Introduction
Aortic dissection (AD) is a life-threatening disease
that requires early diagnosis and treatment to prevent death. The
estimated incidence is 2.5 to 3.5 per 100,000 person-years
(1). The mortality rate for acute
AD increases by 1% per hour during the first 24 h (2), and the overall mortality rate for
patients with acute AD complicated by adverse perfusion syndrome
may be as high as 45% (3). The
high mortality rate of AD underscores the need for rapid
identification and treatment.
The Stanford classification has been used in
clinical practice for a number of years. Type A dissection involves
the ascending aorta and type B dissection begins in the descending
aorta. The most typical presentation is a sudden onset of tearing
pain in the chest or back (4).
Type A is considered to be a surgical emergency, while type B is
more often treated with medication. Painless AD is relatively rare,
but is more likely to be missed or misdiagnosed due to the atypical
presentation. Numerous patients die before they reach a hospital or
obtain a definitive diagnosis (1).
For such cases, it is necessary to maintain a high degree of
vigilance to avoid misdiagnosis and mistreatment. The present study
report a case of painless AD in a patient who was admitted to
hospital with symptoms of flatulence and a reduced defecation
frequency. The preliminary diagnosis was ileus, but the patient was
eventually diagnosed with painless type B AD.
Case report
A 45-year-old man was admitted to Bethune
International Peace Hospital (Shijiazhuang, China) in October 2015
due to flatulence accompanied by reduced defecation for 8 days. At
8 days prior to admission, the patient felt continuous abdominal
fullness without obvious inducement and the defecation frequency
was reduced, but there was no abdominal pain, nausea or vomiting.
Abdominal distension could be relieved after flatus. Food intake
was significantly reduced, with no aversion to greasy food,
abdominal enlargement was not observed and the urine was normal. At
5 days prior to admission, the patient came to the Outpatient
Department of the Bethune International Peace Hospital, and at that
time, blood routine examination, electrolyte levels, liver
function, renal function and blood lipid levels were normal. Double
adrenal color ultrasound indicated no obvious abnormalities in both
renal adrenal areas and double renal arteries. An abdominal
vertical X-ray showed gas accumulation in the intestine without an
air-fluid level. Laxatives were not very effective. After oral
administration of 250 ml mannitol 3 days before admission, yellow
sparse defecation occurred 4-5 times. However, the abdominal
distention became significantly worse, and the patient stopped
defecating and passing gas 2 days before admission. Due to these
symptoms, the patient attended the Department of Gastroenterology
in the Bethune International Peace Hospital. Since the onset of the
disease, the patient had consumed a poor diet and had lost 6 kg of
body weight. The patient had a history of hypertension for >10
years, with levels up to 180/100 mmHg. Intermittent oral
administration of indapamide had resulted in poor blood pressure
control, fluctuating at ~160/100 mmHg. At the physical examination
on admission, the patient had a body temperature of 36.6˚C, a pulse
rate of 104 beats/min, a respiration rate of 18 breaths/min and a
blood pressure of 156/102 mmHg. There was no obvious abnormality of
the heart and lungs, and no obvious tenderness and rebound pain in
the whole abdomen. No mass was palpable and auscultation showed
hypoactive bowel sounds.
After admission, laboratory tests showed no
abnormalities in the blood, urine and stool routine examinations,
or in liver and kidney function, electrolyte levels, coagulation
and tumor markers, blood gas analysis and lactic acid levels. No
abnormality was found upon cardiac, abdominal or pelvic ultrasound.
After clean enema and catharsis treatment, there was still no
flatus or defecation. An AD was incidentally found on abdominal
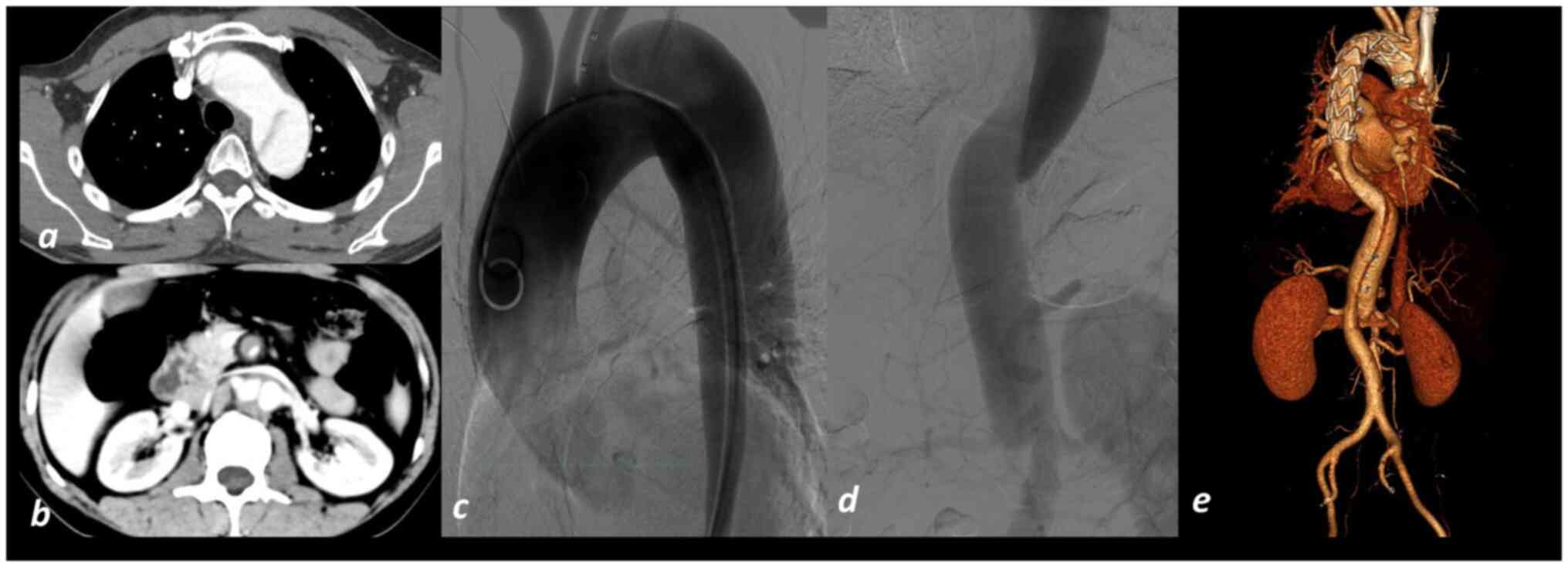
computed tomography angiography (CTA). To further delineate the
extent of the dissection, CTA of the aorta was performed, which
confirmed a type B AD where the AD extended from the arch of the
aorta to the superior mesenteric artery (Fig. 1a and b). The diagnosis of type B AD was
definitive, and the patient was treated using a micropump to
continuously administer intravenous urapidil hydrochloride (50 mg)
and sodium nitroprusside (50 mg) for blood pressure and heart rate
control. Aortic angiography and a thoracic aorta coated stent
implantation were performed (Fig.
1c-e). At 11 days post-surgery, the patient had recovered and
was discharged. Follow-up was performed every 3-6 months, and after
5 years of follow-up, the general condition of the patient was
good, and the blood pressure was well controlled by oral
antihypertensive drugs.
Discussion
AD, also known as an aortic dissecting aneurysm,
refers to a serious aortic disease in which the blood in the aortic
intima passes through the intima tear and enters the middle layer
of the arterial wall to form a dissecting hematoma, which expands
along the long axis of blood vessels and forms pathological changes
in the true and false lumens of the artery (5). The typical clinical presentation of
AD is a tearing pain in the chest, abdomen or back, but 4.5-6% of
AD is painless, and these cases have a very high rate of missed
diagnosis (6). The preponderant
use of medication for type B AD does not mean that these patients
do not have a complex dissection, as they may still experience poor
perfusion and aortic rupture. Tolenaar et al (7) found an increased trend in in-hospital
mortality among patients with type B AD, which may be the result of
the delayed diagnosis and treatment due to a lack of typical
symptoms. Park et al (8)
found that patients with painless type B AD had significantly
higher rates of mortality and aortic rupture than patients with
type A AD with pain. Since painless type B AD is associated with
increased mortality, timely identification of these patients is
important.
Hypertension is the most important risk factor for
AD. In total, 65-75% of patients with AD have hypertension, and
their blood pressure is poorly controlled. In addition to the
absolute increase in blood pressure, the increase in blood pressure
change rate is also a factor causing AD. In addition
atherosclerosis and aging are also important risk factors for AD
(6). In the present case, the
patient had a long history of hypertension, which was poorly
controlled, and this was considered as the main cause of the
AD.
Retrospectively, although the patient had no obvious
abdominal pain, the clinical manifestations of type B AD were more
like ileus; however, x-rays did not support this diagnosis. Until
CT found an AD involving the superior mesenteric artery, AD-related
hypoperfusion was considered to be the primary cause of the ileus
symptoms. At 2 days prior to admission, the patient took mannitol
orally to relieve the constipation, and after discharging loose
stools several times, defecation and flatus stopped completely. It
was considered that the aggravation of the condition, perhaps
related to the mannitol stimulating gastrointestinal peristalsis,
lead to the expansion and progression of the dissection, which
increased the risk of intestinal ischemia, intestinal necrosis and
dissection rupture. Although mesenteric complications occur in only
5% of patients with type B AD, the mortality rate is higher at
≤33.3% (9).
The reasons for the absence of pain in patients with
AD are unknown, but according to reports, there are several
possible reasons: i) The hematoma dilates into the aortic lumen,
lateral pressure is low for the outer membrane with plexus
distribution and there is no pain caused by the outer membrane
protruding; ii) the dissection progresses slowly, and chronic
stretch stimulation may increase the pain threshold (7); iii) the severe spinal cord ischemia
that has occurred makes the viscera, spinal cord and thalamus lose
their ability to sense pain, thus the ability of the patient to
sense pain is reduced (10); iv)
the gap between the false cavity and the true cavity is large and
numerous, the pressure in the false cavity is low and no obvious
pain is caused. A previous stud has also shown that patients with
AD who have a history of Marfan syndrome may also present with
painless symptoms (11).
There were a number of deficiencies in the diagnosis
and treatment process of this case, such as the pre-existing
diagnosis of ileus, ignoring hypertension as the main cause,
failing to compare the blood pressure of both upper limbs and
failing to pay attention to vascular murmurs due to insufficient
physical examination. Painless AD is rare in the clinic. The
present case report suggested that there was an insufficient blood
supply, mainly due to the tear of the intima and media of the
vessel, and blood flowing into the interlayer from its own tubular
channels, resulting in hemodynamic changes. An insufficient distal
blood supply may lead to reduced or absent arterial pulsation,
ischemia of the head may lead to lethargy, apathy and syncope, and
ischemia of the spinal cord may lead to limb weakness and
paraplegia (12,13). When dissection occurs in the aortic
arch, there is a significant difference in the blood supply between
the brachiocephalic trunk and the left subclavian artery, which may
result in a significant difference in blood pressure between the
left and right upper limbs. The same thing can happen in other
parts of the body to cause huge differences between the upper and
lower limbs (14). As the torn
intima forms a ‘reservoir sac’, this portion of blood may fall back
during diastole to cause aortic valve murmurs (12). Therefore, a new aortic murmur is
also a high-risk sign for AD; however, it has been reported that
32% of patients with painless AD have no vascular murmur (15). Clinical attention to the physical
examination can be lead to a more timely diagnosis of painless AD.
Fortunately, in the present study, the AD was incidentally found
during abdominal CTA, which resulted in the correct diagnosis and
treatment, and avoided adverse consequences. Therefore, physicians
should be aware of this relatively rare presentation of painless
type B AD.
In conclusion, the clinical manifestations of
typical AD are easy to recognize, but the hidden and complex
clinical manifestations are easy to ignore or misdiagnose. The rate
of missed diagnosis and misdiagnosis in patients with first
painless AD is higher than that in patients with pain, and the
mortality rate is higher. Therefore, clinicians should be alert to
painless symptoms and subtle signs related to AD, and should pay
enough attention to them.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
CW was responsible for the clinical management of
the patient, and the processing and scientific interpretation of
the data. BW, JZ and YH were responsible for evaluating the data,
revising the manuscript, analyzing the specialized literature and
writing the manuscript. ZL and JY were responsible for data
processing and revising the manuscript. CW was responsible for
analyzing the specialized literature, interpreting and evaluating
the data, and revising the manuscript. JY, JZ and YH confirm the
authenticity of all the raw data. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent was obtained from the
patient.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Khan IA and Nair CK: Clinical, diagnostic,
and management perspectives of aortic dissection. Chest.
122:311–328. 2002.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Braverman AC: Acute aortic dissection:
Clinician update. Circulation. 122:184–188. 2010.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Juraszek A, Czerny M and Rylski B: Update
in aortic dissection. Trends Cardiovasc Med: Aug 16, 2021 (Epub
ahead of print).
|
|
4
|
Erbel R, Aboyans V, Boileau C, Bossone E,
Bartolomeo RD, Eggebrecht H, Evangelista A, Falk V, Frank H,
Gaemperli O, et al: 2014 ESC Guidelines on the diagnosis and
treatment of aortic diseases: Document covering acute and chronic
aortic diseases of the thoracic and abdominal aorta of the adult.
The Task Force for the Diagnosis and Treatment of Aortic Diseases
of the European Society of Cardiology (ESC). Eur Heart J.
35:2873–2926. 2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Clouse WD, Hallett JW Jr, Schaff HV,
Spittell PC, Rowland CM, Ilstrup DM and Melton LJ III: Acute aortic
dissection: Population-based incidence compared with degenerative
aortic aneurysm rupture. Mayo Clin Proc. 79:176–180.
2004.PubMed/NCBI View
Article : Google Scholar
|
|
6
|
Alter SM, Eskin B and Allegra JR:
Diagnosis of aortic dissection in emergency department patients is
rare. West J Emerg Med. 16:629–631. 2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Tolenaar JL, Hutchison SJ, Montgomery D,
O'Gara P, Fattori R, Pyeritz RE, Pape L, Suzuki T, Evangelista A,
Moll FL, et al: Painless type B aortic dissection: Insights from
the International registry of acute aortic dissection. Aorta
(Stamford). 1:96–101. 2013.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Park SW, Hutchison S, Mehta RH,
Isselbacher EM, Cooper JV, Fang J, Evangelista A, Llovet A,
Nienaber CA, Suzuki T, et al: Association of painless acute aortic
dissection with increased mortality. Mayo Clin Proc. 79:1252–1257.
2004.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Fukunaga N, Konishi Y, Matsuo T, Saji Y
and Koyama T: Paralytic ileus as the first presentation in type A
acute aortic dissection. J Med Invest. 64:286–287. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Gerber O, Heyer EJ and Vieux U: Painless
dissections of the aorta presenting as acute neurologic syndromes.
Stroke. 17:644–647. 1986.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Algarni KD, Arafat AA, Adam AI and
Pragliola C: Giant ascending aortic aneurysm with painless
dissection in a patient with marfan syndrome. J Saudi Heart Assoc.
32(284)2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Marroush TS, Boshara AR, Parvataneni KC,
Takla R and Mesiha NA: Painless aortic dissection. Am J Med Sci.
354:513–520. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Hdiji O, Bouzidi N, Damak M and Mhiri C:
Acute aortic dissection presenting as painless paraplegia: A case
report. J Med Case Rep. 10(99)2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Levy D, Goyal A, Grigorova Y, Farci F and
Le JK: Aortic Dissection. In: StatPearls. StatPearls Publishing.
Copyright©. 2020, StatPearls Publishing LLC, Treasure
Island, FL, 2020.
|
|
15
|
Hagan PG, Nienaber CA, Isselbacher EM,
Bruckman D, Karavite DJ, Russman PL, Evangelista A, Fattori R,
Suzuki T, Oh JK, et al: The International registry of acute aortic
dissection (IRAD): New insights into an old disease. JAMA.
283:897–903. 2000.PubMed/NCBI View Article : Google Scholar
|