1. Introduction
Coronavirus disease 2019 (COVID-19) is an emerging
and rapidly evolving situation, causing an unprecedented continuous
impact on survival and daily life worldwide (1). Severe acute respiratory syndrome
coronavirus 2 (SARS-CoV-2) is the pathogen that causes
COVID-19(2). Furthermore,
SARS-CoV-2 is mutating, and it appears that numerous vaccines have
been developed and are now available. But most vaccines that
specifically target the existing SARS-CoV-2 may not be effective
against a mutant SARS-CoV-2(3). In
the context of viral infection, the body mainly relies on immune
function to eventually eliminate the virus, but treatment for the
series of consequences of infection is also essential. Exploration
of more effective treatment methods, including comprehensive and
combined therapies, is urgently required. Previous studies have
indicated that phosphodiesterase-5 inhibitors (PDE5Is) exhibit
important anti-infectious disease efficacy and may have multiple
mechanisms different from those of direct antiviral therapy. The
present study systematically summarized the mechanisms of action
and potential combined application value of PDE5Is in the treatment
of COVID-19.
2. Adverse effects of COVID-19
Unfavourable impacts of COVID-19 on
the respiratory system and general health
SARS-CoV-2 invades human cells through the receptor
angiotensin-converting enzyme 2 (ACE2). During virus entry into
host cells, the spike protein on the envelope of SARS-CoV-2 is
cleaved into the S1 and S2 subunits. S1 contains a receptor-binding
domain (RBD) that directly binds to the peptidase domain (PD) of
ACE2 to allow entry into the host cell (4). The peptidase activity of ACE2 is
required for SARS-CoV-2 to enter the host cell's cytoplasm. The
cleavage of the RBD in the C-terminus of the S1 subunit initiates
the interaction with the PD of ACE2(5). The binding of S1 to the receptor ACE2
triggers the cleavage of ACE2 via a disintegrin and metalloprotease
17 (ADAM17)/tumour necrosis factor (TNF)-converting enzyme in the
outer domain. In addition, a transmembrane serine protease
(TMPRSS2) cleaves the C-terminal domain of ACE2 and the cleavage of
ADAM17 and TMPRSS2 (outer domain and inner domain) promotes
effective viral entry (6). This
process appears to result in the loss of ACE2 receptor function and
systemic release of the S1/ACE2 complex.
ACE receptors are expressed in almost all tissues,
while ACE2 is expressed in alveolar epithelial cells and capillary
endothelial cells. ACE2 is highly expressed in capillary-rich
organs, including the lungs and kidneys, as well as in the
intestine and brain (7). ACE
regulates the renin-angiotensin-aldosterone system (RAAS). ACE2
balances the harmful effects of the ACE/renin-angiotensin system
(RAS) pathway through its downstream ACE2/angiotensin/MAS axis
(1-7). The classic activation of angiotensin II (AngII) depends on
renin and ACE activity. When the juxtaglomerular apparatus of the
afferent glomerular artery is activated, a special protease cleaves
prorenin into renin. Renin decomposes angiotensinogen into AngI and
ACE catalyses the conversion of AngI into AngII. After AngI is
converted into AngII, AngII binds to AngII receptors in the
kidneys, adrenal cortex, arterioles and brain. AngII acts on the
adrenal cortex and stimulates the release of aldosterone, which
leads to the retention of sodium and water, and the binding of
AngII to angiotensin II type (AT) receptors leads to
vasoconstriction, endothelial damage, intravascular thrombosis and
an increased blood volume (8).
Moreover, AngII may also effectively induce interleukin (IL)-6 and
TNF-α through serine tyrosine kinase, ERK/JNK-MAPK, G
protein-coupled receptor activation or corticosteroid receptor
interactions (4). AngII is an
effective activator of NADPH oxidase, so it is also an inducer of
reactive oxygen species. In addition, AngII may activate
neutrophils and macrophages to migrate to affected tissues and
inhibit the production of nitric oxide (NO), thus promoting
vascular injury (9).
ACE2 acts as a ligand through the MAS1 receptor,
which is a G protein-coupled receptor. ACE2 is a single
carboxypeptidase that is able to cleave AngI into Ang1-9 and AngII
into Ang1-7(10). These two
peptides have vasodilating, antiproliferative and protective
effects mediated by activating the MAS/G receptor. The
ACE2/Ang1-7/MAS1 axis provides an endogenous reverse regulation in
the RAAS to balance the harmful effects of the ACE/AngII/AT1
receptor axis. ACE2 activation may prevent the harmful effects of
AngII on cells and organisms, such as cell death, fibrosis,
vasoconstriction and thrombosis, that lead to alveolar epithelial
injury and respiratory failure (11,12).
When SARS-CoV-2 binds to ACE2 and enters pulmonary epithelial
cells, the receptor ACE2 is released from the surface of host cells
and loses its protective function. The loss of ACE2 activity blocks
the production of Ang1-9 and Ang1-7, resulting in an imbalance in
the AngII level and the upregulation of RAS/AngII pathway activity.
The upregulation of the AngII level leads to vasoconstriction,
thrombosis, alveolar epithelial injury and respiratory failure,
which eventually leads to the symptoms of COVID-19 in the lungs and
even the whole body.
COVID-19 may have adverse effects on
the testes and reproductive system
Certain studies have examined the expression pattern
of ACE2 in adult human testes at the level of single-cell
transcriptomes. ACE2 was indicated to be mainly enriched in
spermatogonia, Leydig cells and Sertoli cells. Gene ontology
analysis suggested that genes related to virus propagation and
transmission were highly enriched in ACE2-positive spermatogonia,
while the expression of genes related to male gametogenesis was
downregulated. ACE2-positive Leydig and Sertoli cells exhibited
increased levels of intercellular junction- and immune-related
genes, while the levels of mitochondria- and reproduction-related
genes were decreased (13). These
results suggested that the human testes are a potential target for
SARS-CoV-2. SARS-CoV-2 is likely to bind to the receptor ACE2 on
testicular cells, which leads to an imbalance in the AngII level
and the upregulation of RAS/AngII pathway activity, resulting in
vasoconstriction, thrombosis and activation of the inflammatory
system, thus causing damage to testicular cells (14).
A study has reported that SARS-CoV-2 may be detected
in the semen of certain patients (15.8%) and that SARS-CoV-2 may
persist in the male reproductive system, causing harm to this
system (15). In another study, a
mild COVID-19 infection was not likely to affect testis and
epididymis function, but semen parameters did seem impaired after a
moderate infection. There is no evidence of SARS-CoV-2 shedding in
semen of recovered men or men with an acute COVID-19 infection
after a recovery time of 32.7 days on average (16). In addition, pathological analysis of
testicular tissues from patients with COVID-19 revealed obvious
damage to the seminiferous tubules, decreased interstitial cell
counts, as well as mild lymphocyte infiltration and obviously
damage to the testicular parenchyma. Although SARS-CoV-2 was not
detected in the testes of the vast majority of autopsy cases (90%)
by reverse transcription PCR, there were still certain cases (10%)
in which testis autopsy provided evidence of SARS-CoV-2(17). Therefore, it is likely that COVID-19
may have adverse effects on the male reproductive system.
3. PDE5Is and their anti-infection
mechanisms
General
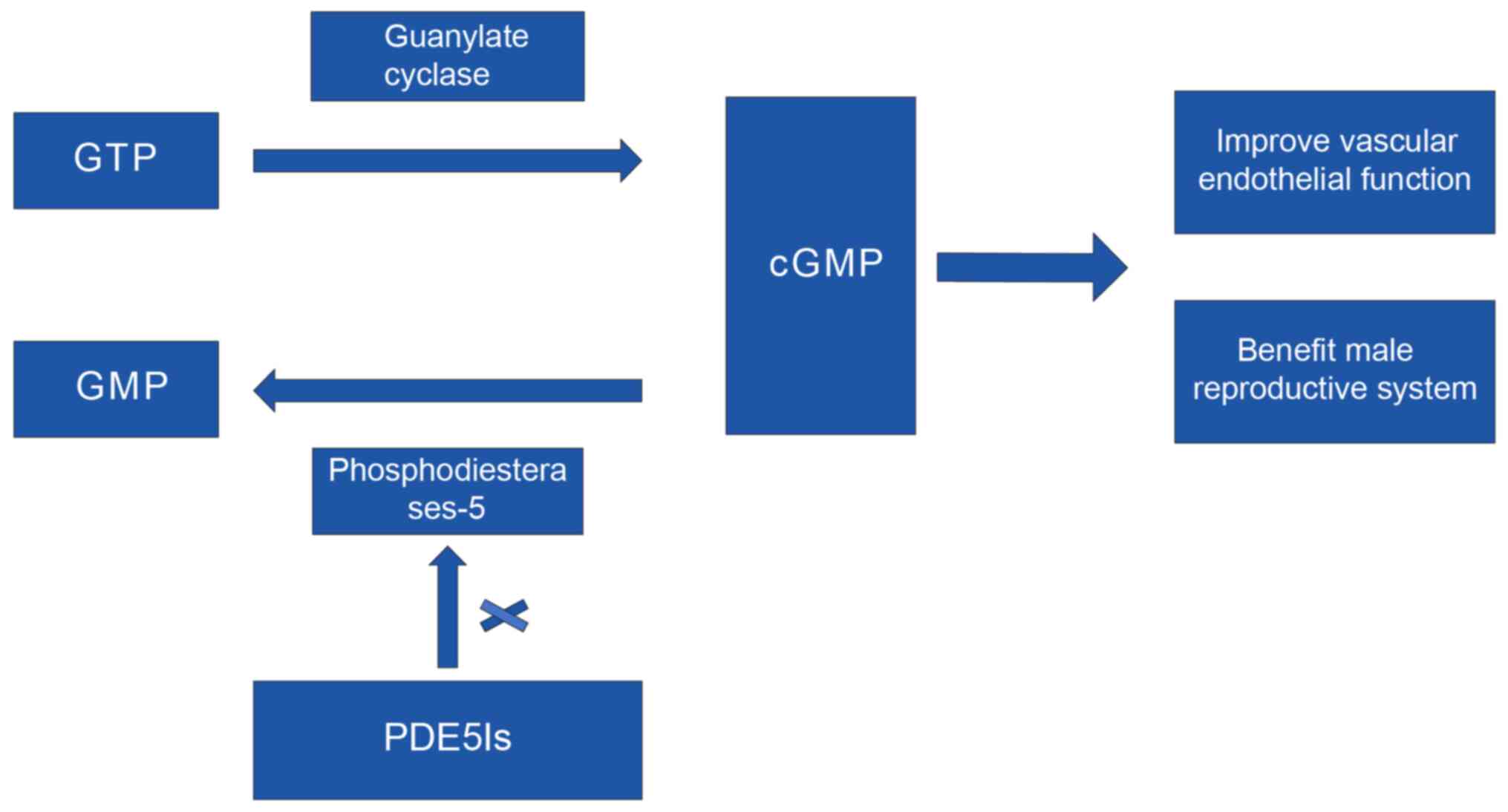
PDE5Is are drugs that effectively improve erectile
function. PDE5Is increase the concentration of cyclic guanosine
monophosphate (cGMP) by inhibiting the activity of PDE5, resulting
in relaxation of the smooth muscle of the penis, which increases
the blood flow of the artery in the cavernous body of the penis and
produces an erection (18). In
addition, PDE5Is protect and repair the vascular endothelium and
improve the blood circulation; therefore, they are used in the
treatment of cardiovascular diseases (19). The detailed mechanism of action of
PDE5Is is presented in Fig. 1.
Compared with other similar drugs that improve vascular endothelial
function, such as ACE inhibitors, statins, calcium antagonists and
β-receptor blockers, PDE5Is have unique mechanisms to improve
vascular endothelial function and numerous advantages, including
high effectiveness, more diverse indications, benefits for male
reproductive health, as well as only a small number of side
effects, which are tolerable. The commonly used PDE5Is include
sildenafil, vardenafil and tadalafil.
At present, there is no satisfactory specific drug
for COVID-19; thus, it is important to improve systemic immune
function in the context of viral infection. Furthermore, it is
necessary to effectively intervene in a series of
pathophysiological processes and inflammatory products caused by
infection, including improvement of local tissue blood circulation
disorder, tissue hypoxia, vascular endothelial damage and oxidative
stress injury (20). Based on the
pharmacological mechanism of action of PDE5Is, these drugs have
unique value in the treatment of infectious diseases and may be
effective in combination with direct antiviral drugs. The
anti-infection mechanism of PDE5Is in COVID-19 is presented in
Fig. 2. The mechanism shown in the
Fig. 2 is as follow: COVID-19 can
cause adverse effects on human body by reducing immunity, enhancing
inflammatory responses and reducing vascular endothelial function.
But PDE5Is can synergistically treat COVID-19 by enhancing
immunity, reducing inflammatory response and enhancing vascular
endothelial function.
Enhancement of immunity
PDE5Is are able to enhance immune function. In
healthy male mice treated with sildenafil, the percentage of
CD4+ T cells was indicated to be decreased, while the
percentage of CD8+ T cells was increased. It was also
observed that the levels of effector memory and central memory T
cells were decreased in male mice but increased in female mice. The
percentages of activated natural killer (NK) cells and T cells were
increased in female mice but decreased in male mice. In addition,
sildenafil decreased the level of serum IL-6 and increased the
level of IL-2. These results suggest that sildenafil has
sex-specific immunoregulatory properties (21). Furthermore, the effects of
sildenafil on thymocytes, splenocytes and T cells isolated from the
lymph nodes of mice were studied. Oral sildenafil (1 mg/kg)
temporarily reduced the percentage of
CD4+CD8+ thymocytes and increased that of
CD8+ cells. In lymphocytes isolated from the mesenteric
lymph nodes, the percentage of CD19+ cells was decreased
and the percentage of CD3+ cells was increased 72 h
after the last administration (22). Certain studies have indicated that
sildenafil increases the production of IL-1β and NO in peritoneal
macrophages and the percentage of phagocytes and decreases the
percentage of monocytes (23). In a
study on experimental autoimmune encephalomyelitis, a multiple
sclerosis model, it was determined that sildenafil was able to
reduce cell infiltration in the spinal cord and white matter of
mice. Sildenafil increased the number of YM-1 cells (which is a
marker of the alternative macrophage/microglial M2 phenotype that
has neuroprotective and regenerative properties) and the expression
of the granzyme B cluster in regulatory T cells (Tregs) (24). In addition, sildenafil was able to
prolong the survival time of tumour-bearing mice by enhancing
antitumour immunity, which is achieved by inhibiting
CD11b+/Gr-1+ myeloid-derived suppressor cells
(MDSCs) and downregulating IL-4 receptor α expression (25,26).
Certain studies have indicated that sildenafil may
significantly reduce the activity of NK cells in the peripheral
blood of females with recurrent abortion and endometrial thickness
significantly increases after treatment (27). A case of B-cell chronic
lymphoblastic leukaemia (B-CLL) treated with sildenafil alone was
reported. During the 3.5-year period of treatment with sildenafil
(50 mg/week), the lymphocyte count of the patient decreased
significantly and sildenafil induced apoptosis in B-CLL cells in a
caspase-3-dependent manner (28).
In addition, sildenafil may affect angiogenesis, platelet
activation, Treg proliferation, proinflammatory cytokines and
autoantibodies, thus affecting the innate and adaptive immune
systems of experimental animals and patients (29).
Anti-inflammatory responses
PDE5Is may also limit inflammatory factor activity
and inflammatory responses and have anti-infection effects. It has
been reported that F508del-CF mice exhibit a potential inflammatory
state characterized by neutrophil infiltration and mouse macrophage
inflammatory protein 2 and TNF-α expression. Intraperitoneal
administration of vardenafil was indicated to significantly reduce
the inflammatory response of F508del-CF mice and decrease
macrophage infiltration, macrophage chemokine levels and IL-1β
levels. These results suggest that vardenafil may significantly
reduce lung inflammation in F508del-CF mice, particularly in terms
of macrophage markers (30).
Another study indicated that vardenafil is able to reduce the
proinflammatory and fibrotic functions of fibroblasts in mice with
cystic fibrosis, providing convincing novel support for developing
a drug therapy to target the cGMP signalling pathway in cystic
fibrosis (31). It has also been
reported that tadalafil is able to reduce the levels of
malondialdehyde and NO in the kidneys of rats with acute
pyelonephritis and enhance the activities of superoxide dismutase
(SOD) and catalase. These results suggest that tadalafil may
improve the levels of circulating inflammatory cytokines, reverse
oxidative dysfunction and ultimately protect the kidneys (32). In addition, in an acute lung injury
(ALI) model, sildenafil treatment reduced cell leakage,
particularly neutrophil infiltration into the lungs, decreased the
release of proinflammatory mediators (TNF-α, IL-8 and IL-6) and the
levels of nitrite/nitrate and oxidative damage markers, reduced the
formation of pulmonary oedema and improved respiratory parameters.
Therefore, sildenafil also has a potential role in the treatment of
ALI (33).
In a rat model of bronchopulmonary dysplasia,
prophylactic treatment with sildenafil was indicated to
significantly increase cGMP levels in the lungs, reduce the
pulmonary inflammatory response, fibrin deposition and right
ventricular hypertrophy and to stimulate alveolarization (34). In addition, oral administration of
tadalafil (1 mg/kg) for 8 weeks was able to significantly improve
silica-induced lung injury. The protective effect of tadalafil is
related to the inhibition of cytokine expression in inflammation
and fibrosis, recovery of the antioxidant capacity and promotion of
angiogenesis (35). It has been
indicated that in a rat model of lipopolysaccharide-induced sepsis,
sildenafil may increase the level of glutathione, reduce the
activities of myeloperoxidase (MPO), lipid peroxidase and SOD and
to decrease the level of serum TNF-α and the inflammatory score.
Therefore, sildenafil has protective effects on the lung and kidney
damage caused by sepsis (36). In
addition, tadalafil may prevent or slow ureteral inflammation and
urothelial degeneration in rats with ureteral obstruction and
reduce the expression of α-smooth muscle actin and transforming
growth factor-β (37).
T helper 1 (Th1)-type cytokines and chemokines are
bioactive mediators in the inflammatory response and the Th1
chemokine C-X-C motif chemokine ligand (CXCL)10 is involved in the
occurrence and development of cardiac injury. In diabetic subjects
with cardiomyopathy, sildenafil was able to significantly reduce
the protein secretion and gene expression of CXCL10 in human
cardiomyocytes and reduce the level of CXCL10 in patients (38). Cavernitis is a rare acute infection
of the penis. If not treated properly, cavernitis may lead to
cavernous abscess formation, fibrosis and subsequent erectile
dysfunction. In a clinical study, two patients with cavernitis were
treated with antibiotics and tadalafil and their clinical symptoms
improved rapidly. After 3 months of follow-up, the erectile
function of the patients was normal and there was no penile
scarring. This suggests that tadalafil may have a synergistic
effect with antibacterial therapy (39). In addition, the therapeutic effect
of vardenafil combined with antibiotics in the treatment of
patients with chronic bacterial prostatitis (CBP) was studied. The
National Institutes of Health-Chronic Prostatitis Symptom Index
(NIH-CPSI) scale, urinary bacteria and leukocyte counts and the
urine flow rate were used to evaluate the therapeutic effect. It
was indicated that the combination of antibiotics and vardenafil
achieved a significantly improved efficacy in CBP (40). In addition, a study reported that in
a patient with a severe chronic foot wound, the wound healed almost
completely after treatment with tadalafil (20 mg three times a day)
and was stable for 2 years, which unexpectedly revealed a role for
tadalafil in chronic wound infection treatment (41).
Improvement of vascular endothelial
function
PDE5Is relax vascular smooth muscle and improve the
local blood circulation. Certain studies have explored the
therapeutic effect of sildenafil in the early stage of sepsis.
Sildenafil may increase the renal blood flow, decrease pulmonary
MPO activity and reduce the levels of plasma creatinine, lactate
and creatine kinase. Therefore, sildenafil may be a treatment for
early sepsis (42). In addition,
the effects of intraperitoneal injection of sildenafil on left
colonic anastomosis healing and intra-abdominal adhesion formation
were also studied. The healing of an anastomotic stoma in model
rats was significantly improved and histopathological results
indicated that angiogenesis was increased and intra-abdominal
adhesion was significantly reduced (43). Severe endothelial cell injury is the
key event in the process of ischaemia-reperfusion (I/R) injury. It
was indicated that sildenafil may dilate blood vessels and increase
the cGMP level, but was not able to affect the recovery of renal
function or I/R injury (44). In
addition, sildenafil may prevent early transplantation failure,
prolong ischaemic tolerance and improve lung function in pigs with
lung I/R injury (45). Furthermore,
a study reported that for patients with benign prostatic
hyperplasia, 5 mg tadalafil may relax the vascular smooth muscle,
improve vascular endothelial function and increase vascular
elasticity (46). The effect of
sildenafil on the microvascular blood flow in patients with
systemic sclerosis secondary to Raynaud's phenomenon (RP) was also
explored in one study. The results suggested that sildenafil
improved the finger artery blood flow and RP symptoms after 8 weeks
of treatment, which indicates that sildenafil is a good choice for
the treatment of secondary RP (47).
Studies have also investigated the effect of
sildenafil on AngII-dependent hypertension and renal vascular
dysfunction. It was indicated that sildenafil is able to lower
AngII-dependent hypertension and enhance NO-mediated vasodilation,
which is a key regulator of renal haemodynamics in patients with
hypertension (48). In addition,
there the effects of sildenafil on irisin levels in the heart,
liver and kidney tissues were explored and it was evaluated whether
sildenafil impacts serum irisin or NO levels in rats with induced
experimental myocardial ischaemia. Sildenafil was effective in
eliminating ischaemia and may be the first choice for offsetting
the effects of ischaemia in the future (49). Studies have also explored the
effects of vardenafil on the systemic endothelial function of
diabetic patients with erectile dysfunction. Chronic vardenafil
administration was able to significantly improve endothelial
parameters in diabetic patients (50). Whether sildenafil is able to improve
tissue perfusion and neovascularization and downregulate
proinflammatory molecules following limb ischaemia has also been
examined. Sildenafil was able to significantly improve tissue
perfusion and the inflammatory status after limb ischaemia, which
implies that sildenafil potentially has properties related to
neovascularization and vascular protection (51). Studies exploring the effects of
sildenafil on cerebrovascular function and cerebral metabolism in
patients with Alzheimer's disease (AD) have also been performed.
After sildenafil administration, the cerebral blood flow and
cerebral oxygen metabolism rate of the patients were significantly
increased (52). Furthermore,
numerous studies have confirmed that sildenafil is able to
significantly improve pulmonary hypertension and relax blood
vessels (53,54).
4. Roles of PDE5Is in COVID-19
Combination treatment of COVID-19
COVID-19 may promote the induction of endothelial
inflammation in multiple organs, which is the direct result of
SARS-CoV-2 infection and host inflammatory response activation.
This progressive endothelial thrombotic inflammatory syndrome
involves the microvessels of numerous important organs, leading to
multiorgan failure and death; this syndrome is also the mechanism
underlying interstitial pneumonia developing into pulmonary
failure. As a vasodilator, tadalafil may be administered once a day
to improve tissue angiogenesis and anti-fibrotic function, thus
blocking or preventing vascular endothelial sclerosis in patients
with COVID-19(55). There is a
strong correlation between the severity of infection caused by
COVID-19 and the presence of hypertension and numerous studies have
demonstrated that hypertension is related to NO. Considering the
pharmacology of PDE5Is, these drugs may be used as early
complimentary drugs in the treatment of COVID-19(56). Furthermore, numerous patients with
COVID-19 have symptoms of bilateral diffuse alveolar injury and
pulmonary oedema, increased levels of proinflammatory mediators and
elevated pulmonary capillary pressure. Whether PDE5Is should be
included in combination treatments of COVID-19 to facilitate the
recovery of patients has also been considered (57). Certain studies have indicated that
NO may alleviate lung injury, reduce the concentrations of
proinflammatory cytokines and decrease the number of neutrophils
entering the lungs. Therefore, PDE5Is may be used to increase the
level of NO as part of the treatment of COVID-19(58). The use of PDE5Is to reduce pulmonary
hypertension and relieve the symptoms of acute respiratory distress
syndrome and the used of PDE5Is for synergistic treatment of
COVID-19 have also been proposed (59).
Therefore, PDE5Is also have important roles in
protecting against infectious diseases. PDE5Is may improve the
treatment of infectious diseases by enhancing immunity and
anti-inflammatory responses and improving vascular endothelial
function. In patients with COVID-19, PDE5Is may be considered as
synergistic drugs for combined treatment.
Multiple beneficial effects for
COVID-19
Various studies have indicated that COVID-19 has
potential adverse effects on the male reproductive system (60,61).
ACE2 is expressed in the spermatogonia, Leydig cells and Sertoli
cells of the testes. SARS-CoV-2 may cause reproductive dysfunction
in patients via the ACE2 pathway (62). Certain studies have indicated that
patients with COVID-19 have symptoms of viral orchitis and scrotal
discomfort, which have a negative impact on male reproductive
function and spermatogenesis (63).
Certain studies have also summarized the adverse effects of
SARS-CoV-2 on male reproductive health, which may lead to
testicular damage and male infertility and suggest the theoretical
possibility of sexual transmission, as SARS-CoV-2 has been
identified in the semen of infected patients. It is also necessary
to rehabilitate and monitor the reproductive system of patients
with COVID-19 (64,65).
PDE5Is may improve vascular endothelial function,
promote the testicular and systemic blood circulation and
testosterone secretion and improve epididymal function, sperm
maturation and capacitation (66,67).
Therefore, in view of the possible damage to the male reproductive
system and whole body caused by SARS-CoV-2, PDE5Is may be
considered for inclusion in combined therapy to treat COVID-19,
which may improve orchitis and sperm quality while enhancing
systemic antiviral effects. In general, PDE5Is are safe in clinical
practice. The incidence of side effects is low (from 9 to 25%), the
symptoms are not serious and most symptoms are tolerable, and
PDE5Is may be obtained from a pharmacy with a prescription. The
potential side effects of PDE5Is include headache, flushing,
dyspepsia, nasal congestion, dizziness, abnormal vision, back pain
and myalgia. The absolute contraindication is combined use with
nitrates (68). For patients with
COVID-19 with damage to male reproductive function or erectile
dysfunction, the combined use of PDE5Is may not only help to treat
the viral infection but also benefit reproductive function. For
patients with COVID-19 without damage to male reproductive function
or erectile dysfunction, the combined use of PDE5Is may prevent
damage to the male reproductive system.
5. Conclusion
COVID-19 has certain adverse effects on numerous
parts of the human body, including the respiratory system and male
reproductive system. PDE5Is with a variety of anti-infection
mechanisms may become important drugs for combination therapy,
particularly in patients with male genital infection, and the
combined use of PDE5Is may achieve multiple beneficial effects.
Acknowledgements
Not applicable.
Funding
Funding: This study was supported by a grant from the CAMS
Innovation Fund for Medical Sciences CIFMS (no.
2018-I2M-1-004).
Availability of data and materials
Not applicable.
Authors' contributions
HS, CM and HL were involved in the acquisition,
analysis and interpretation of the data. HS and HL also contributed
to the design and conception of the study. HS drafted the
manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Wu F, Zhao S, Yu B, Chen YM, Wang W, Song
ZG, Hu Y, Tao ZW, Tian JH, Pei YY, et al: A new coronavirus
associated with human respiratory disease in China. Nature.
579:265–269. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Chakraborty C, Sharma AR, Sharma G,
Bhattacharya M and Lee SS: SARS-CoV-2 causing pneumonia-associated
respiratory disorder (COVID-19): Diagnostic and proposed
therapeutic options. Eur Rev Med Pharmacol Sci. 24:4016–4026.
2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Liu X, Liu C, Liu G, Luo W and Xia N:
COVID-19: Progress in diagnostics, therapy and vaccination.
Theranostics. 10:7821–7835. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Samavati L and Uhal BD: ACE2, much more
than just a receptor for SARS-COV-2. Front Cell Infect Microbiol.
10(317)2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Yan R, Zhang Y, Li Y, Xia L, Guo Y and
Zhou Q: Structural basis for the recognition of SARS-CoV-2 by
full-length human ACE2. Science. 367:1444–1448. 2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Hoffmann M, Kleine-Weber H, Schroeder S,
Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH,
Nitsche A, et al: SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2
and is blocked by a clinically proven proteaseinhibitor. Cell.
181:271–280.e8. 2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Roca-Ho H, Riera M, Palau V, Pascual J and
Soler MJ: Characterization of ACE and ACE2 expression within
different organs of the NOD mouse. Int J Mol Sci.
18(563)2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Das UN: Renin-angiotensin-aldosterone
system in insulin resistance and metabolic syndrome. J Transl Int
Med. 4:66–72. 2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Kato H, Hou J, Chobanian AV and Brecher P:
Effects of angiotensin II infusion and inhibition of nitric oxide
synthase on the rat aorta. Hypertension. 28:153–158.
1996.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Santos RA, Simoese Silva AC, Maric C,
Silva DM, Machado RP, de Buhr I, Heringer-Walther S, Pinheiro SV,
Lopes MT, Bader M, et al: Angiotensin-(1-7) is an endogenous ligand
for the G protein-coupled receptor Mas. Proc Natl Acad Sci USA.
100:8258–8263. 2003.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Fraga-Silva RA, Sorg BS, Wankhede M,
Dedeugd C, Jun JY, Baker MB, Li Y, Castellano RK, Katovich MJ,
Raizada MK, et al: ACE2 activation promotes antithrombotic
activity. Mol Med. 16:210–215. 2010.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Tikellis C and Thomas MC:
Angiotensin-converting enzyme 2 (ACE2) is a key modulator of the
renin angiotensin system in health and disease. Int J Pept.
2012(256294)2012.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang Z and Xu X: scRNA-seq profiling of
human testes reveals the presence of the ACE2 receptor, a target
for SARS-CoV-2 infection in Spermatogonia, Leydig and Sertoli
cells. Cells. 9(920)2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Illiano E, Trama F and Costantini E: Could
COVID-19 have an impact on male fertility? Andrologia.
52(e13654)2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Li D, Jin M, Bao P, Zhao W and Zhang S:
Clinical characteristics and results of semen tests among men with
coronavirus disease 2019. JAMA Netw Open. 3(e208292)2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Holtmann N, Edimiris P, Andree M, Doehmen
C, Baston-Buest D, Adams O, Kruessel JS and Bielfeld AP: Assessment
of SARS-CoV-2 in human semen-a cohort study. Fertil Steril.
114:233–238. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Yang M, Chen S, Huang B, Zhong JM, Su H,
Chen YJ, Cao Q, Ma L, He J, Li XF, et al: Pathological findings in
the testes of COVID-19 patients: Clinical implications. Eur Urol
Focus. 6:1124–1129. 2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Lue TF: Erectile dysfunction. N Engl J
Med. 342:1802–1813. 2000.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Cai Z, Zhang J and Li H: Two birds with
one stone: Regular use of PDE5 inhibitors for treating male
patients with erectile dysfunction and cardiovascular diseases.
Cardiovasc Drugs Ther. 33:119–128. 2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Tay MZ, Poh CM, Rénia L, MacAry PA and Ng
LF: The trinity of COVID-19: Immunity, inflammation and
intervention. Nat Rev Immunol. 20:363–374. 2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Karakhanova S, Yang Y, Link J, Soltek S,
von Ahn K, Umansky V, Werner J and Bazhin AV: Gender-specific
immunological effects of the phosphodiesterase 5 inhibitor
sildenafil in healthy mice. Mol Immunol. 56:649–659.
2013.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Szczypka M and Obmińska-Mrukowicz B:
Modulating effects of nonselective and selective phosphodiesterase
inhibitors on lymphocyte subsets and humoral immune response in
mice. Pharmacol Rep. 62:1148–1158. 2010.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Szczypka M and Obmińska-Mrukowicz B: The
effects of selective and nonselective phosphodiesterase inhibitors
on phagocytic cells in mice. Immunopharmacol Immunotoxicol.
32:507–513. 2010.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Pifarré P, Gutierrez-Mecinas M, Prado J,
Usero L, Roura-Mir C, Giralt M, Hidalgo J and García A:
Phosphodiesterase 5 inhibition at disease onset prevents
experimental autoimmune encephalomyelitis progression through
immunoregulatory and neuroprotective actions. Exp Neurol.
251:58–71. 2014.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Serafini P, Meckel K, Kelso M, Noonan K,
Califano J, Koch W, Dolcetti L, Bronte V and Borrello I:
Phosphodiesterase-5 inhibition augments endogenous antitumor
immunity by reducing myeloid-derived suppressor cell function. J
Exp Med. 203:2691–2702. 2006.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Meyer C, Sevko A, Ramacher M, Bazhin AV,
Falk CS, Osen W, Borrello I, Kato M, Schadendorf D, Baniyash M, et
al: Chronic inflammation promotes myeloid-derived suppressor cell
activation blocking antitumor immunity in transgenic mouse melanoma
model. Proc Natl Acad Sci USA. 108:17111–17116. 2011.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Jerzak M, Kniotek M, Mrozek J, Górski A
and Baranowski W: Sildenafil citrate decreased natural killer cell
activity and enhanced chance of successful pregnancy in women with
a history of recurrent miscarriage. Fertil Steril. 90:1848–1853.
2008.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Sarfati M, Mateo V, Baudet S, Rubio M,
Fernandez C, Davi F, Binet JL, Delic J and Merle-Beral H:
Sildenafil and vardenafil, types 5 and 6 phosphodiesterase
inhibitors, induce caspase-dependent apoptosis of B-chronic
lymphocytic leukemia cells. Blood. 101:265–269. 2003.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Kniotek M and Boguska A: Sildenafil can
affect innate and adaptive immune system in both experimental
animals and patients. J Immunol Res. 2017(4541958)2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Lubamba B, Huaux F, Lebacq J, Marbaix E,
Dhooghe B, Panin N, Wallemacq P and Leal T: Immunomodulatory
activity of vardenafil on induced lung inflammation in cystic
fibrosis mice. J Cyst Fibros. 11:266–273. 2012.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Huaux F, Noel S, Dhooghe B, Panin N, Lo Re
S, Lison D, Wallemacq P, Marbaix E, Scholte BJ, Lebecque P, et al:
Dysregulated proinflammatory and fibrogenic phenotype of
fibroblasts in cystic fibrosis. PLoS One. 8(e64341)2013.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Zhu CY, Liu M, Liu YZ, Li W, Zhai W, Che
JP, Yan Y, Wang GC and Zheng JH: Preventive effect of
phosphodiesterase 5 inhibitor tadalafil on experimental
post-pyelonephritic renal injury in rats. J Surg Res. 186:253–261.
2014.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Kosutova P, Mikolka P, Balentova S,
Kolomaznik M, Adamkov M, Mokry J, Calkovska A and Mokra D: Effects
of phosphodiesterase 5 inhibitor sildenafil on the respiratory
parameters, inflammation and apoptosis in a saline lavage-induced
model of acute lung injury. J Physiol Pharmacol. 69:2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
de Visser YP, Walther FJ, Laghmani H,
Boersma H, van der Laarse A and Wagenaar GT: Sildenafil attenuates
pulmonary inflammation and fibrin deposition, mortality and right
ventricular hypertrophy in neonatal hyperoxic lung injury. Respir
Res. 10(30)2009.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Abdelaziz RR, Elkashef WF and Said E:
Tadalafil reduces airway hyperactivity and protects against lung
and respiratory airways dysfunction in a rat model of silicosis.
Int Immunopharmacol. 40:530–541. 2016.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Cadirci E, Halici Z, Odabasoglu F,
Albayrak A, Karakus E, Unal D, Atalay F, Ferah I and Unal B:
Sildenafil treatment attenuates lung and kidney injury due to
overproduction of oxidant activity in a rat model of sepsis: A
biochemical and histopathological study. Clin Exp Immunol.
166:374–384. 2011.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Köktürk S, Benli E, Ayyıldız A, Cırrık S,
Çetinkol Y, Ayyıldız SN and Noyan T: Positive outcomes of
phosphodiesterase type 5 inhibitor on histopathologic and
biochemical changes induced by ureteral obstruction. Rev Assoc Med
Bras (1992). 65:388–393. 2019.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Di Luigi L, Corinaldesi C, Colletti M,
Scolletta S, Antinozzi C, Vannelli GB, Giannetta E, Gianfrilli D,
Isidori AM, Migliaccio S, et al: Phosphodiesterase type 5 inhibitor
sildenafil decreases the proinflammatory chemokine CXCL10 in human
cardiomyocytes and in subjects with diabetic cardiomyopathy.
Inflammation. 39:1238–1252. 2016.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Bakhsh A, Daqqaq T, Alhasan A, Alnazari M
and Rajih E: The role of phosphodiesterase inhibitors in the
management of cavernositis with multifocal abscesses: A report of
case series. Sex Med. 8:318–322. 2020.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Aliaev IuG, Vinarov AZ and Akhvlediani ND:
Wardenafil in combined treatment of patients with chronic bacterial
prostatitis. Urologiia. 6:52–55. 2008.PubMed/NCBI(In Russian).
|
|
41
|
Davenport C and Dubin A: Tadalafil therapy
and severe chronic foot wound resolution. Int Wound J. 12:733–736.
2015.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Kovalski V, Prestes AP, Oliveira JG, Alves
GF, Colarites DF, Mattos JE, Sordi R, Vellosa JC and Fernandes D:
Protective role of cGMP in early sepsis. Eur J Pharmacol.
807:174–181. 2017.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Ayten R, Cetinkaya Z, Girgin M, Ozercan I,
Ustundag B and Aygen E: The effects of intraperitoneal sildenafil
administration on healing of left colonic anastomoses and
intra-abdominal adhesion formation in the presence of
intra-abdominal infection. Dis Colon Rectum. 51:1837–1841.
2008.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Hosgood SA, Randle LV, Patel M, Watson CJ,
Bradley JA and Nicholson ML: Sildenafil citrate in a donation after
circulatory death experimental model of renal ischemia-reperfusion
injury. Transplantation. 98:612–617. 2014.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Korom S, Hillinger S, Cardell M, Zhai W,
Tan Q, Dutly A, Leskosek B and Weder W: Sildenafil extends survival
and graft function in a large animal lung transplantation model.
Eur J Cardiothorac Surg. 29:288–293. 2006.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Amano T, Earle C, Imao T, Matsumoto Y and
Kishikage T: Administration of daily 5 mg tadalafil improves
endothelial function in patients with benign prostatic hyperplasia.
Aging Male. 21:77–82. 2018.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Andrigueti FV, Ebbing PCC, Arismendi MI
and Kayser C: Evaluation of the effect of sildenafil on the
microvascular blood flow in patients with systemic sclerosis: A
randomised, double-blind, placebo-controlled study. Clin Exp
Rheumatol. 35 (Suppl 106):151–158. 2017.PubMed/NCBI
|
|
48
|
Thieme M, Sivritas SH, Mergia E, Potthoff
SA, Yang G, Hering L, Grave K, Hoch H, Rump LC and Stegbauer J:
Phosphodiesterase 5 inhibition ameliorates angiotensin II-dependent
hypertension and renal vascular dysfunction. Am J Physiol Renal
Physiol. 312:F474–F481. 2017.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Aydin S, Kuloglu T, Aydin S, Yardim M,
Azboy D, Temizturk Z, Kalkan AK and Eren MN: The effect of iloprost
and sildenafil, alone and in combination, on myocardial ischaemia
and nitric oxide and irisin levels. Cardiovasc J Afr. 28:389–396.
2017.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Santi D, Granata AR, Guidi A, Pignatti E,
Trenti T, Roli L, Bozic R, Zaza S, Pacchioni C, Romano S, et al:
Six months of daily treatment with vardenafil improves parameters
of endothelial inflammation and of hypogonadism in male patients
with type 2 diabetes and erectile dysfunction: A randomized,
double-blind, prospective trial. Eur J Endocrinol. 174:513–522.
2016.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Valatsou A, Briasoulis A, Vogiatzi G,
Pantopoulou A, Oikonomou E, Miliou A, Perrea D and Tousoulis D:
Beneficial effects of sildenafil on tissue perfusion and
inflammation in a murine model of limb ischemia and
atherosclerosis. Curr Vasc Pharmacol. 15:282–287. 2017.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Sheng M, Lu H, Liu P, Li Y, Ravi H, Peng
SL, Diaz-Arrastia R, Devous MD and Womack KB: Sildenafil improves
vascular and metabolic function in patients with Alzheimer's
disease. J Alzheimers Dis. 60:1351–1364. 2017.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Palma G, Giordano R, Russolillo V, Cioffi
S, Palumbo S, Mucerino M, Poli V and Vosa C: Sildenafil therapy for
pulmonary hypertension before and after pediatric congenital heart
surgery. Tex Heart Inst J. 38:238–242. 2011.PubMed/NCBI
|
|
54
|
Kelly LE, Ohlsson A and Shah PS:
Sildenafil for pulmonary hypertension in neonates. Cochrane
Database Syst Rev. 8(CD005494)2017.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Mondaini N: Phosphodiesterase type 5
inhibitors and COVID-19: are they useful in disease management?
World J Mens Health. 38:254–255. 2020.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Dal Moro F and Livi U: Any possible role
of phosphodiesterase type 5 inhibitors in the treatment of severe
COVID19 infections? A lesson from urology. Clin Immunol.
214(108414)2020.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Solaimanzadeh I: Acetazolamide, nifedipine
and phosphodiesterase inhibitors: Rationale for their utilization
as adjunctive countermeasures in the treatment of coronavirus
disease 2019 (COVID-19). Cureus. 12(e7343)2020.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Dal Moro F, Vendramin I and Livi U: The
war against the SARS-CoV2 infection: Is it better to fight or
mitigate it? Med Hypotheses. 143(110129)2020.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Reinert JP and Reinert NJ: The role of
phosphodiesterase-5 inhibitors in COVID-19: An exploration of
literature from similar pathologies. J Intensive Care Med. 36:3–8.
2020.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Groner MF, de Carvalho RC, Camillo J,
Ferreira PR and Fraietta R: Effects of Covid-19 on male
reproductive system. Int Braz J Urol. 47:185–190. 2021.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Batiha O, Al-Deeb T, Al-Zoubi E and
Alsharu E: Impact of COVID-19 and other viruses on reproductive
health. Andrologia. 52(e13791)2020.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Shen Q, Xiao X, Aierken A, Yue W, Wu X,
Liao M and Hua J: The ACE2 expression in Sertoli cells and germ
cells may cause male reproductive disorder after SARS-CoV-2
infection. J Cell Mol Med. 24:9472–9477. 2020.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Pan F, Xiao X, Guo J, Song Y, Li H, Patel
DP, Spivak AM, Alukal JP, Zhang X, Xiong C, et al: No evidence of
severe acute respiratory syndrome-coronavirus 2 in semen of males
recovering from coronavirus disease 2019. Fertil Steril.
113:1135–1139. 2020.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Fraietta R, Pasqualotto FF, Roque M and
Taitson PF: SARS-COV-2 and male reproductive health. JBRA Assist
Reprod. 24:347–350. 2020.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Khalili MA, Leisegang K, Majzoub A,
Finelli R, Panner Selvam MK, Henkel R, Mojgan M and Agarwal A: Male
fertility and the COVID-19 pandemic: Systematic review of the
literature. World J Mens Health. 38:506–520. 2020.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Sofikitis N, Kaltsas A, Dimitriadis F,
Rassweiler J, Grivas N, Zachariou A, Kaponis A, Tsounapi P,
Paterakis N, Karagiannis A, et al: The Effect of PDE5 inhibitors on
the male reproductive tract. Curr Pharm Des: Feb 26, 2020 (Epub
ahead of print). doi: 10.2174/1381612826666200226121510.
|
|
67
|
Corvasce A, Albino G, Leonetti T, Buonomo
AF and Marucco EC: Once-a-day Tadalafil administration improves the
spermogram parameters in fertile patients. Arch Ital Urol Androl.
87:210–213. 2015.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Yuan J, Zhang R, Yang Z, Lee J, Liu Y,
Tian J, Qin X, Ren Z, Ding H, Chen Q, et al: Comparative
effectiveness and safety of oral phosphodiesterase type 5
inhibitors for erectile dysfunction: A systematic review and
network meta-analysis. Eur Urol. 63:902–912. 2013.PubMed/NCBI View Article : Google Scholar
|