Introduction
Hepatitis C virus (HCV) is a single-stranded RNA
virus, a source of debilitating liver disease ranging from
hepatitis to cirrhosis and HCC. Approximately 170 million people
are infected with HCV worldwide (1).
Among the extrahepatic manifestations of HCV
infection, an important role is played by renal involvement, being
described as a series of histopathological forms: Proliferative
membrane glomerulonephritis, IgA nephropathy, focal segmental
glomerulosclerosis, fibrillar glomerulonephritis,
tubulointerstitial nephropathy and thrombosis (2,3). The
prevalence of HCV infection in patients with chronic kidney disease
(CKD) is between 5 and 60% in developed countries, with a certain
predominance in hemodialysis patients (3). HCV-positive hemodialysis patients show
a decreased survival rate; the risk of death associated with liver
disease being aggravated by the increased risk of cardiovascular
death attributed to malnutrition and chronic inflammation (4).
HCV induces a low-amplitude T and B
lymphocyte-mediated immune response, which leads to long-lasting
infection and production of non-specific biochemical markers
(3-5).
Thus, disease progression is difficult to predict. Efforts should
be made to identify patients with HCV and liver inflammation
secondary to the viral infection before progression to a more
difficult to treat illness such as HCC and cirrhosis (6). The use of routine biochemical markers
as the tools for therapeutic response is essential for disease
prognosis, similar to biomarker applicability in other pathologies
(7,8).
There are different non-specific clinical,
biochemical and sonographic tools for the evaluation of the
clinical impact of HCV infection: Age, social status, smoking
status, platelet count, serum cholesterol and triglycerides,
albumin, creatinine, bilirubin, fibrinogen, hemostasis parameters,
liver aminotransferases [aspartate transaminase (AST), alanine
aminotransferase (ALT)], alkaline phosphatase (AP), γ-glutamyl
transferase (GGT), mixed cryoglobulinemia (MC), α-fetoprotein
(AFP), and assessment of liver fibrosis by non-invasive methods
(sonographic) (9-11).
AFP is a fetal protein synthesized in the same
chromosome location as serum albumin. AFP is considered similar to
fetal serum albumin, achieving high levels during fetal development
and sharing almost the same functions, binding heavy metals, fatty
acids and bilirubin. This protein can be found in monomeric,
dimeric or trimeric forms. In rodents, it has an important
anti-estrogenic property by binding maternal estradiol and
preventing virilization of the fetus. The exact role in human
adults, however, is not yet elucidated (12). This protein is considered an
important serological marker in various hepatic and non-hepatic
pathologies such as abnormalities of fetal development
(aneuploidies), ataxia-telangiectasia, and various malignancies
(yolk sac tumor, HCC, liver metastasis, non-seminomatous germ cell
tumors, teratomas, hepatoblastomas, Gruber-Frantz tumor) (13).
Elevated AFP is independently associated with a
decreased level of albumin (<3.5 g/dl) (14). Elevated AFP has also been shown to
be independently associated with elevated uric acid (10). Hyperuricemia is both a marker of
cardiovascular risk and an independent risk factor for the
occurrence of high blood pressure and heart failure (15). Arterial stiffness and vascular
calcification are major contributors to cardiovascular disease and
are independent predictors of cardiovascular mortality in patients
with end-stage renal disease (ESRD) (16-18).
Although the role of AFP in HCC is well established,
low to moderate plasma levels of AFP in patients with chronic
hepatitis C in the absence of HCC have also been described
(12). A plasma level higher that
200 ng/ml is considered highly sensitive for HCC in patients with
HCV, while levels of 5 to 60 ng/ml are routinely found in patients
with HCV, but lacking clinical significance (12).
Correlative biochemical and clinical interactions
between AFP and other routinely used markers in HCV infection have
been poorly studied. HCV could induce, for example, a greater
plasma increase in AFP compared to hepatitis B virus (HBV)
(19). The degree of fibrosis
and/or inflammation also influences AFP plasma concentration
(20).
Research has also revealed correlations between AST,
ALT, AST/ALT ratio, bilirubin, platelet count, and the HCV genotype
and plasma AFP (20).
AFP could also be used as a marker of antiviral
therapy response in HCV infection and could represent a good
biochemical tool in quantifying the sustained viral response to
therapy (21,22).
Hematologic and metabolic alterations ranging from
minor to severe, in accordance with disease progression are
generally accepted in the literature. There are, however,
contradictory data regarding correlations of AFP levels in HCV
patients, without cirrhosis or HCC.
Mixed cryoglobulinemia is another biochemical
feature of HVC infection, having a partially elucidated
significance (2). According to
Schamberg and Lake-Bakaar (2) and
Ozkok and Yildiz (23), mixed
cryoglobulinemia is related to virus replication,
membranoproliferative glomerulonephritis, and therapy response.
Cryoglobulinemia is present in a wide range of hematologic,
infectious, and rheumatologic diseases. Moreover, mixed
cryoglobulinemia is almost consistently present in HCV infections,
more so than in HBV infections or other non-infectious diseases
(24). The correlative potential of
AFP with mixed cryoglobulinemia in patients with HCV infection has
not yet been fully explored.
Sonographic imaging of patients with chronic
hepatitis is a reliable and non-invasive tool for assessing liver
progression to cirrhosis. Using modern interfaces such as
Fibroscan®, it is currently able to quantify the degree
of fibrosis (25,26). However, the relationship between
biochemical and sonographic data in HCV patients is still an
unresolved issue.
This study aimed to identify novel clinical,
biochemical and sonographic paradigms, which correlate with the
plasma levels of AFP in patients with chronic hepatitis C. We
considered demographic data (age, social status, smoking status,
antiviral therapies, weight and height), biochemical markers [total
hemoglobin, white blood cell count, platelet count, serum
cholesterol, triglycerides, blood urea nitrogen, creatinine, direct
bilirubin (DB), total bilirubin (TB), fibrinogen, activated partial
thromboplastin time (APTT), international normalized ratio (INR),
Na, K, C-reactive protein (CRP), erythrocyte sedimentation rate
(ESR), procalcitonin (PCT), AST, ALT, AP, GGT, MC] and sonographic
data (Fibroscan®).
Patients and methods
Ethic statement
This study is a monocentric, randomized,
double-blinded, prospective, clinical trial, which focused on the
comparison and correlation data regarding AFP in HCV patients,
without cirrhosis or HCC. All clinical procedures were carried out
following approval of the local Ethics Committee of Fundeni
Clinical Institute (Romania) (no. 48358/01.10.2019) for clinical
trials in accordance with the European Communities Council
Directive 2001/20/EC and with respect to personal data privacy
(European Directive 95/46/EC).
Demographic data collection and blood
sampling
After obtaining informed consent for sampling and
collecting the data, 37 healthy volunteers were enrolled in the
control group (C group). Another 108 patients were initially
enrolled in the study group in accordance with the inclusion
criteria: Detectible HCV infection over a six-month period, life
expectancy over 12 months, and no decompensated cirrhosis.
In order to minimize the confounding factors, we
excluded the patients with structural renal illness, decompensated
heart failure, concurrent alcohol and drug use, extrahepatic
manifestation of HCV Infection, HBV co-infection and HIV
co-infection. After exclusion, we integrated the data of 35 HCV
patients, designing an HCV group. For more accuracy in comparing
the data, a further 20 HCV patients with sustained viral response
(SVR) to antiviral therapy were included in the SVR group. We
considered SVR the absence of virus in the blood 12 weeks after
treatment cessation (27). The HCV
and SVR groups were matched with the control group in regards to
age and sex.
Demographic data such age, sex, social status,
smoking status, weight and height were obtained using a
questionnaire. The Body-Mass Index was calculated for each
patient.
All biochemical markers were assessed in the
clinical laboratory following a local sampling protocol. Whole
blood was collected in sampling tubes provided with clotting
accelerator and maintained at 4˚C. After clotting, the samples were
centrifuged for 20 min at approximately 1,000 x g and assessed
immediately with no freeze/thaw cycles for the markers detailed
below.
We assessed, spectrophotometrically, the levels of
albumin (g/dl), AST (U/l), ALT (U/l), GGT (U/l), AP (U/l), DB
(mg/dl), TB (mg/dl), CRP (mg/l), triglyceride (mg/dl), and serum
cholesterol (mg/dl) using compatible biochemistry kits for Siemens
Advia 1800 Reader (Siemens, Erlangen-Germany). Using
chemiluminescence, plasmatic AFP (ng/ml) and the presence of mixed
cryoglobulinemia was determined on an Advia Centaur XPT Reader
(Siemens). Plasmatic Na and K were determined using a
potentiometric method (ion selective electrode) on an Advia 1800
Reader (Siemens). The results are expressed in mmol/l. Complete
blood count analysis was performed with dedicated kits with the
means of an automatic flow cytometry technique (Sysmex XN-1000
Reader) (Sysmex, Kobe, Japan) and the results are expressed per ml
blood.
For the coagulation analysis, we sampled whole blood
using EDTA sampling tubes and following the manufacturer's
recommendation, we assessed using a StaR Max 3 Reader (Stago,
France) the following coagulation parameters: aPTT (sec), PT/INR
(sec), and plasmatic fibrinogen (mg/dl).
The virus replication level was assessed using
RT-PCR (Montania 4896 Real Time PCR Reader; Anatolia, Istanbul,
Turkey) using Bosphore HCV quantification kit (Anatolia) in
accordance with the manufacturer's recommendation. The main steps
include d: Total RNA collection, reverse transcription and
amplification in accordance with the local protocol. All plasma
samples were performed in triplicate and the viral load was
expressed per ml.
Non-invasive assessment of liver
fibrosis
The non-invasive measurement of liver fibrosis was
performed using Fibroscan® technology, Model 530 compact
(Echosens, France). The results were expressed automatically in kPa
and staged in 4 degrees according to the literature, with 0
representing no fibrosis and 4 representing major fibrosis
(28).
Data analysis
Data analysis and graphical representations were
performed using GraphPad Prism 6.00 (GraphPad Software Inc.). We
assessed the groups for normal distribution using D'
Agostino-Pearson omnibus normality test and Shapiro-Wilk test. For
the comparative analysis we considered demographic, biochemical and
sonographic data and for numerical results, we used one-way measure
ANOVA followed by post-hoc Tukey's test for interactions between
groups. The data are expressed as mean ± SD. A two-sided P-value
<0.05 was considered statistically significant. For binary data
(social status, smoking status and MC), we used the Chi-squared
test.
For the correlative data, we used a linear
correlation model, between AFP and all other variables including:
DB, TB, CRP, serum cholesterol, triglycerides, mixed
cryoglobulinemia, Na, K, complete blood count, aPTT, PT/INR,
fibrinogen, viral load and Fibroscan®. The Pearson
correlation coefficient, r, and R squared (R2) were
determined. We considered a two-sided P-value <0.05
statistically significant, for each correlation. We assigned
R2 values under 0.15 to have negligible correlative
effect.
Results
Demographic data
We did not observe any differences among groups
concerning age, sex or smoking status. The results are revealed in
Table I.
 | Table IGroup distribution according to age,
sex, and smoking status. |
Table I
Group distribution according to age,
sex, and smoking status.
| Parameters | Control group
(n=37) | HCV group (n=35) | SVR group (n=20) | P-value |
|---|
| Mean age (years) | 56.3±1.34 | 62.2±1.98 | 64.5±3.04 | NS |
| Sex (F/M %) | 59.45/40.54 | 65.42/34.58 | 67.5/32.5 | NS |
| Smoking (%) | 13.51 | 11.42 | 18.18 | NS |
AFP, cryoglobulinemia and
FibroScan® dynamics between groups
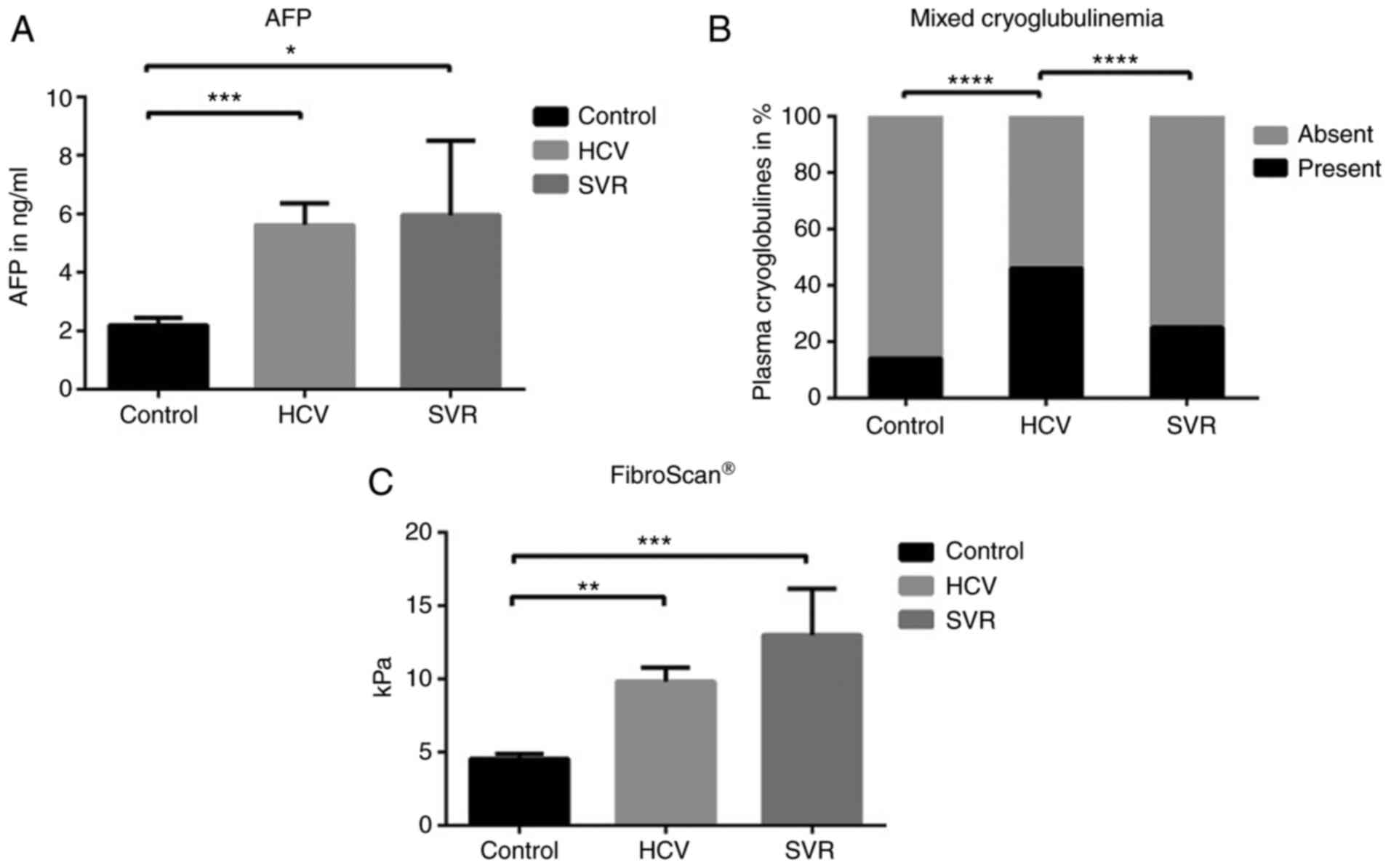
The plasma levels of AFP between groups are
illustrated in Fig. 1A. The plasma
levels of AFP in the HCV and SVR groups were significantly
increased when compared to the control group (5.79±0.7 vs. 2.4±0.3,
P=0.0009 and 5.9±2.5 vs. 2.4±0.27, P=0.042, respectively). No
difference in AFP was observed between the HCV and SVR group
[5.79±0.73 vs. 5.9±2.5, P>0.05 (NS, not significant)]. The
presence of mixed cryoglobulinemia in plasma among the groups is
shown in Fig. 1B. We observed a
statistically increased incidence of mixed cryoglobulinemia in the
HCV group when compared to both the control group (46 vs. 14%,
P<0.0001) and SVR group (46 vs. 25%, P<0.0001).
The Fibroscan® results are revealed in
Fig. 1C. We identified
significantly increased fibrosis in both the HCV and SVR groups
when compared to the control group: 9.8±0.9 vs. 4.5±0.37, P=0.001
and 13±3.2 vs. 4.5±0.37, P=0.0006, respectively. No significant
difference was observed between the HCV and SVR groups (9.8±1 vs.
13±3.2, NS).
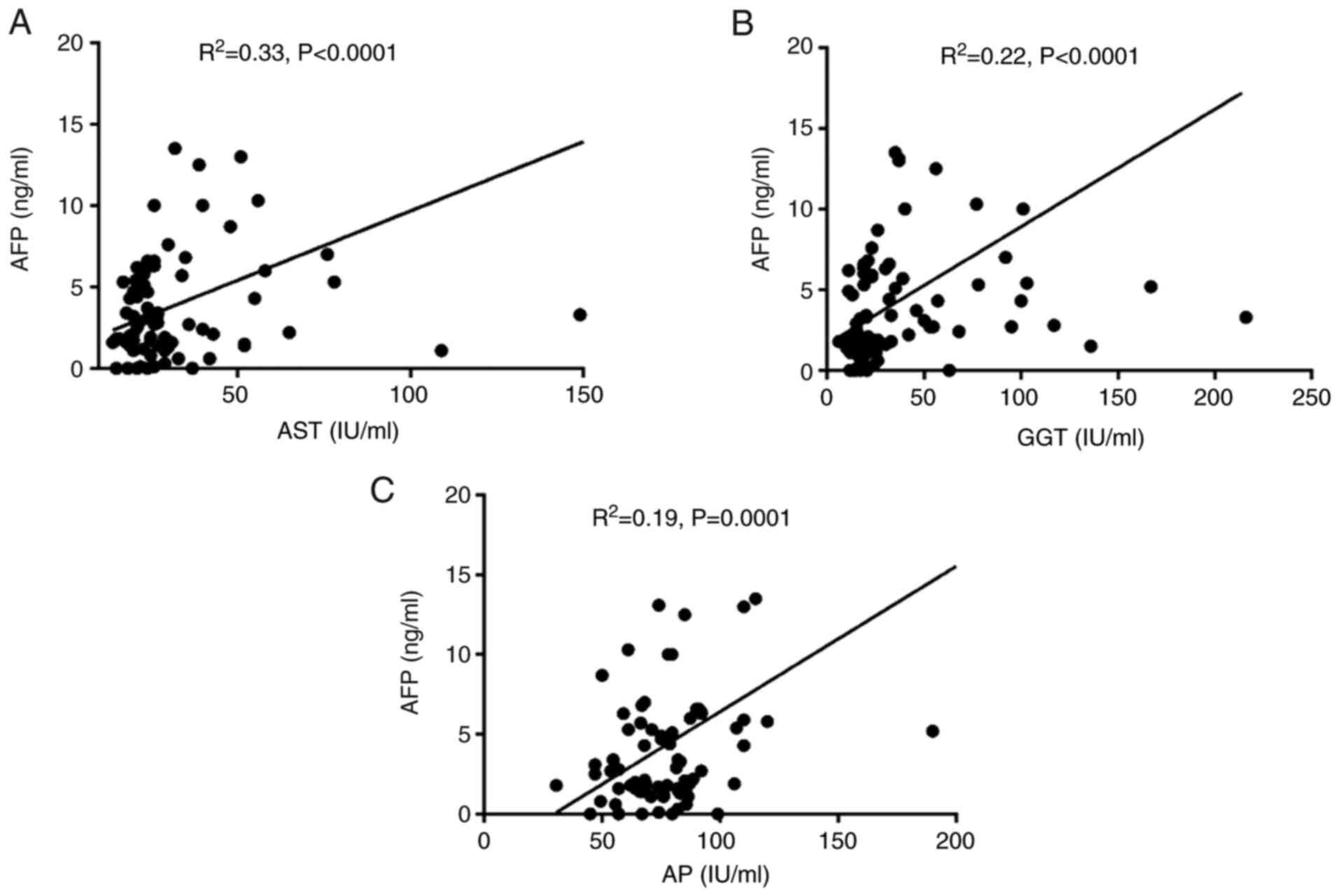
AFP depends mainly on serum AST, GGT,
and AP
By means of a linear correlation model between AFP
and all other biochemical and sonographic variables, we observed
statistically significant minor positive correlations between AFP
and AST (R2=0.33, P<0.0001), GGT (R2=0.22,
P<0.0001), and AP (R2=0.19, P=0.0001) in the
HCV-infected patients (HCV and SVR group respectively) (Fig. 2A-C). All other variables had either
negligible (R2<0.15) or no correlative effect
(P>0.05, NS).
Discussion
α-fetoprotein (AFP) is a well-known diagnostic tool
for hepatitis C virus (HCV) patients when associated with
hepatocellular carcinoma (HCC). AFP is, in fact, a protein present
in high concentrations in a wide range of diseases, having a liver
origin in the fetus (13,29). The cut-off level of AFP in
predicting HCC is, however, controversial. Values between 17.4
ng/ml for early-stage HCC, to 200 ng/ml for recurrent HCC after
liver transplantation are advocated (30,31).
Considering that AFP stratifies the risk of developing HCC in
patients with HCV, we observed that AFP values were statistically
higher in patients with HCV unrelated to viral load or the presence
of sustained viral response (SVR) after therapy in patients without
HCC. Although we detected plasma AFP values under the cut-off
values advocated in the literature as highly sensitive for HCC, our
study reveals a controversial mean of this protein in HCV patients
in the absence of HCC. Moreover, AFP could be an important outcome
marker in hepatitis C, even if the patients are not yet diagnosed
with HCC, regardless of the clinical response to therapy. The
persistence of plasmatic AFP in patients with HCV and SVR suggests
that the likelihood of developing HCC in the future despite
long-term response to antiviral therapy could be a significant
clinical issue. Conversely, a decreased clearance of AFP after SVR
could be advocated. According to Chen et al, a longer
follow-up period after SVR might influence the plasmatic levels of
AFP (32).
Additionally, we observed a statistically
significant direct correlation between AFP and aspartate
transaminase (AST). Thus, AFP could quantify, indirectly, the
magnitude of HCV-induced liver cytolysis. Our data are in
accordance with Chen et al, whose study identified the
predictive value of AFP to therapy response and the role of AST
elevation as an independent risk factor for plasma AFP presence
(32). Considering that cholestasis
syndrome [high alkaline phosphatase (AP), γ-glutamyl transferase
(GGT)] could be an independent factor for HCC in patients with
viral hepatitis, especially in those with cirrhosis, the cumulative
biochemical evidence of low to moderate plasmatic AFP and elevated
GGT and AP could represent a future biochemical algorithm for
identifying patients with HCV at risk of developing HCC (33). There are, however, inconsistent
data, regarding patients with HCV in the absence of cirrhosis and
their risk to develop HCC, when low to moderate levels of AFP are
associated with high plasmatic GGT and FA levels. We identified a
strong correlation between AFP levels, GGT, and FA as predictive
variables in HCV patients without cirrhosis or HCC. The clinical
significance should be, however, further explored.
Regarding mixed cryoglobulinemia, we observed a
statistically significant increase in the HCV group compared to the
control group. Mixed cryoglobulinemia in HCV infection is an
important qualitative serologic tool of viral replication and
HCV-related renal injury (2,22).
Thus, we observed the presence of mixed cryoglobulinemia in
patients with active disease conditions, unrelated to viral load.
Moreover, patients with SVR showed a decreased incidence of
plasmatic mixed cryoglobulinemia, confirming the role of this
diagnostic tool in the qualitative assessment of disease activity.
According to Landau et al, MC in the context of SVR is more
an exception (34).
Furthermore, patients with HCV-related
glomerulonephritis were not included in the present study.
According to Santoriello et al, the presence of
membranoproliferative glomerulonephritis is typically secondary to
HCV but not to HBV and the presence of mixed cryoglobulinemia
correlates with this renal condition (35).
Regarding Fibroscan® examination, our
study results are in accordance with the literature. We detected an
increased fibrosis score in HCV patients when compared to healthy
volunteers. The fibrosis degree was, however, the same between the
HCV and the SVR group. This paradox could be explained due the
longer time until fibrosis resolution could be present in HCV
patients after SVR (36). There are
controversial data in the literature regarding the reversibility of
liver fibrosis secondary to HCV by means of Fibroscan®.
Some data advocate a relatively slow resolution of fibrosis in time
after SVR. According to studies, the resolution could occur 1 year
after SVR and is less predictive of successful therapy compared to
liver biopsy (36,37). Furthermore, it is likely that
earlier stages of liver fibrosis are more easily reversible than
the later stages (38).
In conclusion, our study opens a new research
pathway in quantifying HCV-related liver lesions by means of modern
serologic biomarkers and sonographic assessment including AFP, MC,
and Fibroscan® in the absence of a diagnosed neoplastic
state.
Acknowledgements
The authors would like to thank Ileana
Constantinescu for her help with blood test analysis.
Funding
Funding: No funding was received.
Availability of data and materials
Due to confidentiality reasons data generated or
analyzed during this study are not included in this published
article.
Authors' contributions
TI conributed to all of the following aspects
including design of the study, collection and analysis of the data,
writing and reviewing of the manuscript. SIs, SIo and MDT wrote and
reviewed the manuscript, analyzed the data, and contributed to the
study design. EM, DGB, and AT performed the literature search and
selected the studies to be included. LI coordinated and designed
the study. All authors read and approved the manuscript and agree
to be accountable for all aspects of the research in ensuring that
the accuracy or integrity of any part of the work are appropriately
investigated and resolved.
Ethics approval and consent to
participate
This study was approved by the local Ethics
Committee of Fundeni Clinical Institute (Romania), approval no.
48358/01.10.2019. Written informed consent was obtained from all
patients prior to publication.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Petruzziello A, Marigliano S, Loquercio G,
Cozzolino A and Cacciaputi C: Global epidemiology of hepatitis C
virus infection: An up-date of the distribution and circulation of
hepatitis C virus genotypes. World J Gastroenterol. 22:7824–7840.
2016.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Schamberg NJ and Lake-Bakaar GV: Hepatitis
C virus-related mixed cryoglobulinemia: Pathogenesis, Clinica
manifestations, and new therapies. Gastroenterol Hepatol (NY).
3:695–703. 2007.PubMed/NCBI
|
|
3
|
Timofte D, Dragos D, Balcangiu-Stroescu
AE, Tanasescu MD, Gabriela Balan D, Avino A, Tulin A, Stiru O and
Ionescu D: Infection with hepatitis C virus in hemodialysis
patients: An overview of the diagnosis and prevention rules within
a hemodialysis center (Review). Exp Ther Med. 20:109–116.
2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Balcangiu-Stroescu AE, Tanasescu MD,
Diaconescu AC, Raducu L, Constantin AM, Balan DG, Tarmure V and
Ionescu D: Cardiovascular comorbidities, inflammation and serum
albumin levels in a group of hemodialysis patients. Rev Chim
Buchar. 69:926–929. 2018.
|
|
5
|
Dustin LB: Innate and adaptive immune
responses in chronic HCV infection. Curr Drug Targets. 18:826–843.
2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Suceveanu AI, Stoian AP, Mazilu L, Voinea
F, Hainarosie R, Diaconu CC, Pituru S, Nitipir C, Badiu DC, Ceausu
I and Suceveanu AP: Interferon-free therapy is not a trigger for
hepatocellular carcinoma in patients with chronic infection with
hepatitis C virus. Farmacia. 66:904–908. 2018.
|
|
7
|
Marinescu I, Schenker RA, Stovicek PO,
Marinescu D, Ciobanu CF, Papacocea SI, Manea MC, Papacocea RI,
Manea M, Chirita R and Ciobanu AM: Biochemical factors involved in
the unfavorable evolution of prostate cancer. Rev Chim.
70:3343–3347. 2019.
|
|
8
|
Papacocea MT, Badarau IA, Radoi M and
Papacocea IR: The predictive role of biochemical plasma factors in
patients with severe traumatic brain injuries. Rev Chim Buchar.
70:1754–1757. 2019.
|
|
9
|
Sheridan DA, Aithal G, Alazawi W, Allison
M, Anstee Q, Cobbold J, Khan S, Fowell A, McPherson S, Newsome PN,
et al: Care standards for non-alcoholic fatty liver disease in the
United Kingdom 2016: A cross-sectional survey. Frontline
Gastroenterol. 8:252–259. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Sagnelli E, Santantonio T, Coppola N,
Fasano M, Pisaturo M and Sagnelli C: Acute hepatitis C: Clinical
and laboratory diagnosis, course of the disease, treatment.
Infection. 42:601–610. 2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Fierbinteanu-Braticevici C, Papacocea R,
Tribus L and Baicus C: Role of 13C methacetin breath test for non
invasive staging of liver fibrosis in patients with chronic
hepatitis C. Indian J Med Res. 140:123–129. 2014.PubMed/NCBI
|
|
12
|
Christiansen M, Høgdall CK, Andersen JR
and Nørgaard-Pedersen B: Alpha-fetoprotein in plasma and serum of
healthy adults: Preanalytical, analytical and biological sources of
variation and construction of age-dependent reference intervals.
Scand J Clin Lab Invest. 61:205–215. 2001.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Spătaru RI, Enculescu A and Popoiu MC:
Gruber-Frantz tumor: A very rare pathological condition in
children. Rom J Morphol Embryol. 55:1497–1501. 2014.PubMed/NCBI
|
|
14
|
Chen CH, Lin ST, Kuo CL and Nien CK:
Clinical significance of elevated alpha-fetoprotein (AFP) in
chronic hepatitis C without hepatocellular carcinoma.
Hepatogastroenterology. 55:1423–1427. 2008.PubMed/NCBI
|
|
15
|
Timofte D, Mandita A, Balcangiu-Stroescu
AE, Balan D, Raducu L, Tanasescu MD, Diaconescu A, Dragos D,
Cosconel CI, Stoicescu SM and Ionescu D: Hyperuricemia and
cardiovascular diseases-clinical and paraclinical correlations. Rev
Chim. 70:1045–1046. 2019.
|
|
16
|
Timofte D, Dragoș D, Balcangiu-Stroescu
AE, Tănăsescu MD, Gabriela Bălan D, Răducu L, Tulin A, Stiru O and
Ionescu D: Abdominal aortic calcification in predialysis patients:
Contribution of traditional and uremia-related risk factors. Exp
Ther Med. 20:97–102. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Timofte D, Ionescu D, Medrihan L, Mandita
A, Rasina A and Damian L: Vascular calcification and bone disease
in hemodialysis patient, assessment, association and risk factors.
Nephrol Dial Transplant. 22:325–326. 2007.
|
|
18
|
Gaman MA, Dobrica EC, Pascu EG, Cozma MA,
Epingeac ME, Gaman AM, Pantea Stoian AM, Bratu OG and Diaconu CC:
Cardio metabolic risk factors for atrial fibrillation in type 2
diabetes mellitus: Focus on hypertension, metabolic syndrome and
obesity. J Mind Med Sci. 6:157–161. 2019.
|
|
19
|
Emokpae MA, Adejumol BG, Abdu A and Sadiq
NM: Serum alpha-fetoprotein level is higher in hepatitis C than
hepatitis B infected chronic liver disease patients. Niger Med J.
54:426–429. 2013.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Tai WC, Hu TH, Wang JH, Hung CH, Lu SN,
Changchien CS and Lee CM: Clinical implications of
alpha-fetoprotein in chronic hepatitis C. J Formos Med Assoc.
108:210–218. 2009.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Di Bisceglie AM, Sterling RK, Chung RT,
Everhart JE, Dienstag JL, Bonkovsky HL, Wright EC, Everson GT,
Lindsay KL, Lok AS, et al: Serum alpha-fetoprotein levels in
patients with advanced hepatitis C: Results from the HALT-C trial.
J Hepatol. 43:434–441. 2005.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Abdoul H, Mallet V, Pol S and Fontanet A:
Serum alpha-fetoprotein predicts treatment outcome in chronic
hepatitis C patients regardless of HCV genotype. PLoS One.
3(e2391)2008.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Ozkok A and Yildiz A: Hepatitis C virus
associated glomerulopathies. World J Gastroenterol. 20:7544–7554.
2014.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Morra E: Cryoglobulinemia. Hematology Am
Soc Hematol Educ Program: 368-372, 2005.
|
|
25
|
Gamil M, Alboraie M, El-Sayed M,
Elsharkawy A, Asemn N, Elbaz T, Mokey M, Abbas B, Mehrez M and
Esmat G: Novel scores combining AFP with non-invasive markers for
prediction of liver fibrosis in chronic hepatitis C patients. J Med
Virol. 90:1080–1086. 2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Fierbinteanu-Braticevici C, Baicus C,
Tribus L and Papacocea R: Predictive factors for nonalcoholic
steatohepatitis (NASH) in patients with nonalcoholic fatty liver
disease (NAFLD). J Gastrointestin Liv Dis. 20:153–159.
2011.PubMed/NCBI
|
|
27
|
Martinot-Peignoux M, Stern C, Maylin S,
Ripault MP, Boyer N, Leclere L, Castelnau C, Giuily N, El Ray A,
Cardoso AC, et al: Twelve weeks posttreatment follow-up is as
relevant as 24 weeks to determine the sustained virologic response
in patients with hepatitis C virus receiving pegylated interferon
and ribavirin. Hepatology. 51:1122–1126. 2010.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Lucero C and Brown RS Jr: Noninvasive
measures of liver fibrosis and severity of liver disease.
Gastroenterol Hepatol (NY). 12:33–40. 2016.PubMed/NCBI
|
|
29
|
He Y, Lu H and Zhang L: Serum AFP levels
in patients suffering from 47 different types of cancers and
noncancer diseases. Prog Mol Biol Transl Sci. 162:199–212.
2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Yoo T, Lee KW, Yi NJ, Choi YR, Kim H, Suh
SW, Jeong JH, Lee JM and Suh KS: Peri-transplant change in AFP
level: A useful predictor of hepatocellular carcinoma recurrence
following liver transplantation. J Korean Med Sci. 31:1049–1054.
2016.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Ahn DG, Kim HJ, Kang H, Lee HW, Bae SH,
Lee JH, Paik YH and Lee JS: Feasibility of α-fetoprotein as a
diagnostic tool for hepatocellular carcinoma in Korea. Korean J
Intern Med. 31:46–53. 2016.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Chen TM, Huang PT, Tsai MH, Lin LF, Liu
CC, Ho KS, Siauw CP, Chao PL and Tung JN: Predictors of
alpha-fetoprotein elevation in patients with chronic hepatitis C,
but not hepatocellular carcinoma, and its normalization after
pegylated interferon alfa 2a-ribavirin combination therapy. J
Gastroenterol Hepatol. 22:669–675. 2007.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Yang JG, He XF, Huang B, Zhang HA and He
YK: Rule of changes in serum GGT levels and GGT/ALT and AST/ALT
ratios in primary hepatic carcinoma patients with different AFP
levels. Cancer Biomark. 21:743–746. 2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Landau DA, Saadoun D, Halfon P,
Martinot-Peignoux M, Marcellin P, Fois E and Cacoub P: Relapse of
hepatitis C virus-associated mixed cryoglobulinemia vasculitis in
patients with sustained viral response. Arthritis Rheum.
58:604–611. 2008.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Santoriello D, Pullela NK, Uday KA, Dhupar
S, Radhakrishnan J, D'Agati VD and Markowitz GS: Persistent
hepatitis C Virus-associated cryoglobulinemic glomerulonephritis in
patients successfully treated with direct-acting antiviral therapy.
Kidney Int Rep. 3:985–990. 2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Pan JJ, Bao F, Du E, Skillin C, Frenette
CT, Waalen J, Alaparthi L, Goodman ZD and Pockros PJ: Morphometry
confirms fibrosis regression from sustained virologic response to
direct-acting antivirals for hepatitis C. Hepatol Commun.
2:1320–1330. 2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Ilie M, Rusu M, Rosianu C, Neagu TP,
Motofei IG, Bratu OG, Socea B, Stanescu AMA, Gherghiceanu F, Pantea
Stoian A, et al: Ultrasound-guided biopsy in focal liver lesions.
Arch Balk Med Union. 53:364–368. 2018.
|
|
38
|
Wang JH, Changchien CS, Hung CH, Tung WC,
Kee KM, Chen CH, Hu TH, Lee CM and Lu SN: Liver stiffness decrease
after effective antiviral therapy in patients with chronic
hepatitis C: Longitudinal study using FibroScan. J Gastroenterol
Hepatol. 25:964–969. 2010.PubMed/NCBI View Article : Google Scholar
|