Introduction
With high morbidity and mortality rates,
hepatocellular carcinoma (HCC) is the fifth most commonly diagnosed
cancer type and the third leading cause of cancer-associated
mortality worldwide (1). Multiple
treatment interventions, such as liver resection, transarterial
chemoembolization, radiotherapy and chemotherapy with sorafenib,
have been used to treat HCC. However, the prognosis of patients
with HCC remains poor due to difficulties making a prognosis, high
recurrence and early vascular invasion (2,3). As a
type of cancer, HCC is associated with multi-gene mutations
(4). Molecular targeting is
currently used as a novel therapy for advanced HCC, as it has been
indicated to acquire favorable curative effects that significantly
prolong the survival time of patients (5). Thus, the obtainment of a detailed
understanding of HCC development and identification of novel
molecular targets for HCC treatment have become a significant
research focus. In particular, the discovery of reliable biological
markers for the diagnosis, treatment and prognosis of HCC is
required.
MicroRNAs (miRNAs/miRs) are endogenous small
non-coding RNAs with a length of 19-25 nucleotides. The expression
of various miRNAs and their functions in tumor progression have
been extensively elucidated. Abnormal expression of miRNAs has been
indicated to regulate cellular processes such as cell
proliferation, differentiation, development and apoptosis in
multiple cancer types (6-8).
Furthermore, an increasing number of studies have demonstrated the
diagnostic, prognostic and biological functions of miRNAs in
patients with HCC (9-11).
Several studies have reported that the expression of
miR-3677 is negatively associated with the survival of patients
with HCC and that miR-3677 is one of the tumor-specific miRNAs in
HCC (12-15).
In addition, the preliminary prognostic role of miR-3677 has been
determined in colon cancer (16,17).
As a novel miRNA, miR-3677-5p has not been previously reported in
neoplasms, to the best of our knowledge. In particular, the
prognostic and biological roles of miR-3677-5p in HCC have remained
elusive.
In the present study, the prognostic value and
biological functions of miR-3677-5p in HCC and its involvement in
HCC progression were explored. It was determined that miR-3677-5p
was upregulated in human primary HCC tissues and cell lines.
Furthermore, high expression of miR-3677-5p was revealed to be
associated with unfavorable prognosis of patients with HCC. In
addition, in vitro functional assays demonstrated that
miR-3677-5p has a role in promoting the proliferation, migration
and invasion of HCC cells.
Materials and methods
Clinical samples
A total of 80 patients with HCC from the Ningbo
Medical Treatment Center Li Huili Hospital (Ningbo, China) between
January 2013 and January 2016 were enrolled in the present study.
With HCC being pathologically confirmed, all patients underwent
routine surgery without receiving any pre-surgical anticancer
treatments, such as chemotherapy or radiotherapy. All HCC tissues
and their adjacent normal liver tissues were collected and then
immediately frozen in liquid nitrogen for further analyses.
Clinicopathological information and survival information were
recorded during follow-ups. To determine tumor recurrence, regular
physical examinations were performed for all patients. Overall
survival (OS) was defined as the time interval from the date of
surgery to the date of death or the last follow-up. Recurrence-free
survival (RFS) was defined as the time interval from the date of
surgery to the date of death, recurrence or the last follow-up. The
follow-up data in this present study was censored in December 2019.
All patients included provided written informed consent. The
present study was approved by the ethics committee of the Ningbo
Medical Treatment Center Li Huili Hospital (Ningbo, China) and was
performed in accordance with the Declaration of Helsinki.
Cell culture
The human HCC cell lines Hep3B, LM3 and MHCC-97H and
the cell line transformed human liver epithelial-2 (THLE-2) were
obtained from the Cell Bank of the Type Culture Collection of the
Chinese Academy of Sciences. HCC cells were cultured in DMEM
(Gibco; Thermo Fisher Scientific, Inc.) supplemented with 10% fetal
bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.), 100 U/ml
penicillin and 100 g/ml streptomycin (Sangong Biotech, Inc.) at
37˚C in a humidified atmosphere containing 5% CO2.
RNA extraction and reverse
transcription-quantitative (RT-q)PCR
Total RNA samples from clinical tissues and cell
lines were extracted using TRIzol® reagent (Invitrogen;
Thermo Fisher Scientific, Inc.). The RNA concentration was
determined using a BioDrop (Biochrom Ltd). RT was performed using
the PrimeScript™ RT reagent kit (Takara Bio Inc.) in accordance
with the manufacturer's protocol, following which qPCR was
performed using a SYBR Green I Mastermix (cat. no. SY1020; Beijing
Solarbio Science & Technology Co., Ltd.) on an ABI PRISM 7500
Sequence Detection System (Applied Biosystems; Thermo Fisher
Scientific, Inc.) using the following thermocycling conditions:
Initial denaturation at 95˚C for 4 min, followed by 40 cycles of
95˚C for 15 sec, 60˚C for 30 sec and 72˚C for 30 sec, final
extension at 72˚C for 2 min and a hold at 4˚C. The relative
expression of miR-3677-5p was normalized to that of U6 using the
2-∆∆Cq method (18). All
oligonucleotides used for RT-qPCR were synthesized by RiboBio. The
primer sequences used were as follows: miR-3677-5p forward,
5'-GGGGTACCCCCTGGCTGGAACAGAAGAT-3' and reverse,
5'-CCCAAGCTTCCCTGGTCTTGGCTGGGATC-3' and U6 forward,
5'-CGCTTCGGCAGCACATATAC-3' and reverse,
5'-TTCACGAATTTGCGTGTCATC-3'.
Cell transfection
miR-3677-5p mimics, miR-3677-5p inhibitor and their
corresponding negative controls (NCs) were purchased from Shanghai
GenePharma Co., Ltd. HCC cells were seeded into six-well plates at
a density of 5x105 cells per well and cultured for 24 h
prior to transfection using Lipofectamine® 2000
(Invitrogen; Thermo Fisher Scientific, Inc.) in accordance with the
manufacturer's protocol. After transfection for 48 h, the cells
were used in subsequent experiments. The primer sequences used were
as follows: miR-3677-5p mimics, 5'-UGUUUGGUGUCACACGACGAC-3' and its
NC, 5'-UUCUCCGAACGUGUCACGUTT-3'; miR-3677-5p inhibitor,
5'-UCACAAGUUAGGGUCUCAGGG-3' and its NC,
5'-UUCUCCGAACGUGUCACGUGGC-3'.
Cell Counting Kit-8 (CCK-8) assay
Transfected HCC cells were seeded into 96-well
plates at a density of 5x103 cells in 100 µl culture
medium per well. A total of three replicates were set up for each
experimental condition. Following culture for 24, 48, 72 or 96 h,
10 µl CCK-8 solution (Dojindo Molecular Technologies, Inc.) was
added to the medium and the cells were then incubated at 37˚C for
another 2 h. Optical density (OD) values were measured at 450 nm
using an automatic microplate reader (Bio-Rad Laboratories,
Inc.).
Crystal violet assay
Transfected HCC cells were seeded into six-well
plates at a density of 1,000 cells per well and then cultured in
DMEM medium with 10% FBS. The culture medium was changed every 3
days for 2 weeks at 37˚C in a humidified atmosphere containing 5%
CO2. Following removal of the medium, cells were stained
with crystal violet (1 ml 0.5% crystal violet solution in 10%
formalin) for 10 min at room temperature. Subsequently, these fixed
cells were washed with PBS and images were acquired. The OD was
measured at 570 nm using an automatic microplate reader (Bio-Rad
Laboratories, Inc.).
Transwell assays
The migratory and invasive ability of transfected
HCC cells was examined by Transwell assays using polyethylene
terephthalate membranes (24-well inserts; 8.0 µm pore size;
Corning, Inc.). A total of 150 µl cell suspension containing
1x105 cells in FBS-free medium was added to the upper
chambers. The lower chambers contained 500 µl medium supplemented
with 10% FBS. For the invasion assay, the membrane was pre-coated
with 50 µl Matrigel® (BD Biosciences) at 37˚C for 3 h.
Cells were maintained in a humidified atmosphere with 5%
CO2 at 37˚C for 48 h. Cells that had transgressed
through the membrane to reach the lower side were fixed with 100%
methanol for 20 min at room temperature prior to being stained with
0.1% crystal violet for 10 min at room temperature. Cells were
counted in five randomly selected fields under a light microscope
(Olympus Corp.) at x400 magnification.
Statistical analysis
Values are expressed as the mean ± standard
deviation. All experiments were performed in ≥ three replicates.
Statistical analyses were performed using GraphPad Prism 5.0
(GraphPad Software, Inc.) and SPSS 20.0 (IBM Corp.). Differences
between two groups were analyzed by unpaired Student's t-tests,
while the expression of miR-3677-5p in tumor tissues and matched
normal tissues was compared using a paired Student's t-test.
Comparisons of multiple groups were performed using one-way ANOVA
followed by Dunnett's test. Categorical data were compared using a
χ2 test. OS and RFS of patients with HCC of patients
were evaluated by Kaplan-Meier curves and compared with log-rank
tests. Prognostic factors were analyzed by univariate and
multivariate logistic regression analysis using the Cox
proportional hazards model. P<0.05 was considered to indicate a
statistically significant difference.
Results
miR-3677-5p is upregulated in HCC
tissues and cell lines
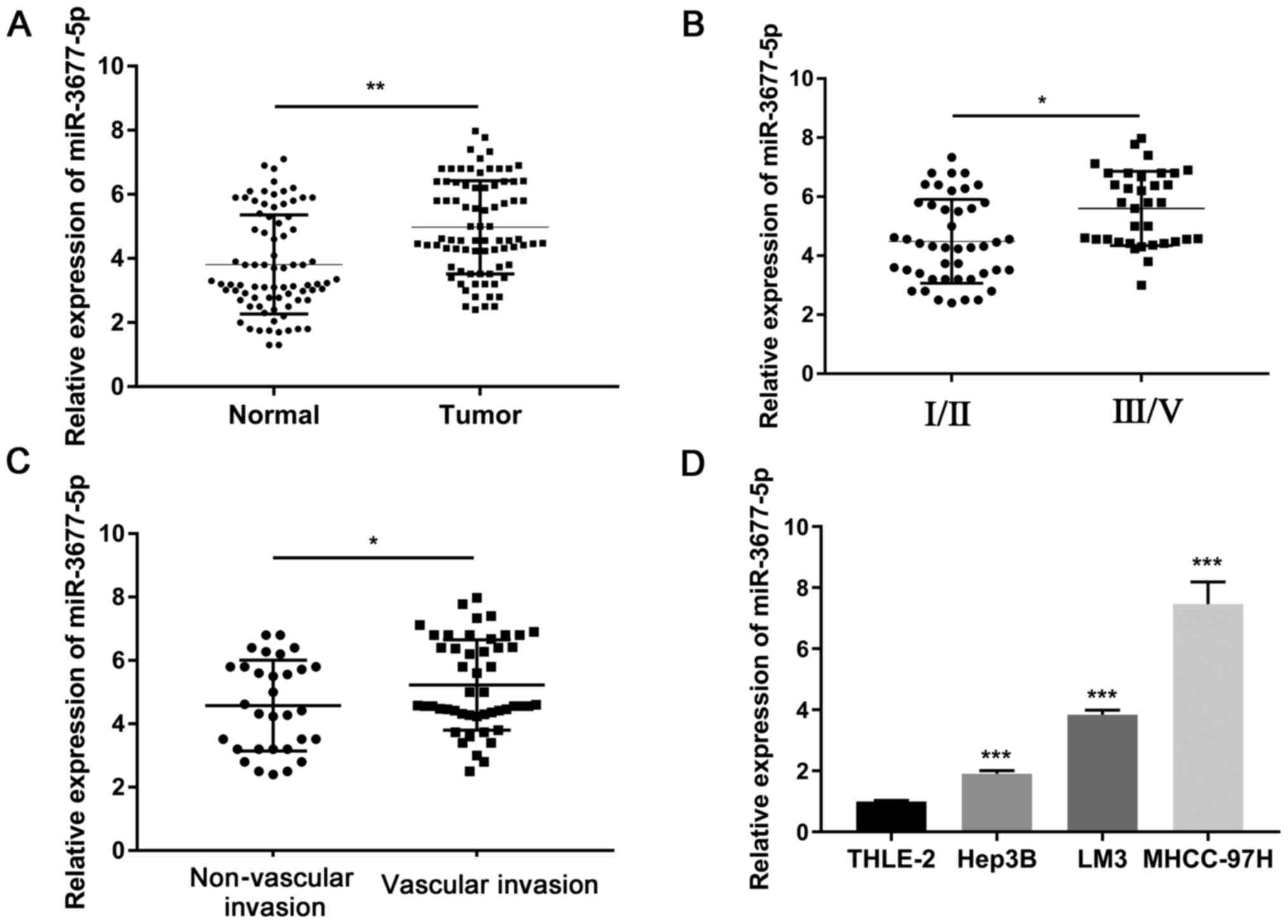
First, the expression levels of miR-3677-5p in 80
pairs of HCC tissues and paired adjacent normal tissues were
determined using RT-qPCR. As presented in Fig. 1A, the expression of miR-3677-5p was
significantly elevated in tumor tissues as compared with that in
matched normal tissues (P<0.01). Furthermore, the expression of
miR-3677-5p was significantly higher in the tumor tissues of
patients with TNM III/V than in those with TNM Ⅰ/II (P<0.05;
Fig. 1B). The expression of
miR-3677-5p was significantly higher in the tumor tissues of
patients with vascular invasion than in those without vascular
invasion (P<0.05; Fig. 1C). In
addition, the expression levels of miR-3677-5p were evaluated in
several HCC cell lines (Hep3B, LM3 and MHCC-97H) and in the
non-cancerous cell line THLE-2. As presented in Fig. 1D, the expression of miR-3677-5p was
significantly higher in HCC cell lines than that in THLE-2 cells
(all P<0.001).
Clinical significance of miR-3677-5p
expression in patients with HCC
Next, the association between miR-3677-5p expression
and clinicopathologic features was analyzed in patients with HCC.
Using the median value of miR-3677-5p expression in HCC tumor
tissues being as the cut-off value, patients with HCC were divided
into two groups: miR-3677-5p low (below the median, n=40 patients)
and miR-3677-5p high (above the median, n=40 patients). As
indicated in Table I, the number of
patients with α-fetoprotein (AFP)≥400 ng/ml and tumor size≥5 cm was
higher in the high miR-3677-5p expression group than that in the
low miR-3677-5p expression group (P=0.008 and 0.026, respectively).
The number of patients with TNM stage III/IV was significantly
higher in the high miR-3677-5p expression group (P=0.001). In
addition, patients with vascular invasion were more common in the
high miR-3677-5p expression group than in the low miR-3677-5p
expression group (P=0.012). However, there was no significant
difference between the two groups in terms of age, sex, hepatitis B
surface antigen (HBsAg) status, tumor number and satellite
nodules.
 | Table IAssociation between miR-3677-5p
expression and the clinicopathologic features in hepatocellular
carcinoma. |
Table I
Association between miR-3677-5p
expression and the clinicopathologic features in hepatocellular
carcinoma.
| | miR-3677-5p | |
|---|
| Item | Total (n=80) | Low expression
(n=40) | High expression
(n=40) | P-value |
|---|
| Age (years) | | | | 0.370 |
|
<60 | 38 | 17 (42.5) | 21 (52.5) | |
|
≥60 | 42 | 23 (57.5) | 19 (47.5) | |
| Sex | | | | 0.556 |
|
Male | 14 | 6 (15.0) | 8 (20.0) | |
|
Female | 66 | 34 (85.0) | 32 (80.0) | |
| HBsAg | | | | 0.390 |
|
Negative | 15 | 9 (22.5) | 6 (15.0) | |
|
Positive | 65 | 31 (77.5) | 34 (85.0) | |
| AFP (ng/ml) | | | | 0.008 |
|
<400 | 25 | 18 (45.0) | 7 (17.5) | |
|
≥400 | 55 | 22 (55.0) | 33 (82.5) | |
| Tumor number | | | | 0.478 |
|
Single | 53 | 28 (70.0) | 25 (62.5) | |
|
Multiple | 27 | 12 (30.0) | 15 (37.5) | |
| Tumor size
(cm) | | | | 0.026 |
|
<5 | 40 | 16 (69.6) | 7 (42.1) | |
|
≥5 | 40 | 24 (30.4) | 33 (57.9) | |
| Satellite
nodules | | | | 0.626 |
|
Negative | 56 | 29 (72.5) | 27 (67.5) | |
|
Positive | 24 | 11 (27.5) | 13 (32.5) | |
| TNM stage | | | | 0.001 |
|
Ⅰ/II | 45 | 30 (75.0) | 15 (37.5) | |
|
III/V | 35 | 10 (25.0) | 25 (62.5) | |
| Vascular
invasion | | | | 0.012 |
|
Negative | 31 | 21 (52.5) | 10 (25.0) | |
|
Positive | 49 | 19 (47.5) | 30 (75.0) | |
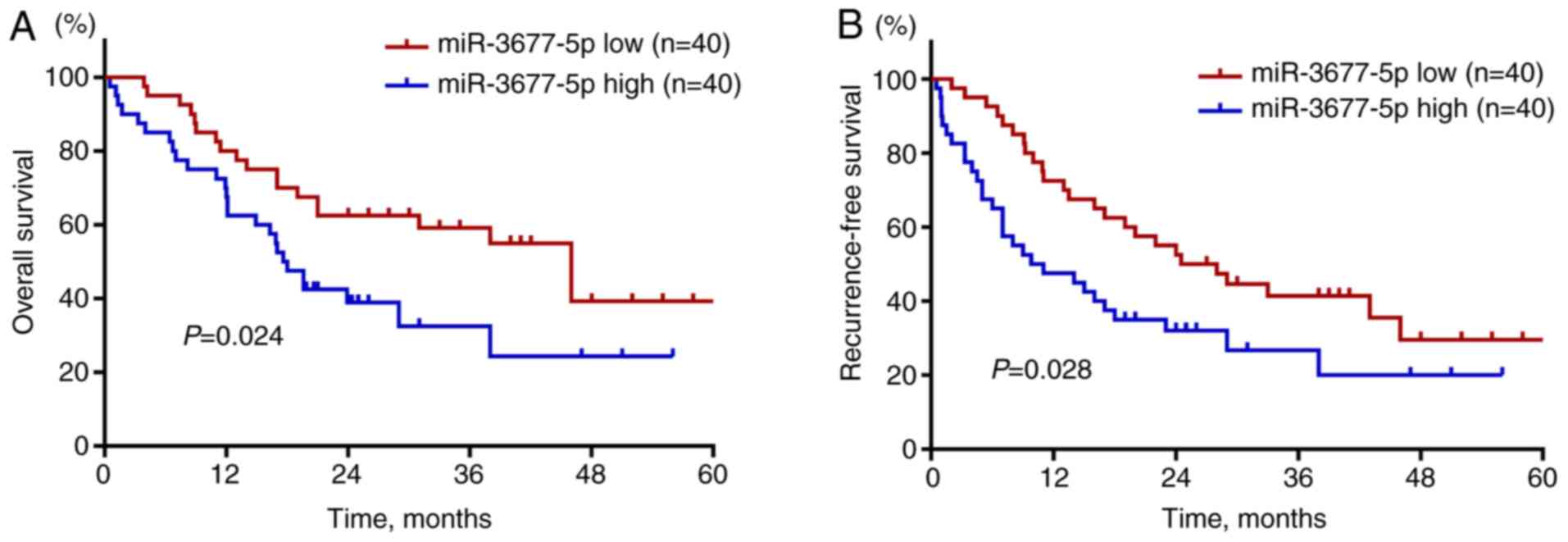
The prognostic value of miR-3677-5p in patients with
HCC was then investigated. As indicated by Kaplan-Meier curves,
patients with high expression of miR-3677-5p in their HCC tissues
were indicated to have poor OS (P=0.024; Fig. 2A) and RFS (P=0.028; Fig. 2B). Next, the prognostic factors
affecting OS and RFS were evaluated by multivariate Cox regression
analysis. The prognostic factors for OS (Table II) are summarized as follows:
AFP≥400 ng/ml [hazard ratio (HR): 1.221, 95% confidence interval
(CI): 1.101-1.413, P=0.031], TNM stage III/V (HR: 2.119, 95% CI:
1.885-2.429, P<0.001), vascular invasion (HR: 2.675, 95% CI:
2.553-2.764, P<0.001) and high expression of miR-3677-5p (HR:
1.632, 95% CI: 1.287-2.015, P=0.019). The prognostic factors for
RFS (Table III) were as follows:
TNM stage III/V (HR: 2.101, 95% CI: 1.801-2.410, P<0.001),
vascular invasion (HR: 2.591, 95% CI: 2.391-2.846, P<0.001) and
high expression of miR-3677-5p (HR: 1.781, 95% CI: 1.210-2.337,
P=0.033). Taken together, the above results suggested that high
expression of miR-3677-5p is associated with unfavorable prognosis
of patients with HCC.
 | Table IIUnivariate and multivariate analysis
of clinical features influencing overall survival. |
Table II
Univariate and multivariate analysis
of clinical features influencing overall survival.
| | Univariate | Multivariate |
|---|
| Factor | HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Age, ≥60 years | 1.021
(0.812-1.191) | 0.667 | | |
| Sex, male | 1.008
(0.818-1.411) | 0.501 | | |
| HBsAg,
positive | 1.312
(0.901-1.714) | 0.091 | | |
| AFP, ≥400
ng/ml | 1.263
(1.113-1.498) | 0.001 | 1.221
(1.101-1.413) | 0.031 |
| Tumor number,
multiple | 1.283
(0.816-1.964) | 0.274 | | |
| Tumor size, ≥5
cm | 1.512
(0.912-2.321) | 0.012 | | |
| Satellite nodules,
positive | 1.612
(0.712-2.265) | 0.865 | | |
| TNM stage,
III/V | 2.248
(1.969-2.576) | <0.001 | 2.119
(1.855-2.429) | <0.001 |
| Vascular invasion,
positive | 2.985
(2.960-3.014) | <0.001 | 2.675
(2.533-2.764) | <0.001 |
| miR-3677-5p
expression, high | 1.798
(1.314-2.579) | <0.001 | 1.632
(1.287-2.015) | 0.019 |
 | Table IIIUnivariate and multivariate analysis
of clinical features influencing recurrence-free survival. |
Table III
Univariate and multivariate analysis
of clinical features influencing recurrence-free survival.
| | Univariate | Multivariate |
|---|
| Factor | HR (95% CI) | P-value | HR (95% CI) | P-value |
|---|
| Age, ≥60 years | 1.041
(0.856-1.215) | 0.312 | | |
| Sex, male | 1.011
(0.876-1.398) | 0.156 | | |
| HBsAg,
positive | 1.212
(0.816-1.651) | 0.187 | | |
| AFP, ≥400
ng/ml | 1.119
(0.876-1.374) | 0.056 | | |
| Tumor number,
multiple | 1.339
(0.781-1.816) | 0.671 | | |
| Tumor size, ≥5
cm | 1.831
(0.761-2.879) | 0.287 | | |
| Satellite nodules,
positive | 1.871
(0.691-2.995) | 1.010 | | |
| TNM stage,
III/V | 2.345
(1.919-2.567) | <0.001 | 2.101
(1.801-2.410) | <0.001 |
| Vascular invasion,
positive | 2.771
(2.461-3.006) | <0.001 | 2.591
(2.391-2.846) | <0.001 |
| miR-3677-5p
expression, high | 1.910
(1.417-2.671) | 0.001 | 1.781
(1.210-2.337) | 0.033 |
Overexpression of miR-3677-5p promotes
the proliferation, migration and invasion of HCC cells
Based on the clinical findings, further experiments
were performed to explore the biological functions of miR-3677-5p
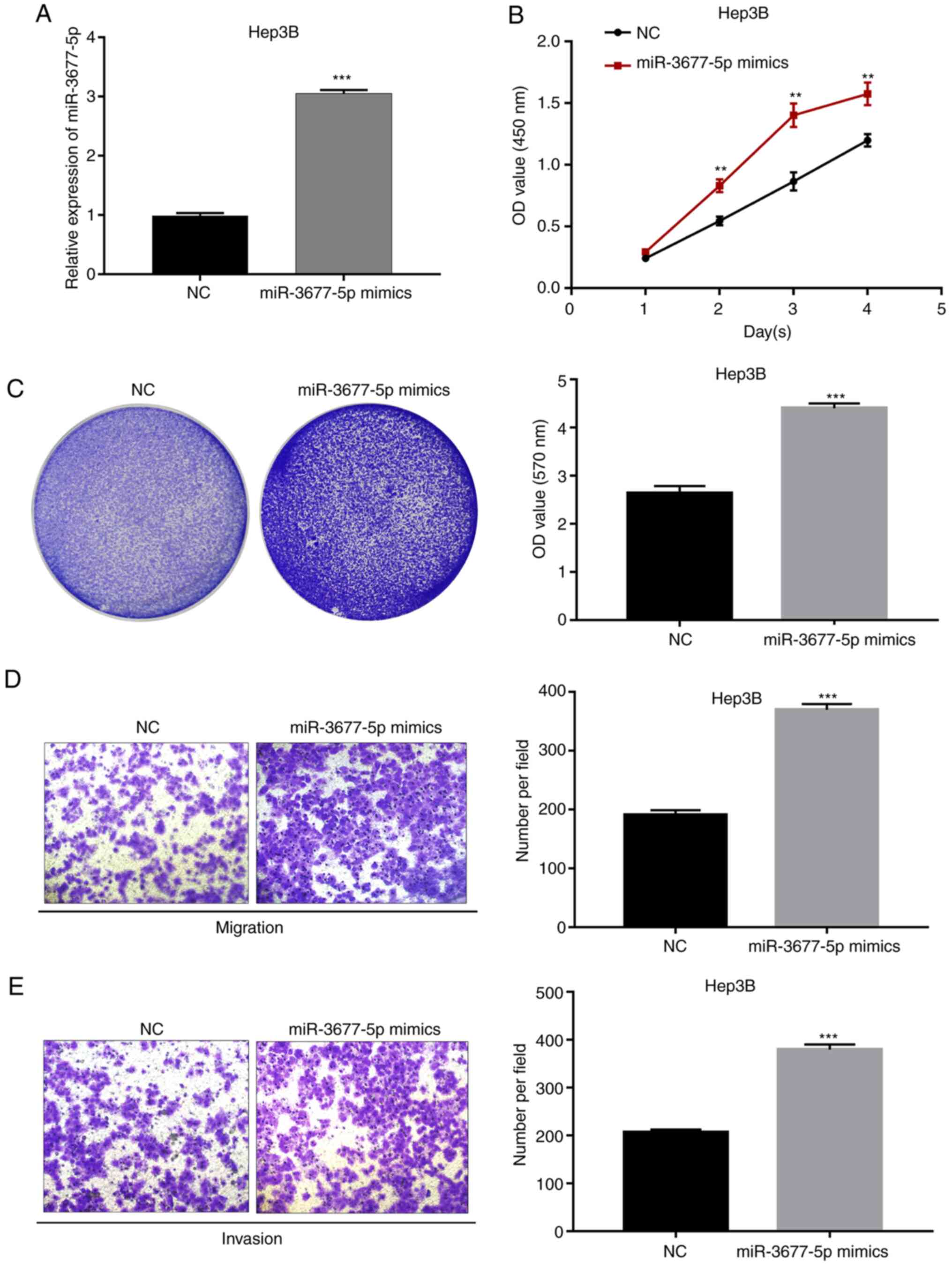
in the progression of HCC. Using miR-3677-5p mimics, miR-3677-5p
was first overexpressed in Hep3B cells; this cell line was chosen
because the native cells had a relatively low expression of
miR-3677-5p. Significant miR-3677-5p overexpression induced by
miR-3677-5p mimics in Hep3B cells was verified by RT-qPCR
(P<0.001; Fig. 3A). Subsequent
evaluation by a CCK-8 assay revealed that the OD values for the
miR-3677-5p mimics-transfected group of Hep3B cells at 2, 3 and 4
days were significantly higher than those of miR-NC-transfected
cells (all P<0.01; Fig. 3B).
Next, the crystal violet assay indicated that miR-3677-5p
overexpression significantly promoted the proliferation of Hep3B
cells (P<0.001; Fig. 3C). In
order to explore the effect of miR-3677-5p on the migratory and
invasive capacities of Hep3B cells, Transwell assays were
performed. As presented in Fig. 3D
and E, miR-3677-5p overexpression
significantly promoted the migration and invasion of Hep3B cells
(all P<0.001).
Knockdown of miR-3677-5p inhibits the
proliferation, migration and invasion of HCC cells
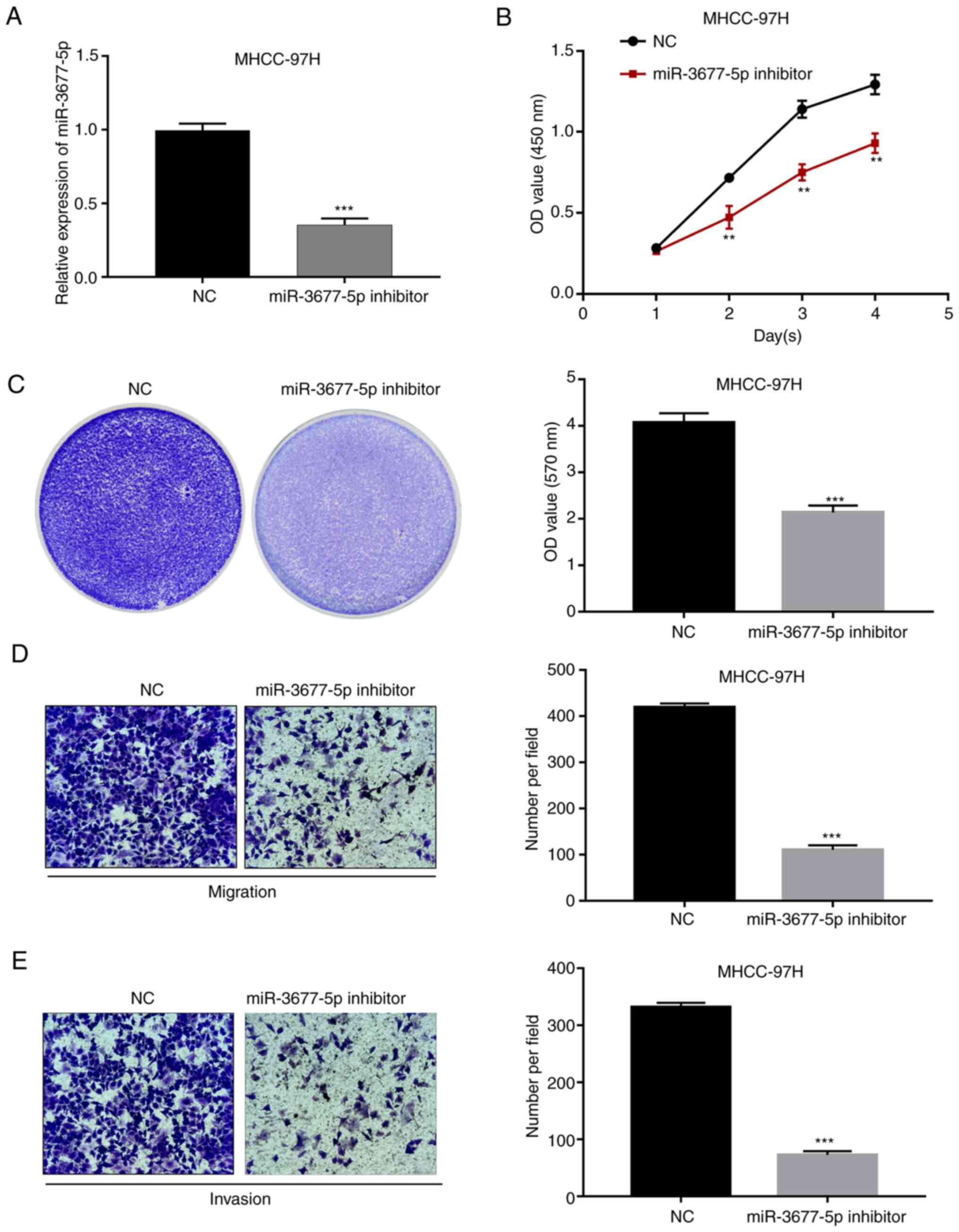
miR-3677-5p inhibitor was further used to knock down
the expression of miR-3677-5p in MHCC-97H cells. This cell line was
chosen as the native cells exhibited relatively high expression of
miR-3677-5p. The knockdown efficacy was confirmed by RT-qPCR
(P<0.001; Fig. 4A). Next,
evaluation by CCK-8 and crystal violet assays indicated that
miR-3677-5p downregulation significantly inhibited the
proliferation of MHCC-97H cells (all P<0.01; Fig. 4B and C). In addition, analysis by Transwell
assays indicated a significantly decreased cell migratory and
invasive capacity of miR-3677-5p inhibitor-transfected MHCC-97H
cells compared with that in cells from the NC group (all
P<0.001; Fig. 4D and E).
Discussion
To date, a variety of miRNAs that exert fundamental
roles in the progression of HCC have been proposed as promising
prognostic predictors (19). For
instance, Budhu et al (20)
have reported a 20-miRNA metastasis signature capable of predicting
HCC with vascular metastases that is associated with patient
survival. It has also been demonstrated that the combination of
miR-10b, miR-106b and miR-181a is able to discriminate patients
with HCC from normal controls (21). In addition, miRNAs have been
reported to function as oncogenes or tumor suppressors in the
regulation of fundamental biological processes, such as cell
proliferation, migration and invasion, of HCC cells (22). Xie et al (23) demonstrated that miR-6875-3p promotes
the proliferation, invasion and metastasis of HCC cells.
Consistently, You et al (24) reported that ectopic miR-766-3p
expression inhibits HCC cell proliferation, colony formation,
migration and invasion.
Previously, the functions of miR-3677 in
tumorigenesis were poorly understood. Zhang et al (12) have generated a 7-miRNA signature in
patients with HCC based on The Cancer Genome Atlas database, in
which miR-3677 has been identified as one of the significant miRNAs
that are able predict poor survival of patients with HCC. In
agreement with this, Lu et al (14) also reported that miR-3677 is
significantly associated with survival of patients with HCC.
Through microarray analysis, Żorniak et al (25) determined that miR-3667 is highly
expressed in cirrhotic patients with gastric antral vascular
ectasia. miR-3677 has also been identified as a key oncogenic miRNA
in breast cancer and significant differences in prognosis were
obtained when stratifying patients into high-risk and low-risk
groups (26). Furthermore, Peng
et al (27) revealed that
overexpression of miR-3677 promoted the proliferation, migration
and invasion of breast cancer cells. Recently, Yao et al
(28) reported that miR-3677-3p
overexpression promoted the malignant behavior and invasiveness of
HCC via suppression of sirtuin 5. Another study that determined the
oncogenic role of miR-3677-3p in HCC indicated that knockdown of
miR-3677-3p inhibited the proliferation of HCC cells via directly
targeting the 3'untranslated region of glycogen synthase kinase 3-β
(29).
In the present study, it was determined that
miR-3677-5p expression was upregulated in human HCC tissues and
cell lines compared with that in adjacent normal tissues and a
non-cancerous liver cell line, respectively. Furthermore, it was
demonstrated that the expression of miR-3677-5p is closely
associated with AFP, tumor size, TNM stage and vascular invasion.
Of note, high miR-3677-5p expression was indicated to be capable of
predicting unfavorable clinical prognosis in patients with HCC.
These results demonstrated the prognostic value, as well as the
oncogenic role of miR-3677-5p in HCC. Subsequently, a functional
in vitro study was performed and miR-3677-5p was
overexpressed or knocked down in two HCC cell lines using
miR-3677-5p mimics or inhibitor, respectively. Subsequently, the
effects of miR-3677-5p on cell proliferation, migration and
invasion were investigated. The results suggested that miR-3677-5p
overexpression promoted the proliferation, migration and invasion
of HCC cells. Conversely, miR-3677-5p knockdown resulted in an
inhibitory effect. To the best of our knowledge, the present study
was the first to determine the biological roles of miR-3677-5p in
HCC and its progression, as well as its prognostic value. However,
the present study remains preliminary and the clinical application
of miR-3677-5p and the mechanisms of how it promotes the
progression of HCC cells have yet to be established. Further
studies that focus on the identification of direct target genes of
miR-3677-5p, as well as the underlying regulatory mechanisms, are
required. In addition, animal models may be used to validate the
functions of miR-3677-3p in vivo (30).
In conclusion, the present study demonstrated that
miR-3677-5p acts as an oncogene and has a critical role in the
regulation of HCC cell proliferation and progression. The present
results indicated that miR-3677-5p may be a valuable prognostic
biomarker and may facilitate the development of a promising
treatment strategy for patients with HCC.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HXM, KJQ and BYC designed the study and performed
the experiments. JW, CYM and YCG collected the clinical samples and
analyzed the data. All authors read and approved the final version
of the manuscript. HXM and KJQ checked and approved the
authenticity of the raw data.
Ethics approval and consent to
participate
All included patients provided written informed
consent. The present study was approved by the ethics committee of
the Ningbo Medical Treatment Center Li Huili Hospital (Ningbo,
China) and was conducted in accordance with the Declaration of
Helsinki.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Lu XJ, Shi Y, Chen JL and Ma S:
Kruppel-like factors in hepatocellular carcinoma. Tumour Biol.
36:533–541. 2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Yu WB, Rao A, Vu V, Xu L, Rao JY and Wu
JX: Management of centrally located hepatocellular carcinoma:
Update 2016. World J Hepatol. 9:627–634. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Portolani N, Coniglio A, Ghidoni S,
Giovanelli M, Benetti A, Tiberio GA and Giulini SM: Early and late
recurrence after liver resection for hepatocellular carcinoma:
Prognostic and therapeutic implications. Ann Surg. 243:229–235.
2006.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Hu J and Gao DZ: Distinction immune genes
of hepatitis-induced heptatocellular carcinoma. Bioinformatics.
28:3191–3194. 2012.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Marquardt JU, Galle PR and Teufel A:
Molecular diagnosis and therapy of hepatocellular carcinoma (HCC):
An emerging field for advanced technologies. J Hepatol. 56:267–275.
2012.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Tutar Y: miRNA and cancer; computational
and experimental approaches. Curr Pharm Biotechnol.
15(429)2014.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Qadir MI and Faheem A: miRNA: A diagnostic
and therapeutic tool for pancreatic cancer. Crit Rev Eukaryot Gene
Expr. 27:197–204. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Sun Z, Shi K, Yang S, Liu J, Zhou Q, Wang
G, Song J, Li Z, Zhang Z and Yuan W: Effect of exosomal miRNA on
cancer biology and clinical applications. Mol Cancer.
17(147)2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Sartorius K, Sartorius B, Winkler C,
Chuturgoon A and Makarova J: The biological and diagnostic role of
miRNA's in hepatocellular carcinoma. Front Biosci (Landmark Ed).
23:1701–1720. 2018.PubMed/NCBI View
Article : Google Scholar
|
|
10
|
Mao B and Wang G: MicroRNAs involved with
hepatocellular carcinoma (Review). Oncol Rep. 34:2811–2820.
2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
He S, Zhang DC and Wei C: MicroRNAs as
biomarkers for hepatocellular carcinoma diagnosis and prognosis.
Clin Res Hepatol Gastroenterol. 39:426–434. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zhang J, Chong CC, Chen GG and Lai PB: A
Seven-microRNA expression signature predicts survival in
hepatocellular carcinoma. PLoS One. 10(e0128628)2015.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Qin L, Huang J, Wang G, Huang J, Wu X, Li
J, Yi W, Qin F and Huang D: Integrated analysis of clinical
significance and functional involvement of microRNAs in
hepatocellular carcinoma. J Cell Physiol. 234:23581–2395.
2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Lu M, Kong X, Wang H, Huang G, Ye C and He
Z: A novel microRNAs expression signature for hepatocellular
carcinoma diagnosis and prognosis. Oncotarget. 8:8775–8784.
2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Nagy A, Lanczky A, Menyhart O and Gyorffy
B: Validation of miRNA prognostic power in hepatocellular carcinoma
using expression data of independent datasets. Sci Rep.
8(9227)2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Chen W, Gao C, Liu Y, Wen Y, Hong X and
Huang Z: Bioinformatics analysis of prognostic miRNA signature and
potential critical genes in colon cancer. Front Genet.
11(478)2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Zhang H, Wang Z, Ma R, Wu J and Feng J:
MicroRNAs as biomarkers for the progression and prognosis of colon
carcinoma. Int J Mol Med. 42:2080–2088. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Villanueva A, Hoshida Y, Toffanin S,
Lachenmayer A, Alsinet C, Savic R, Cornella H and Llovet JM: New
strategies in hepatocellular carcinoma: Genomic prognostic markers.
Clin Cancer Res. 16:4688–4694. 2010.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Budhu A, Jia HL, Forgues M, Liu CG,
Goldstein D, Lam A, Zanetti KA, Ye QH, Qin LX, Croce CM, et al:
Identification of metastasis-related microRNAs in hepatocellular
carcinoma. Hepatology. 47:897–907. 2008.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Jiang L, Cheng Q, Zhang BH and Zhang MZ:
Circulating microRNAs as biomarkers in hepatocellular carcinoma
screening: A validation set from China. Medicine (Baltimore).
94(e603)2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Petrini E, Caviglia GP, Abate ML, Fagoonee
S, Smedile A and Pellicano R: MicroRNAs in HBV-related
hepatocellular carcinoma: Functions and potential clinical
applications. Panminerva Med. 57:201–209. 2015.PubMed/NCBI
|
|
23
|
Xie Y, Du J, Liu Z, Zhang D, Yao X and
Yang Y: miR-6875-3p promotes the proliferation, invasion and
metastasis of hepatocellular carcinoma via BTG2/FAK/Akt pathway. J
Exp Clin Cancer Res. 38(7)2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
You Y, Que K, Zhou Y, Zhang Z, Zhao X,
Gong J and Liu Z: MicroRNA-766-3p inhibits tumour progression by
targeting Wnt3a in hepatocellular carcinoma. Mol Cells. 41:830–841.
2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Żorniak M, Garczorz W, Wosiewicz P, Marek
T, Błaszczyńska M, Waluga M, Kukla M, Kimsa-Furdzik M, Francuz T
and Hartleb M: Mucosal miR-3677 is over-expressed in cirrhotic
patients with gastric antral vascular ectasia (GAVE). Scand J
Gastroenterol. 53:1503–1508. 2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Lu DC, Han W and Lu K: Identification of
key microRNAs involved in tumorigenesis and prognostic microRNAs in
breast cancer. Math Biosci Eng. 17:2923–2935. 2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Peng LN, Deng XY, Gan XX, Zhang JH, Ren
GH, Shen F, Feng JH, Cai WS and Xu B: Targeting of TLE3 by miR-3677
in human breast cancer promotes cell proliferation, migration and
invasion. Oncol Lett. 19:1409–1417. 2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Yao B, Li Y, Niu Y, Wang L, Chen T, Guo C
and Liu Q: Hypoxia-induced miR-3677-3p promotes the proliferation,
migration and invasion of hepatocellular carcinoma cells by
suppressing SIRT5. J Cell Mol Med. 24:8718–8731. 2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Li Y, Zhou Y, Ma L, Liu D, Dai Z and Shen
J: miR-3677-3p promotes hepatocellular carcinoma progression via
inhibiting GSK3β. Acta Biochim Biophys Sin (Shanghai).
52:1404–1412. 2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Robinson NB, Krieger K, Khan FM, Huffman
W, Chang M, Naik A, Yongle R, Hameed I, Krieger K, Girardi LN and
Gaudino M: The current state of animal models in research: A
review. Int J Surg. 72:9–13. 2019.PubMed/NCBI View Article : Google Scholar
|