Subgroup analysis according to the type of GnRH
analogue used for LH suppression did not change the direction or
the magnitude of the effect observed; there were no significant
differences between the GnRH-a (P=0.080; RR=1.16, 95% CI=0.98–1.37)
(19,20,24) and
GnRH-A groups (P=0.420; RR=1.19, 95% CI=0.78–1.84; Fig. 2) (19,21,23,26).
However, there was high heterogeneity in the studies
using a GnRH-A ovarian stimulation protocol (P=0.008;
I2=75%). Therefore, sensitivity analysis under exclusion
of the study by Ata and Urman (23),
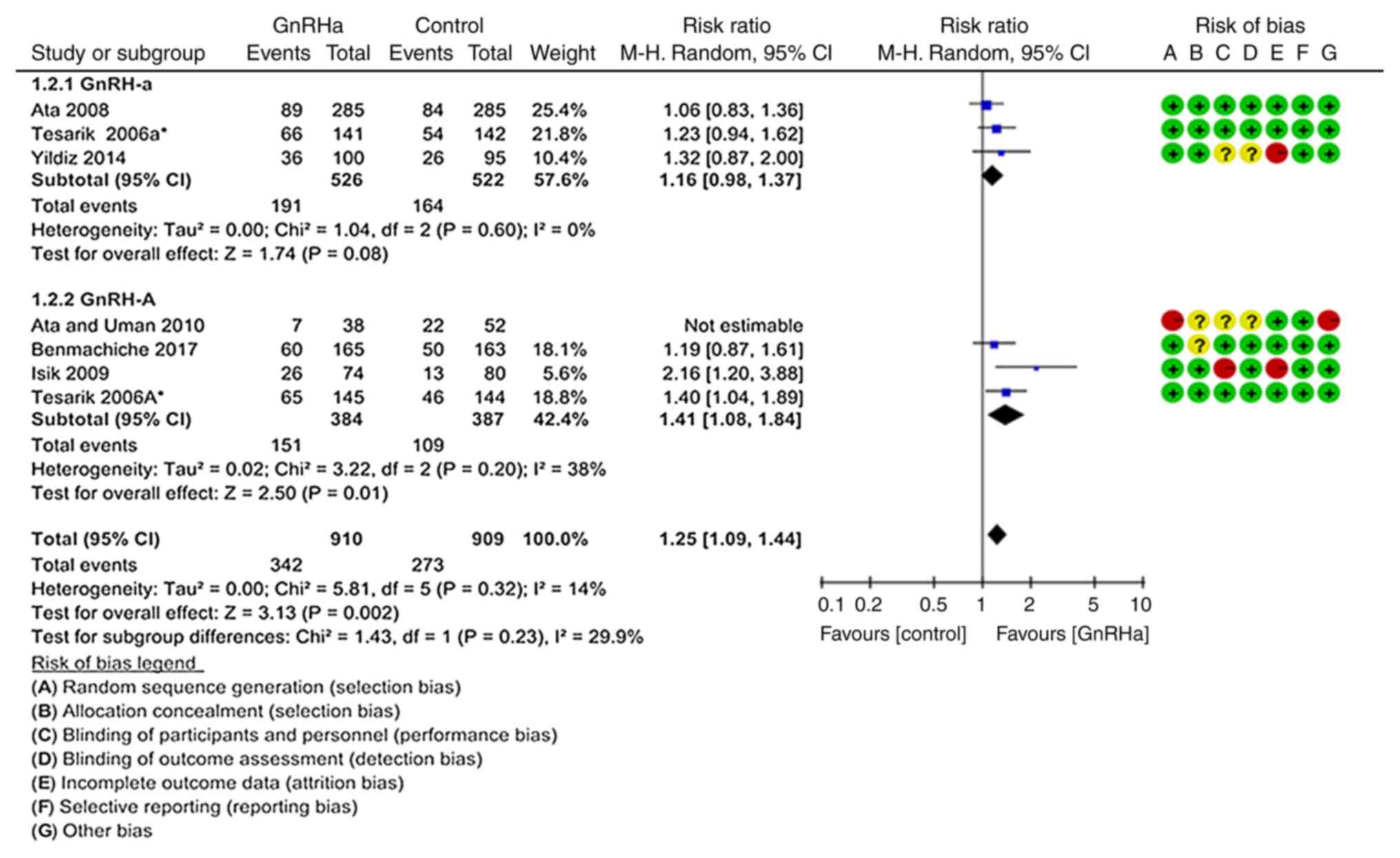
the source of heterogeneity, was performed (Fig. 3). The results suggested that the
ongoing pregnancy or live birth rate per transfer was significantly
higher in the GnRHa group (37.5%; 342/910) compared with that in
the control group (30.03%; 273/909; P=0.002; RR=1.25, 95%
CI=1.09–1.44) (19–21,24,26).
This trend was more pronounced in the GnRH-A ovarian stimulation
protocol (P=0.010; RR=1.41, 95% CI=1.08–1.84) (19,21,26).
However, there were no significant differences in the GnRH-a
ovarian treatment protocol compared with control (P=0.080; RR=1.16,
95% CI=0.98–1.37; Fig. 3) (19,20,24).
CPR per transfer
In total, nine records from eight studies were
included to calculate the CPR (Fig.
4) (19–26). The pooled CPR per transfer was
significantly higher in the GnRHa group (41.58%; 437/1,051)
compared with that in the control group (33.64%; 367/1,091;
P=0.010; RR=1.28; 95% CI=1.06–1.55). There was significant
heterogeneity in this comparison (P=0.020; I2=57%).
However, in the subgroup of trials where the long GnRH-a ovarian
stimulation protocol was used, the pooled CPR per transfer did not
differ significantly between the GnRHa group (41.72%; 257/616) and
the control group (35.62%; 218/612; P=0.090; RR=1.24, 95%
CI=0.97–1.58), and the comparison did not exhibit significant
heterogeneity (P=0.070; I2=57%) (19,20,22,24). On
the other hand, in the subgroup of trials where the GnRH-A ovarian
stimulation protocol was used, the CPR per transfer was
significantly superior in the GnRHa group (41.38%; 180/435) than in
the control group (31.11%; 149/479; P=0.040; RR=1.32, 95%
CI=0.96–1.81; Fig. 4) (19,21,23,25,26).
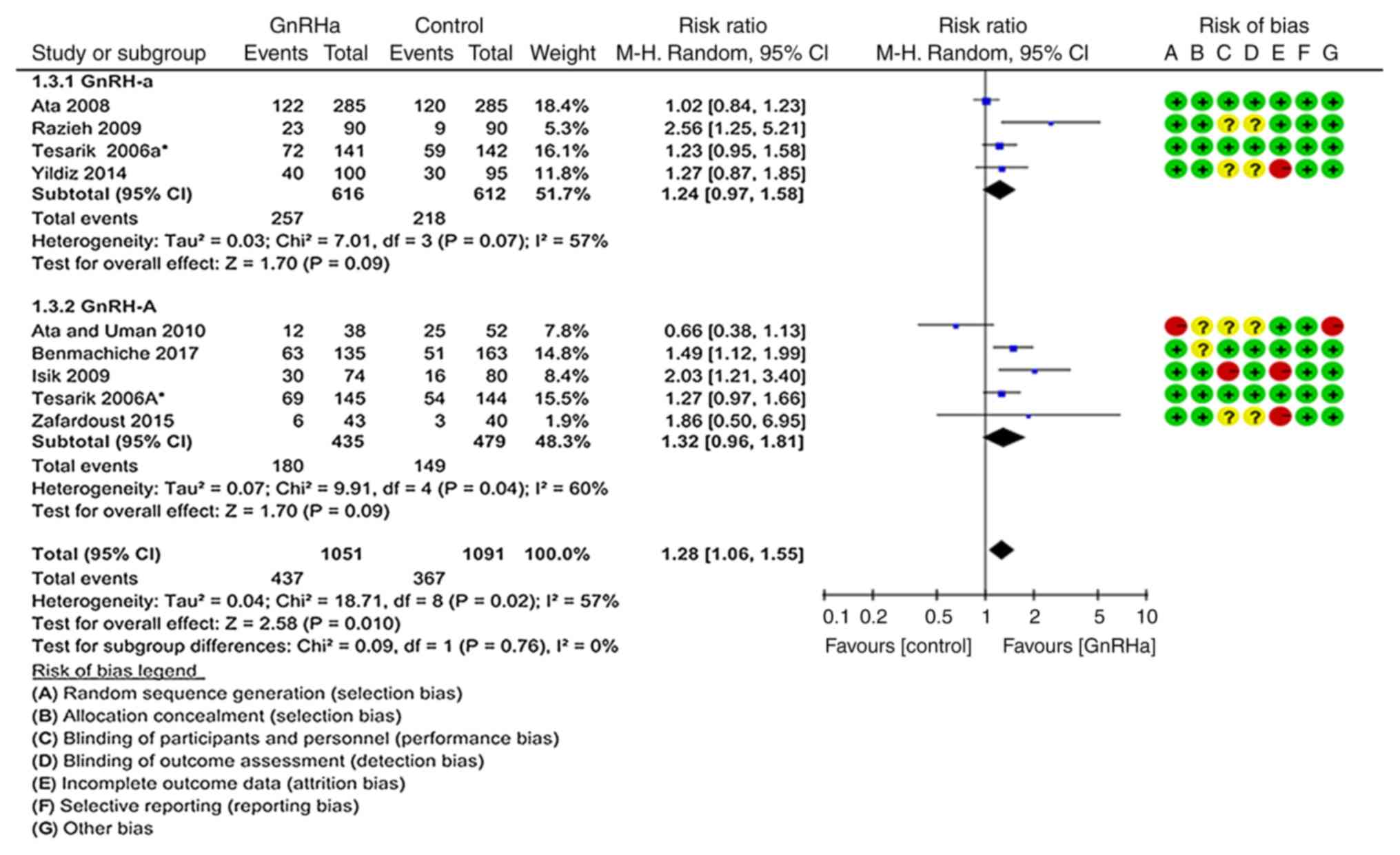 | Figure 4.Forest plot for the CPR. In total,
nine records from eight studies were included. The pooled CPR per
transfer rate was significantly higher in the GnRHa group than in
the control group. There was heterogeneity in this comparison.
However, in the subgroup of trials using the long GnRH-a ovarian
stimulation protocol, the pooled CPR per transfer did not differ
significantly between the GnRHa group and the control group
(35.62%; 218/612). Furthermore, this comparison exhibited a certain
heterogeneity (P=0.070; I2=57%) (19,20,22,24). By
contrast, in the subgroup of trials using the GnRH-A ovarian
stimulation protocol, the CPR per transfer was significantly
different in the GnRHa group compared with that in the control
group. CPR, clinical pregnancy rate; GnRH, gonadotropin-releasing
hormone; GnRH-a, long GnRH agonist ovarian stimulation protocol;
GnRH-A, GnRH antagonist multi low-dose ovarian stimulation
protocol; GnRHa, single-dose GnRH agonist for luteal support;
Control: Regular luteal support; MH, Mantel-Haentzel; df, degrees
of freedom. |
As above, a sensitivity analysis excluding the study
by Ata and Urman (23) was performed
(Fig. 5), and the pooled CPR per
transfer was significantly higher in the GnRHa group (41.95%;
425/1,013) than that in the control group (32.92%; 342/1,039;
P=0.001; RR=1.34, 95% CI=1.12–1.59) (19–22,24–26).
This trend was more evident with the GnRH-A ovarian stimulation
protocol (P<0.0001; RR=1.45, 95% CI=1.20–1.74) (19,21,25,26).
However, there were no significant differences in the GnRH-a
ovarian stimulation protocol compared with control (P=0.090;
RR=1.24, 95% CI=0.97–1.58; Fig. 5)
(19,20,22,24).
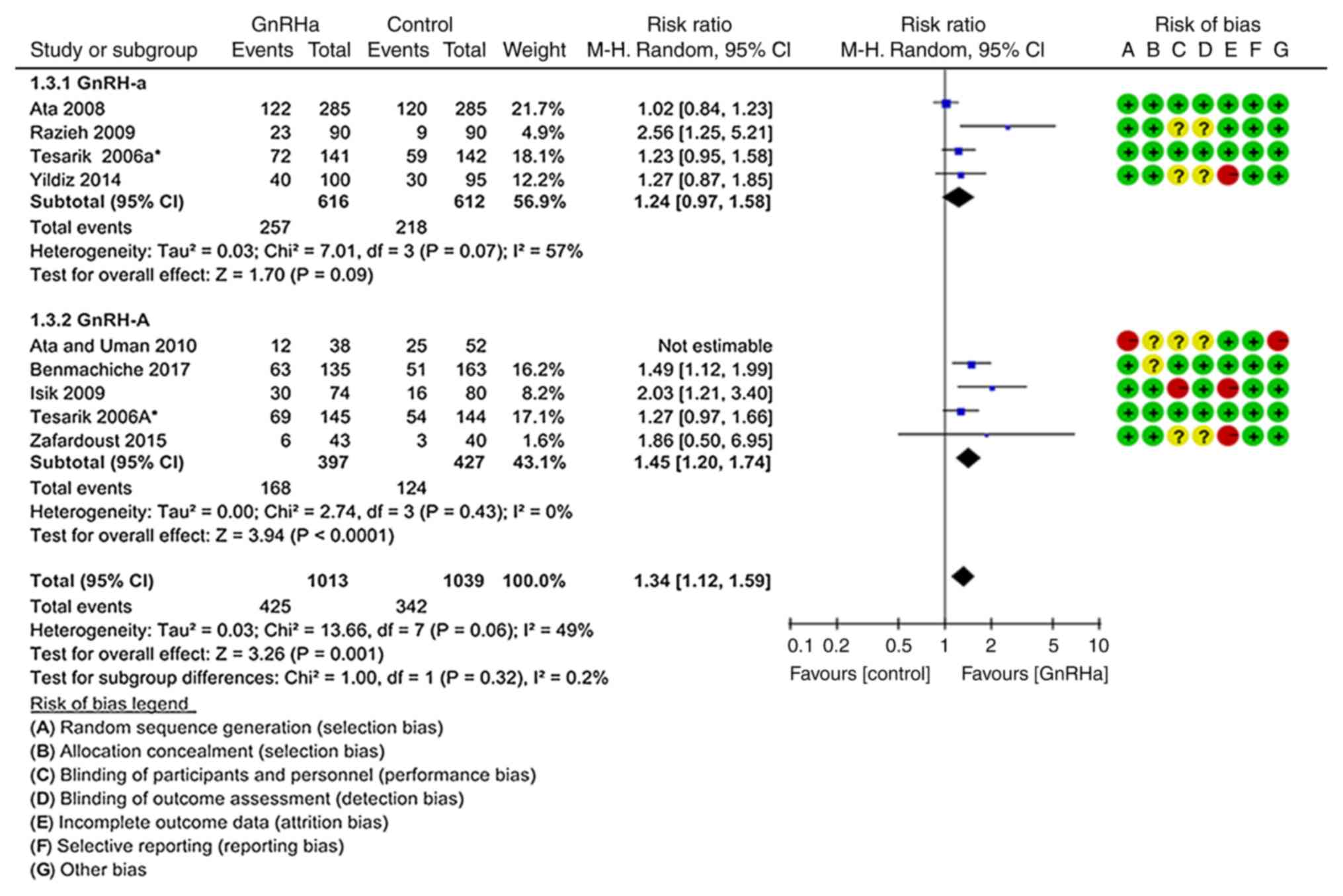 | Figure 5.Forest plot for the sensitivity
analysis for the CPR. Sensitivity analysis excluding the study by
Ata and Urman was performed, and the results suggested that the
pooled CPR per transfer was significantly higher in the GnRHa group
compared with that in the control group. This trend was more
significant following the GnRH-A ovarian stimulation protocol.
However, there were no significant differences in the GnRH-a
ovarian stimulation protocol compared with the control group. CPR,
clinical pregnancy rate; GnRH, gonadotropin-releasing hormone;
GnRH-a, long GnRH agonist ovarian stimulation protocol; GnRH-A,
GnRH antagonist multi low-dose ovarian stimulation protocol; GnRHa,
single-dose GnRH agonist for luteal support; Control, regular
luteal support; MH, Mantel-Haentzel; df, degrees of freedom. |
Multiple pregnancy rate per
pregnancy
As indicated in Fig.
6, the rate of multiple pregnancy per pregnancy was
significantly higher in the GnRHa group (32.82%; 106/323) compared
with that in the control group (19.17%; 51/266; P=0.020; RR=2.37,
95% CI=1.17–4.81). In total, four studies were included (19–21,24). In
the subgroup of trials where the GnRH-A ovarian stimulation
protocol was used, the multiple pregnancy rate per pregnancy was
significantly higher in the GnRH-A group (33.68%; 32/95) than that
in the control group (8.06%; 5/62; P=0.003; RR=3.70; 95%
CI=1.57–8.69) (19,21). However, the difference between the
GnRH-a ovarian stimulation protocol and the control group was not
significant (P=0.140; RR=1.88; 95% CI=0.82–4.34; Fig. 6) (19,20,24).
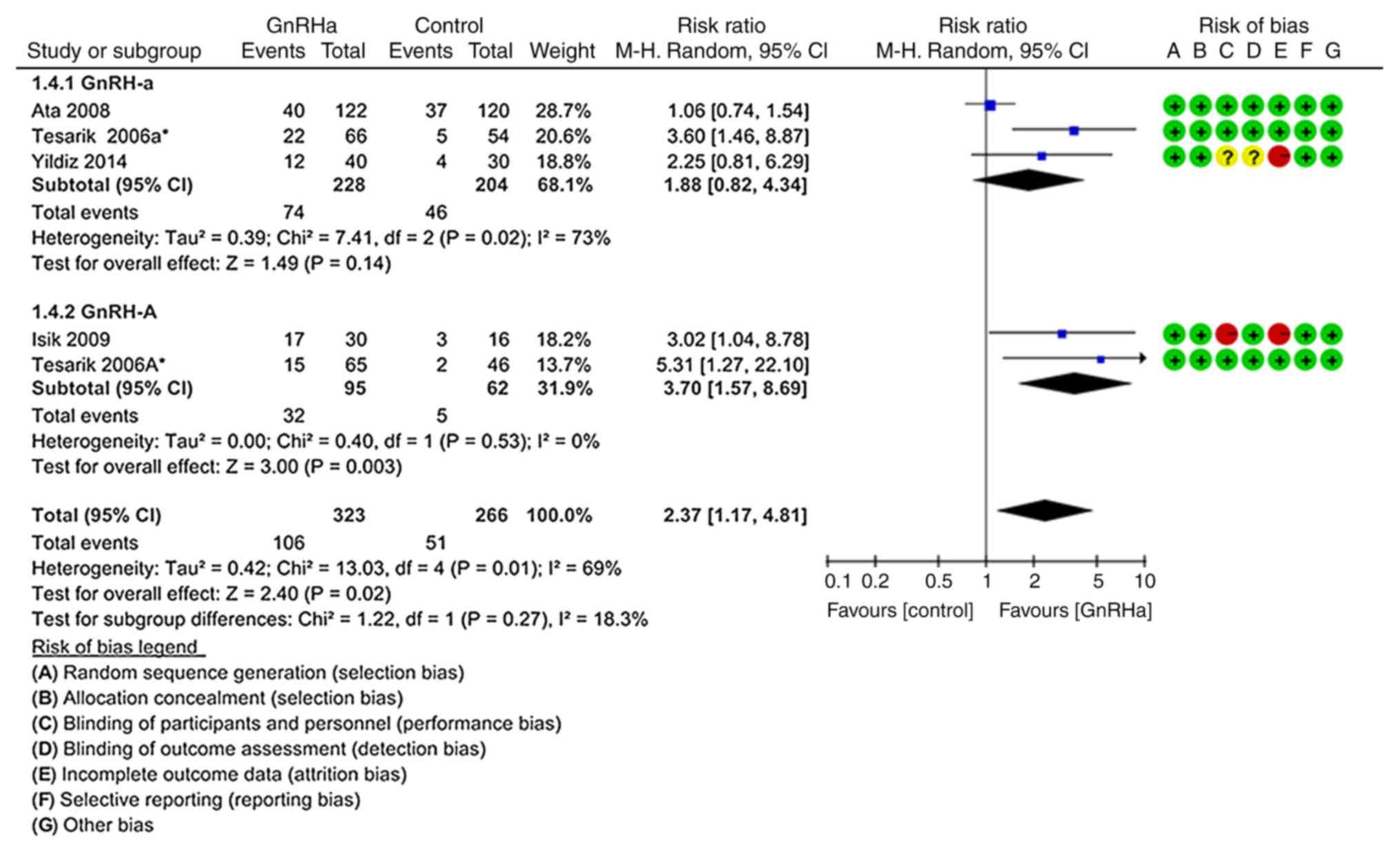 | Figure 6.Forest plot for the multiple
pregnancy rate. The probability of multiple pregnancy per pregnancy
was significantly higher in the GnRHa group compared with that in
the control group. In total, four studies were included. In the
subgroup of trials using the GnRH-A ovarian stimulation protocol,
the multiple pregnancy rate was higher in the GnRHa group than in
the control group. However, there were no significant differences
in the GnRH-a ovarian stimulation protocol. GnRH,
gonadotropin-releasing hormone; GnRH-a, long GnRH agonist ovarian
stimulation protocol; GnRH-A, GnRH antagonist multi low-dose
ovarian stimulation protocol; GnRHa, single-dose GnRH agonist for
luteal support; Control, regular luteal support; MH,
Mantel-Haentzel; df, degrees of freedom. |
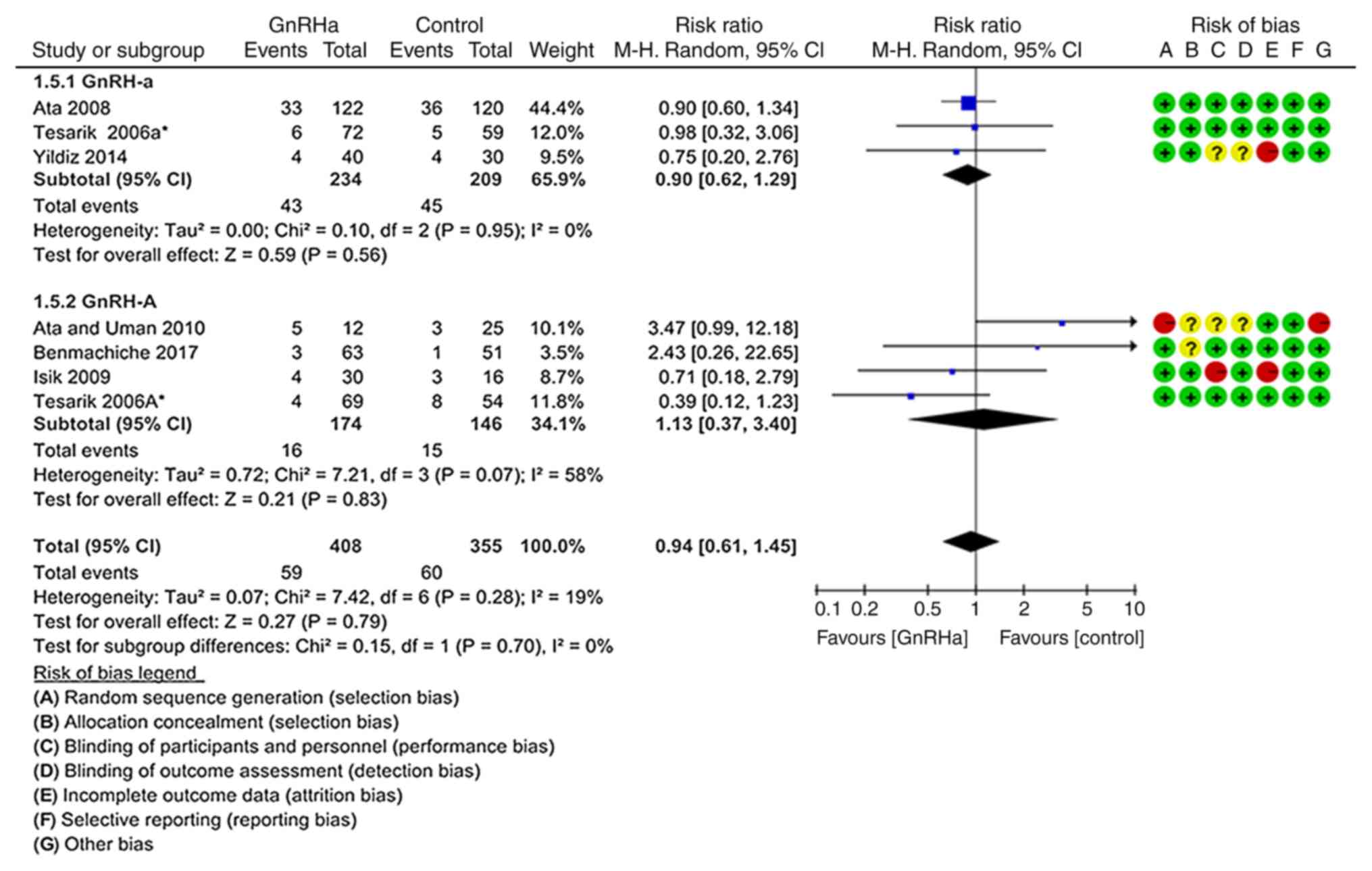
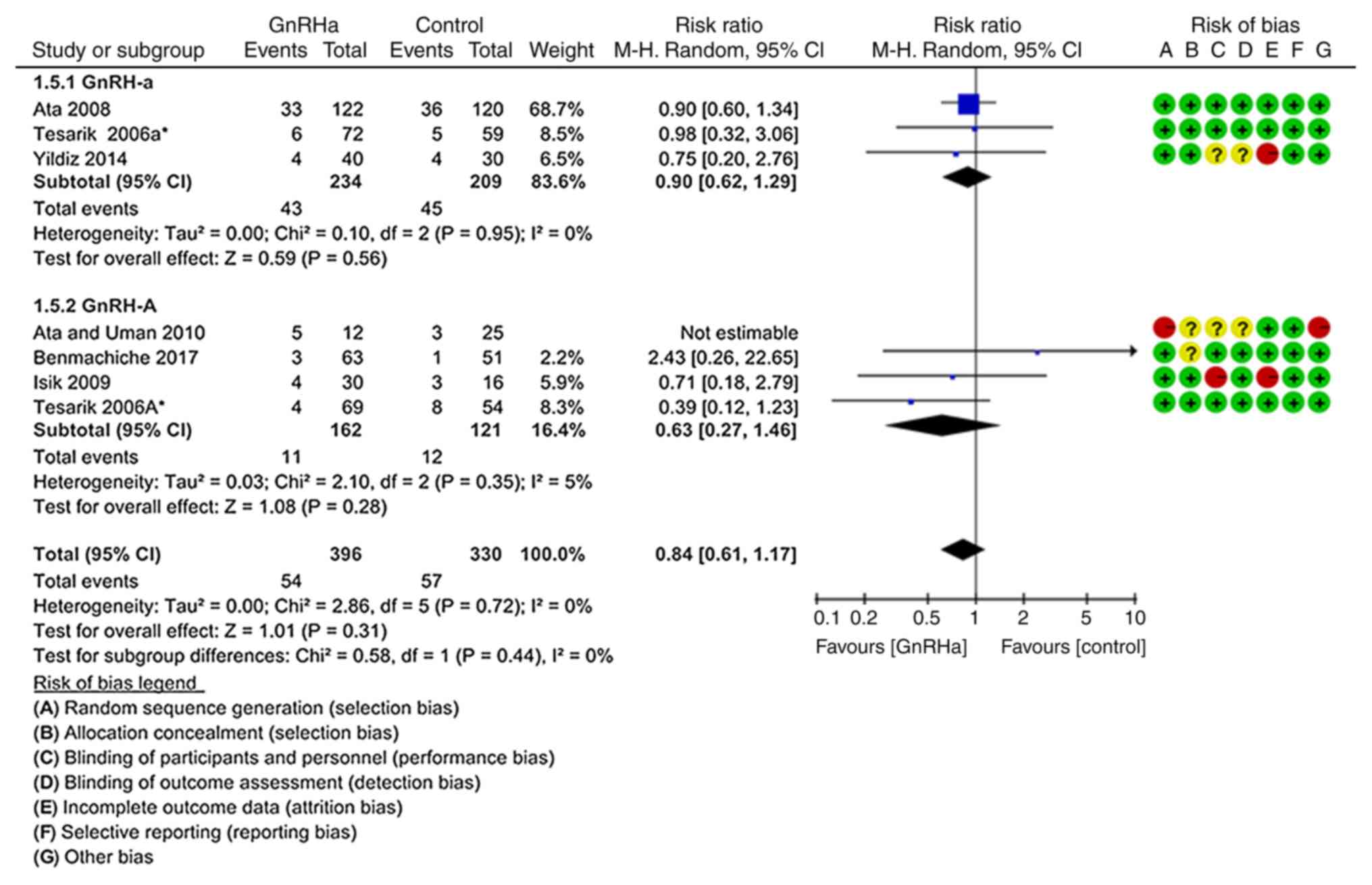
Clinical abortion rate
As indicated in Fig.
7, the clinical abortion rate was not significantly different
between the GnRHa group (14.46%; 59/408) and the control group
(16.90%; 60/355; P=0.790; RR=0.94; 95% CI=0.61–1.45). These results
were obtained from six studies (19–21,23,24,26).
There were also no significant differences in the subgroup
analyses.
According to the sensitivity analysis excluding the
study by Ata and Urman (23), the
clinical abortion rates were not significantly different between
the GnRHa group and the control group (P=0.310; RR=0.84, 95% CI=
0.61–1.17; I2=0%; Fig.
8). In addition, the subgroup analyses did not provide any
significant differences.
Risk of bias of the individual studies are presented
in the forest plots shown in Figs.
2–8. There was ‘high risk’ and
high heterogeneity in the study by Ata and Urman (23). The high risk of this study mainly
comes from selection bias (random sequence generation and
allocation concealment) and other bias.
Discussion
In 1993, Wilshire et al (27) reported that the use of GnRH agonist
during early pregnancy did not have any adverse effect on pregnancy
outcomes. Over the past years, the question regarding whether GnRH
agonist may be used during LPS to improve pregnancy outcomes has
attracted increasing attention. Zafardoust et al (25) indicated that subcutaneous injection
of 0.1 mg triptorelin on the 6th day after oocyte collection
increased the rate of embryo implantation and pregnancy rate in
ICSI patients who were downregulated following GnRH antagonist
regimen. A previous study identified increased levels of LH, as
well as increased embryo implantation and CPRs, following
intranasal inhalation of buserelin as LPS treatment compared with
those following vaginal administration of progesterone, but the
differences were not statistically significant (28). However, Aboulghar et al
(29) indicated that continuous
injection of GnRH agonist at 0.1 mg/day from the luteal phase to
hCG test day (14 days after embryo transfer) was not sufficient to
increase the rate of ongoing pregnancy and CPR, and exhibited a
negative effect; therefore, the effect of GnRH treatment in LPS
remains controversial.
The studies included were single-center RCTs with a
small sample size, and the present study included nine records from
eight studies with a total of 2,142 embryo transfer cycles. The
present results suggested that addition of GnRH agonist during LPS
significantly increased the ongoing pregnancy or live birth rate
per transfer, CPR per transfer and multiple pregnancy rate per
pregnancy. The present meta-analysis suggested that the majority of
the present results exhibited high heterogeneity. Therefore, a
sensitivity analysis was performed using Revman 5.3 software, and
it was indicated that the high heterogeneity was primarily caused
by the study by Ata and Urman (23).
Sensitivity analysis was performed following the exclusion of this
study, resulting in significantly higher rates of ongoing pregnancy
or live birth per transfer (P=0.002), clinical pregnancy per
transfer (P=0.001) and multiple pregnancy per pregnancy (P=0.020)
in the GnRHa group compared with those in the control group. In
addition, meta-analysis was performed for subgroups of trials that
had used a long GnRH-a ovarian stimulation protocol, indicating
that ongoing pregnancy or live birth rate per transfer (P=0.080),
CPR per transfer (P=0.090) and multiple pregnancy rate per
pregnancy (P=0.140) were not significantly different between the
two groups. Furthermore, the results from trials that had used
GnRH-A multi-dose ovarian stimulation protocols indicated
significantly higher ongoing pregnancy or live birth rate per
transfer (P=0.010), CPR per transfer (P<0.0001) and multiple
pregnancy rate per pregnancy (P=0.003) compared with those in the
control group. Therefore, the present results suggested that the
addition of single-dose GnRH agonist during LPS was clinically
beneficial for pregnancy outcomes.
At present, the mechanisms underlying the use of
GnRH agonist to improve pregnancy outcomes remain to be fully
elucidated. Previous studies have suggested various possible
mechanisms of action of GnRH agonist. GnRH agonist may act on the
pituitary gland or ovary during the luteal phase (30–32);
GnRH agonist may stimulate the ovaries to produce estrogen and P by
stimulating the pituitary to produce LH, and GnRH agonist may
produce estrogen and P by acting directly on the corpus luteum
(20). Furthermore, GnRH agonist may
directly act on the embryo and placenta during implantation
(8); GnRH agonist promotes the
expression of GnRH agonist receptor in placental cytotrophoblasts
and syncytiotrophoblasts and increases serum hCG levels by
upregulating GnRH agonist receptor levels and stimulating placental
production of hCG, thus improving the implantation ability of the
embryo (33,34). In addition, GnRH agonist promoted the
growth of mouse embryos when it was added in the culture medium;
however, GnRH antagonist had a detrimental effect on mouse embryos
(11). Therefore, it is possible
that pre-implantation mouse embryos may express the GnRH agonist
receptor. Furthermore, GnRH agonist may directly act via the GnRH
agonist receptor on the surface of the endometrium (8,9).
The major limitation of the present meta-analysis is
that there may be publication bias in the studies analyzed, and
this may be due to a number of reasons; the present analysis only
included studies including full texts, and abstract-only papers
were excluded due to the lack of complete statistical data. In
addition, three of articles included in the meta-analysis (21,24,25)
exhibited a high risk of bias. In addition, the evidence is limited
to recommend the use of GnRHa in the luteal phase support.
Therefore, multi-center randomized controlled studies following a
unified standardized scheme are required.
In conclusion, the present meta-analysis study
suggested that administration of single-dose GnRH agonist for LPS
in females partaking in IVF/ICSI was able to increase the ongoing
pregnancy or live birth rate per transfer, CPR per transfer and
multiple pregnancy rate per pregnancy. However, GnRH agonist did
not affect the clinical abortion rate. In addition, since the
multiple pregnancy risk increased significantly, GnRH agonist
administration may be a better option for LPS with single embryo
transfer. Therefore, GnRH agonist treatment may be an ideal choice
for LPS in patients undergoing IVF/ICSI. However, further RCTs or
multi-center randomized controlled studies are required prior to
clinical application of GnRH agonist.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Natural
Science Foundation of Ningxia (grant no. NZ17180).
Availability of data and materials
All data generated or analyzed during the present
study are included in this published article.
Authors' contributions
MLS conceived the study and was a major contributor
in writing the manuscript. CLL participated in the design and
coordination of the study and helped to draft the manuscript. RH
performed the statistical analysis. FMW and ZHH contributed to the
collection, analysis of data and edited the manuscript. All authors
have read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Tavaniotou A, Albano C, Smitz J and
Devroey P: Impact ofovarian stimulation on corpus luteum function
and embryonicimplantation. J Reprod Immunol. 55:123–130. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pritts EA and Atwood AK: Luteal phase
support in infertilitytreatment: A meta-analysis of the randomized
trials. Hum Reprod. 17:2287–2299. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
van der Linden M, Buckingham K, Farquhar
C, Kremer JA and Metwally M: Luteal phase support for assisted
reproduction cycles. Cochrane Database Syst Rev.
CD0091542015.PubMed/NCBI
|
|
4
|
Fatemi HM: The luteal phase after 3
decades of IVF: What do we know? Reprod Biomed Online. 19 (Suppl
4):S43312009. View Article : Google Scholar
|
|
5
|
Devroey P, Bourgain C, Macklon NS and
Fauser BC: Reproductive biology and IVF: Ovarian stimulationand
endometrial receptivity. Trends Endocrinol Metab. 15:84–90. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gizzo S, Andrisani A, Esposito F, Noventa
M, Di Gangi S, Angioni S, Litta P, Gangemi M and Nardelli GB: Which
luteal phase support is better for each IVF stimulation protocol to
achieve the highest pregnancy rate? A superiority randomized
clinical trial. Gynecol Endocrinol. 30:902–908. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fujii S, Sato S, Fukui A, Kimura H, Kasai
G and Saito Y: Continuous administration of gonadotrophin-releasing
hormone agonist during the luteal phase in IVF. Hum Reprod.
16:1671–1675. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tesarik J, Hazout A and Mendoza C:
Enhancement of embryo developmental potential by a single
administration of GnRH agonist at the time of implantation. Hum
Reprod. 19:1176–1180. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Casan EM, Raga F and Polan ML: GnRH mRNA
and protein expression in human preimplantation embryos. Mol Hum
Reprod. 5:234–239. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Nam DH, Lee SH, Kim HS, Lee GS, Jeong YW,
Kim S, Kim JH, Kang SK, Lee BC and Hwang WS: The role of
gonadotropin-releasing hormone (GnRH) and its receptor in
development of porcine preimplantation embryos derived from in
vitro fertilization. Theriogenology. 63:190–201. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Raga F, Casan EM, Kruessel J, Wen Y,
Bonilla-Musoles F and Polan ML: The role of gonadotropin- releasing
hormone in murine preimplantation embryonic development.
Endocrinology. 140:3705–3712. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kyrou D, Kolibianakis EM, Fatemi HM,
Tarlatzi TB, Devroey P and Tarlatzis BC: Increased live birth rates
with GnRH agonist addition for luteal support in ICSI/IVF cycles: A
systematic review and meta-analysis. Hum Reprod Update. 17:734–740.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Oliveira JB, Baruffi R, Petersen CG, Mauri
AL, Cavagna M and Franco JG Jr: Administration of single-dose GnRH
agonist in the luteal phase in ICSI cycles: A meta-analysis. Reprod
Biol Endocrinol. 8:1072010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yu LP, Liu N and Liu Y: Effect of
luteal-phase gonadotropin-releasing hormone agonist administration
on pregnancy outcome in IVF/ICSI cycles: A systematic review and
Meta-analysis. Zhonghua Fu Chan Ke Za Zhi. 51:850–858. 2016.(In
Chinese). PubMed/NCBI
|
|
15
|
Zhou W, Zhuang Y, Pan Y and Xia F: Effects
and safety of GnRH-a as a luteal support in women undertaking
assisted reproductive technology procedures: Follow-up results for
pregnancy, delivery, and neonates. Arch Gynecol Obstet.
295:1269–1275. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Martins WP, Ferriani RA, Navarro PA and
Nastri CO: GnRH agonist during luteal phase for women undergoing
assisted reproductive techniques: Systematic review and
meta-analysis of randomized controlled trials. Ultrasound Obstet
Gynecol. 47:144–151. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gordon A, Raynes-Greenow C, McGeechan K,
Morris J and Jeffery H: Risk factors for antepartum stillbirth and
the influence of maternal age in New South Wales Australia: A
population based study. BMC Pregnancy Childbirth. 13:122013.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Reddy UM, Laughon SK, Sun L, Troendle J,
Willinger M and Zhang J: Prepregnancy risk factors for antepartum
stillbirth in the United States. Obstet Gynecol. 116:1119–1126.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tesarik J, Hazout A, Mendoza-Tesarik R,
Mendoza N and Mendoza C: Beneficial effect of luteal-phase GnRH
agonist administration on embryo implantation after ICSI in both
GnRH agonist- and antagonist-treated ovarian stimulation cycles.
Hum Reprod. 21:2572–2579. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ata B, Yakin K, Balaban B and Urman B:
GnRH agonist protocol administration in the luteal phase in ICSI-ET
cycles stimulated with the long GnRH agonist protocol: A
randomized, controlled double blind study. Hum Reprod. 23:668–673.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Isik AZ, Caglar GS, Sozen E, Akarsu C,
Tuncay G, Ozbicer T and Vicdan K: Single-dose GnRH agonist
administration in the luteal phase of GnRH antagonist cycles: A
prospective randomized study. Reprod Biomed Online. 19:472–477.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Razieh DF, Maryam AR and Nasim T:
Beneficial effect of luteal-phase gonadotropin-releasing hormone
agonist administration on implantation rate after intracytoplasmic
sperm injection. Taiwan J Obstet Gynecol. 48:245–248. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ata B and Urman B: Single dose GnRH
agonist administration in the luteal phase of assisted reproduction
cycles: Is the effect dependent on the type of GnRH analogue used
for pituitary suppression? Reprod Biomed Online. 20:165–166. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yildiz GA, Şükür YE, Ateş C and Aytaç R:
The addition of gonadotrophin releasing hormone agonist to routine
luteal phase support in intracytoplasmic sperm injection and embryo
transfer cycles: A randomized clinical trial. Eur J Obstet Gynecol
Reprod Biol. 182:66–70. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zafardoust S, Jeddi-Tehrani M, Akhondi MM,
Sadeghi MR, Kamali K, Mokhtar S, Badehnoosh B, Arjmand-Teymouri F,
Fatemi F and Mohammadzadeh A: Effect of administration of single
dose GnRH agonist in luteal phase on outcome of ICSI-ET cycles in
women with previous history of IVF/ICSI failure: A randomized
controlled trial. J Reprod Infertili. 16:116–120. 2015.
|
|
26
|
Benmachiche A, Benbouhedja S, Zoghmar A,
Boularak A and Humaidan P: Impact of mid-luteal phase GnRH agonist
administration on reproductive outcomes in GnRH agonist-triggered
cycles: A randomized controlled trial. Front Endocrinol (Lausanne).
8:1242017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wilshire GB, Emmi AM, Gagliardi CC and
Weiss G: Gonadotropin-releasing hormone agonist administration in
early human pregnancy is associated with normal outcomes. Fertil
Steril. 60:980–983. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Pirard C, Loumaye E, Laurent P and Wyns C:
Contribution to more patient-friendly ART treatment: Efficacy of
continuous low-dose GnRH agonist as the only luteal support-results
of a prospective, randomized, comparative study. Int J Endocrinol.
2015:7275692015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Aboulghar MA, Marie H, Amin YM, Aboulghar
MM, Nasr A, Serour GI and Mansour RT: GnRH agonist plus vaginal
progesterone for luteal phase support in ICSI cycles: A randomized
study. Reprod Biomed Online. 30:52–56. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chillik CF, Itskovitz J, Hahn DW, McGuire
GL, Danforth DR and Hodgen GD: Characterizing pituitary response to
a gonadotropin-releasing hormone (GnRH) antagonist in monkeys:
Tonic follicle-stimulating hormone/luteinizing hormone secretion
versus acute GnRH challenge tests before, during, and after
treatment. Fertil Steril. 48:480–485. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gordon K, Williams RF, Danforth DR and
Hodgen GD: A novel regimen of gonadotropin-releasing hormone (GnRH)
antagonist plus pulsatile GnRH: Controlled restoration of
gonadotropin secretion and ovulation induction. Fertil Steril.
54:1140–1145. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Felberbaum RE, Reissmann T, Küpker W,
Bauera O, al Hasania S, Diedrich C and Diedricha K: Preserved
pituitary response under ovarian stimulation with HMG and GnRH
antagonists (Cetrorelix) in women with tubal infertility. Eur J
Obstet Gynecol Reprod Biol. 61:151–155. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Iwashita M, Kudo Y, Shinozaki Y and Takeda
Y: Gonadotropin-releasing hormone increases serum human chorionic
gonadotropin in pregnant women. Endocr J. 40:539–544. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lin LS, Roberts VJ and Yen SS: Expression
of human gonadotropin-releasing hormone receptor gene in the
placenta and its functional relationship to human chorionic
gonadotropin secretion. J Clin Endocrinol Metab. 80:580–585. 1995.
View Article : Google Scholar : PubMed/NCBI
|