Introduction
Chronic hepatitis B (CHB) is a major public health
concern with a global prevalence. Approximately 65,000 patients die
of liver failure, liver cirrhosis and hepatocellular carcinoma
caused by hepatitis B virus (HBV) infection each year (1). Significant liver fibrosis and cirrhosis
cause severe liver injury in patients with CHB and are important
indicators for anti-viral therapy in those patients. At present,
liver biopsy is considered the gold standard for staging of the
degree of liver injury, but it is not widely used due to its
invasiveness and risk of complications. Even if several
non-invasive tests are applied for evaluating liver injury, in
general, the early diagnosis of cirrhosis still depends on liver
biopsy, as these non-invasive indicators are currently not
ideal.
Platelets contain numerous growth factors that are
required for organ development, tissue regeneration and repair
(2,3). It was reported that in chronic liver
diseases, platelets participate not only in liver injury through T
cell-mediated immune responses but also in tissue repair through
secretion of growth factors (4,5).
Although thrombocytopenia is one of the common complications in
patients with chronic liver disease in the clinic, there is no
available data supporting that the degree of liver injury in
patients with chronic HBV infection or cirrhosis associated with
HBV may be evaluated based on the platelet count. Only several
studies have suggested that the etiology of thrombocytopenia in
liver disease may be attributed to splenomegaly, hypersplenism,
portal hypertension and decreased thrombopoietin production
(6,7). A recent study reported that in patients
with CHB with significant fibrosis receiving anti-viral therapy,
the increase of platelets was associated with the improvement of
liver fibrosis (8). For hepatitis C,
a clinical study has suggested that the platelet count may predict
significant hepatitis C-associated fibrosis (9), and recently, the platelet count was
reported to be able to indicate the degree of liver steatosis and
fibrosis (10,11), and may serve as a clue regarding the
severity of disease in patients with non-alcoholic fatty liver
disease (12).
Overall, the association between thrombocytopenia
and hepatic pathogenesis remains obscure and the role of platelets
in chronic liver disease has remained largely elusive. There are
currently no available data to support that the platelet count is
able to evaluate the severity of liver injury in patients with
chronic HBV infection. Using the results of liver biopsy as the
gold standard, the present cross-sectional study aimed to evaluate
the role of the platelet count in estimating liver injury in
patients with chronic HBV infection.
Materials and methods
Patients
In the present study, 1,597 patients with chronic
HBV infection at the First Hospital of Lanzhou University (Lanzhou,
China), who met the inclusion criterion of positive serum HBV
surface antigen (HBsAg) for >6 months, were enrolled between
June 2016 and August 2017. The exclusion criteria were as follows:
HBsAg-negative status; age <18 years; co-infection with
hepatitis A, C and/or D virus; autoimmune hepatitis; alcoholic
liver disease; non-alcoholic fatty liver disease; Wilson's disease;
primary biliary cholangitis; drug-induced liver injury; co-existent
hematologic diseases; history of splenectomy; history of therapy
with anti-platelet drugs, immune inhibitors, molecular targeted
drugs and hepatotoxic drugs; incomplete data; liver
transplantation; extrahepatic tumor; multiple organ failure and
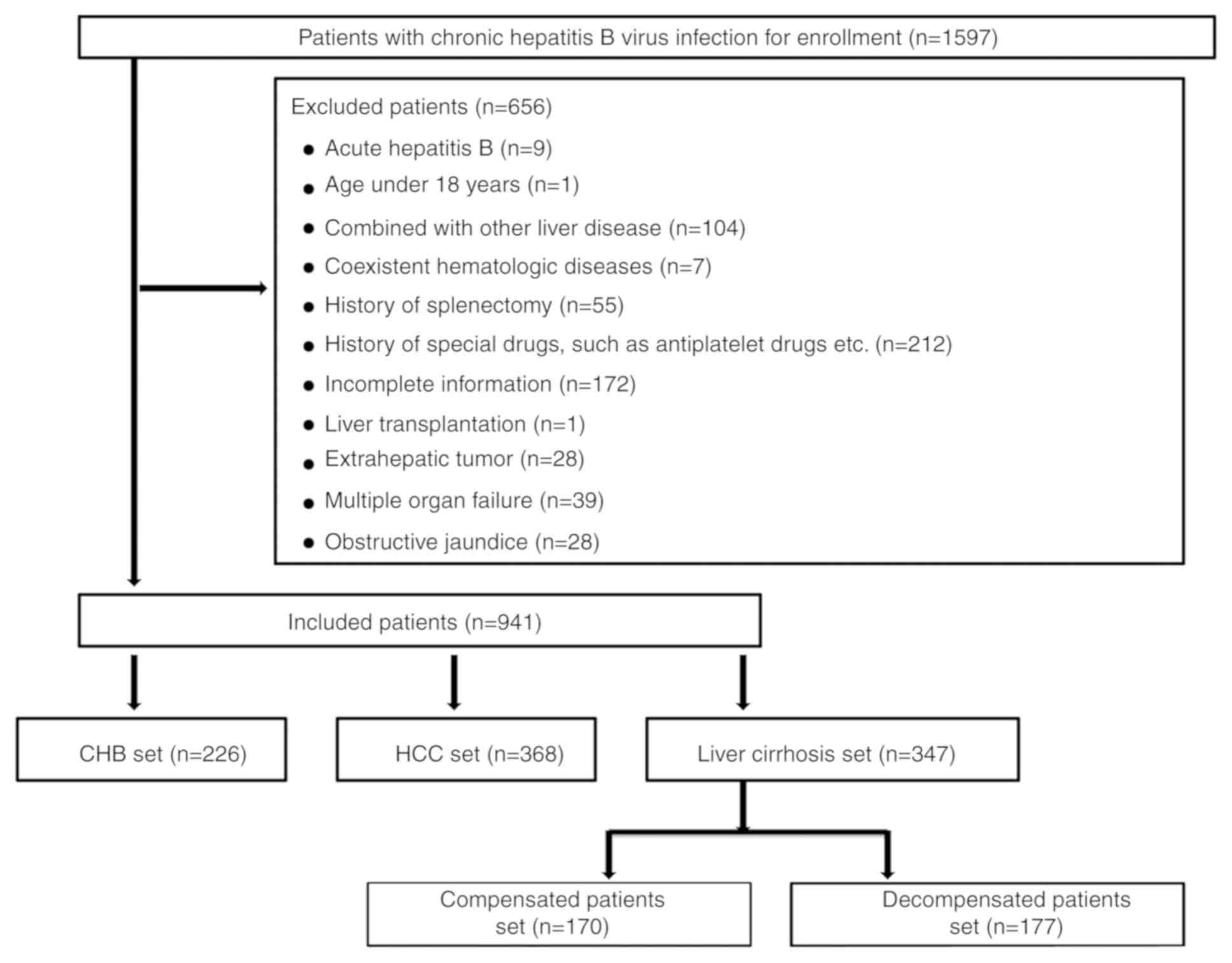
obstructive jaundice. In total, 941 patients were finally included,
of whom 347 and 226 patients were diagnosed with cirrhosis and CHB,
respectively, based on the diagnostic criteria (1,13).
Diagnostic data were obtained from clinical, imaging examination or
liver biopsy results. Cirrhosis was classified as the compensated
(n=170) or decompensated stage (n=177). This classification depends
on the presence or absence of clinically evident decompensating
events (specifically ascites, variceal hemorrhage and
encephalopathy) (13). A flow chart
of the patients included in the study is provided in Fig. 1.
This study was approved by the Ethics Committee of
the First Hospital of Lanzhou University (Lanzhou, China; no.
LDYYLL2019-209).
Laboratory tests and imaging
examination
All patients included had complete biochemical,
routine blood and coagulation test results, which were respectively
detected by an automatic biochemical analyzer (AU400; Olympos
Corp.), an automatic blood cell analyzer (BC-5390CRP; Mindray) and
an automatic coagulation Analyzer (Precil C3510; Beijing Precil
Instrument Co., Ltd.). The serum levels of alanine aminotransferase
(ALT) and aspartate aminotransferase (AST) >40 IU/l were
considered abnormal. Splenic thickness, portal vein diameter and
portal vein velocity were measured on an Ultrasound Machine (GE
Logiq E9; GE Healthcare) by an experienced ultrasonographist.
Pathological assessment and indices
calculated
Liver pathological assessment was based on the
METAVIR scoring system (14). In the
present study, F4 was defined as cirrhosis. The formulas for
calculating the platelet count to splenic thickness ratio (PC/ST
ratio), fibrosis index based on four factors (FIB-4) and
AST-to-platelet ratio index (APRI) were as follows (15,16):
Platelets/splenic thickness (PC/ST) ratio=platelet count
(109/l)/splenic thickness (mm); APRI=AST (IU/l)/its
upper limit of normal (ULN)/platelet count (109/l) x100.
The ULN for AST was 40 IU/l. FIB-4=age (years) x AST/platelet count
x [ALT (IU/l)]1/2.
Statistical analysis
Epidata Software (version 3.1; Epidata) was used to
manage data. SPSS software (version 17.0; SPSS, Inc.) was used to
perform statistical analysis. Continuous variables are expressed as
the mean ± standard deviation and categorical variables as n (%).
One-sample Kolmogorov-Smirnov test was applied to determine if a
set of data was normally distributed. For intergroup comparisons of
continuous variables, variables with a normal and non-normal
distribution were assessed by the independent-samples t-test and
the non-parametric U Mann-Whitney test, respectively. The
χ2 test was used for categorical variables. To assess
the association between variables and the Child-Turcotte-Pugh (CTP)
score (17) or pathological stage,
Spearman's rank correlation coefficient was determined.
Multivariate logistic regression analysis was performed to identify
the independent factors predicting liver function impairment.
Finally, receiver operating characteristic (ROC) curve analysis was
used to evaluate the diagnostic performance of the platelet count.
P<0.05 was considered to indicate statistical significance.
Results
Patient characteristics
The present study included 941 patients with a mean
age of 50.29±10.93 years (range, 18-90 years). The study population
comprised 657 (69.82%) male patients and 284 (30.18%) female
patients. These patients were stratified into a CTP class A group
and a CTP class B/C group using the CTP scoring system. The
characteristics of the patients included are summarized in Table SI. Compared with those of CTP class
A, those with CTP class B/C had a lower age (P=0.009), platelet
count (P<0.001), albumin (P<0.001) and prothrombin activity
(PTA; P<0.001), and had a greater splenic thickness (P<0.001)
and portal vein diameter (P<0.001). Similarly, in patients with
normal ALT levels, age, platelet count, albumin and PTA were
significantly lower, whilst splenic thickness and portal vein
diameter were significantly higher in patients with CTP class B/C
vs. A (Table I).
 | Table IBaseline characteristics of the study
population of patients of chronic hepatitis B virus infection with
normal ALT. |
Table I
Baseline characteristics of the study
population of patients of chronic hepatitis B virus infection with
normal ALT.
| Variable | CTP class A
(n=395) | CTP class B/C
(n=137) | P-value |
|---|
| Age (years) | 51.61±11.19 | 49.22±11.59 | 0.028 |
| Male sex | 250 (63.29) | 84 (61.31) | 0.757 |
| Platelet count
(x109/l) | 124.76±67.60 | 64.18±44.70 | <0.001 |
| PC/ST ratio | 172,2.80±2.15 | 92,1.12±0.94 | <0.001 |
| AST (U/l) | 33.21±16.77 | 49.68±49.11 | <0.001 |
| TP (g/l) | 69.91±7.56 | 62.08±10.20 | <0.001 |
| Albumin (g/l) | 43.24±4.28 | 34.30±6.20 | <0.001 |
| TBIL (µmol/l) | 20.10±8.64 | 46.54±34.41 | <0.001 |
| DBIL (µmol/l) | 4.66±2.15 | 15.09±15.48 | <0.001 |
| CHE (KU/l) | 6.19±1.88 | 3.28±1.56 | <0.001 |
| TC (mmol/l) | 3.78±0.92 | 2.55±0.95 | <0.001 |
| PT (sec) | 12.41±1.17 | 16.42±4.38 | <0.001 |
| PTA (%) | 86.32±14.57 | 59.29±14.67 | <0.001 |
| APTT (sec) | 33.86±5.58 | 38.67±15.65 | <0.001 |
| Splenic thickness
(n, mm) |
172,45.09±11.82 | 92,57.49±10.78 | <0.001 |
| Portal vein
diameter (n, mm) | 211,11.71±1.95 | 90,13.48±2.30 | <0.001 |
| FIB-4 | 4.14±4.75 | 9.63±7.43 | <0.001 |
| APRI | 1.02±1.31 | 2.56±2.00 | <0.001 |
Platelet count is associated with the
severity of liver function in patients with chronic HBV
infection
In terms of liver function, correlation analysis
revealed that the CTP score was inversely correlated with the
platelet count, albumin, cholinesterase (CHE) and PTA (Table SII). In addition, the platelet count
correlated positively with albumin (r=0.350, P<0.001), CHE
(r=0.500, P<0.001) and PTA (r=0.503, P<0.001), and correlated
inversely with prothrombin time (PT; r=-0.522, P<0.001; Table SIII). Multivariate logistic
regression analysis verified that the platelet count [odds ratio
(OR), 0.994; P=0.009], albumin (OR, 0.703; P<0.001), total
bilirubin (OR, 1.146; P<0.001) and age (OR, 0.971; P=0.010) were
independently associated with CTP class B/C (Table SIV). Furthermore, in patients with
normal ALT levels, the platelet count, albumin and PTA were also
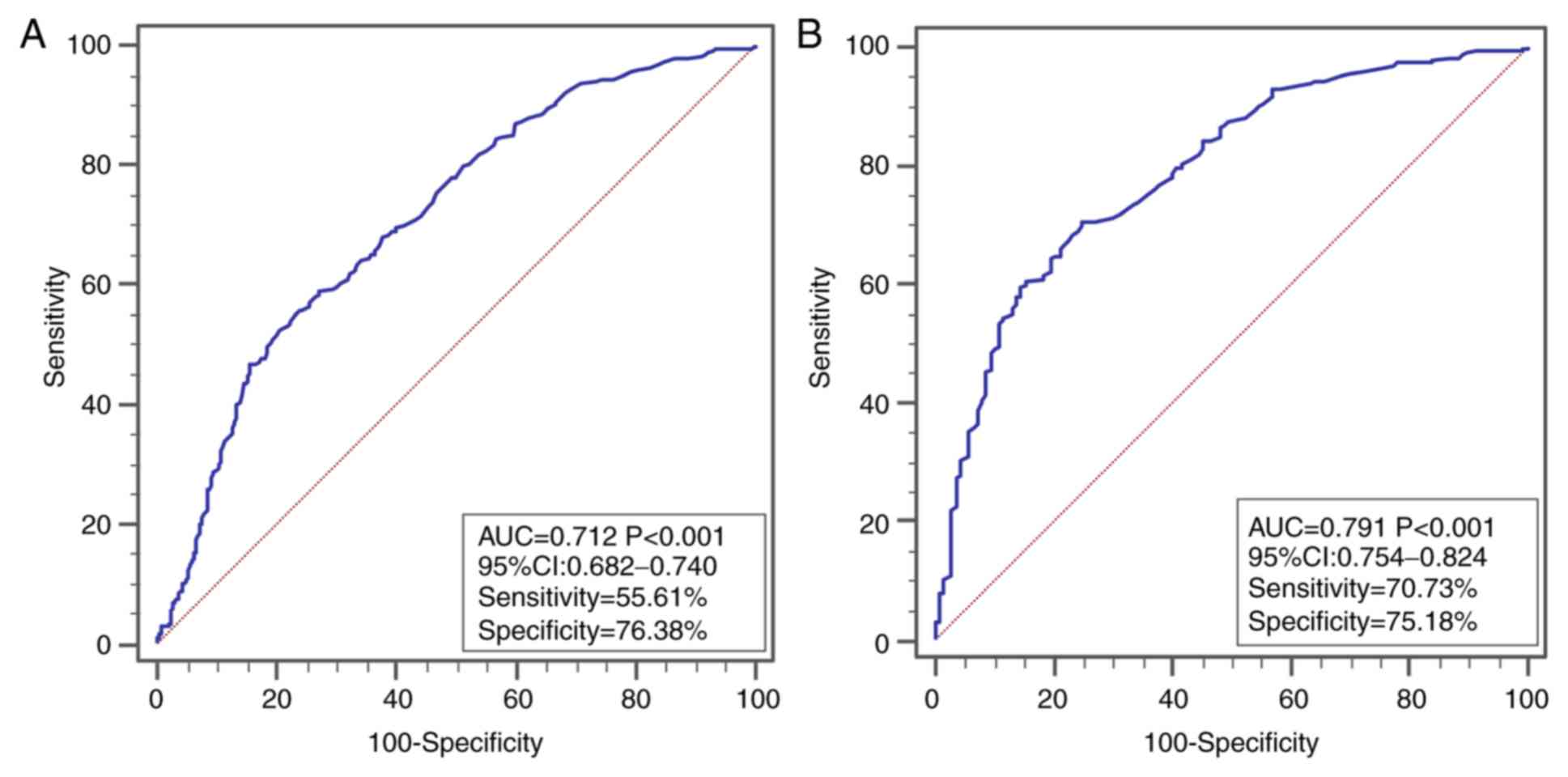
independently associated with CTP class B/C (Table II). ROC curve analysis indicated
that the area under the curve (AUC) of the platelet count to
distinguish CTP class B/C from A were 0.712 (Fig. 2A) and 0.791 (Fig. 2B) in patients with chronic HBV
infection and normal ALT levels, respectively.
 | Table IIIndependently associated indicators
of moderate to severe liver function impairment in patients with
normal alanine aminotransferase obtained by multivariate analysis
of the indicators with statistical differences in Table I. |
Table II
Independently associated indicators
of moderate to severe liver function impairment in patients with
normal alanine aminotransferase obtained by multivariate analysis
of the indicators with statistical differences in Table I.
| Variable | OR | 95% CI | P-value |
|---|
| Platelet count | 0.991 | 0.985-0.998 | 0.007 |
| Albumin | 0.773 | 0.719-0.831 | <0.001 |
| PTA | 0.902 | 0.875-0.931 | <0.001 |
| Age | 0.973 | 0.946-1.002 | 0.067 |
The platelet count is able to identify
patients with cirrhosis among those with chronic HBV infection
Among the patients with chronic HBV infection, those
with cirrhosis had a significantly lower age, platelet count,
albumin and PTA, and significantly higher PT, splenic thickness,
portal vein diameter, CTP score, APRI and FIB-4 than those with no
cirrhosis (Table SV). Correlation
analysis demonstrated that the platelet count was inversely
correlated with FIB-4 (r=-0.855, P<0.001) and APRI (r=-0.741,
P<0.001), whereas splenic thickness and CTP scores correlated
positively with FIB-4 (Table SVI).
These three indicators and age were then included in a multivariate
analysis. The results suggested that the platelet count, CTP score
and splenic thickness were independently associated with cirrhosis
(Table SVII). The ROC curve
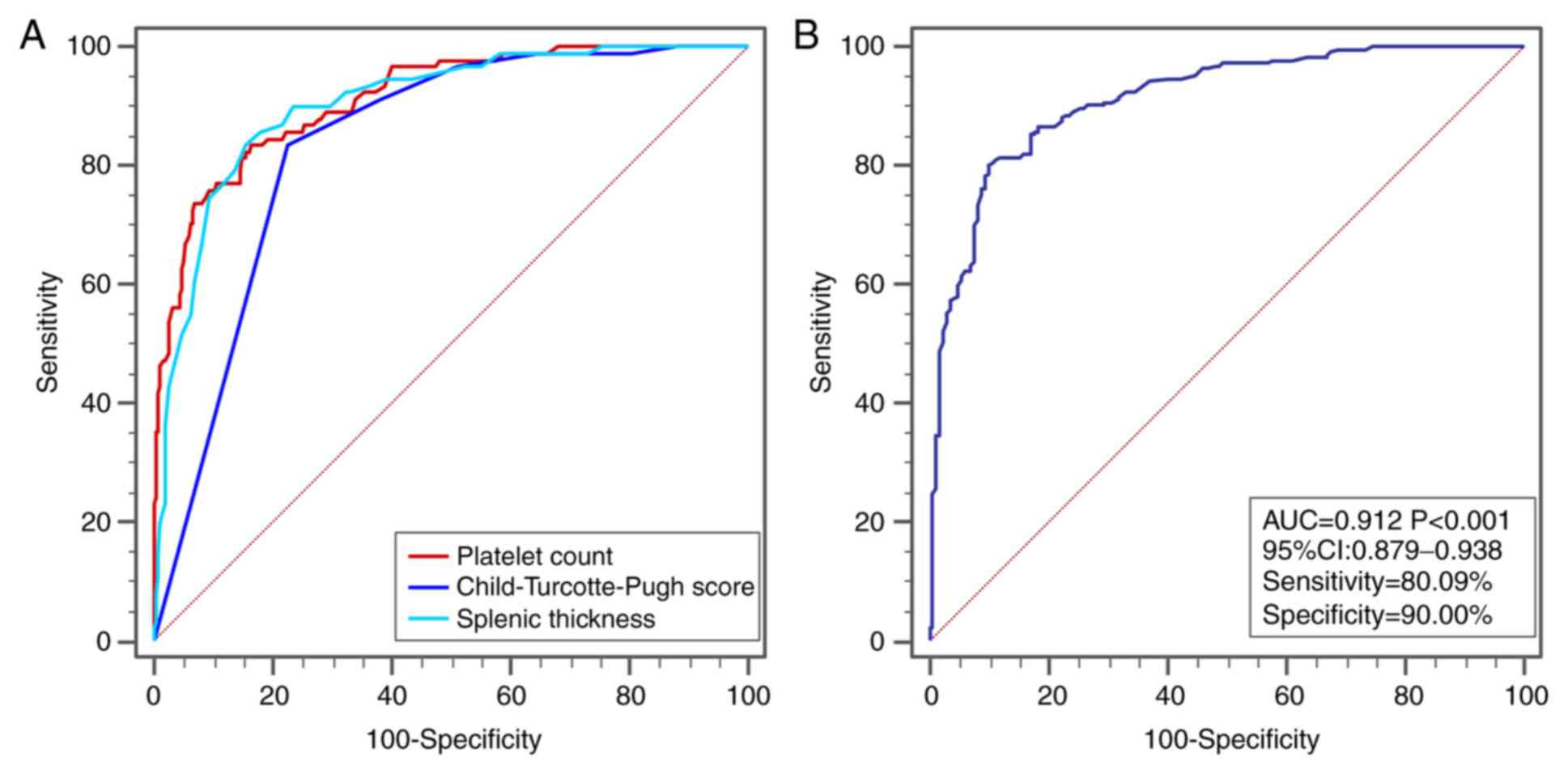
analysis indicated that the AUC of the platelet count for cirrhosis
was 0.927 (P<0.001), the AUC of splenic thickness for cirrhosis
was 0.903 (P<0.001), the AUC of CTP score for cirrhosis was
0.838 (P<0.001; Fig. 3A and
Table SVIII).
The platelet count has potential
diagnostic value for early-stage cirrhosis
The stage of CHB and compensated cirrhosis are
indistinguishable if no invasive examination is performed. In the
present study, patients with compensated cirrhosis, compared to
those with CHB, also had a lower age, platelet count and albumin,
and higher splenic thickness, APRI, FIB-4 and CTP score
(P<0.001). The differences in these groups are provided in the
supplementary material (Table SIX).
Logistic regression analysis indicated that the platelet count was
also independently correlated with compensated cirrhosis (OR,
0.966; P<0.001; Table SX). The
AUC of the platelet count for compensated cirrhosis was 0.912 and
the optimal cut-off value was 117x109/l, with a
sensitivity of 80.09% and a specificity of 90.00% (Fig. 3B).
Platelet count is closely associated
with histological severity in patients with chronic HBV
infection
A total of 53 patients included underwent liver
biopsy and were stratified into the ≤F3 set (n=6) and F4 set (n=47)
according to the METAVIR scoring system. The study population with
a mean age of 50.15±8.55 years comprised 40 (75.47%) male patients
and 13 (24.53%) female patients. The baseline characteristics of
these patients are provided in Table
III. The patients with F4 had a significantly lower platelet
count, albumin, CHE and PTA (P<0.05), and correlation analysis
suggested that these parameters were significantly negatively
correlated with all pathological stages (Table IV). The multivariate logistic
regression analysis revealed that only the platelet count (OR,
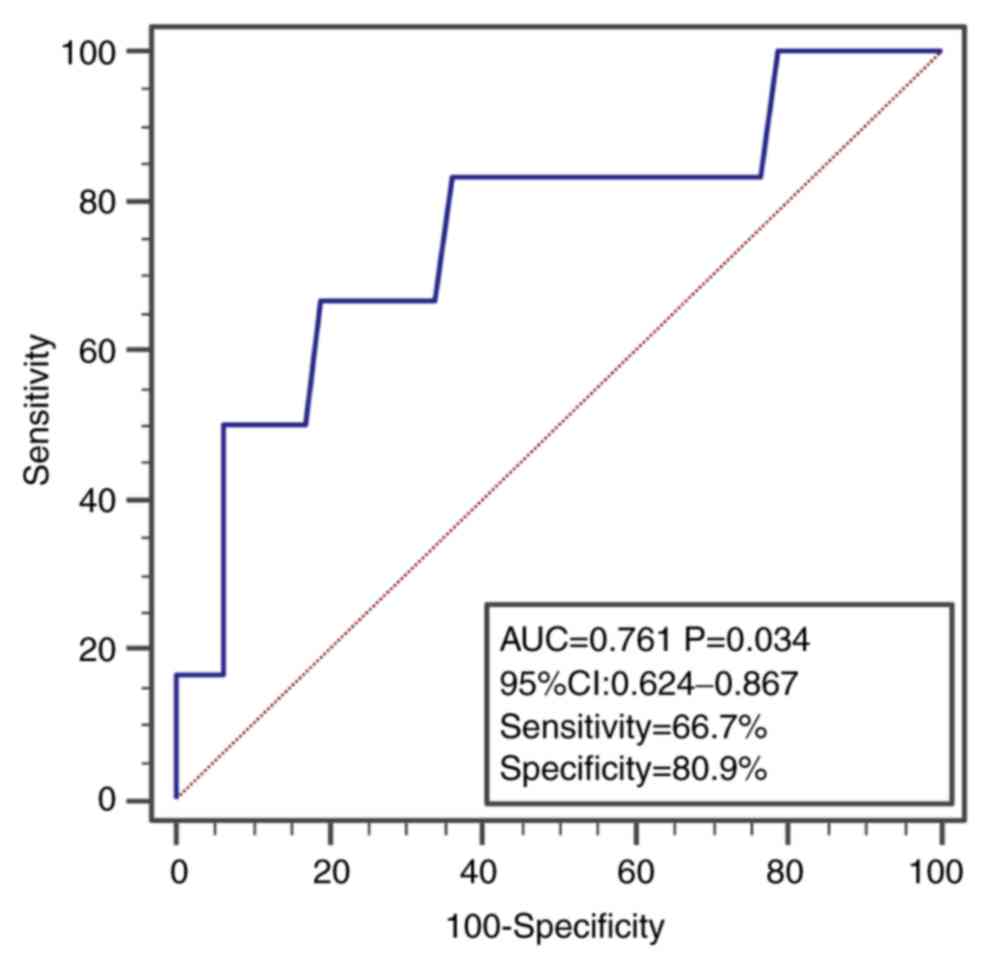
0.978; P=0.026) was independently associated with F4 (Table V). The AUC of the platelet count to
distinguish F4 from ≤F3 was 0.761 with a sensitivity of 66.7% and
specificity of 80.9% (Fig. 4).
 | Table IIIBaseline characteristics of patients
who underwent liver biopsy. |
Table III
Baseline characteristics of patients
who underwent liver biopsy.
| Variable | ≤F3 (n=6) | F4 (n=47) | P-value |
|---|
| Age (years) | 47.83±5.15 | 50.45±8.88 | 0.486 |
| Male sex | 5 (83.33) | 35 (74.47) | 0.635 |
| Platelet count
(x109/l) | 152.83±58.78 | 103.15±42.97 | 0.013 |
| AST (U/l) | 40.67±22.93 | 44.11±26.71 | 0.764 |
| ALT (U/l) | 59.50±39.07 | 41.15±23.94 | 0.107 |
| TP (g/l) | 74.66±5.08 | 70.10±7.62 | 0.162 |
| Albumin (g/l) | 47.10±4.52 | 42.88±4.26 | 0.027 |
| TBIL (µmol/l) | 20.08±8.69 | 21.32±12.17 | 0.902 |
| DBIL (µmol/l) | 3.77±1.56 | 5.87±7.41 | 0.207 |
| IBIL (µmol/l) | 16.32±7.16 | 15.45±6.41 | 0.758 |
| CHE (KU/l) | 8.05±1.22 | 5.85±1.55 | 0.002 |
| TC (mmol/l) | 4.40±0.73 | 3.58±0.76 | 0.016 |
| PT (sec) | 11.53±1.18 | 12.91±1.62 | 0.049 |
| PTA (%) | 99.18±19.54 | 81.87±13.43 | 0.007 |
| APTT (sec) | 34.22±6.70 | 33.69±5.91 | 0.840 |
| Splenic thickness
(n, mm) | 2,38.50±7.78 | 19,47.47±8.00 | 0.147 |
| Portal vein
diameter (n, mm) | 5,10.80±1.48 | 24,11.79±1.32 | 0.145 |
 | Table IVCorrelation between variables and the
pathological stage. |
Table IV
Correlation between variables and the
pathological stage.
| Variable | r-value | P-value |
|---|
| Platelet count | -0.295 | 0.032 |
| Albumin | -0.279 | 0.043 |
| CHE | -0.392 | 0.004 |
| PT | 0.334 | 0.014 |
| PTA | -0.328 | 0.016 |
 | Table VIndependently associated indicators
for cirrhosis (pathological stage IV) obtained by multivariate
analysis of the indicators with statistical differences in Table III. |
Table V
Independently associated indicators
for cirrhosis (pathological stage IV) obtained by multivariate
analysis of the indicators with statistical differences in Table III.
| Variable | OR | 95% CI | P-value |
|---|
| Platelet count | 0.978 | 0.960-0.997 | 0.026 |
| ALT | 0.980 | 0.950-1.010 | 0.170 |
| Age | 1.034 | 0.913-1.171 | 0.425 |
Discussion
Several clinical studies indicated that the platelet
count is able to distinguish significant fibrosis in patients with
hepatitis C infection and may be used to assess the degree of liver
steatosis and fibrosis in patients with non-alcoholic fatty liver
disease (11,18). Furthermore, it has been indicated
that the platelet count is able to modulate the quantitative
association between HBV DNA and surface antigen concentrations
(19), and HBsAg levels were able to
predict the degree of fibrosis in patients with CHB (20). Therefore, the platelet count is
closely linked to the HBV infection status. Furthermore, a study
reported that the platelet count was associated with inflammation
and fibrosis of the liver in patients with chronic HBV infection
(21). However, at present, only a
small amount of evidence is available to support the association of
the platelet count with the severity of liver injury in patients
with chronic HBV infection. In the present study, a cohort of
patients with chronic HBV infection that met the study requirements
based on rigorous criteria were ultimately included. Based on the
above standards and the liver biopsy data, the ability of the
platelet count to evaluate chronic liver injury in patients with
HBV infection was confirmed. The results suggested that the
platelet count was closely linked to the severity of liver injury
in patients with chronic HBV infection.
Over the past decades, certain studies have
attempted to demonstrate that certain non-invasive indicators are
able to distinguish liver fibrosis, liver inflammation or liver
function (22,23). Furthermore, the platelet count was
able to indicate the degree of fibrosis in non-alcoholic fatty
liver disease (10,11), and the platelet count was associated
with the degree of hepatic fibrosis in chronic hepatitis C via the
mRNA expression of platelet-derived growth factor A (24). An animal study also suggested that
platelets produce platelet-derived growth factor-b to activate
hepatic stellate cells and promote fibrosis (25). Thus, these studies demonstrated that
the platelet count was closely linked to liver function and liver
fibrosis.
In the present study, the platelet count was
significantly lower in patients with chronic HBV infection and
CTP-B/C, and inversely correlated with the CTP score. In
particular, the platelet count was found to be independently
associated with CTP-B/C and used to distinguish CTP-B/C with an AUC
of 0.791, optimal cut-off value of 73x109/l, sensitivity
of 70.73% and specificity of 75.18% in patients with normal ALT
levels. Therefore, platelets were confirmed to be linked to the
severity of liver function in patients with chronic HBV infection.
Another study suggested that the increase of platelets was
associated with the improvement of liver fibrosis in patients with
CHB with significant fibrosis receiving anti-viral therapy
(8). This also indirectly indicated
the close association between platelets and fibrosis in patients
with hepatitis B.
The stage of compensated cirrhosis in CHB is
difficult to distinguish without performing an invasive
examination. Furthermore, compensated cirrhosis is associated with
an increased risk of developing varices, overt clinical
decompensation (ascites, variceal hemorrhage, hepatic
encephalopathy), post-surgical decompensation and hepatocellular
carcinoma (26). Therefore, the
present study focused on the association between platelets and
cirrhosis, particularly compensated cirrhosis. The results
indicated that the platelet count was inversely correlated with
FIB-4 and APRI, which are a non-invasive diagnostic model for
hepatic fibrosis. The platelet count was independently associated
with cirrhosis. The AUC for the platelet count to indicate
cirrhosis (0.927) was significantly higher than that for splenic
thickness and CTP score. For early-stage cirrhosis, the platelet
count had the best discriminative ability in distinguishing
compensated cirrhosis. The AUC was 0.912 with an optimal cut-off
value of 117x109/l, sensitivity of 80.09% and
specificity of 90.00%. Therefore, it may be speculated that
patients with chronic HBV infection with platelet counts of
<117x109/l were likely to already have early-stage
cirrhosis based on the results.
In addition, the liver biopsy results were used to
confirm the above-mentioned association. The results indicated that
the platelet count was inversely correlated with the histological
stage of fibrosis and independently associated with F4. ROC curve
analysis demonstrated that the platelet count is an effective
diagnostic indicator for the F4 stage with an AUC of 0.761.
Therefore, the present study further confirmed that the platelet
count is closely associated with histological severity in patients
with chronic HBV infection.
Of note, the present study had certain limitations.
First, no basic experiments were performed to confirm the relevant
mechanisms. Additionally, the predictive value of the platelet
count to assess the severity of liver injury was not proven.
However, based on the rigorous research design and associated
results, it was possible to draw conclusions.
In conclusion, the present study confirmed for the
first time that the platelet count is independently associated with
moderate to severe liver function impairment and cirrhosis in
patients with chronic HBV infection. The platelet count may be used
to determine the severity of liver injury and liver fibrosis.
Supplementary Material
Baseline characteristics of the study
population in patients of chronic hepatitis B virus infection.
Correlation between variables and
Child-Turcotte-Pugh score in patients with chronic hepatitis B
virus infection.
Correlation between platelet count and
variables associated with liver function.
Independently associated indicators
for moderate to severe liver function impairment in patients with
chronic hepatitis B virus infection obtained by multivariate
analysis of the indicators with statistical differences in Table SI.
Characteristics of patients stratified
into CHB set and cirrhosis set.
Association among variables and FIB-4,
APRI and splenic thickness.
Independently associated indicators of
cirrhosis obtained by multivariate analysis of the indicators with
statistical differences in Table
SV.
Diagnostic performance of platelet
count and other variables for cirrhosis in chronic hepatitis B
virus infection.
Characteristics of patients stratified
into CHB set and compensated cirrhosis set.
Independently associated indicators of
compensated cirrhosis obtained by multivariate analysis of the
indicators with statistical differences in Table SIX.
Acknowledgements
Not applicable.
Funding
The present study was financially supported by the
National Natural Science Foundation (grant no. 81800528), the
Hospital Fund from the First Hospital of Lanzhou University (grant
no. ldyyyn2017-17), the Gansu Health Industry Research Project
(grant no. GSWSKY2018-24), the Science and Technology Development
Project of Chengguan District (grant no. 2018SHFZ0023), the
National Science and Technology Key Project (grant nos.
2017ZX10201201, 2017ZX10203201-005, 2017ZX10202203-006-001 and
2017ZX10302201-004-002) and the Beijing Municipal Administration of
Hospital's Ascent Plan (grant no. DFL20151601).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
JFL and ZPD designed the study; YTY, LTY, LLW, WZ
and YC collated the data; YTY, LTZ, SJZ and QFC analyzed the data;
YTY, LLW and ZPD wrote the paper; JFL, YC, SJZ and ZPD revised the
paper. All authors approved the final version of the
manuscript.
Ethics approval and consent to
participate
The current study meets the ethical requirements of
the Ethics Committee of the First Hospital of Lanzhou University
(Lanzhou, China) and received approval. Patients were not required
to give informed consent, as the analysis used anonymous clinical
data obtained after each patient agreed to treatment by written
informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
World Health Organization: In: Guidelines
for the prevention, care and treatment of persons with chronic
hepatitis B infection. Geneva, World Health Organization, 2015.
|
|
2
|
Kurokawa T, Zheng YW and Ohkohchi N: Novel
functions of platelets in the liver. J Gastroenterol Hepatol.
31:745–751. 2016.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Minamino T, Ito Y, Ohkubo H, Shimuzu Y,
Kojo K, Nishizwa N, Amano H, Narumiya S, Koizumi W and Majima M:
Adhesion of platelets through thromboxane A(2) receptor signaling
facilitates liver repair during acute chemical-induced
hepatotoxicity. Life Sci. 132:85–92. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Meyer J, Lejmi E, Fontana P, Morel P,
Gonelle-Gispert C and Bühler L: A focus on the role of platelets in
liver regeneration: Do platelet-endothelial cell interactions
initiate the regenerative process? J Hepatol. 63:1263–1271.
2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Nowatari T, Murata S, Fukunaga K and
Ohkohchi N: Role of platelets in chronic liver disease and acute
liver injury. Hepatol Res. 44:165–172. 2014.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Afdhal N, McHutchison J, Brown R, Jacobson
I, Manns M, Poordad F, Weksler B and Esteban R: Thrombocytopenia
associated with chronic liver disease. J Hepatol. 48:1000–1007.
2008.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Peck-Radosavljevic M: Thrombocytopenia in
liver disease. Can J Gastroenterol. 14 (Suppl D):60D–66D.
2000.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Wang L, Wang B, You H, Wu X, Zhou J, Ou X
and Jia J: Platelets' increase is associated with improvement of
liver fibrosis in entecavir-treated chronic hepatitis B patients
with significant liver fibrosis. Hepatol Int. 12:237–243.
2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Myers RP, De Torres M, Imbert-Bismut F,
Ratziu V, Charlotte F and Poynard T: MULTIVIRC Group. Biochemical
markers of fibrosis in patients with chronic hepatitis C: A
comparison with prothrombin time, platelet count, and age-platelet
index. Dig Dis Sci. 48:146–153. 2003.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Milovanovic Alempijevic T, Stojkovic
Lalosevic M, Dumic I, Jocic N, Pavlovic Markovic A, Dragasevic S,
Jovicic I, Lukic S, Popovic D and Milosavljevic T: Diagnostic
accuracy of platelet count and platelet indices in noninvasive
assessment of fibrosis in nonalcoholic fatty liver disease
patients. Can J Gastroenterol Hepatol. 2017(6070135)2017.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Yoneda M, Fujii H, Sumida Y, Hyogo H, Itoh
Y, Ono M, Eguchi Y, Suzuki Y, Aoki N, Kanemasa K, et al: Platelet
count for predicting fibrosis in nonalcoholic fatty liver disease.
J Gastroenterol. 46:1300–1306. 2011.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Garjani A, Safaeiyan A and Khoshbaten M:
Association between platelet count as a noninvasive marker and
ultrasonographic grading in patients with nonalcoholic Fatty liver
disease. Hepat Mon. 15(e24449)2015.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Garcia-Tsao G, Abraldes JG, Berzigotti A
and Bosch J: Portal hypertensive bleeding in cirrhosis: Risk
stratification, diagnosis, and management: 2016 practice guidance
by the American Association for the study of liver diseases.
Hepatology. 65:310–335. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Bedossa P and Poynard T: An algorithm for
the grading of activity in chronic hepatitis C. The METAVIR
cooperative study group. Hepatology. 24:289–293. 1996.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wai CT, Greenson JK, Fontana RJ,
Kalbfleisch JD, Marrero JA, Conjeevaram HS and Lok AS: A simple
noninvasive index can predict both significant fibrosis and
cirrhosis in patients with chronic hepatitis C. Hepatology.
38:518–526. 2003.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Sterling RK, Lissen E, Clumeck N, Sola R,
Correa MC, Montaner J, S Sulkowski M, Torriani FJ, Dieterich DT,
Thomas DL, et al: Development of a simple noninvasive index to
predict significant fibrosis in patients with HIV/HCV coinfection.
Hepatology. 43:1317–1325. 2006.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Samiullah S, Qasim R, Khalid S, Hussain
BG, Mukhtair J and Akbar Y: Evaluation of creatinine-modified child
pugh score for predicting short-term prognosis of patients with
decompensated cirrhosis of liver as compare to original child pugh
score. J Ayub Med Coll Abbottabad. 21:64–67. 2009.PubMed/NCBI
|
|
18
|
Khokhar N: Serum aminotransferase levels
and platelet count as predictive factor of fibrosis and cirrhosis
in patients with chronic hepatitis C infection. J Pak Med Assoc.
53:101–104. 2003.PubMed/NCBI
|
|
19
|
Hsu CW, Liang KH, Lin CL, Wang TH and Yeh
CT: Platelet counts modulate the quantitative relationship between
hepatitis B viral DNA and surface antigen concentrations: A
cross-sectional study of hematological, histological and viral
factors. BMC Infect Dis. 17(9)2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Seto WK, Wong DK, Fung J, Ip PP, Yuen JC,
Hung IF, Lai CL and Yuen MF: High hepatitis B surface antigen
levels predict insignificant fibrosis in hepatitis B e antigen
positive chronic hepatitis B. PLoS One. 7(e43087)2012.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Pan Y, Muheremu A, Wu X and Liu J:
Relationship between platelet parameters and hepatic pathology in
patients with chronic hepatitis B infection-a retrospective cohort
study of 677 patients. J Int Med Res. 44:779–786. 2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Liu DP, Lu W, Zhang ZQ, Wang YB, Ding RR,
Zhou XL, Huang D and Li XF: Comparative evaluation of GPR versus
APRI and FIB-4 in predicting different levels of liver fibrosis of
chronic hepatitis B. J Viral Hepat. 25:581–589. 2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Li Q, Lu C, Li W, Huang Y and Chen L:
Globulin-platelet model predicts significant fibrosis and cirrhosis
in CHB patients with high HBV DNA and mildly elevated alanine
transaminase levels. Clin Exp Med. 18:71–78. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Tanikawa AA, Grotto RM, Silva GF, Ferrasi
AC, Sarnighausen VC and Pardini MI: Platelet-derived growth factor
A mRNA in platelets is associated with the degree of hepatic
fibrosis in chronic hepatitis C. Rev Soc Bras Med Trop. 50:113–116.
2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Yoshida S, Ikenaga N, Liu SB, Peng ZW,
Chung J, Sverdlov DY, Miyamoto M, Kim YO, Ogawa S, Arch RH, et al:
Extrahepatic platelet-derived growth factor-β, delivered by
platelets, promotes activation of hepatic stellate cells and
biliary fibrosis in mice. Gastroenterology. 147:1378–1392.
2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
de Franchis R: Baveno VI Faculty.
Expanding consensus in portal hypertension: Report of the Baveno VI
consensus workshop: Stratifying risk and individualizing care for
portal hypertension. J Hepatol. 63:743–752. 2015.PubMed/NCBI View Article : Google Scholar
|