Introduction
Pulmonary tuberculosis is mainly an infectious
disease caused by Mycobacterium tuberculosis infection.
Relevant research data have shown that 10.4 million new cases of
tuberculosis occurred worldwide in 2015, and 1.4 million people
died of the disease in the same year (1,2). Studies
have found that pulmonary tuberculosis is highly infectious and can
seriously affect the physical and mental health and quality of life
of patients. Some symptoms (expectoration, emaciation and chest
pain) are the main clinical manifestations of pulmonary
tuberculosis (3,4). In recent years, because of the specific
expression of inflammatory factors, immune function cytokines and
other related indicators in pulmonary tuberculosis patients, it has
become a ‘hot spot’ in clinical research (5,6).
Pulmonary tuberculosis often leads to the decline of immune
function. In addition, pulmonary tuberculosis itself is a
consumptive disease. Patients often suffer from a decline in
immunity. Excessive inflammatory reaction will aggravate the
severity of pulmonary tuberculosis and complications, resulting in
a poor therapeutic effect (7,8).
Therefore, studying the changes in inflammatory factors, immune
function and other indicators in the treatment process of pulmonary
tuberculosis is helpful for the selection of indicator schemes and
treatment.
At present, there are many types of drugs used in
the treatment of pulmonary tuberculosis, among which first-line
antituberculosis drugs are the first choice, including isoniazid
and rifampicin. The treatment scheme combined with multiple drugs
is also often used in clinical treatment (9). Literature has demonstrated the effect
of rifampicin combined with acetylcysteine and vitamin C on the
antibacterial activity of Mycobacterium tuberculosis and
Staphylococcus aureus, respectively. It has shown that the
combined application of antituberculosis drugs and vitamin C can
effectively eradicate microbial infection (10). Vitamin D is an essential nutrient
element in human body and belongs to one of the fat-soluble
vitamins (11). Recently, it was
shown that vitamin D not only plays a role in calcium phosphate and
bone metabolism, but also has antibacterial effect (12). Many studies have indicated that the
rise in tuberculosis risk is related to deficiency of vitamin D
(13). However, there is little
research on the effect of vitamin D-assisted anti-tuberculosis
drugs on the expression of immune cells and inflammatory factors in
patients with pulmonary tuberculosis.
The present study was mainly designed to explore the
application value of vitamin D-assisted antituberculosis drugs in
pulmonary tuberculosis by detecting the levels of inflammatory
factors and immune function in pulmonary tuberculosis patients.
Materials and methods
Baseline data
A total of 256 patients with pulmonary tuberculosis
who were admitted to Changchun Hospital for Infectious Diseases
were collected as research participants; 120 patients who were
treated with conventional antituberculosis drugs were taken as the
control group (CG). There were 66 males and 54 females, with a mean
age of 55.66±6.83 years (age range, 45-68 years). A total of 136
patients who were treated with vitamin D-assisted antituberculosis
drugs were taken as the research group (RG). There were 74 males
and 62 females, with a mean age of 54.62±6.41 years (age range,
42-66 years). The research study was approved by the Ethics
Committee of Changchun Hospital for Infectious Diseases (Changchun,
Jilin, China). Patients and their families were informed in
advance. Informed consent was signed after the consent of all of
the participants.
Inclusion criteria
For inclusion into the study, all patients were
diagnosed with pulmonary tuberculosis by clinical diagnosis; They
were accompanied by family members on admission. There were no
other malignant tumors and coronary heart disease diagnosed. The
patients did not present with basic diseases before admission, and
the body was relatively healthy. The subjects showed tuberculosis
toxic symptoms such as cough, hemoptysis and low fever to varying
degrees. There were no hematological diseases diagnosed.
Exclusion criteria
Subjects were excluded from the study if they
presented with a history of mental diseases, clinical data were
incomplete and the subjects did not actively cooperate when they
were admitted to the hospital. Patients with severe organ disease
or liver dysfunction were excluded. Patient who were allergic to
the treatment drug were also exluded.
Therapeutic schemes
Patients in the CG received anti-tuberculosis
treatment and received 0.3 g/day of isoniazid orally (Beijing
Yongkang Pharmaceutical Co., Ltd.; SFDA approval no. H11020585).
Patients were given rifampicin (Beijing Shuguang Pharmaceutical
Co., Ltd.; SFDA approval no. H11021062), 0.15 g once (1 tablet),
3-4 times a day. Ethambutol (Shanxi Zhendong Taisheng
Pharmaceutical Co., Ltd.; SFDA approval no. H14021899) was taken
orally once a day at a dose of 15 mg/kg. Pyrazinamide (Ningbo
Tianheng Pharmaceutical Co., Ltd.; H33021921) was orally
administered at 15-30 mg/kg daily, or 50-70 mg/kg, 2-3 times a
week. Patients were treated continuously for 60 days, and then
treated with isoniazid and rifampicin (usage and dosage were the
same as above) for 4 months. Patients in the RG received routine
antituberculosis drug therapy, supplemented by vitamin D therapy.
During the acute attack of pulmonary tuberculosis, the patient was
given 300,000 IU vitamin D3 (Zhejiang Xianju Pharmaceutical Co.,
Ltd.; SFDA approval no. H20058981) by intramuscular injection. The
intervention was implemented once a day, and the specific dose was
adjusted according to the actual situation of the patient. Since
the patient's pulmonary tuberculosis condition was stable, the
patient was given vitamin D3 (Beijing Zizhu Pharmaceutical Co.,
Ltd.; SFDA approval no. H11022062) for intervention by oral
administration with a daily dosage of 0.25 mg. All the patients
were treated for 6 months.
Index of inspection
Before and after treatment for 3 months, 5 ml of
peripheral venous blood of patients in the two groups were
collected, respectively, for detection and was centrifuged at 3,000
x g for 10 min. The serum was stored for later use. HR-801
Enzyme-Labeled Analyzer (Shenzhen Huakerui Technology Co., Ltd.)
was used to detect the expression levels of serum inflammatory
factors [interleukin (IL)-6, matrix metallopeptidase (MMP)-9, IL-4,
tumor necrosis factor (TNF)-α)], surfactant protein (SP-A, SP-D),
soluble selectin (sE-seletin, sP-seletin, sL-seletin) in patients
in the two groups. The operational steps were strictly carried out
in accordance with the instructions outlined in the enzyme-linked
immunosorbent assay (ELISA) kits. ELISA kits were purchased from
Beijing Baiolebo Technology Co., Ltd. FACSCalibur full-automatic
flow cytometer (Becton Dickinson and Flowjo v10.4.2 software
(Flowjo LLC) were used to measure the immune function indexes
(CD3+, CD4+, CD4+/CD8+)
of patients in the two groups after operation. Fluorescence coupled
monoclonal antibodies (CD3+, CD4+,
CD8+) were used to analyze the surface. Extracellular
staining was performed according to the manufacturer's
instructions. First, the coupled fluorescent antibody (different
combination of surface markers) was added to each peripheral blood
sample without plasma and incubated in dark for 15 min. Then
erythrocyte lysate was added, placed at room temperature for 10 min
and centrifuged at 350 x g for 5 min, and then the supernatant was
discarded to terminate cell lysis. Finally, cells were washed twice
with cell staining buffer by 350 x g centrifugation for 5 min and
the supernatant was discarded. The sample was resuspended in
staining buffer in preparation for flow cytometric analysis, in
which CD4+ and CD8+ T cells were gated on
CD3+ cells and expressed in lymphocyte percentage.
Evaluation standard Efficacy
evaluation
For a markedly effective evaluation, the clinical
symptoms of the patient disappeared. After continuous sputum
examination, the patient was negative. After imaging examination,
it was found that the lesion site had disappeared, the lesion was
obviously narrowed and the cavity was closed. For an effective
evaluation, the clinical symptoms of the patient were significantly
improved. The sputum examination results turned negative. After
examination, it was found that the lesion site had significantly
disappeared, the lesion was reduced and the cavity was basically
closed. For an ineffective evaluation, the patient's sputum
examination was positive, the lesion was not significantly
improved, and the cavity was enlarged or a new cavity appeared.
Total effective rate /%=markedly effective /% + effective /%.
Outcome measures
The improvement of symptoms after treatment was
compared between the two groups. The X-ray chest plain film and
sputum examination results were observed in the two groups. The
related factors (inflammatory factors, immune cytokines) were
detected in the two groups. In the treatment process, the
occurrence of adverse reactions were classified and counted in the
two groups, and the adverse reaction rates in the treatment process
were compared in the two groups. According to SF-36 quality of life
score (14), the living standards of
the two groups were compared after treatment.
Statistical methods
SPSS v19.0 (Asia Analytics formerly SPSS) was used
in this study. The counting data are expressed as [n (%)]. The
Chi-square test was used for inter-group comparison. The
measurement data are expressed as [mean ± standard deviation (SD)].
t-test was used to compare two groups. Repetitive measurement and
analysis of variance was used to analyze multiple groups. LSD-t
test was used for back testing. P<0.05 was considered to
indicate a statistically significant difference.
Results
Comparison of baseline clinical data
in both groups
The baseline clinical data of patients in the two
groups were statistically analyzed, as shown in Table I. There were no significant
differences in sex, age, body mass index (BMI), education level,
average duration, smoking or not and household registration of
patients between the two groups, which were comparable
(P>0.05).
 | Table IComparison of baseline clinical data
in the research group (RG) and control group (CG). |
Table I
Comparison of baseline clinical data
in the research group (RG) and control group (CG).
| | RG (n=136) | CG (n=120) |
t/χ2 | P-value |
|---|
| Sex | | | 0.009 | 0.925 |
|
Male | 74 (54.41) | 66 (55.00) | | |
|
Female | 62 (45.59) | 54 (45.00) | | |
| Mean age
(years) | 54.62±6.41 | 55.66±6.83 | 1.295 | 0.197 |
| Age (years) | | | 0.058 | 0.809 |
|
>50 | 83 (61.03) | 75 (62.50) | | |
|
≤50 | 53 (38.97) | 45 (37.50) | | |
| BMI
(kg/m2) | 22.84±2.56 | 22.94±2.47 | 0.317 | 0.752 |
| Education
level | | | 0.107 | 0.744 |
|
Junior high
school or below | 56 (41.18) | 47 (39.17) | | |
|
High school
or above | 80 (58.82) | 73 (60.83) | | |
| Average duration of
disease (months) | 5.01±2.13 | 4.97±2.32 | 0.144 | 0.886 |
| Smoking | | | 0.204 | 0.651 |
|
Yes | 87 (63.97) | 80 (66.67) | | |
|
No | 49 (36.03) | 40 (33.33) | | |
| Household
registration | | | 0.031 | 0.861 |
|
Resident
population | 79 (58.09) | 71 (59.17) | | |
|
Floating
population | 57 (41.91) | 49 (40.83) | | |
Comparison of improvement time of
clinical symptoms in the two groups
According to the results in Table II, the disappearance time of
wheezing and cough in RG was shorter than that in CG (P<0.001),
while the disappearance time of pulmonary rales was not
significantly different in the two groups (P>0.05).
 | Table IIComparison of improvement time (days)
of clinical symptoms in the research group (RG) and the control
group (CG). |
Table II
Comparison of improvement time (days)
of clinical symptoms in the research group (RG) and the control
group (CG).
| | Disappearance time
of pulmonary rales | Disappearance time
of cough | Disappearance time
of wheezing |
|---|
| RG (n=136) | 5.78±1.34 | 4.02±1.65 | 3.97±1.51 |
| CG (n=120) | 6.10±1.72 | 5.24±1.33 | 5.86±1.45 |
| t | 1.670 | 6.457 | 10.180 |
| P-value | 0.096 | <0.001 | <0.001 |
Comparison of X-ray chest plain film
and sputum examination results of patients in the two groups
Through X-ray chest plain film and sputum
examination, it was found (Table
III) that patients in the two groups had improvements in the
number of cavity closure, lesion absorption and the sputum
examinations were negative. There were no significant differences
in examination results between the two groups (P>0.05).
 | Table IIIComparison of the X-ray chest plain
film and sputum examination results of the patients in the research
group (RG) and control group (CG). |
Table III
Comparison of the X-ray chest plain
film and sputum examination results of the patients in the research
group (RG) and control group (CG).
| | No. of cavity
closures | No. of lesion
absorptions | No. of sputum
examination turning negative |
|---|
| RG (n=136) | 125 (91.91) | 124 (91.18) | 126 (92.65) |
| CG (n=120) | 106 (88.33) | 107 (89.17) | 109 (90.83) |
| χ2 | 0.926 | 0.292 | 0.279 |
| P-value | 0.336 | 0.589 | 0.598 |
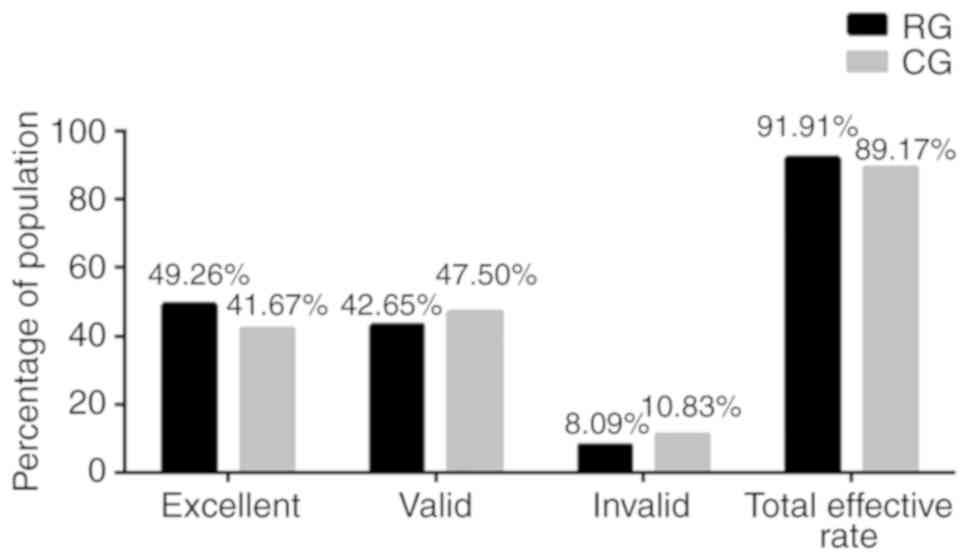
Comparison of the efficacy of
treatment in the two groups
Following comparison of the efficacy of patients in
the RG and CG groups after treatment, it was found (Fig. 1) that there were 67 cases with
markedly effective results, 58 cases with effective results and 11
cases with ineffective results, with a total effective rate of
91.91% in RG, while there were 50 cases with markedly effective
results, 57 cases with effective results and 13 cases with
ineffective results, with a total effective rate of 89.17% in CG.
There was no significant difference in total effective rate between
the two groups (P>0.05).
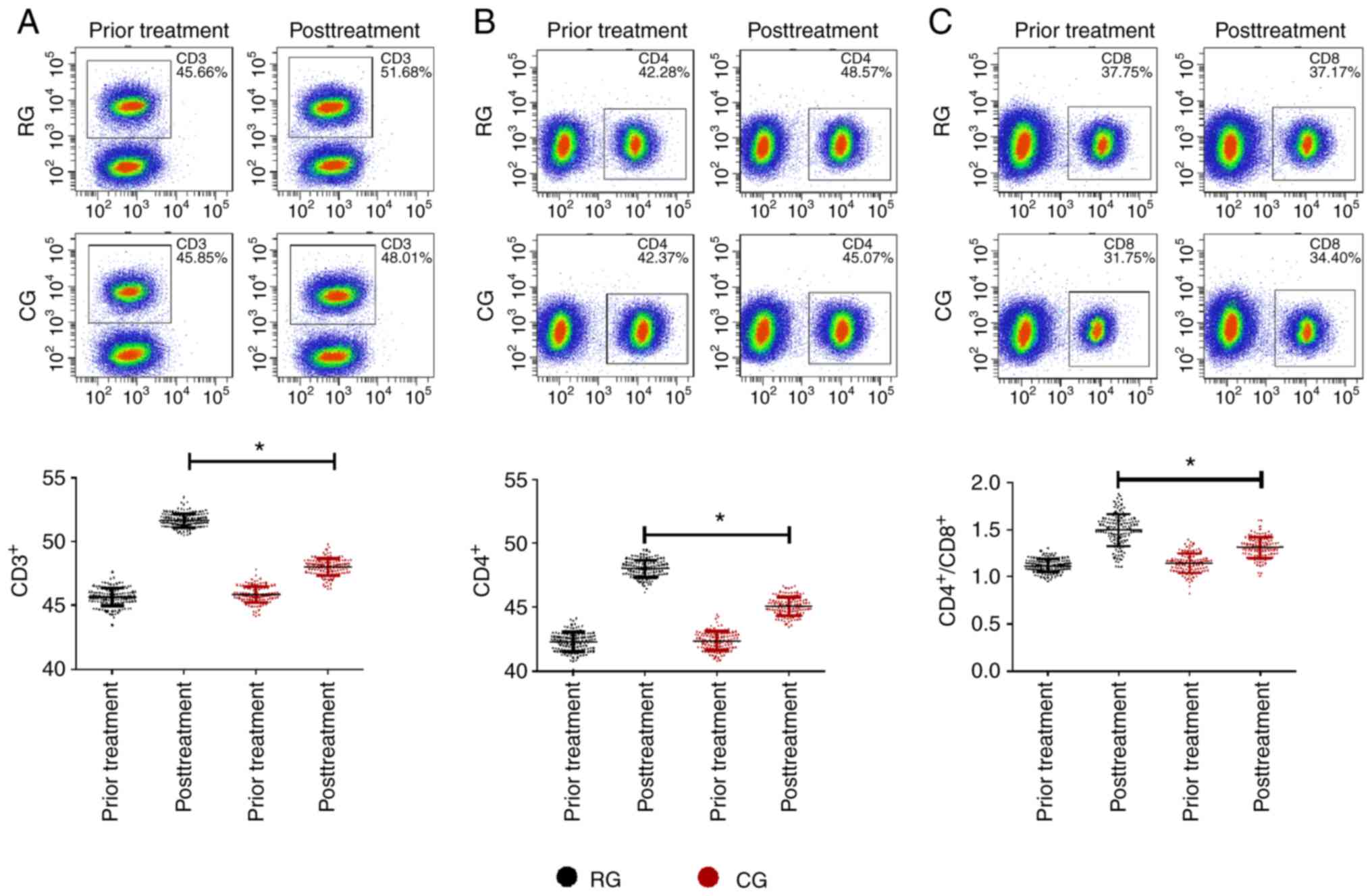
Comparison of immune cytokines in the
two groups
Comparing the changes of immune function indexes
between the RG and CG after different treatment methods, it was
found that there were no significant differences in immune function
indexes between the two groups before treatment (P>0.05). After
treatment, the immune function indexes of the two groups were both
upregulated (P<0.05), while CD3+, CD4+,
CD4+/CD8+ in the RG were significantly higher
than those in the CG (P<0.05; Fig.
2).
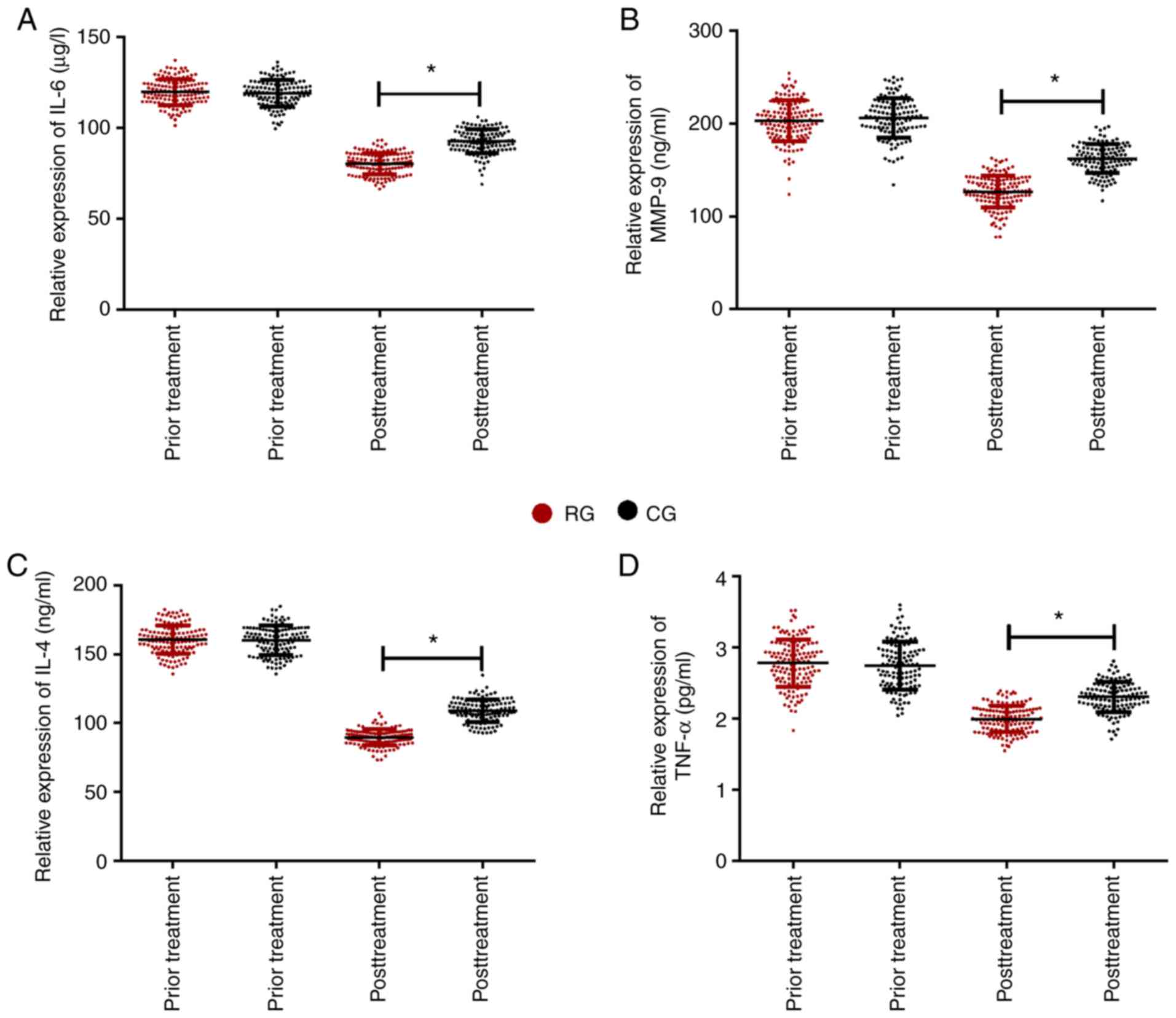
Comparison of levels of inflammatory
factors in the two groups
Following comparison of the changes in inflammatory
factors between the RG and CG after different treatment methods, it
was found that there were no significant differences in the levels
of inflammatory factors before treatment between the two groups.
After treatment, the inflammatory factors in the two groups were
improved compared with those before treatment. Serum inflammatory
factors (IL-6, MMP-9, IL-4, TNF-α) in RG were significantly lower
than those in CG (P<0.05; Fig.
3).
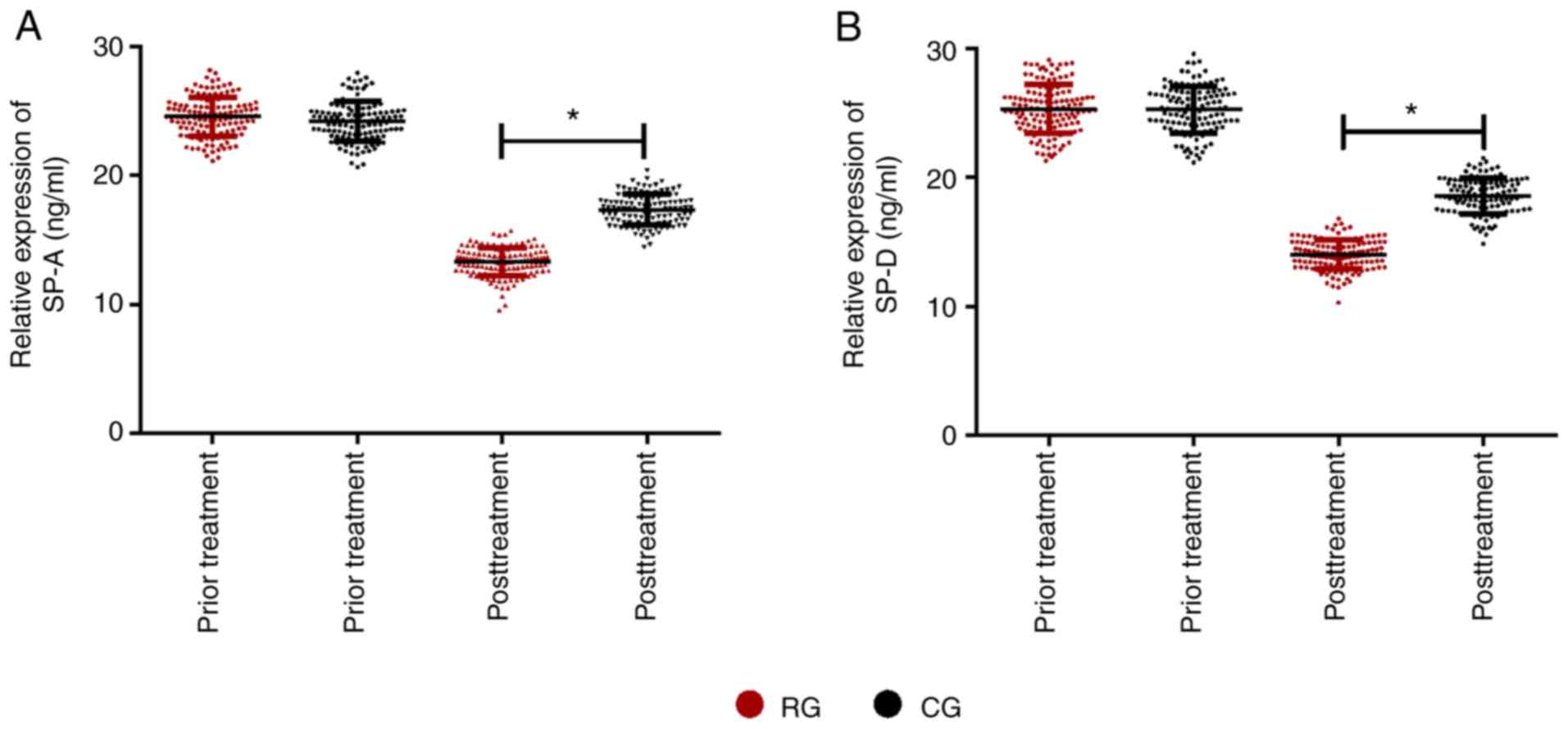
Comparison of surfactant proteins in
the two groups
Following comparison of the changes of surfactant
protein in the RG and CG after different treatment methods, there
was no difference in the comparison of surfactant protein between
the two groups before treatment (P>0.05). After treatment, the
surfactant protein in the two groups was lower than that before
treatment, while SP-A and SP-D in RG were significantly lower than
those in CG (P<0.05; Fig. 4).
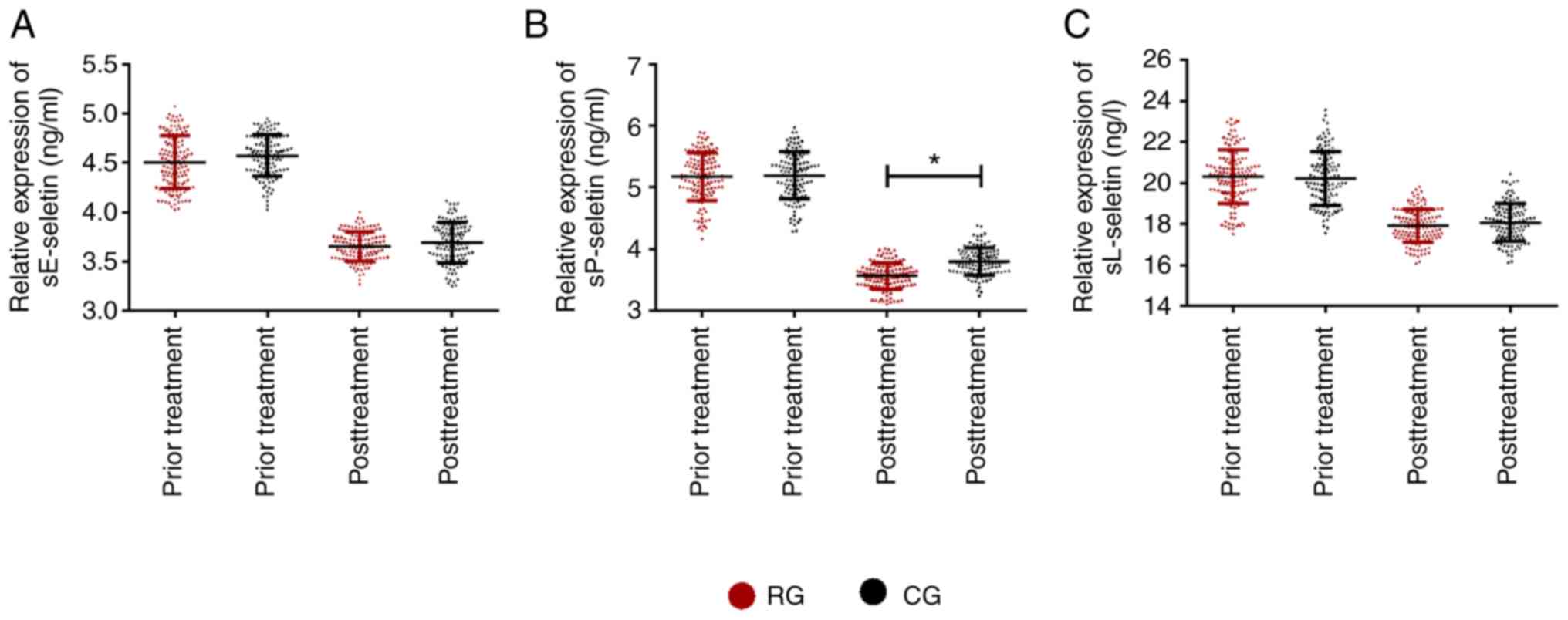
Comparison of levels of soluble
selectins in the two groups
Following comparison of the levels of soluble
selectins (sE-seletin, sP-seletin, sL-seletin) before and after
treatment in the RG and CG (Fig. 5),
it was found that there was no significant difference between the
two groups before treatment (P>0.05). After treatment, soluble
selectins improved significantly in the both groups. The levels of
soluble selectins in RG were slightly lower than those in CG, while
the levels of sP-seletin in RG were significantly lower than those
in CG (P<0.05).
Comparison of adverse reactions in the
two groups
Adverse reactions of patients during treatment was
counted, as shown in Table IV. It
could be found that the incidence of adverse reactions in RG was
lower than that in CG (15.45% vs. 25.84%).
 | Table IVComparison of adverse reactions in
the research group (RG) and control group (CG). |
Table IV
Comparison of adverse reactions in
the research group (RG) and control group (CG).
| | Nausea and
vomiting | Fever | Gastrointestinal
reaction | Rash | Dizziness | Overall
incidence |
|---|
| RG (n=136) | 4 (2.94) | 3 (2.21) | 6 (4.41) | 1 (0.74) | 7 (5.15) | 15.45% |
| CG (n=120) | 6 (5.00) | 5 (4.17) | 8 (6.67) | 3 (2.50) | 9 (7.50) | 25.84% |
| χ2 | | | | | | 4.253 |
| P-value | | | | | | 0.039 |
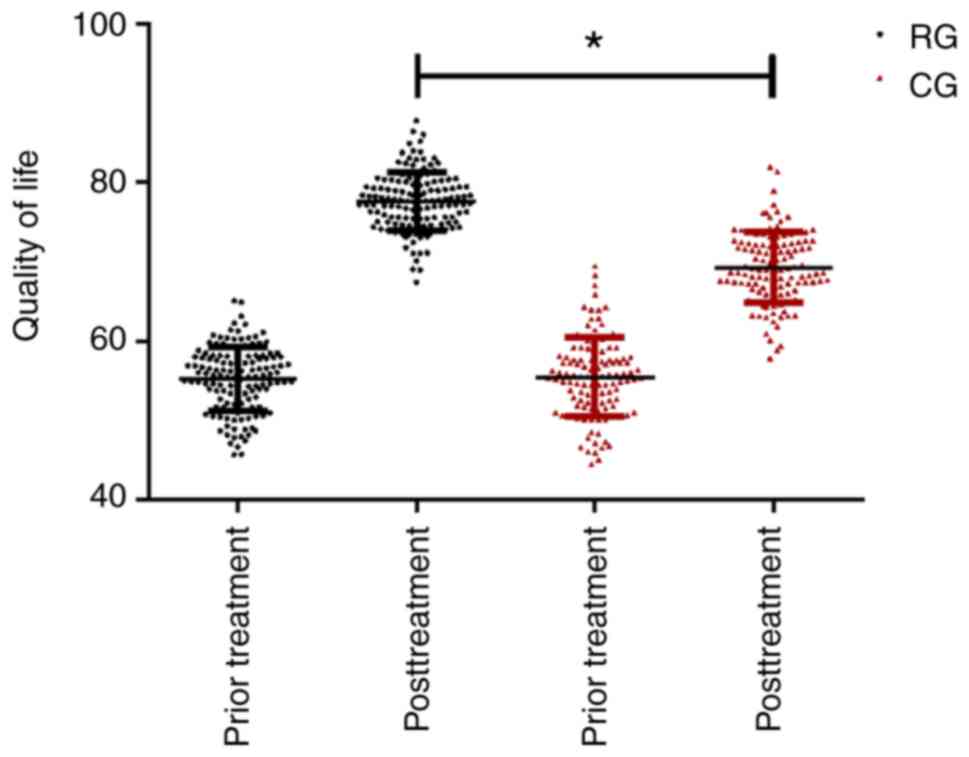
Comparison of life quality score in
the two groups
The life quality score in the RG and CG were
statistically recorded. There was no difference in the two groups
before treatment. After different treatments, the life quality
score were significantly improved in the two groups, and the life
quality score of patients in RG were significantly higher than
those in CG (P<0.05; Fig. 6).
Discussion
Pulmonary tuberculosis is a chronic infectious
disease transmitted through the respiratory system (15). At present, pulmonary tuberculosis is
still a global health problem with a huge disease burden. In the
new cases of pulmonary tuberculosis, 10% are children and 12% are
human immunodeficiency virus (HIV) co-infection with high mortality
rate (16,17). Therefore, pulmonary tuberculosis has
become the main cause of death from infectious diseases. Common
drugs for the treatment of pulmonary tuberculosis include
rifampicin, ethambutol, isoniazid and pyrazinamide. Although
isoniazid and ethambutol have ideal therapeutic effects, resistance
of drugs will appear after long-term use, resulting in drug
resistance. The therapeutic effect of pyrazinamide is easily
affected by pH value. Therefore, when the pH value is slightly
acidic, the drug has a better curative effect (18-21).
Rifampicin has better antibacterial activity, but there are many
adverse reactions after long-term use (18-21).
Therefore, at present, most of the patients with pulmonary
tuberculosis are treated with a combination of drugs, which can
significantly improve the inflammatory levels and quality of life
of patients (22).
Pulmonary tuberculosis is often accompanied by
typical respiratory symptoms. The signs are not very obvious in the
early stage, but obvious moist rales, increased vocal fremitus and
percussion dullness can be found in the middle and late stages
(23,24). The results of the present study
indicated that both treatment methods improved the clinical
symptoms of pulmonary tuberculosis patients, and the effect was
better with vitamin D supplementation. In addition, the therapeutic
effects were also compared in the research group (RG) and control
group (CG) in this study. The results indicated that both treatment
methods improved the number of cavity closures, lesion absorption
and the sputum examination results turned negative, but there was
no significant difference in efficacy between the two groups.
Studies have indicated no improvement in clinical parameters
(mortality, sputum smear positivity and sputum culture positivity)
in patients with pulmonary tuberculosis compared to placebo
(25). Since this study mainly
focused on the short-term efficacy of patients and did not deal
with the long-term efficacy, the effect of vitamin D on the
efficacy of pulmonary tuberculosis patients needs to be confirmed
by a large number of studies. T lymphocyte subsets are one type of
human immune response cells and is an important cell tissue of the
body's immune system. CD3+ is expressed in T cells and
it is also the basis of the body's cellular immunity.
CD4+ is considered to be helper T (Th) cells, and
CD4+/CD8+ can reflect cellular immune
dysfunction, thus the level of T cell subsets can fully reflect the
body's immune function and degree of disease development (26-28).
In the literature, it has been reported that vitamin D is a key
regulator for host defense against infection in patients infected
with human HIV (29). In combination
of the enhancement of innate and adaptive immunity by activating
genes and pathways, supplementation of vitamin D and restoring it
to a normal value can improve immune recovery during antiretroviral
therapy, reduce inflammation and immune activation levels, and
improve immunity to pathogens (29).
The present study revealed that there was no significant difference
in immune function indexes in the RG and CG before treatment. After
treatment, immune function indexes were upregulated in both groups,
while CD3+, CD4+,
CD4+/CD8+ in the RG were higher than those in
the CG. The results suggest that vitamin D assistance could
alleviate the immune dysfunction of pulmonary tuberculosis
patients. Interleukin (IL-6), matrix metalloproteinase (MMP)-9,
IL-4 and tumor necrosis factor (TNF)-α are important inflammatory
factors with strong biological effects, which can participate in
the regulation of immune response and are upregulated when the body
is in a pathological state (30,31).
Vitamin D has been shown to inhibit pro-inflammatory cytokine
responses and enhance anti-inflammatory responses (32). This study revealed that the levels of
IL-6, MMP-9, IL-4 and TNF-α were significantly reduced after
vitamin D adjuvant treatment, and the therapeutic effect was more
obvious than that of conventional antituberculosis drugs.
Serum SP-A and SP-D belong to surfactant proteins
and have been confirmed to be useful biomarkers for the severity of
pneumonia, chronic obstructive pulmonary disease and tuberculosis.
They are highly expressed in pulmonary diseases (33,34). The
present study was designed to compare the content of surfactant
protein before and after treatment in the RG and CG. Both treatment
methods improved the content of surfactant protein after treatment,
and vitamin D auxiliary treatment effectively reduced the levels of
SP-A and SP-D. Animal experimental research has revealed that SP-D
levels in mice with sufficient vitamin D before and after infection
are lower than those in mice with insufficient vitamin D (35). Since the serum level of the patients
was tested in this study at 3 months after treatment, we did not
know the long-term changing trend of surfactant protein, thus the
effect of vitamin D on the content of surfactant protein still
needs further research to confirm the long-term impact. Soluble
selectin (sE-seletin, sP-se-letin, sL-seletin) is highly expressed
in patients with pulmonary tuberculosis, which may be related to
pulmonary inflammatory stress of the patient (36,37),
thus regulating its expression has high value in the diagnosis and
treatment of diseases. The results of the present study showed that
both treatments improved the level of soluble selectins and
regulated the inflammatory stress state of pulmonary tuberculosis
patients. A related meta-analysis (a study of children) showed that
vitamin D supplementation had no beneficial effect on improving
sputum smear, culture transformation and adverse reactions in
children with pulmonary tuberculosis (38). In the present study, the total
incidence of adverse reactions in RG was relatively low. It may be
that vitamin D plays an auxiliary role and it effectively relieved
the occurrence of various adverse reactions. Many studies have
shown that adequate vitamin D supplementation can improve the
quality of life of patients presenting with various diseases. For
example, Bai and Dai (39) proposed
that increased serum vitamin A and 25-hydroxyvitamin D3
(25OHD3) levels can reflect the lung function and
quality of life of asthmatic children. Vitamin D supplementation
can improve the quality of life of patients with chronic pulmonary
obstruction (40). In this study,
quality of life scores were observed and counted in the RG and CG.
It was found that the use of vitamin D combined with
antituberculosis drugs significantly improved the quality of life
of the pulmonary tuberculosis patients, and had better application
value.
In the present study, the application value of
vitamin D-assisted antituberculosis drug treatment in tuberculosis
patients was compared from several aspects. Due to the short
follow-up period in this study, it was not possible to follow up
the long-term efficacy of patients, changes in immune cells and
inflammatory factors, thus there are certain limitations. In the
future, we can strengthen the research in this direction and
further study the complex factors affecting the efficacy of
tuberculosis treatment.
To sum up, vitamin D-assisted antituberculosis drugs
can effectively improve the immune function indexes and expression
level of inflammatory factors in pulmonary tuberculosis patients
and reduce the adverse reactions associated with treatment.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
CC conceived the research design and wrote the
manuscript. WP and XL were responsible for the treatment and
evaluation. HQ performed statistical analysis and designed the
tables and figures. All authors read and approved the manuscript
and agree to be accounTable for all aspects of the research in
ensuring that the accuracy or integrity of any part of the work are
appropriately investigated and resolved.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
Changchun Hospital for Infectious Diseases (Changchun, Jilin,
China). Patients who participated in this research, signed the
informed consent and had complete clinical data. Signed written
informed consents were obtained from the patients and/or
guardians.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Guo Z, Xiao D, Wang X, Wang Y and Yan T:
Epidemiological characteristics of pulmonary tuberculosis in
mainland China from 2004 to 2015: A model-based analysis. BMC
Public Health. 19(219)2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Xu Y, Wu J, Liao S and Sun Z: Treating
tuberculosis with high doses of anti-TB drugs: Mechanisms and
outcomes. Ann Clin Microbiol Antimicrob. 16(67)2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Carvalho AC, Cardoso CA, Martire TM,
Migliori GB and Sant'Anna CC: Epidemiological aspects, clinical
manifestations, and prevention of pediatric tuberculosis from the
perspective of the end TB strategy. J Bras Pneumol. 44:134–144.
2018.PubMed/NCBI View Article : Google Scholar : (In Portuguese,
English).
|
|
4
|
Rao VG, Bhat J, Yadav R, Muniyandi M,
Bhondeley MK and Wares DF: Yield of pulmonary tuberculosis cases by
symptoms: Findings from a community survey in Madhya Pradesh,
central India. Indian J Tuberc. 62:121–123. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Shen X, Zhang J, Tang P, Song H, Liu X,
Huang Z, Zhang X, Wang X and Wu M: Expression and clinical
significance of B and T lymphocyte attenuator on CD4+
and CD8+ T cells from patients with pulmonary
tuberculosis. Indian J Pathol Microbiol. 62:232–238.
2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Peng X, Luo T, Zhai X, Zhang C, Suo J, Ma
P, Wang C and Bao L: PPE11 of Mycobacterium tuberculosis can alter
host inflammatory response and trigger cell death. Microb Pathog.
126:45–55. 2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Rekha RS, Mily A, Sultana T, Haq A, Ahmed
S, Mostafa Kamal SM, van Schadewijk A, Hiemstra PS, Gudmundsson GH,
Agerberth B and Raqib R: Immune responses in the treatment of
drug-sensitive pulmonary tuberculosis with phenylbutyrate and
vitamin D3 as host directed therapy. BMC Infect Dis.
18(303)2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Tomio J, Yamana H, Matsui H, Yamashita H,
Yoshiyama T and Yasunaga H: Tuberculosis screening prior to
anti-tumor necrosis factor therapy among patients with
immune-mediated inflammatory diseases in Japan: A longitudinal
study using a large-scale health insurance claims database. Int J
Rheum Dis. 20:1674–1683. 2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Tornheim JA and Dooley KE: The global
landscape of tuberculosis therapeutics. Annu Rev Med. 70:105–120.
2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Khameneh B, Fazly Bazzaz BS, Amani A,
Rostami J and Vahdati-Mashhadian N: Combination of
anti-tuberculosis drugs with vitamin C or NAC against different
Staphylococcus aureus and Mycobacterium tuberculosis strains.
Microb Pathog. 93:83–87. 2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Zittermann A: Vitamin D in preventive
medicine: Are we ignoring the evidence? Br J Nutr. 89:552–572.
2003.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Syal K, Chakraborty S, Bhattacharyya R and
Banerjee D: Combined inhalation and oral supplementation of Vitamin
A and Vitamin D: A possible prevention and therapy for
tuberculosis. Med Hypotheses. 84:199–203. 2015.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Huang SJ, Wang XH, Liu ZD, Cao WL, Han Y,
Ma AG and Xu SF: Vitamin D deficiency and the risk of tuberculosis:
A meta-analysis. Drug Des Devel Ther. 11:91–102. 2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Li CT, Chu KH, Reiher B, Kienene T and
Chien LY: Evaluation of health-related quality of life in patients
with tuberculosis who completed treatment in Kiribati. J Int Med
Res. 45:610–620. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Ossalé Abacka KB, Koné A, Akoli Ekoya O,
Bopaka RG, Lankoandé Siri H and Horo K: Extrapulmonary tuberculosis
versus pulmonary tuberculosis: Epidemiological, diagnosis and
evolutive aspects. Rev Pneumol Clin. 74:452–457. 2018.PubMed/NCBI View Article : Google Scholar : (In French).
|
|
16
|
Maitra A, Kamil TK, Shaik M, Danquah CA,
Chrzastek A and Bhakta S: Early diagnosis and effective treatment
regimens are the keys to tackle antimicrobial resistance in
tuberculosis (TB): A report from Euroscicon's international TB
Summit 2016. Virulence. 8:1005–1024. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Churchyard G, Kim P, Shah NS, Rustomjee R,
Gandhi N, Mathema B, Dowdy D, Kasmar A and Cardenas V: What we know
about tuberculosis transmission: An overview. J Infect Dis. 216
(suppl 6):S629–S635. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Reynolds IR and Chan ED: An update on the
trends and treatment in multidrug-resistant tuberculosis. Am J
Respir Crit Care Med. 193:A5533. 2016.
|
|
19
|
An J, Bai X, Gao M, Chu N, Huang H, Xu H,
Zhang X, Li B, Huang C, Cai H, et al: Antituberculosis drug
prescribing for inpatients in a national tuberculosis hospital in
China, 2011-2015. J Glob Antimicrob Resist. 14:17–22.
2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Gegia M, Winters N, Benedetti A, van
Soolingen D and Menzies D: Treatment of isoniazid-resistant
tuberculosis with first-line drugs: A systematic review and
meta-analysis. Lancet Infect Dis. 17:223–234. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Hu Y, Wu X, Luo J, Fu Y, Zhao L, Ma Y, Li
Y, Liang Q, Shang Y and Huang H: Detection of pyrazinamide
resistance of Mycobacterium tuberculosis using nicotinamide as a
surrogate. Clin Microbiol Infect. 23:835–838. 2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Alessandra C Pinheiro and Marcus V N de
Souza. The relevance of new drug combinations for modern
tuberculosis treatment-a patent perspective. Recent Pat Antiinfect
Drug Discov. 8:130–138. 2013.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Li C, Liu L and Tao Y: Diagnosis and
treatment of congenital tuberculosis: A systematic review of 92
cases. Orphanet J Rare Dis. 14(131)2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Lewinsohn DM, Leonard MK, LoBue PA, Cohn
DL, Daley CL, Desmond E, Keane J, Lewinsohn DA, Loeffler AM,
Mazurek GH, et al: Official American Thoracic Society/Infectious
Diseases Society of America/Centers for disease control and
prevention clinical practice guidelines: Diagnosis of tuberculosis
in adults and children. Clin Infect Dis. 64:–e1. –e33.
2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Haris R, Irbaz R, Tanvir A, Maaz B and
Abul M: Vitamin D as a supplementary agent in the treatment of
pulmonary tuberculosis: A systematic review and meta-analysis of
randomized controlled trials. Eur Respir J. 42(4623)2013.
|
|
26
|
Li C, Zhou HC, Nie YL, Zhao BY and Wu CC:
Effects of lipopolysaccharide on T lymphocyte cell subsets and
cytokine secretion in mesenteric lymph nodes of mice: Histological
and molecular study. Environ Toxicol Pharmacol.
71(103214)2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Pérez-Antón E, Egui A, Thomas MC, Puerta
CJ, González JM, Cuéllar A, Segovia M and López MC: Impact of
benznidazole treatment on the functional response of Trypanosoma
cruzi antigen-specific CD4+CD8+ T cells in
chronic Chagas disease patients. PLoS Negl Trop Dis.
12(e0006480)2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Muller L, Mitsuhashi M, Simms P, Gooding
WE and Whiteside TL: Tumor-derived exosomes regulate expression of
immune function-related genes in human T cell subsets. Sci Rep.
6(20254)2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Jiménez-Sousa MÁ, Martínez I, Medrano LM,
Fernández-Rodríguez A and Resino S: Vitamin D in human
immunodeficiency virus infection: Influence on immunity and
disease. Front Immunol. 9(458)2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Naik SP, Mahesh PA, Jayaraj BS,
Madhunapantula SV, Jahromi SR and Yadav MK: Evaluation of
inflammatory markers interleukin-6 (IL-6) and matrix
metalloproteinase-9 (MMP-9) in asthma. J Asthma. 54:584–593.
2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Acharya AB, Thakur S, Muddapur MV and
Kulkarni RD: Tumor necrosis factor-α, interleukin-4 and -6 in the
serum of health, chronic periodontitis, and type 2 diabetes
mellitus. J Indian Soc Periodontol. 20:509–513. 2016.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Selvaraj P, Harishankar M and Afsal K:
Vitamin D: Immuno-modulation and tuberculosis treatment. Can J
Physiol Pharmacol. 93:377–384. 2015.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Güzel A, Karadağ A, Okuyucu A, Alaçam H
and Küçük Y: The evaluation of serum surfactant protein D (SP-D)
levels as a biomarker of lung injury in tuberculosis and different
lung diseases. Clin Lab. 60:1091–1098. 2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Enomoto Y, Hagiwara E, Komatsu S,
Nishihira R, Baba T and Ogura T: Comparison of biomarkers of
pulmonary tuberculosis activity-serum surfactant proteins A and D,
KL-6, C-reactive protein, and erythrocyte sedimentation rate.
Kekkaku. 89:637–642. 2014.PubMed/NCBI(In Japanese).
|
|
35
|
Serré J, Mathyssen C, Ajime TT, Korf H,
Maes K, Heulens N, Gysemans C, Mathieu C, Vanaudenaerde B, Janssens
W and Gayan-Ramirez G: Data on inflammatory cytokines and pathways
involved in clearance of nontypeable Haemophilus influenzae
from the lungs during cigarette smoking and vitamin D deficiency.
Data Brief. 22:703–708. 2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Mukae H, Ashitani J, Tokojima M, Ihi T,
Kohno S and Matsukura S: Elevated levels of circulating adhesion
molecules in patients with active pulmonary tuberculosis.
Respirology. 8:326–331. 2003.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Nowak JK, Wojsyk-Banaszak I, Mądry E,
Wykrętowicz A, Krzyżanowska P, Drzymała-Czyż S, Nowicka A,
Pogorzelski A, Sapiejka E, Skorupa W, et al: Increased soluble
VCAM-1 and normal P-selectin in cystic fibrosis: A cross-sectional
study. Lung. 195:445–453. 2017.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Xia J, Shi L, Zhao L and Xu F: Impact of
vitamin D supplementation on the outcome of tuberculosis treatment:
A systematic review and meta-analysis of randomized controlled
trials. Chin Med J (Engl). 127:3127–3134. 2014.PubMed/NCBI
|
|
39
|
Bai YJ and Dai RJ: Serum levels of vitamin
A and 25-hydroxyvitamin D3 (25OHD3) as reflectors of
pulmonary function and quality of life (QOL) in children with
sTable asthma: A case-control study. Medicine (Baltimore).
97(e9830)2018.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Alavi Foumani A, Mehrdad M, Jafarinezhad
A, Nokani K and Jafari A: Impact of vitamin D on spirometry
findings and quality of life in patients with chronic obstructive
pulmonary disease: A randomized, double-blinded, placebo-controlled
clinical trial. Int J Chron Obstruct Pulmon Dis. 14:1495–1501.
2019.PubMed/NCBI View Article : Google Scholar
|