Introduction
Trends pertaining to antimicrobial endodontic
irrigation solutions and of nanoparticles used in root canal
filling materials are focused on disinfection of the endodontic
system and prevention of the development of bacterial biofilm.
Since failures of endodontic treatment are present in clinical
practice, there is continuous development of endodontic irrigating
solutions and of root canal filling materials. More specifically,
nanotechnology helps to create new materials with biologic
applications, including dental biomaterials (1).
Dental nano-biomaterials are based on polymeric or
metallic nanoparticles, with demonstrated antimicrobial effect,
which are incorporated in filling materials or are used as
adjuvants in root canal cleaning to eliminate the presence of
microorganisms (2). Silver colloid
is of special interest because of its distinctive properties as
good conductivity, chemical stability, and catalytic and
antibacterial activity. Silver diamine fluoride
[Ag(NH3)2F] solution has been used in the
prevention and progression of dental caries. When silver diamine
fluoride is used at a concentration of 3.8% as a root canal
irrigant or as interim medication, it has potential activity in the
reduction of bacterial adhesion (3).
Silver colloid is considered a ‘natural antibiotic’
from antiquity, being non-toxic for human cells, but highly toxic
for some bacterial species, such as E. coli,
Staphylococcus aureus, and others (2). It is an aqueous solution of
ultra-small silver nanoparticles, with 1-9 nanometers diameter in
distilled water, with the antimicrobial activity of silver colloid
being very similar to that of antibiotics with large spectrum. Its
presence activates an enzyme with local action against each
bacteria, virus, and fungi. Silver colloid establishes the
acid-base equilibrium and initiates external resistance. At this
colloid, polymorph or mutant microorganism is extremely sensitive
(2).
Gold nanoparticles are generally 1-100 nm particles
usually suspended in a fluid (colloidal gold). Known as Nanogold,
colloidal gold suspension is usually burgundy red (for spherical
gold nanoparticles with a diameter <100 nm), or yellow brown
(for higher diameter of the nanoparticles). The diameter of
nanoparticles plays an indispensable role in nanotechnology and is
crucial for all biological applications. In recent years, gold
nanoparticles have been intensely studied because of their optical
properties, with large applications in biomedicine (4-10).
As elementary silver liberates ions and gold is known for its
optical properties and the absorption of proteins, the combination
of these two elements, especially in one single material, has been
tested to obtain a synergistic effect of their properties. For
example, the utilization of silver nanoparticles in combination
with gold can create new possibilities for materials with
antibacterial activity (1).
Two new irrigating solutions based on gold and
silver nanoparticles have been introduced in endodontic practice:
NanoCare Plus Silver Gold and NanoCare Gold (Nanotechnology Dental,
Poland). NanoCare Plus Silver Gold is a pharmaceutical complex with
long-lasting bacteriostatic activity, indicated as a final irrigant
in root canal treatment. The aim of this solution is to complete
the cleaning of the endodontic system obtained after conventional
irrigation protocol. Sodium hypochlorite is used as the main
endodontic irrigant as, owing to its germicide and proteolitic
activity, it eliminates most of the organic substances from the
root canals. Nanocare Plus Silver Gold acts as a supplementary
cleaner of organic rests and because of the gold and silver
nanoparticles contained, prevents the bacterial re-colonization
inside the root canal system. The final step of the root canal
filling is not negatively influenced by the corrosion of gold and
silver nanoparticles and by their minimal concentration in the
irrigating solution (11,12).
NanoCare Gold was developed with the purpose of
immediate use prior to filling, cementation of a prosthetic
restoration, veneer or inlay/onlay, or covering the entire surface
of the tooth with a layer of nanoparticles. Due to the chemical
neutrality of silver and gold nanoparticles, the components of
NanoCare Gold do not interact with the products usually used in
dental treatment, thus, their properties are preserved (11,12).
Moreover, the nanosilver and nanogold particles from its structure
do not deactivate by the light used in photo polymerization of the
restoration. NanoCare Gold has a positive influence on the adhesion
of composite materials to dentin, which increases the longevity of
the restoration (a lower risk of micro-fissure occurrence). It is
very important that NanoCare Gold does not change the color of the
final restoration (11,12).
Optical Coherence Tomography (OCT) was first
reported as a new imagistic method by Huang et al (13) in 1991. Since then, it has a lot of
clinical applications in medical diagnosis, including the field of
dentistry. OCT is a non-invasive, non-radioactive optical
diagnostic method based on low-coherence interferometry, which
achieves images with different orientation, with a depth resolution
of 10 µm; after their importation in numerical simulation softs, it
creates a three-dimensional reconstruction and interpretation of
the analyzed structures. In dentistry, the major advantage of OCT
is represented by the localization and characterization of the
smallest defects in hard dental tissues or dental materials, and of
the smallest details in dental anatomy, such as supplementary
canals, recesses, isthmuses, or intra-radicular connections.
Structural defects existent in the root canal filling material,
rests of materials or debris, even at a level of nanoparticles,
present in the canal lumen can be observed by using this method.
Another advantage of this method is that it is total non-invasive
and does not deteriorate the analyzed samples (12-15).
The aim of the present in vitro study was to
evaluate using c-scan en-face optical coherence tomography
(OCT) the optical opacity and the distribution inside the root
canal lumen of several extracted human teeth, of silver and gold
nanoparticles from special irrigating solutions used in endodontic
treatment.
Materials and methods
Materials
Twelve root canals with different apical curvature
from 5 human teeth extracted for periodontal reasons, after
informed consent of the patients was obtained (1 monoradicular-1
canal, 1 first upper premolar-2 canals, 1 upper molar-3 canals and
2 lower molars-each with 3 canals) were instrumented using the
ProTaper Universal system (Dentsply Maillefer) after initial
negotiation with hand K-files ISO #10 and rotary nickel-titanium
(NiTi) PathFile instruments (Dentsply Maillefer). The root canals
have been shaped in a crown-down technique, using the following
working sequence of the ProTaper system: S1-S2-F1-F2-F3, at 300 rpm
and torque settings according to the manufacturer's instructions,
using a conventional irrigation protocol, with common use in
clinical endodontic practice. The main irrigant used for each root
canal during shaping procedures was sodium hypochlorite NaOCl 5.25%
(10 ml), in alternation with ethylenediaminetetraacetic acid EDTA
17% solution (2 ml) and distilled water. The root canals were dried
with sterile paper points at the end of cleaning and shaping until
they were completely dried and no irrigant remained inside.
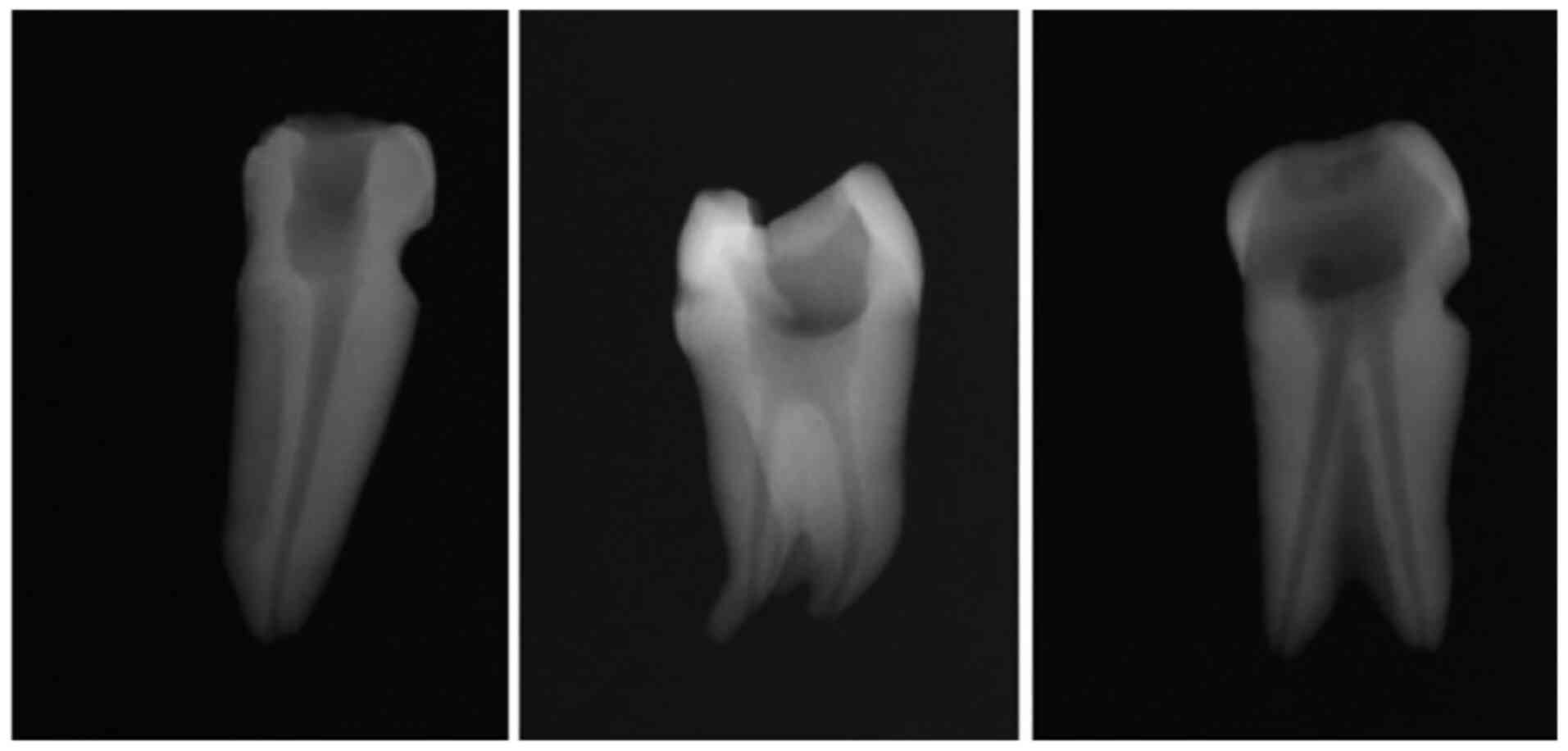
Periapical radiographs (Fig. 1)
were taken for each of the root canals included in the study to
confirm the quality of the endodontic preparation.
Evaluation of root canals
A Time Domain Optical Coherence Tomography (TD-OCT)
setup with the new dynamic focus method was employed for this
evaluation. The output optical power used by the system was between
1 and 3.5 mW, using a Thorlab PM100 device. The system was working
in the range of 1,250-1,360 nm (OSA) (model no. HP70950B) and the
width of the optical band was >25 nm (OSA) (model no. HP70950B).
The dynamic focus method requires only that the object arm (not the
reference arm) is placed on the translation stage. By allowing
physical separation of the two interferometer arms, the dynamic
focus method removes a design constraint which restricts the layout
of more complex systems such as those employing adaptive optics. In
addition, it permits the design of a probe-head which is both
smaller and can be positioned independently of the rest of the OCT
system. The TD-OCT system operates both C-scan (en-face) and
B-scan (cross-sectional) sections.
Grouping
An initial C-scan OCT analysis was performed for
each of the samples to confirm that the root canal lumen is empty
from radiopaque materials: Group 1 (Control group) (Fig. 2). The C-scan OCT parameters used
were: 1,300 nm, 100 sections/1 mm, with a distance of 0.01±0.005
mm. The samples were locked in fixed positions in a block of putty
silicone and placed at 20.5 mm from the OCT lens (Fig. 2). The C-scan OCT investigation
started at the apex of the tooth, at a depth of 1 mm from its tip.
Then, 100 slices of 10 microns were obtained.
The same 12 samples were used as two other study
groups, according to the non-conventional irrigating protocol that
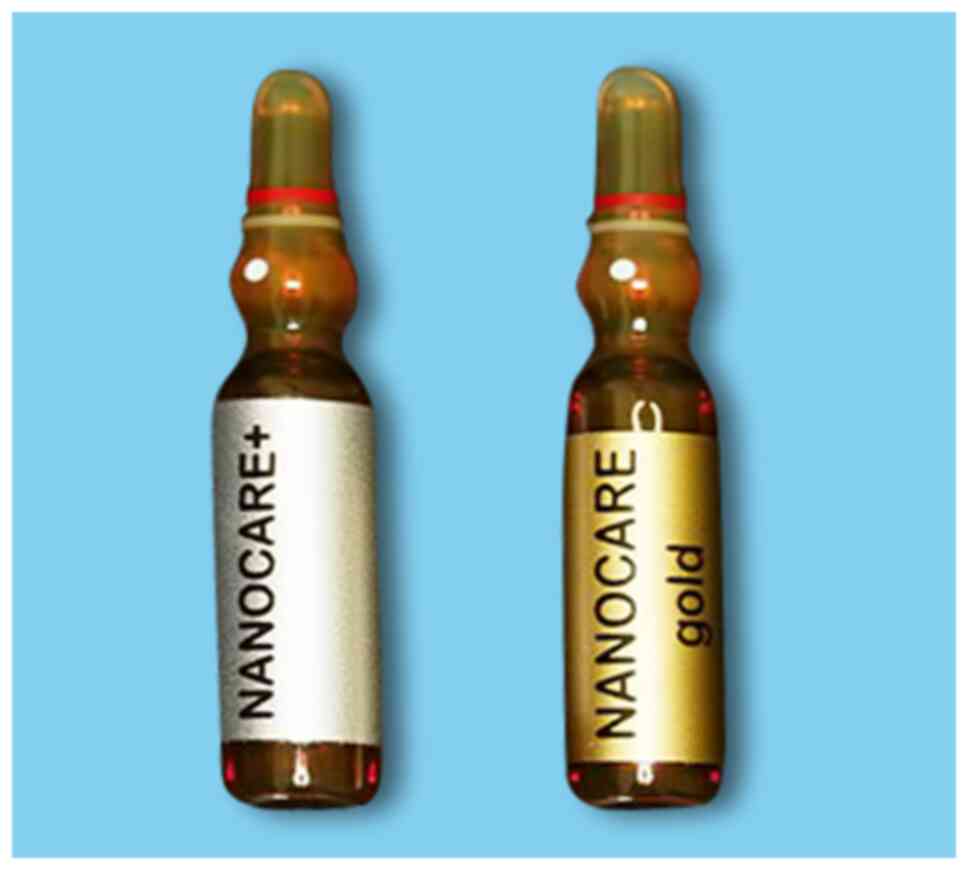
followed, by using the nanoparticle irrigating solutions. In Group
2 (n=12), only 1 ml of NanoCare Plus Silver Gold (Dental
Nanotechnology Company) was used as supplementary irrigation for
each sample (Fig. 3). The irrigant
was placed into the root canal with a 25-Gauge needle introduced on
the entire working length. No aspiration followed, and teeth were
evaluated with another OCT investigation. In Group 3 (n=12), 1 ml
of NanoCare Gold (Dental Nanotechnology Company) was additionally
used for irrigation in each sample (Fig. 3) and C-scan OCT analysis was again
performed to determine whether there is a difference in the
imagistic appearance of the levels of grey in the root canal lumen
comparative to the initial OCT images (Group 1) or with the first
non-conventional irrigation protocol (Group 2). The time between
the irrigation and fixation and OCT evaluation of the samples was
approximately 5 min, until the OCT analysis was completed. OCT
images obtained before and after the two irrigation protocols with
nanoparticles solutions were compared.
Regions of interest
One hundred images representing the OCT-sections
were recorded for each of the 12 root canals and were analyzed,
using the public domain software ImageJ (version 1.33 µ; National
Institute of Health), which is a free-of-charge software widely
used in medical and biological image analysis over a long period of
time, with a wide range of analysis functions (16,17).
Image files can be opened, and regions of interests (ROI) can be
created automatically or by freehand selection. The ROIs can be
saved and transferred to other images and edited afterwards to
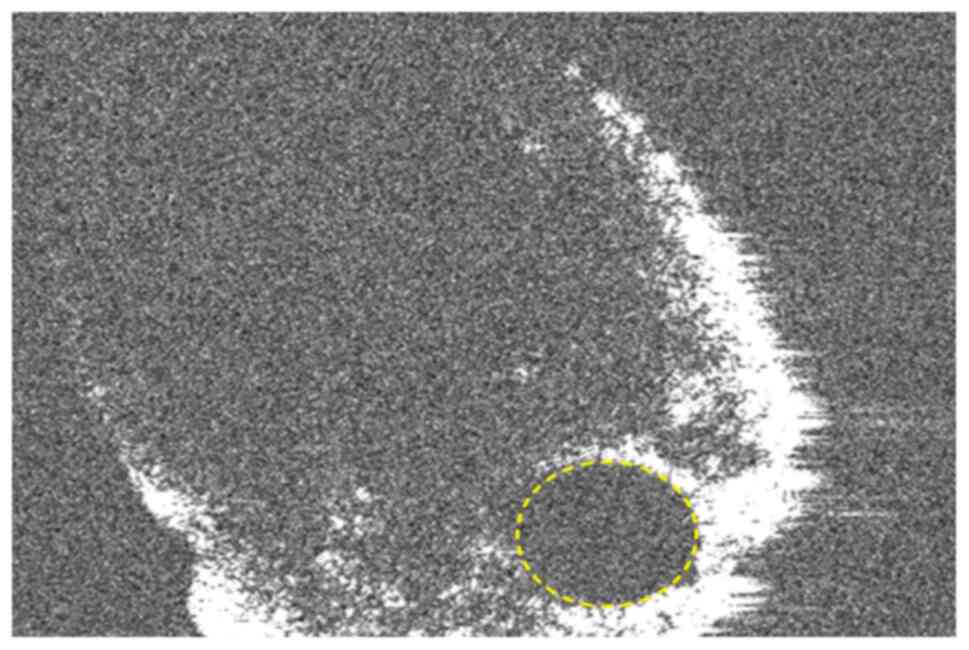
different projection conditions. A region of interest was opened
using as its borders the margins of the root canal of every tooth.
Only the root canal was enclosed in the ROI. Using the histogram
tool, the pixel values of the ROIs were established. The grey
values were related to the absorption of X-rays, the radiologic
density of a certain tissue. The grey values were saved in an 8-bit
color space. Every pixel obtained a value between 0 and 255 in
which 0 stands for black, low radiologic density and 255 for white,
total X-ray absorption (Fig.
4).
Statistical analysis
The maximum, minimum, mean value and the standard
deviation of the grey values were calculated. The Student's t-test
was used to compare the results between the groups to identify the
statistically significant differences. The result was considered
statistically significant for P<0.05.
Results
OCT analysis
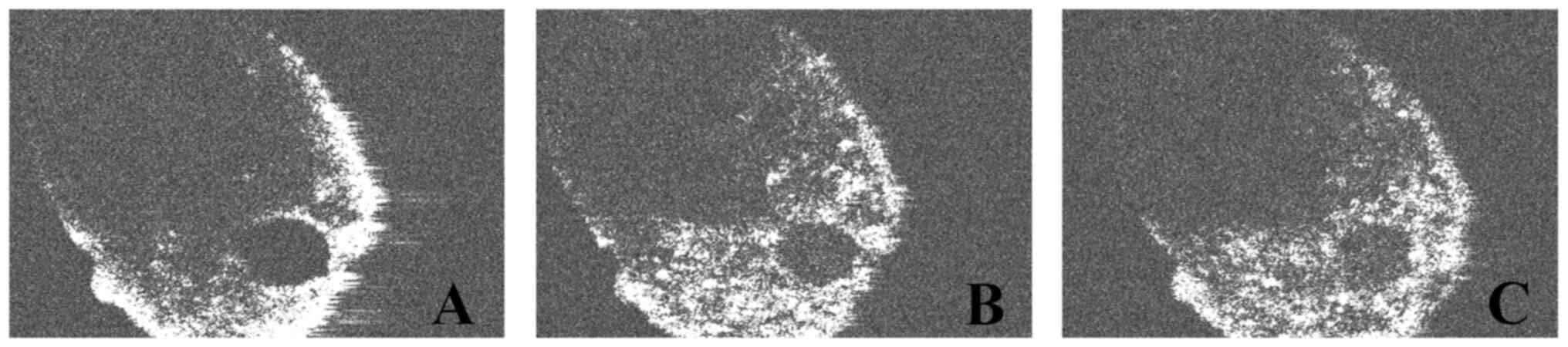
The c-scan OCT analysis allowed the investigation of
the root canal outline and lumen on every section that varies from
0 to 1 mm from the root apex. On the samples without
nanoparticle-based irrigants, the root canal lumen appeared clearly
separated from the dentinal contour, which is almost white on the
OCT images (255 radiological density). The empty root canal lumen
showed the lowest level of grey on the initial OCT images, as it
was empty and almost completely black on the initial OCT-scans.
Grey values and significance
The maximum, minimum, mean value, and the standard
deviation of the grey values on the analyzed OCT images for each
group were recorded in Table I.
 | Table ILevels of grey scores of the OCT scans
before and after irrigation with nanoparticles. |
Table I
Levels of grey scores of the OCT scans
before and after irrigation with nanoparticles.
| Group | Min | Max | Mean | SD |
|---|
| 1 | 9 | 255 | 88.74076 | 4.87669 |
| 2 | 12 | 255 | 102.94019 | 5.82824 |
| 3 | 11 | 255 | 104.31445 | 2.12771 |
Following Student's t-test, a statistically
significant difference was found between Group 1 and Group 2
(P<0.00001) and Group 1 and Group 3 (P<0.00001). However,
between Group 2 and Group 3 even if the data showed a difference,
this was not statistically significant (P>0.05).
Root canal
Analysis of the images showed that the pixel values
in the two groups (where nanoparticle irrigants were used)
increased in comparison with the control side (where only standard
sodium hypochlorite was used). The level of grey inside the root
canal lumen was higher in the samples irrigated with NanoCare Plus
(Group 2) compared to the initial samples (Group 1). It can be
assumed that the nanoparticles present in the irrigating solution
were observed as whiter, dot-like material inside the root canal.
The highest levels of grey were obtained in Group 3, after the
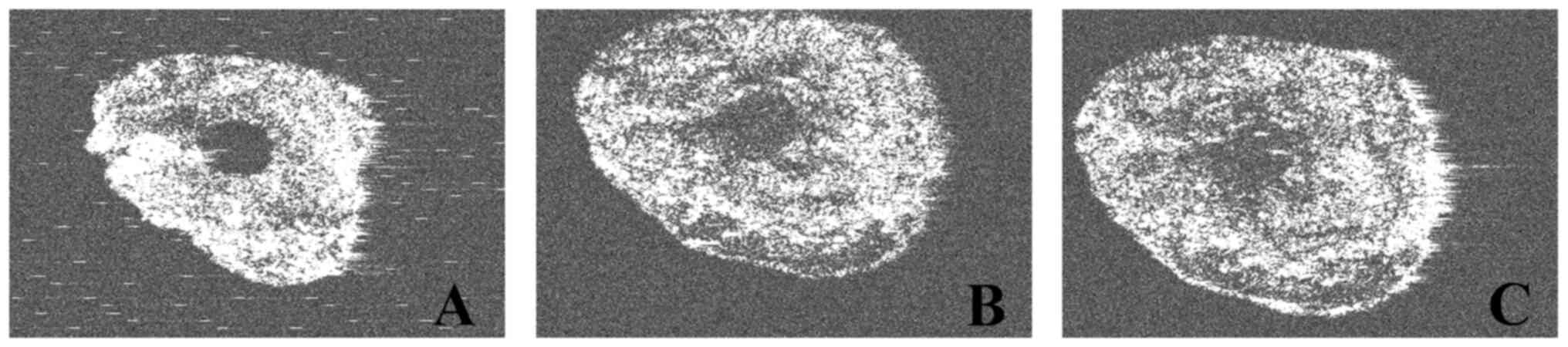
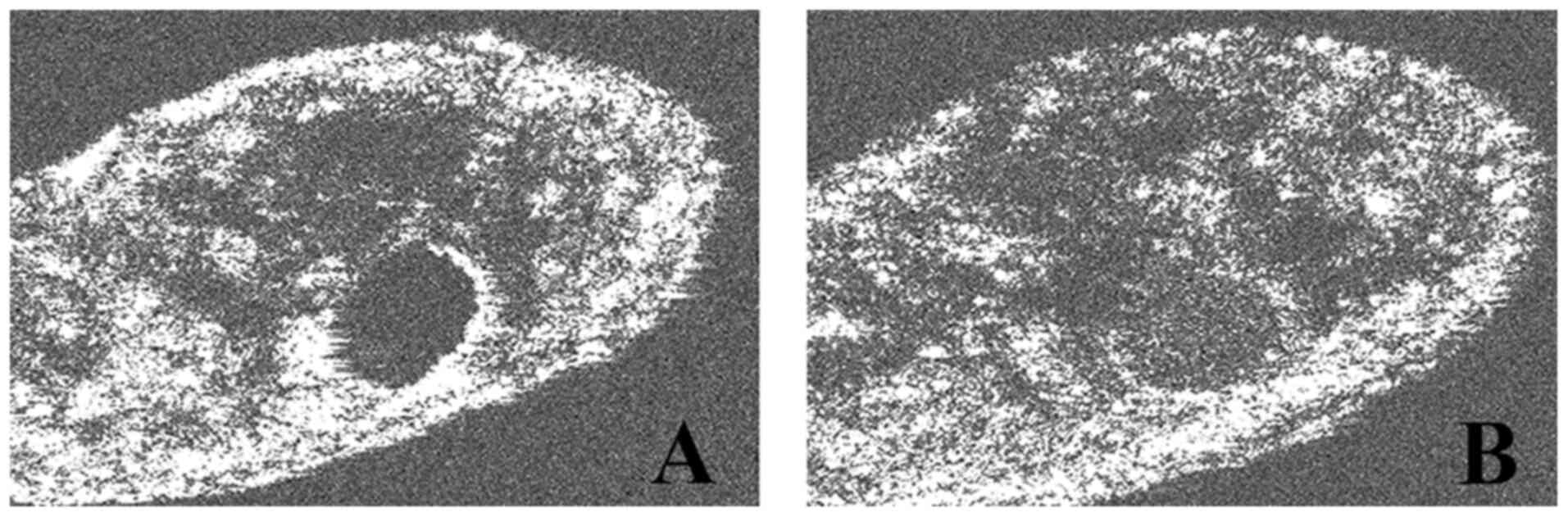
second irrigation with NanoCare Gold (Figs. 5 and 6).
The almost circular shape of the root canal contour,
as the result of the rotary instrumentation with nickel-titanium
instruments, was also observed (Fig.
5). On some of the analyzed samples, the eccentric position of
the apical foramen in relation to the radiological apex was
identified (Figs. 6 and 7).
Discussion
The OCT-based investigation of the current study
revealed that nanoparticles from the irrigating solutions were
observed even at the level of the last apical millimeter of the
root canal, suggesting good diffusion capacity of these endodontic
irrigants. This observation can lead to improvement of the
classical irrigation protocols used in daily endodontic practice,
as the apical area is the most refractory area to cleaning during
treatment, and the presence of the nanoparticles in close contact
with the root canal outline, as observed in the present study, may
lead to improved disinfection efficiency of endodontic
irrigants.
The results obtained in the present study are
similar with literature data and show that the addition of gold and
silver nanoparticles in root canal irrigants increases their
optical properties. It is known that gold addition decreases the
micro-infiltration at the root canal walls level, increasing the
adhesion of filling materials to dentin. Both types of
nanoparticles have proved antimicrobial efficiency. Furthermore,
this property enhances the capacity of the irrigation protocol to
reduce the number of remnant bacteria in the root canal lumen
(Fig. 5, Fig. 6 and Fig.
7).
One of the limitations of the present study could be
the resolution of the OCT investigating system; because of the lack
of a clear differentiation between the tubular dentin and the gold
and silver nanoparticles the present study cannot demonstrate the
penetration capacity of the nanoparticles at a tubular level. A
better resolution of the OCT investigating system to differentiate
between the dentin optical opacity and the one of the nanoparticles
or the performance of other imagistic analysis such as Electronic
Transmission Microscopy could offer more precise results.
Micro-infiltration tests performed on cleaned,
shaped, and filled root canals after irrigation with solutions
supplemented with nanoparticles can be performed in future studies,
to prove the influence of these particles on the adhesion of
various materials to root canal dentin, as shown in the literature
(18-20).
In addition, the present study can be continued and improved by
investigating the efficacy of these irrigants on specific bacteria
including E. faecalis or other species colonizing the root
canal, to prove their antibacterial effect (9,10,21).
The present study proved that gold and silver
nanoparticles introduced in root canal irrigants could be evidenced
through OCT imagistic analysis because of their optical opacity,
different from that of the root canal dentin and from the empty
root canal lumen. The optical opacity of these nanoparticles allows
their ability to be highlighted in an empty root canal lumen, after
the endodontic treatment is performed and the root canal is cleaned
and shaped using specific protocols. There was no possibility to
separately highlight gold from silver nanoparticles using this
technique because both irrigants used in the present study contain
both types of nanoparticles, with a bigger gold concentration for
the NanoCare Gold irrigant.
The OCT investigation performed in the present study
proved that the highest optical opacity in the investigated samples
was obtained after using an alternation of these two irrigants,
explained by a higher concentration of nanoparticles remaining in
the root canal lumen.
Numerous applications of nanomedicine and the
promising results of research regarding the applicability of
nanotechnology in clinical medicine are prerequisites for the
continuous development of this area (22-25).
Therefore, nanotechnology is currently one of the
future directions for investigation in endodontics as well.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used/analyzed in this study are
available from the corresponding author, upon reasonable
request.
Authors' contributions
FT, LMN, DR, MB and RC participated in data
acquisitions and interpretation thereof. AP, MLN, VFD, CS, AM and
SIS participated in the study design. LEC, FT, MB and LMN performed
the statistical analysis. LMN, MB and FT are responsible for
confirming the authenticity of the raw data. All authors drafted
the manuscript. All authors read and approved the final version of
the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Campos PI, La Fuenteha J, Tenorio RF and
Acosta TL: Biocompatible antimicrobial irrigants and
nanoparticles-sealers for endodontics. Entreciencias. 1:9–28.
2013.
|
|
2
|
Kim JS, Kuk E, Yu KN, Kim JH, Park SJ, Lee
HJ, Kim SH, Park YK, Park YH, Hwang CY, et al: Antimicrobial
effects of silver nanoparticles. Nanomedicine. 10(e1119)2014.
|
|
3
|
Hiraishi N, Yiu CK, King NM, Tagami J and
Tay FR: Antimicrobial efficacy of 3.8% silver diamine fluoride and
its effect on root dentin. J Endod. 36:1026–1029. 2010.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Peng G, Tisch U, Adams O, Hakim M, Shehada
N, Broza YY, Billan S, Abdah-Bortnyak R, Kuten A and Haick H:
Diagnosing lung cancer in exhaled breath using gold nanoparticles.
Nat Nanotechnol. 4:669–673. 2009.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Baptista P, Pereira E, Eaton P, Doria G,
Miranda A, Gomes I, Quaresma P and Franco R: Gold nanoparticles for
the development of clinical diagnosis methods. Anal Bioanal Chem.
391:943–950. 2008.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Kumar A, Bhargavi Mazinder B, Liang XL and
Cui D: Gold nanoparticles: Promising nanomaterials fo the
diagnostic of cancer and HIV/AIDS. J Nanomater. 2011:1–17.
2011.
|
|
7
|
Rayavarapu RG, Petersen W, Ungureanu C,
Post JN, van Leeuwen TG and Manohar S: Synthesis and bioconjugation
of gold nanoparticles as potential molecular probes for light-based
imaging techniques. Int J Biomed Imaging.
2007(29817)2007.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Rosi NL and Mirkin CA: Nanostructures in
biodiagnostics. Chem Rev. 105:1547–1562. 2005.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Sofi W, Gowri M, Shruthilaya M, Rayala S
and Venkatraman G: Silver nanoparticles as an antibacterial agent
for endodontic infections. BMC Infect Dis. 12(60)2012.
|
|
10
|
Grade S, Eberhard J, Jakobi J, Winkel A,
Stiesch S and Barcikowski S: Alloying colloidal silver
nanoparticles with gold disproportionally controls antibacterial
and toxic effects. Gold Bull. 47:83–93. 2014.
|
|
11
|
Mackiewicz A and Olczak-Kowalczyk D:
Microscopic evaluation of surface topography and chemical
composition of Nanocare Gold. J Stomatol. 67:826–840. 2014.
|
|
12
|
Idorași L, Cîrligeriu L, Sinescu C,
Zaharia C, Stan AT and Negruţiu ML: Silver nanotechnology-the
future in caries therapy? A report of two cases. J Interdiscip Med.
2:67–71. 2017.
|
|
13
|
Huang D, Swanson EA, Lin CP, Schuman JS,
Stinson WG, Chang W, Hee MR, Flotte T, Gregory K, Puliafito CA, et
al: Optical coherence tomography. Science. 254:1178–1181.
1991.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Sinescu C, Marsavina L, Negrutiu ML, Rusu
LC, Ardelean L, Rominu M, Antoniac I, Topala FI and Podoleanu A:
New metallic nanoparticles modified adhesive used for time domain
optical coherence tomography evaluation of class II direct
composite restoration. Rev Chim. 63:380–383. 2012.
|
|
15
|
Craciunescu MC, Negrutiu ML, Hogea E,
Freiman PC, Boariu M, Craciunescu E and Sinescu C: Bacteriostatic
effect of silver nanoparticles over acrylic resin and composite
dental materials. Mater Plast. 51:414–416. 2014.
|
|
16
|
Schneider CA, Rasband WS and Eliceiri KW:
NIH image to ImageJ: 25 years of image analysis. Nat Methods.
9:671–675. 2012.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Abramoff MD, Magalhães PJ and Ram SJ:
Image processing with ImageJ. Biophoton Int. 11:36–42. 2004.
|
|
18
|
Kesler Shvero D, Abramovitz I, Zaltsman N,
Perez Davidi M, Weiss EI and Beyth N: Towards antibacterial
endodontic sealers using quaternary ammonium nanoparticles. Int
Endod J. 46:747–754. 2013.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Pagonis TC, Chen J, Fontana CR,
Devalapally H, Ruggiero K, Song X, Foschi F, Dunham J, Skobe Z,
Yamazaki H, et al: Nanoparticle-based endodontic antimicrobial
photodynamic therapy. J Endod. 36:322–328. 2010.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Moghadas L, Shahmoradi M and Narimani T:
Antimicrobial activity of a new nanobased endodontic irrigation
solution: In vitro study. Dent Hypotheses. 3:142–146.
2012.
|
|
21
|
Van Dong P, Ha CH, Binh LT and Kasbohm J:
Chemical synthesis and antibacterial activity of novel-shaped
silver nanoparticles. Int Nano Lett. 2(9)2012.
|
|
22
|
Kovvuru SK, Mahita VN, Manjunatha BS and
Babu BS: Nanotechnology: The emerging science in dentistry. J
Orofac Res. 2:33–36. 2012.
|
|
23
|
Verma SK, Prabhat KC, Goyal L, Rani M and
Jain A: A critical review of the implication of nanotechnology in
modern dental practice. Natl J Maxillofac Surg. 1:41–44.
2010.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Martinez-Gutierrez F, Olive PL, Banuelos
A, Orrantia E, Nino N, Sanchez EM, Ruiz F, Bach H and Av-Gay Y:
Synthesis, characterization, and evaluation of antimicrobial and
cytotoxic effect of silver and titanium nanoparticles.
Nanomedicine. 6:681–688. 2010.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Cheon JY, Kim SJ, Rhee YH, Kwon OH and
Park WH: Shape-dependent antimicrobial activities of silver
nanoparticles. Int J Nanomedicine. 14:2773–2780. 2019.PubMed/NCBI View Article : Google Scholar
|