Introduction
Severe preeclampsia is one of the most serious
hypertensive disorders of pregnancy; it is characterized by a
persistent increase in blood pressure after 20 weeks of gestation,
often alongside maternal organ function impairment and
fetal-placental complications (1).
The pathogenesis of this disease has not been fully elucidated. The
fundamental pathophysiological changes are systemic vasospasm and
endothelial injury, but the pathogenesis of severe preeclampsia
caused by vascular endothelial dysfunction is still unclear
(2). Transient receptor potential
cation channel subfamily V member 1 (TRPV1) has attracted
increasing attention in the study of the cardiovascular system in
recent years (3-5).
A study showed that dietary capsaicin could activate TRPV1 on
endothelial cells and promote the endothelial nitric oxide synthase
(eNOS)/NO pathway, which may enhance endothelial production of NO
and reduce blood pressure in rats with spontaneous hypertension
(6). The ATP-sensitive potassium
channel (KATP), a specific type of voltage-dependent potassium ion
channel, is composed of an inwardly rectifying potassium channel
(Kir) and sulfonylurea receptor (SUR), and the SUR2B/Kir6.1 subtype
is also known as the vascular type (7). Studies have shown that the KATP
channel-specific agonist etakarin not only directly relaxes
vascular smooth muscle, but also acts on endothelial cells to
increase eNOS expression and promote the synthesis and release of
NO (8,9). Current correlations have been
reported between transient receptor potential channels and
potassium ion channels (4,10,11).
A study confirmed the involvement of large-conductance K+ (BKca) in
coronary endothelium-dependent relaxation mediated by
TRPV1(4); however, to the best of
our knowledge, there are no reports on the role of TRPV1 and the
KATP subtype SUR2B/Kir6.1 in severe preeclampsia. Since the
placenta is a vital tissue that connects the fetus to the mother,
and once the placenta has been delivered, this illness may be
rapidly treated; there is no doubt that the placenta has an
essential role in the development of this disease (12). As one of the vasoactive substances
regulating blood flow homeostasis in the body, NO must also be one
of the crucial factors regulating blood flow perfusion in the
placental artery (13). Therefore,
the present study aimed to investigate whether severe preeclampsia
was associated with impaired vasodilation mediated by TRPV1-KATP
channels in the placental artery, leading to deficient eNOS/NO
pathway activity and severe maternal vascular endothelial
dysfunction.
Materials and methods
Management of human placental
arteries
The present study was approved by the Clinical Trial
Ethics Committee of the Affiliated Hospital of Southwest Medical
University (approval no. KY2019039). All work was performed in
accordance with the provisions of the Declaration of Helsinki and
its later amendments. All patients provided written informed
consent for specimen collection. A total of 10 pregnant women with
a singleton pregnancy diagnosed with severe preeclampsia were
selected as the experimental group (SP group), whose average
maternal age ranged from 23 to 41 years, with a mean age of
32.20±5.226 years. In addition, 10 pregnant women with a singleton
pregnancy with normotensive pregnancies were selected as the
control group (NP group), whose average maternal age ranged from 22
to 44 years, with a mean age of 30.40±5.475 years (Table I). All patients were hospitalized
and delivered by either vaginal delivery or cesarean section at the
Department of Obstetrics, The Affiliated Hospital of Southwest
Medical University (no. 8, Section 2, Kangcheng Road, Luzhou,
Sichuan, China) between May 2020 and May 2021. The inclusion
criteria for the SP group were based on the American College of
Obstetricians and Gynecologists Guidelines (14). Subjects were excluded if they had
chronic hypertension, kidney disease, cardio-cerebrovascular
disease, severe liver and kidney function impairments, other
primary diseases, systemic diseases or other pregnancy
complications. Patients with a history of smoking, alcohol abuse,
syphilis, hepatitis virus or human immunodeficiency virus were also
excluded. Blood samples were acquired from the mother by peripheric
venous puncture before any medical treatments were administered.
Samples were then placed in anticoagulant tubes, centrifuged at
3,000 x g for 15 min at room temperature and stored in a -20˚C
refrigerator until they were used for extraction. After the
placenta was delivered, the fetal membrane was immediately stripped
under sterile conditions, and 3-5 pieces of placental tissues with
a small placental artery near the edge of the placenta, ~4x1x1
cm3 in size, were extracted and placed in the prepared
specimen box, which was immediately transferred to the laboratory
under low-temperature conditions. Next, the perivascular connective
tissue was rapidly and gently removed under a microscope, taking
care to minimize the damage to the blood vessels, and vessels with
diameters of 0.1-0.2 cm and lengths of 2-3 cm were separated. Some
of the isolated blood vessels were frozen in liquid nitrogen and
then quickly transferred to a -80˚C freezer for cryopreservation,
and these samples were used for subsequent PCR and western blot
analyses, and the nitrate reductase method. The remainder were
fixed in 4% paraformaldehyde for >24 h at room temperature, and
the specimens were used for subsequent hematoxylin-eosin staining
and immunohistochemical analysis.
 | Table IClinical characteristics. |
Table I
Clinical characteristics.
| Category | Severe
preeclampsia | Normotensive
pregnancy | t | P-value |
|---|
| Maternal age,
years | 32.2±5.226 | 30.40±5.475 | 0.9211 | 0.365 |
| Gestational age,
weeks | 33.18±1.102 | 38.78±1.015 | 3.439 |
<0.001a |
| Maximum systolic
pressure, mmHg | 172.0±8.577 | 112.7±8.932 | 18.54 |
<0.001a |
| Maximum diastolic
pressure, mmHg | 117.2±8.711 | 72.93±7.887 | 11.29 |
<0.001a |
Hematoxylin-eosin staining of the
placental arteries
Hematoxylin and eosin (H&E) staining was
performed as previously described (15,16).
To deparaffinize the paraffin sections, slides were submerged for 5
min in a slide jar filled with xylene (Sinopharm Chemical Reagent
Co., Ltd.), repeating this procedure twice and using fresh xylene
each time. Subsequently, sections were rehydrated by running slides
through a series of decreasing concentrations of EtOH (Sinopharm
Chemical Reagent Co., Ltd.). Hematoxylin (Shanghai Jingke Chemical
Technology Co., Ltd.) was used to stain the tissues for 5 min at
room temperature, and after washing with tap water, differentiation
in staining was visualized by submerging for 30 sec in 1% acetic
acid. To visualize the blue staining, the sections were rinsed
under running tap water for 5 min, after which the slices were dyed
in eosin solution (Shanghai Jingke Chemical Technology Co., Ltd.)
for 2 min at room temperature. After dehydration with EtOH and
transparency with xylene, the slices were sealed with neutral
balsam (G8590-100; Solarbio). Finally, tissues were imaged under an
electron microscope (400x magnification; OLYMPUS CX-21; Shanghai
Xinyu Biotechnology Co., Ltd.).
Cell culture
HUVECs (cat. no. CL-0675; Procell Life Science &
Technology Co., Ltd.) were cultured in high-glucose DMEM (AMEKO;
cat. no. FB8025; Shanghai Lianshuobaowei Biological Technology Co.,
Ltd.) supplemented with 10% FBS (Guangzhou Ruite Biological
Technology Co., Ltd.) and 1% penicillin and streptomycin (Shanghai
Biyuntian Biotechnology Co., Ltd.) at 5% CO2 and 37˚C in
a humidified chamber. HUVECs in the logarithmic growth stage were
subcultured and inoculated in six-well plates. The cells were
divided into a control group, a capsaicin group (cat. no. HY-10448;
MedChemExpress) and a capsazepine group (cat. no. HY-15640MCE;
MedChemExpress). After reaching ~80% confluency, high-glucose DMEM
containing capsaicin and capsazepine, prepared in DMSO to a final
concentration of 1 µmol/l, was added. This concentration was based
on a previous study by Yang et al (6), in which NO production was highest
with 1 µM capsaicin in cultured endothelial cells from Wistar rats.
In the control group, high-sugar DMEM with an equivalent volume of
DMSO was used. After 24 h of incubation, total RNA and protein were
extracted from the cells.
Reverse transcription-quantitative
(RT-q)PCR
Total RNA was extracted from tissues and cells using
TRIzol reagent (cat. no. 15596018; Thermo Fisher Scientific, Inc.),
the absorbance of the extracted RNA was measured using a
spectrophotometer on a Bio-Rad 3000 UV-vis spectrophotometer
(Bio-Rad Laboratories, Inc.), and the integrity was examined by gel
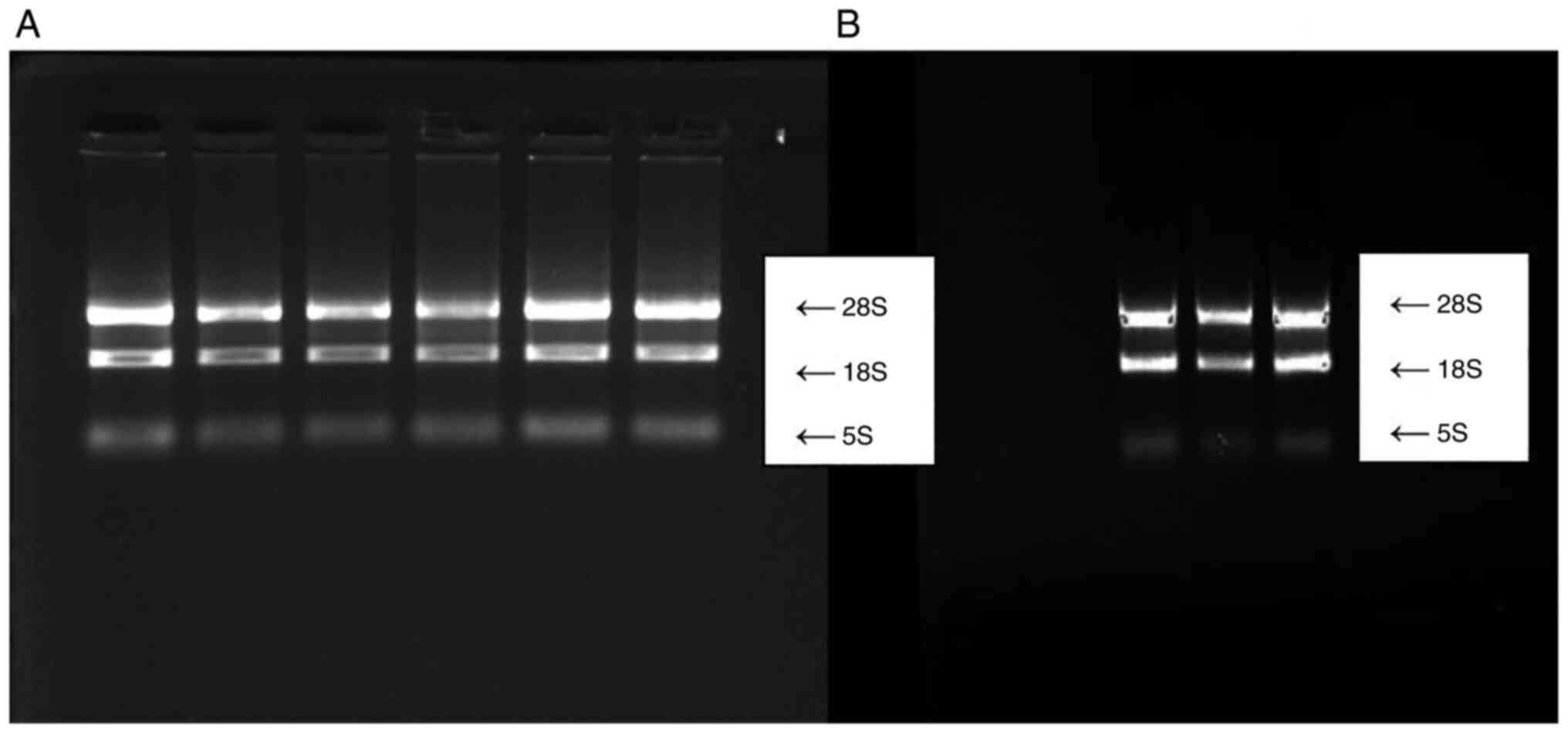
electrophoresis (Bio-Rad Laboratories, Inc.). RNA extracts with an
A260/A280 ratio between 1.8-2.0 and three complete bands of 28S,
18S and 5S, after 2% agarose gel electrophoresis, were chosen for
reverse transcription (Fig. 1A and
B). A cDNA Reverse Transcription
kit (Fuji Biological Co., Ltd.) was used to perform the reverse
transcription procedure using the following reverse transcription
temperature protocol: 42˚C for 15 min, 85˚C for 5 sec and cooling
at 4˚C. A SYBR Green MasterMix kit (Toyobo Co., Ltd.) was used for
qPCR in a StepOnePlus PCR System (Applied Biosystems; Thermo Fisher
Scientific, Inc.), in a 20-µl reaction system with the following
amplification temperature protocol: 95˚C for 10 min, followed by 40
cycles of 95˚C for 5 sec, 58˚C for 20 sec and 72˚C for 10 sec.
GAPDH was used as the internal reference, and the internal
reference and target gene primers were designed and synthesized by
Shanghai Sangon Biological Co., Ltd. (Table II). Finally, the relative
expression of each target gene was calculated using the
2-∆∆Cq method (17).
The experiments were repeated three times.
 | Table IIPrimer sequences. |
Table II
Primer sequences.
| Gene | Primer
sequence | PCR product size,
bp |
|---|
| GAPDH forward |
5'-CCACTCCTCCACCTTTG-3' | 106 |
| GAPDH reverse |
5'-CACCACCCTGTTGCTGT-3' | 106 |
| TRPV1 forward |
5'-CATCATCCTGCTGGCCTATG-3' | 105 |
| TRPV1 reverse |
5'-TTGCTCTGTGCGATCTTGTTG-3' | 105 |
| Kir6.1 forward |
5'-GATCATCTGCCACGTGATTGA-3' | 150 |
| Kir6.1 |
5'-GCAATGTAGGAGGTTCGTGCT-3' | 150 |
| SUR2B forward |
5'-CGGGACATAACCTGAGATGG-3' | 130 |
| SUR2B reverse |
5'-ATCACGGCTGGCATAAAGAG-3' | 130 |
| eNOS forward |
5'-GTGGCTGGTACATGAGCACT -3' | 180 |
| eNOS reverse |
5'-TGGCTAGCTGGTAACTGTGC-3' | 180 |
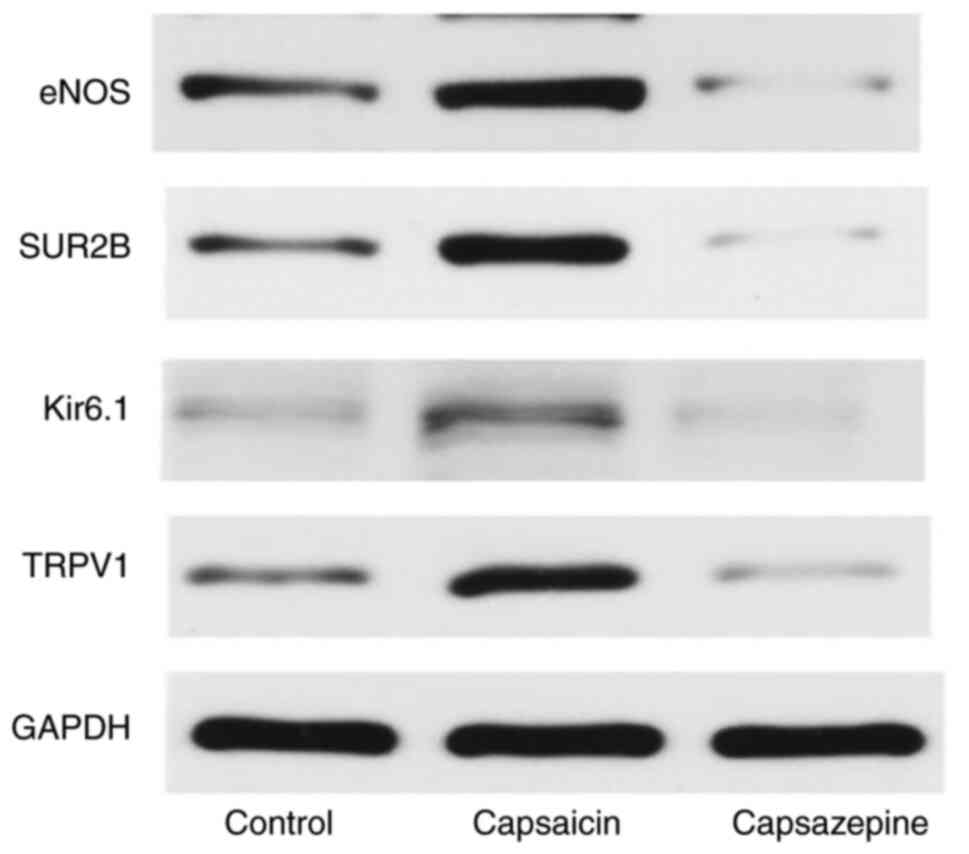
Western blot analysis
Total protein was extracted from tissues and cells
using RIPA lysis buffer (Biyuntian Biotechnology Co., Ltd.)
containing the protease inhibitor phenylmethanesulfonylfluoride
(Biyuntian Biotechnology Co., Ltd.), and protein concentration was
measured using a BCA protein assay kit (Biyuntian Biotechnology
Co., Ltd.). The protein samples were denatured, and 25 µg protein
per sample was loaded per lane on a 10% SDS gel, resolved using
SDS-PAGE and transferred to a PVDF membrane. The membrane was
blocked in 5% skimmed milk powder dissolved in 0.5% Tween-TBS for 2
h at room temperature. After washing the membranes, they were
incubated overnight at 4˚C on an agitator with one of the following
primary antibodies: Mouse anti-GAPDH (1:10,000; cat. no. MB001H;
Bioworld), rabbit anti-TRPV1 (1:1,000; cat. no. KL-KT-008292;
Abcam), rabbit anti-ABCC9 (also known as SUR2B; 1:500; cat. no.
ab84299; Abcam), rabbit anti-Kir6.1 (1:1,000; cat. no. TF6468R;
Crystal Wind Biological Co., Ltd.) or rabbit anti-eNOS (1:1,000;
cat. no. ab15280-eNOS; Abcam). The following day, after the
membranes were washed, the secondary HRP-conjugated Goat
Anti-Rabbit IgG antibody (1:1,000; cat. no. PF01924; Pufei Bio Co.,
Ltd.) was added and incubated at 37˚C for 2 h. Densitometry
analysis was performed using ImageJ version 1.49 (National
Institutes of Health). Experiments were repeated three times.
Immunohistochemistry
Immunohistochemistry was performed using an HRP
Streptavidin Conjugate kit according to the manufacturer's protocol
(DBA cat. no. Im-05818B; Shanghai Caiyou Industrial Co., Ltd.).
After baking at 60˚C for 2 h, the paraffin sections were dewaxed
and hydrated in turn, washed with PBS and incubated with 3%
hydrogen peroxide (TITABIO; cat. no. SY2622; Beijing Ita Biological
Co., Ltd.) for 5-10 min at room temperature to quench endogenous
peroxidase activity. The paraffin sections were subjected to
antigen retrieval in a microwave in EDTA buffer (cat. no. AS1016;
Wuhan Aspen Biotechnology Co., Ltd.). Subsequently, blocking
solution (solution B included in the kit) was added and samples
were incubated at room temperature for 30 min. The primary antibody
working solution containing anti-TRPV1 (1:200 dilution; cat. no.
KL-KT-008292; Abcam), anti-ABCC9 (1:200 dilution; cat. no. ab84299;
Abcam), anti-Kir6.1 (1:200 dilution; cat. no. TF6468R; Crystal Wind
Biological Co., Ltd.) or anti-eNOS (1:2,000 dilution; cat. no.
ab15280-eNOS; Abcam) was added to cover the sections, which were
incubated at 4˚C overnight. The following day, the samples were
rinsed, and to stain the sections, the
streptavidin-biotin-peroxidase complex was used after the sections
had been treated with biotinylated goat anti-rabbit immunoglobulin
G secondary antibody for 30 min at room temperature. A DBA solution
was used to develop the stain. Next, the samples were stained again
with hematoxylin for 5 min at room temperature and differentiated
by hydrochloric acid alcohol, dehydrated, made transparent and
sealed with neutral glue. Finally, the samples were observed and
imaged under a light microscope (400x magnification; Leica Aperio
AT2; Leica Microsystems, Co., Ltd.). The expression levels of
various proteins in the endothelial cells were observed.
Brown-yellow staining indicated positive expression, and the
optical density value represented the relative expression level.
The images were analyzed using Q-IMAGING software (MicroPublisher 6
CCD; Nano Hai Bioscientific Instruments Co., Ltd.).
Measurement of total nitrite
levels
An NO assay kit (Nanjing Jiancheng Biological Co.,
Ltd.) was used in the present study according to the manufacturer's
protocol. Prior to the experiment, solutions III, IV and V were
prepared into chromogenic agents according to the ratio of 2.5:1:1.
A total of 300 µl homogenate supernatant and 100 µl plasma was
deproteinized by adding 200 µl reagent I and stirring, after which
100 µl reagent II was added, the solution was stirred and
centrifuged at 3,500-4,500 x g for 15 min at 4˚C, and the
supernatant was recovered. A preheated microplate reader (BMG
LABTECH, CLARIOstar PLUS) (preheated for >30 min) was used and
the wavelength was adjusted to 550 nm. The samples to be tested
were added to the 96-well plate, mixed, and left for 15 min, after
which the OD value of absorbance in each well was measured using a
microplate reader.
Statistical analysis
All data were analyzed using SPSS version 20.0 (IBM
Corp.) and GraphPad Prism 5.0 (GraphPad Software, Inc.). Normally
distributed data are presented as the mean ± SD. An independent
samples t-test was used to compare the NP and SP groups, and a
one-way ANOVA followed by a Newman-Keuls multiple comparison
post-hoc test was used for multiple comparisons. P<0.05 was
considered to indicate a statistically significant difference.
Results
Patient demographics and clinical
characteristics
Table I presents
the demographic and clinical characteristics of the included
patients when they were admitted to hospital, including maternal
age, gestational age and blood pressure. There was no significant
difference in maternal age between the NP and SP groups
(P>0.05), but the gestational age difference was statistically
significant in the two groups (P<0.01). In addition, systolic
and diastolic blood pressure were significantly higher in the SP
group than in the NP group (P<0.01).
Endothelial layers are impaired in
severe preeclampsia
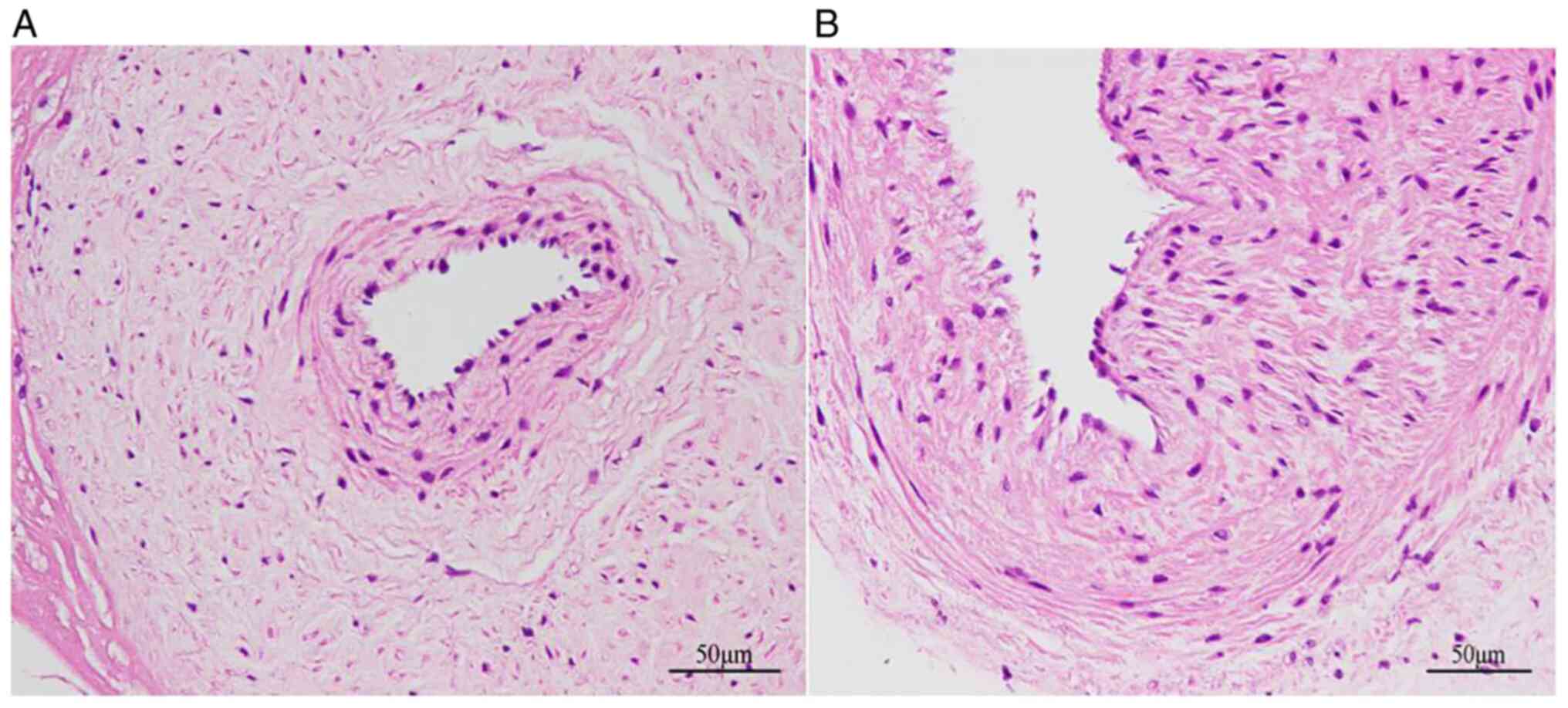
As shown in Fig. 2A
endothelial cells in the NP group were uniformly and regularly
arranged, and the nucleus was oblong, bluish in color and protruded
slightly from the official cavity. In the SP group, the walls of
the placental artery were thickened, the number of collagen fibers
had increased, the artery was hyalinized, the endothelial cells
were arranged in a disorderly manner and the endodermis was notably
damaged (Fig. 2B).
TRPV1, Kir6.1/SUR2B and eNOS
expression levels are decreased in the placental artery of patients
with severe preeclampsia
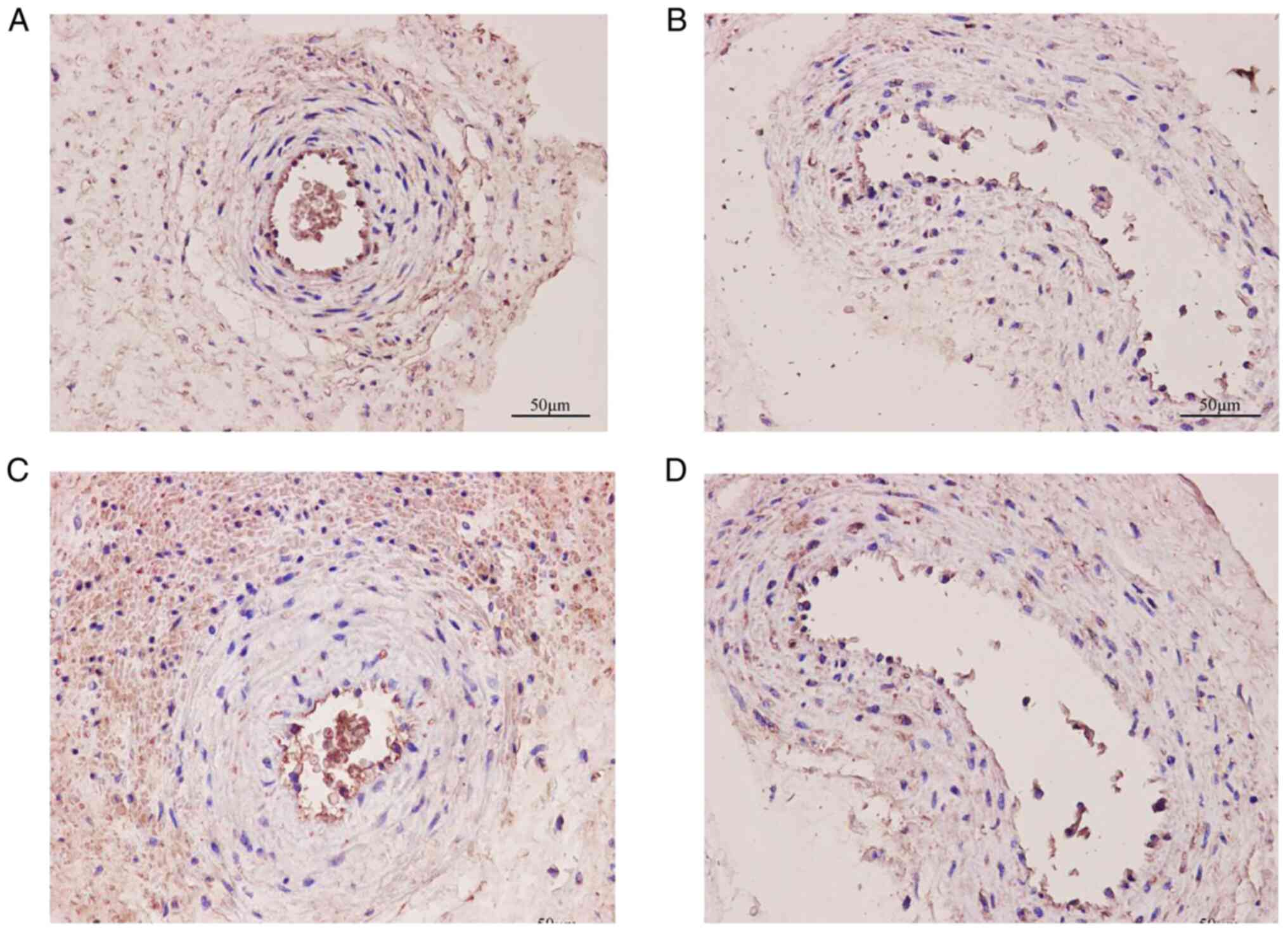
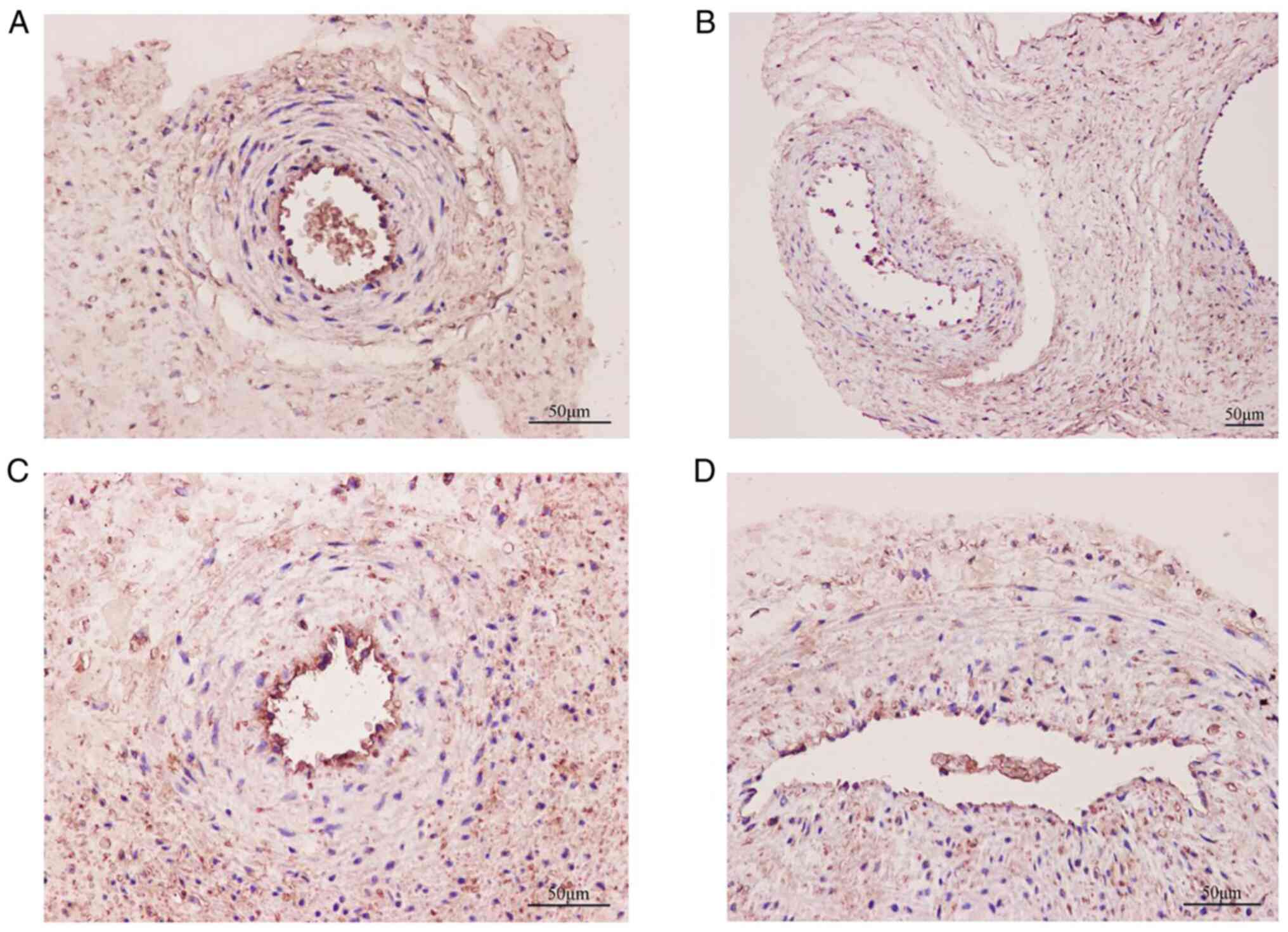
Immunohistochemistry was used to evaluate the
localization of the TRPV1, Kir6.1/SUR2B and eNOS channels in the
placental artery (Figs. 3 and
4). The results showed that each
channel was primarily distributed in the vascular endothelial
layer. It was noted that the number of counterstained nuclei in the
endothelial cell layer was significantly decreased, as was the
membrane staining of each channel in the SP group compared with the
NP group. The OD values of TRPV1, Kir6.1/SUR2B and eNOS in the SP
group were 11,660.0±1,721.0, 2,975.0±505.5, 10,236.0±1,355.0 and
14,663.0±2,320.0, respectively, which were lower than those in the
NP group (24,917.0±2,044.0, 4,495.0±775.0, 14,663.0±2,320.0, and
20,988.0±2,289.0; all P<0.05) (Fig.
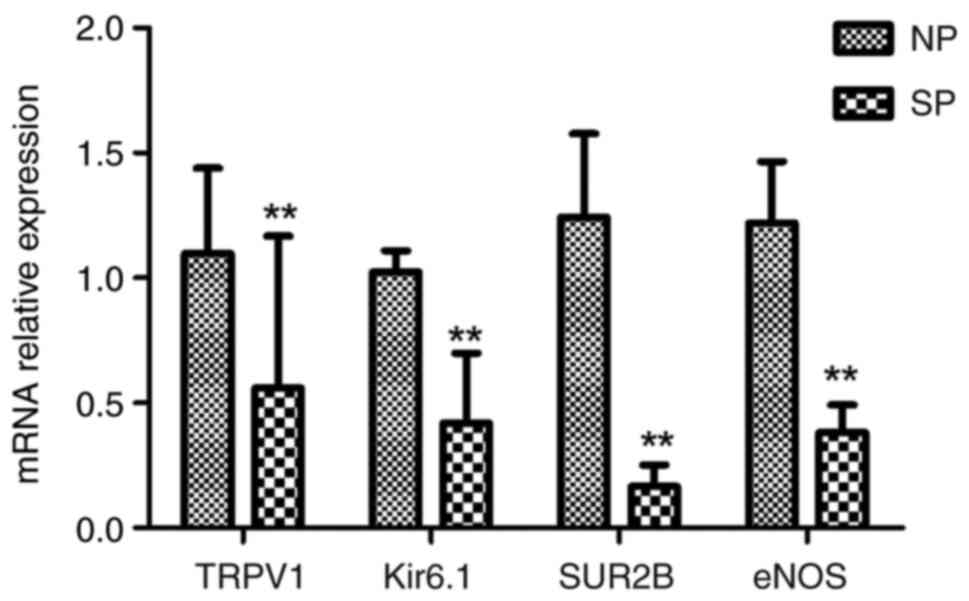
5). RT-qPCR was used to examine the relative expression of the
four-channel genes. The relative expression of TRPV1, Kir6.1, SUR2B
and eNOS in the SP group was 0.559±0.609, 0.419±0.281, 0.166±0.087
and 0.383±0.110, respectively, which was significantly lower than
that in the NP group (1.098±0.341, 1.024±0.085, 1.243±0.335 and
1.219±0.247; all P<0.01) (Fig.
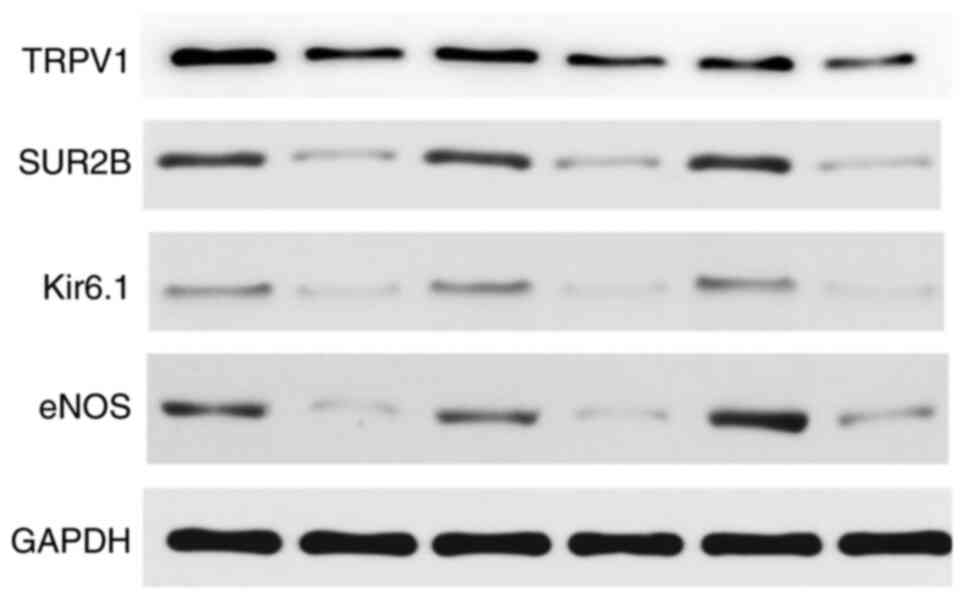
6). Since gene expression does not fully reflect protein
expression, western blotting was used to quantitatively examine
whether this difference was also present at the protein level
(Fig. 7). The relative
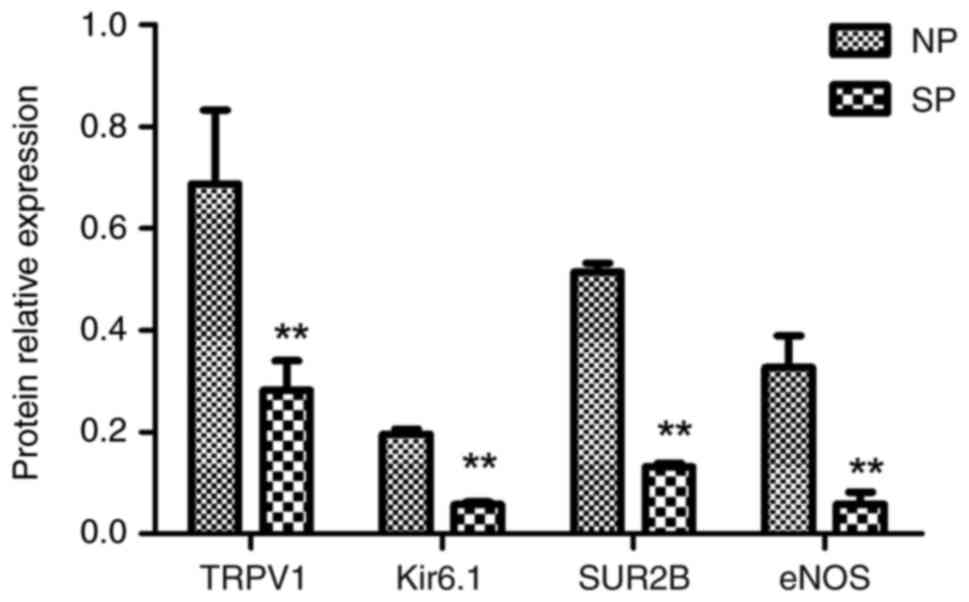
quantitative analysis results showed that the ratio of the gray
value of each target protein to GAPDH in the SP group was
0.282±0.058, 0.058±0.005, 0.132±0.007 and 0.059±0.023,
respectively, which was lower than that in the NP group
(0.688±0.145, 0.196±0.010, 0.514±0.018 and 0.327±0.063; all
P<0.01) (Fig. 8), suggesting
that the relative protein expression of TRPV1, KATP subtype
Kir6.1/SUR2B and eNOS in the SP group was also significantly
downregulated.
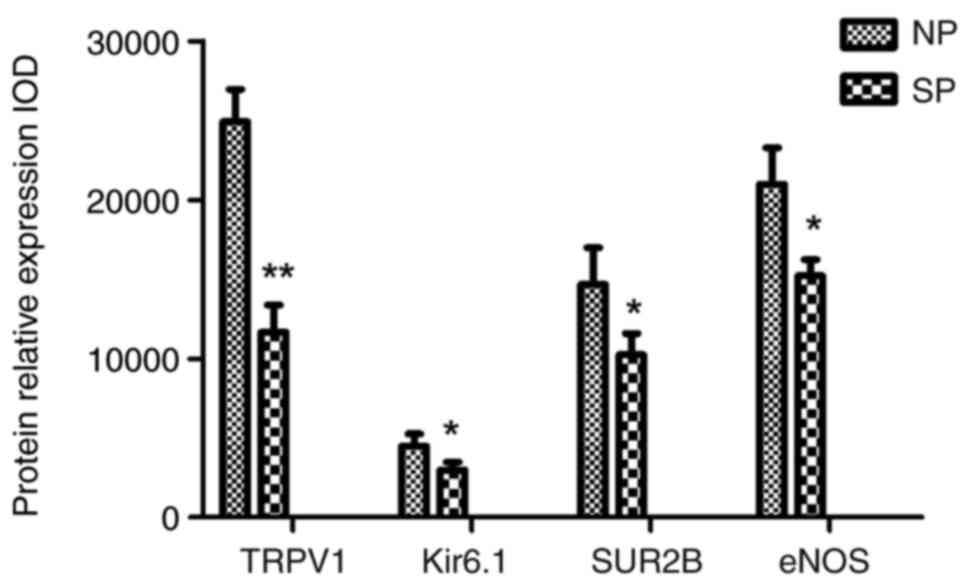 | Figure 5Histogram of the optical density of
TRPV1, Kir6.1/SUR2B and eNOS expression in the two groups based on
immunohistochemistry. TRPV1, Kir6.1/ SUR2B and eNOS levels in the
SP group were significantly lower than those in the NP group.
*P<0.05 and **P<0.01 vs. NP. SP, severe
preeclampsia; NP, normotensive pregnancy; eNOS, endothelial nitric
oxide synthase. SP, severe preeclampsia; NP, normotensive
pregnancy; eNOS, endothelial nitric oxide synthase; TRPV1,
transient receptor potential cation channel subfamily V member 1;
IOD, integrated optical density. |
Roles of eNOS and Kir6.1/SUR2B
channels in capsaicin/ capsazepine-induced relaxation in
HUVECs
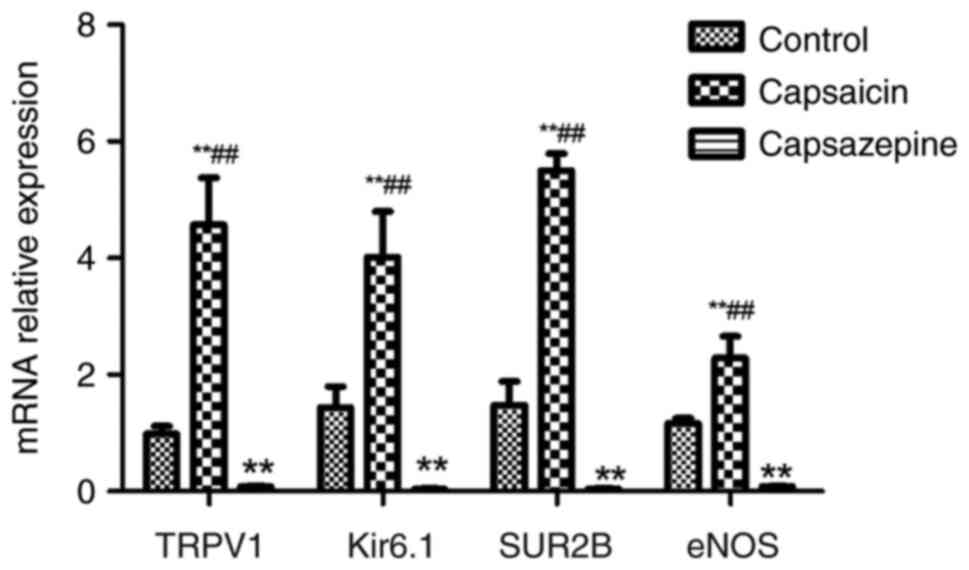
To explore the interactions of the four channels,
further cytological experiments were performed. The relative gene
expression of TRPV1, Kir6.1, SUR2B and eNOS in the control group
was 0.986±0.129, 1.439±0.358, 1.479±0.403 and 1.162±0.090,
respectively, while in the capsaicin group, it was 4.568±0.810,
4.014±0.781, 5.505±0.287 and 2.821±0.377, respectively, and in the
capsazepine group, it was 0.077±0.010, 0.036±0.014, 0.046±0.010 and
0.083±0.005, respectively; the differences between the control
group and capsaicin groups, and the capsazepine group were
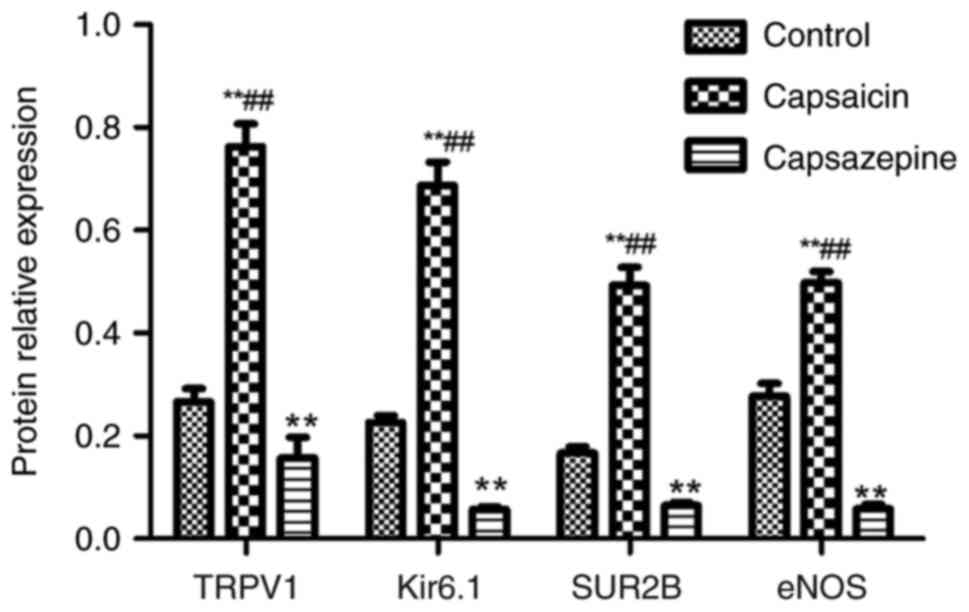
statistically significant (all P<0.01; Fig. 9). The results of western blotting
showed that the relative protein expression of TRPV1, Kir6.1, SUR2B
and eNOS in the control group was 0.266±0.026, 0.226±0.014,
0.166±0.013 and 0.277±0.025, respectively, while the expression in
the capsaicin group was 0.763±0.044, 0.687±0.046, 0.493±0.035 and
0.498±0.022, respectively, and expression in the capsazepine group
was 0.157±0.040, 0.057±0.005, 0.065±0.005 and 0.058±0.008,
respectively (all P<0.01), and these differences between the
control group and capsaicin groups, and the capsazepine group were
statistically significant (P<0.01; Figs. 10 and 11).
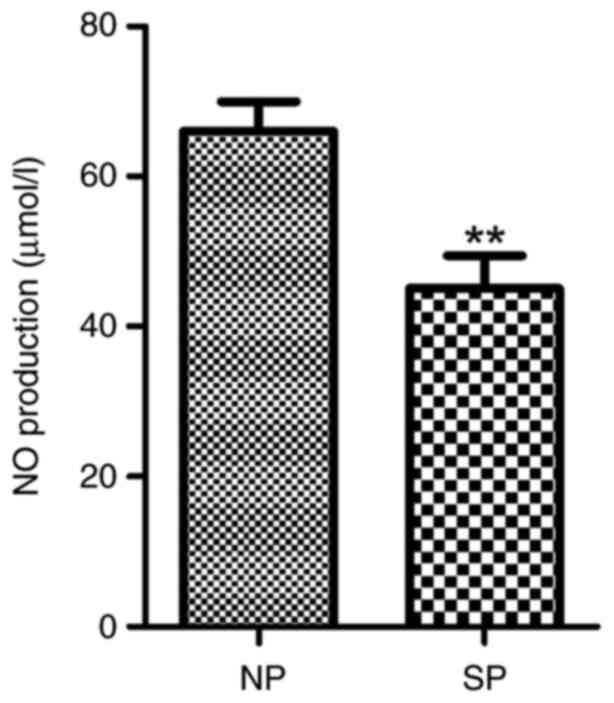
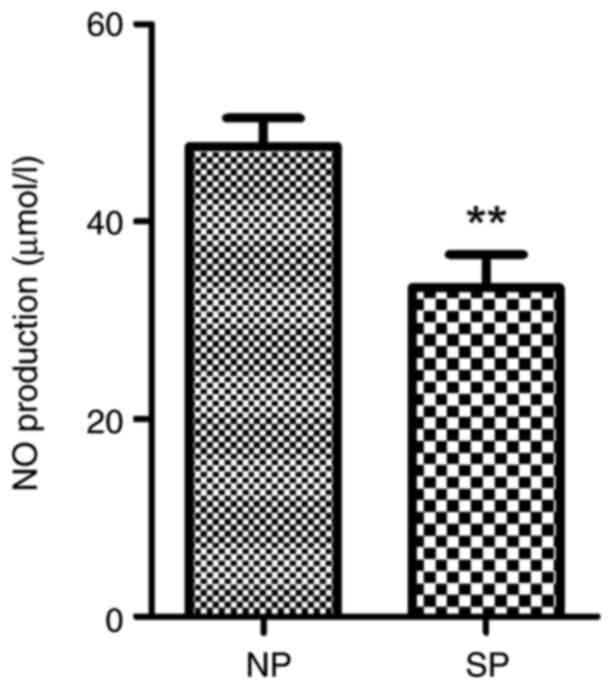
NO levels are decreased in the
placental artery and serum of patients with severe
preeclampsia
The nitrate reduction method indicated that the mean
levels of total nitrites in the placental artery and plasma were
33.31±11.73 and 45.12±14.92, respectively, in severe preeclampsia
patients, which were significantly lower than those in the control
group (47.65±9.81 and 66.01±13.81, respectively; both P<0.01)
(Figs. 12 and 13).
Discussion
The vascular endothelium is present throughout the
body; it acts not only as a mechanical barrier between blood and
smooth muscle cells, but also as the body's largest endocrine
organ, which can synthesize and release various vasoactive
substances through multiple mechanisms to regulate vascular tension
(18-20).
It is generally accepted that endothelial dysfunction promotes the
occurrence of cardiovascular diseases, such as hypertension, and
that the degree of endothelial injury is directly related to the
severity of hypertension. In patients with hypertension,
endothelial cells are damaged due to elevated blood pressure, which
reduces the secretion of NO and other vasoactive substances, thus
leading to endothelial-dependent diastolic dysfunction, which in
turn becomes an initiating factor of various complications of
hypertension, forming a vicious cycle (21). As the leading cause of fetal growth
retardation and infant morbidity and mortality associated with
premature delivery and maternal death, studies have shown that
preeclampsia is a complex obstetric syndrome associated with
maternal vascular dysfunction in which the NO signaling pathway is
a crucial driver of disease progression and severity (22). It is common knowledge that the
placental vasculature plays a crucial role in the placenta's
ability to function normally. The substantial placental
vascularization and the placenta's sophisticated processes that
control vascular tone assist the fetus's growing needs for blood
supply as the pregnancy progresses (23). NO in the syncytiotrophoblast and
endothelial cells of the chorionic plate and stem villous vessels
is suggested to cause the dilatation of the human placental
vasculature (24). The results of
the present study showed that in individuals with severe
preeclampsia, the vascular walls of the placental artery were
thickened, the presence of collagen fibers was increased, the
artery hyaline was altered, and the endothelial cell layer was
notably damaged. Dikensoy et al (25) found that the mean serum levels of
NO were significantly lower in patients with severe or moderate
preeclampsia compared to control subjects, and this decrease was
correlated with the severity of the disease. Similar to these
findings, in the present study, a chemical reagent was used to
determine the amount of NO in the mother's serum and in the
placental artery. It was shown that there were functional
alterations following vascular endothelial injury in patients with
severe preeclampsia, as the NO levels in the patients with severe
preeclampsia were much lower than those in the control group, both
in the serum and placental tissue lysates. In fact, numerous
studies have demonstrated the role of NO in the regulation of
fetal-maternal vascular tone in both animals and humans (25-27).
It was also found that infusion of eNOS inhibitor in mice during
pregnancy resulted in hypertension and fetal growth retardation,
similar to that observed in patients with preeclampsia (26). Boeldt and Bird (28) showed
that maternal peripheral endothelial dysfunction is central to the
disease stage of preeclampsia. However, it is still unclear how
diminished placental perfusion causes severe maternal vascular
endothelial dysfunction. The majority of in vitro studies
claim that Ca2+-dependent eNOS is the most prevalent NOS
isoform in the human placenta and is responsible for placental NO
generation (29). The findings of
the present study revealed that the relative expression of eNOS was
notably downregulated in patients with severe preeclampsia compared
with that in women with normal-term pregnancies. Thus, we
hypothesize that normal placenta formation may require locally
generated NO to promote intravascular infiltration of
cytotrophoblast cells. The decreased expression of eNOS in the
placental artery results in a decrease in locally generated NO
synthesis, leading to the preeclampsia-related impairment of
cytotrophoblast invasion, diastolic endothelial dysfunction and
vasospasm, which form a vicious cycle and participate in the
changes in uterine placental ischemia and hypoxia, and the release
of a variety of placental factors, which enter maternal blood
circulation, activate systemic inflammatory reaction, damage
vascular endothelial cells, and eventually lead to hypertension and
various complications, consistent with the classical theory of
preeclampsia due to poor placentation (30).
TRPV1 was originally discovered by researchers to be
specifically activated by capsaicin, which is why it is also known
as the capsaicin receptor (31).
As the most studied member of the TRPV subfamily, TRPV1 was
originally identified in the nervous system and is present in the
vagus nerve, trigeminal ganglion and dorsal ganglion neurons
(32). In recent years, an
increasing number of studies have shown that TRPV1 also plays an
important role in the regulation of cardiovascular disease and is
primarily expressed in cardiomyocytes, smooth muscle cells,
vascular endothelial cells, inflammatory cells and peripheral
vascular adipose tissue (33-36).
TRPV1 is a non-selective cationic channel that can mediate the
transmembrane flow of cations, dominated by Ca2+, when
ligands bind to the receptor, which can result in changes in
intracellular Ca2+ concentrations and activate a series
of intracellular signals to participate in a variety of
intracellular physiological and pathological processes (33,37).
The synthesis of NO is closely related to the increase in
intracellular Ca2+. Ca2+ binding to
calmodulin in endothelial cells can activate eNOS and promote the
synthesis and release of NO, thereby dilating blood vessels and
decreasing vascular resistance (38-40).
TRPV1 may play an essential role in endothelial cell physiological
and pathological status to allow the maintenance of normal vascular
function and the pathological formation of vascular lesions. It has
been confirmed in animal experiments that TRPV1 can mediate
coronary artery relaxation in an endothelium-dependent manner
(4), and TRPV1 can stimulate NO
synthesis through different signaling pathways (6,41).
It is well established that the structural dysfunction of potassium
channels can disrupt the balance of vasoconstriction and the
diastole of blood vessels themselves, increasing vascular tension
and eventually leading to the pathological state of hypertension
(42,43). KATP is a potassium ion channel;
reports on KATP have primarily focused on vascular smooth muscle
cells, and researchers later showed that KATP in endothelial cells
also participates in regulating vascular tone (44). Studies have shown that the KATP
agonist cromakalim can increase Ca2+ levels in
endothelial cells, which can increase the expression of eNOS,
promote the synthesis and release of NO, and indirectly relax
smooth muscle tissue (8,9). Bratz et al (4) found that endothelium-dependent
dilation mediated by TRPV1 could be attenuated by iberiotoxin, a
selective inhibitor of Ca2+-activated K+
channels (BKca), suggesting that BKca is involved in
capsaicin-induced relaxation. In addition, tetraethylammonium, a
non-specific potassium channel blocker, also attenuates
TRPV1-mediated endothelium-dependent relaxation, suggesting that
multiple potassium channels are involved in TRPV1-mediated
endothelium-dependent relaxation. The present study showed that
TRPV1 and Kir6.1/SUR2B are primarily expressed in the endothelial
cell layer in human placental arteriolar cells, and the relative
gene and protein expression of TRPV1 and Kir6.1/SUR2B in the
experimental group is significantly downregulated compared with
that in the control group. Thus, we hypothesize that there might be
some correlation between TRPV1 and the KATP. Chen et al
(45) conducted animal experiments
on Wistar rats and showed that capsazepine, a selective TRPV1
blocker could be used to prevent endothelium-dependent dilatation
and enhance NO release induced by Natakalim, a novel KATP opener in
the mesenteric arterioles of rats. To further characterize the
signaling cascade behind the TRPV1-KATP mediated relaxation, in the
present study, HUVECs were used and it was shown that in the
agonist and blocker groups, the relative expression of SUR2B/Kir6.1
and eNOS was also significantly upregulated and downregulated,
respectively, compared with that in the control group, indicating
that the activation or inhibition of TRPV1 could upregulate or
downregulate the expression of Kir6.1/SUR2B and eNOS, which
suggests that the TRPV1-specific agonist capsaicin is working via
the KATP channel as a result of a second messenger, most notably
NO. Referring to the previous theory, we hypothesize that TRPV1
activation in endothelial cells activates Kir6.1/SUR2B via an
unknown mechanism, causing hyperdifferentiation of the endothelial
membrane and thereby boosting Ca2+ influx into cells. As
a non-selective cation channel, TRPV1 can also mediate the
transmembrane flow of cations dominated by Ca2+ when the
ligand binds to its receptor. Through the aforementioned two
possible mechanisms, Ca2+ levels can be further elevated
in endothelial cells, enhancing the expression and activity of
eNOS, thereby increasing the synthesis and release of NO, and the
endothelium-dependent vasodilation response. Breyne and Vanheel
(46) demonstrated that
stimulation of TRPV1 may lead to the release of calcitonin
gene-related peptides, which hyperpolarize the smooth muscle cells
in rat mesenteric arteries by activating KATP channels. However,
the present study found that TRPV1 and KATP channels were primarily
distributed in the endothelial cell layer in the placental artery,
which may be caused by species differences. We hypothesize that
TRPV1 channels are functionally expressed in the placental artery
and mediate endothelium-dependent vasodilation through a mechanism
involving NO and KATP channels. In patients with severe
preeclampsia, the expression of TRPV1 and Kir6.1/SUR2B is decreased
due to endothelial cell damage, and the TRPV1-KATP channel-mediated
vasodilation is also impaired, leading to endothelial dysfunction.
In addition, NO diffusion to the vascular smooth muscle is
correspondingly decreased, and vascular smooth muscle cannot relax
properly, resulting in decreased placental perfusion and secondary
systemic circulatory disturbances, which underlie the clinical
symptoms of severe preeclampsia. Animal models are vital to
determine the mechanism of action of TRPV1/KATP in vivo. The
ideal animal experiment would be to place an experimental group of
animals on a capsaicin diet (TRPV1-specific agonist) and ultimately
observe the blood pressure, the size and weight of the placenta,
the birth weight and survival of the pups, and the levels of
certain biochemical markers in the mother. Researchers have found
that infusion of an NO synthesis inhibitor in mice during pregnancy
results in hypertension and fetal growth retardation, which are
similar to the signs of preeclampsia. However, severe preeclampsia
is a spontaneous disease of pregnant women with certain risk
factors after 20 weeks of gestation, while other animals do not
develop preeclampsia and the gestational age of animals is
difficult to match with that of humans; thus, there is no
well-recognized animal model as of yet. Due to the limitations of
experimental conditions, it was not possible to conduct vascular
ring tension tests, additional electrophysiological experiments or
animal experiments, which are significant limitations of this study
and will thus serve as a direction for future studies.
In conclusion, as a specific type of hypertensive
disease during pregnancy, severe preeclampsia may be characterized
by multiple factors, mechanisms and pathways. The present study
showed that the impairment of the endothelial TRPV1-KATP
channel-mediated eNOS/NO pathway may play an important role.
However, further vascular ring tension tests, additional
electrophysiological experiments and animal experiments are still
necessary to further understand the etiology and pathogenesis of
this disease, and to provide a novel theoretical basis for its
prevention, diagnosis and treatment.
Acknowledgements
Not applicable.
Funding
Funding: This study was supported by the Luzhou Science and
Technology Bureau (grant no. 2020-SYF-27) and Sichuan Science and
Technology Bureau (grant no. N0.2021JDR0185).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XZ and XF conceived and designed the study. XZ, LW,
HL and YT performed the experiments and statistical analyses. XZ
and XF wrote the manuscript. HL, YT and XF reviewed and edited the
manuscript. All authors have read and approved the final
manuscript. XZ and HL confirm the authenticity of all the raw
data.
Ethics approval and consent to
participate
This study was approved by the Clinical Trial Ethics
Committee of the Affiliated Hospital of Southwest Medical
University (approval no. KY2019039). All work was undertaken
according to the provisions described in the Declaration of
Helsinki and its later amendments. All patients signed informed
consent for specimen collection.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Agrawal A and Wenger NK: Hypertension
during pregnancy. Curr Hypertens Rep. 22(64)2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Hypertension in pregnancy. Report of the
american college of obstetricians and gynecologists' task force on
hypertension in pregnancy. Obstet Gynecol. 122:1122–1131.
2013.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Zhu Z, Luo Z, Ma S and Liu D: TRP channels
and their implications in metabolic diseases. Pflugers Archiv.
461:211–223. 2011.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Bratz IN, Dick GM, Tune JD, Edwards JM,
Neeb ZP, Dincer UD and Sturek M: Impaired capsaicin-induced
relaxation of coronary arteries in a porcine model of the metabolic
syndrome. Am J Physiol Heart Circ Physiol. 294:H2489–H2496.
2008.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Marshall NJ, Liang L, Bodkin J,
Dessapt-Baradez C, Nandi M, Collot-Teixeira S, Smillie SJ, Lalgi K,
Fernandes ES, Gnudi L and Brain SD: A role for TRPV1 in influencing
the onset of cardiovascular disease in obesity. Hypertension.
61:246–252. 2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Yang D, Luo Z, Ma S, Wong WT, Ma L, Zhong
J, He H, Zhao Z, Cao T, Yan Z, et al: Activation of TRPV1 by
dietary capsaicin improves endothelium-dependent vasorelaxation and
prevents hypertension. Cell Metab. 12:130–141. 2010.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Yokoshiki H, Sunagawa M, Seki T and
Sperelakis N: ATP-sensitive K+ channels in pancreatic, cardiac, and
vascular smooth muscle cells. Am J Physiol. 274:C25–C37.
1998.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Lückhoff A and Busse R: Activators of
potassium channels enhance calcium influx into endothelial cells as
a consequence of potassium currents. Naunyn Schmiedebergs Arch
Pharmacol. 342:94–99. 1990.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Lückhoff A and Busse R: Calcium influx
into endothelial cells and formation of endothelium-derived
relaxing factor is controlled by the membrane potential. Pflugers
Arch. 416:305–311. 1990.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Nelson MT, Cheng H, Rubart M, Santana LF,
Bonev AD, Knot HJ and Lederer WJ: Relaxation of arterial smooth
muscle by calcium sparks. Science. 270:633–637. 1995.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Guarini G, Ohanyan VA, Kmetz JG,
DelloStritto DJ, Thoppil RJ, Thodeti CK, Meszaros JG, Damron DS and
Bratz IN: Disruption of TRPV1-mediated coupling of coronary blood
flow to cardiac metabolism in diabetic mice: Role of nitric oxide
and BK channels. Am J Physiol Heart Circ Physiol. 303:H216–H223.
2012.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Roberts JM and Cooper DW: Pathogenesis and
genetics of pre-eclampsia. Lancet. 357:53–56. 2001.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Vatish M, Randeva HS and Grammatopoulos
DK: Hormonal regulation of placental nitric oxide and pathogenesis
of pre-eclampsia. Trends Mol Med. 12:223–233. 2006.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Gestational hypertension and preeclampsia:
ACOG Practice Bulletin, Number 222. Obstet Gynecol. 135:e237–e260.
2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Zhang Z, Qu J, Zheng C, Zhang P, Zhou W,
Cui W, Mo X, Li L, Xu L and Gao J: Nrf2 antioxidant pathway
suppresses Numb-mediated epithelial-mesenchymal transition during
pulmonary fibrosis. Cell Death Dis. 9(83)2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Li L, Li D, Xu L, Zhao P, Deng Z, Mo X, Li
P, Qi L, Li J and Gao J: Total extract of Yupingfeng attenuates
bleomycin-induced pulmonary fibrosis in rats. Phytomedicine.
22:111–119. 2015.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Moncada S, Palmer RM and Higgs EA: The
discovery of nitric oxide as the endogenous nitrovasodilator.
Hypertension. 12:365–372. 1988.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Sumpio BE, Riley JT and Dardik A: Cells in
focus: Endothelial cell. Int J Biochem Cell Biol. 34:1508–1512.
2002.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Konukoglu D and Uzun H: Endothelial
dysfunction and hypertension. Adv Exp Med Biol. 956:511–540.
2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Al-Magableh MR, Kemp-Harper BK and Hart
JL: Hydrogen sulfide treatment reduces blood pressure and oxidative
stress in angiotensin II-induced hypertensive mice. Hypertens Res.
38:13–20. 2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Osol G, Ko NL and Mandalà M: Altered
endothelial nitric oxide signaling as a paradigm for maternal
vascular maladaptation in preeclampsia. Curr Hypertens Rep.
19(82)2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Reynolds LP and Redmer DA: Utero-placental
vascular development and placental function. J Anim Sci.
73:1839–1851. 1995.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Zhou Y, Fisher SJ, Janatpour M, Genbacev
O, Dejana E, Wheelock M and Damsky CH: Human cytotrophoblasts adopt
a vascular phenotype as they differentiate. A strategy for
successful endovascular invasion? J Clin Invest. 99:2139–2151.
1997.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Dikensoy E, Balat O, Pence S, Balat A,
Cekmen M and Yurekli M: The changes of plasma malondialdehyde,
nitric oxide, and adrenomedullin levels in patients with
preeclampsia. Hypertens Pregnancy. 28:383–389. 2009.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Yallampalli C and Garfield RE: Inhibition
of nitric oxide synthesis in rats during pregnancy produces signs
similar to those of preeclampsia. Am J Obstet Gynecol.
169:1316–1320. 1993.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Kaufmann P, Black S and Huppertz B:
Endovascular trophoblast invasion: Implications for the
pathogenesis of intrauterine growth retardation and preeclampsia.
Biol Reprod. 69:1–7. 2003.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Boeldt DS and Bird IM: Vascular adaptation
in pregnancy and endothelial dysfunction in preeclampsia. J
Endocrinol. 232:R27–R44. 2017.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Sahin-Tóth M, Kukor Z and Tóth M:
Tetrahydrobiopterin preferentially stimulates activity and promotes
subunit aggregation of membrane-bound calcium-dependent nitric
oxide synthase in human placenta. Mol Hum Reprod. 3:293–298.
1997.PubMed/NCBI View Article : Google Scholar
|
|
30
|
BROSENS I: A STUDY OF THE SPIRAL ARTERIES
OF THE DECIDUA BASALIS IN NORMOTENSIVE AND HYPERTENSIVE
PREGNANCIES. J Obstet Gynaecol Br Commonw. 71:222–230.
1964.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Caterina MJ, Schumacher MA, Tominaga M,
Rosen TA, Levine JD and Julius D: The capsaicin receptor: A
heat-activated ion channel in the pain pathway. Nature.
389:816–824. 1997.PubMed/NCBI View
Article : Google Scholar
|
|
32
|
Caterina MJ: Vanilloid receptors take a
TRP beyond the sensory afferent. Pain. 105:5–9. 2003.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Yang XR, Lin MJ, McIntosh LS and Sham JS:
Functional expression of transient receptor potential melastatin-
and vanilloid-related channels in pulmonary arterial and aortic
smooth muscle. Am J Physiol Lung Cell Mol Physiol. 290:L1267–L1276.
2006.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Zsombok A: Vanilloid receptors-do they
have a role in whole body metabolism? Evidence from TRPV1. J
Diabetes Complications. 27:287–292. 2013.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Gunthorpe MJ and Szallasi A: Peripheral
TRPV1 receptors as targets for drug development: New molecules and
mechanisms. Curr Pharm Des. 14:32–41. 2008.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Xin H, Tanaka H, Yamaguchi M, Takemori S,
Nakamura A and Kohama K: Vanilloid receptor expressed in the
sarcoplasmic reticulum of rat skeletal muscle. Biochem Biophys Res
Commun. 332:756–762. 2005.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Song MY and Yuan JX: Introduction to TRP
channels: Structure, function, and regulation. Adv Exp Med Biol.
661:99–108. 2010.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Cai H, Davis ME, Drummond GR and Harrison
DG: Induction of endothelial NO synthase by hydrogen peroxide via a
Ca(2+)/calmodulin-dependent protein kinase II/janus kinase
2-dependent pathway. Arterioscler Thromb Vasc Biol. 21:1571–1576.
2001.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Zhang M and Vogel HJ: Characterization of
the calmodulin-binding domain of rat cerebellar nitric oxide
synthase. J Biol Chem. 269:981–985. 1994.PubMed/NCBI
|
|
40
|
Zhang M, Yuan T, Aramini JM and Vogel HJ:
Interaction of calmodulin with its binding domain of rat cerebellar
nitric oxide synthase. A multinuclear NMR study. J Biol Chem.
270:20901–20907. 1995.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Torres-Narváez JC, Pérez-Torres I,
Castrejón-Téllez V, Varela-López E, Oidor-Chan VH, Guarner-Lans V,
Vargas-González Á, Martínez-Memije R, Flores-Chávez P,
Cervantes-Yañez EZ, et al: The role of the activation of the TRPV1
receptor and of nitric oxide in changes in endothelial and cardiac
function and biomarker levels in hypertensive rats. Int J Environ
Res Public Health. 16(3576)2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Nieves-Cintrón M, Syed AU, Nystoriak MA
and Navedo MF: Regulation of voltage-gated potassium channels in
vascular smooth muscle during hypertension and metabolic disorders.
Microcirculation. 25:2018.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Jackson WF: KV channels and the
regulation of vascular smooth muscle tone. Microcirculation.
25:2018.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Wu Y, He MY, Ye JK, Ma SY, Huang W, Wei
YY, Kong H, Wang H, Zeng XN and Xie WP: Activation of ATP-sensitive
potassium channels facilitates the function of human endothelial
colony-forming cells via Ca2+/Akt/eNOS pathway. J Cell
Mol Med. 21:609–620. 2017.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Chen X, Han W, Zhang Y, Cui W, Pan Z, Jin
X, Long C and Wang H: The molecular pathway of ATP-sensitive
potassium channel in endothelial cells for mediating arteriole
relaxation. Life Sci. 137:164–169. 2015.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Breyne J and Vanheel B: Methanandamide
hyperpolarizes gastric arteries by stimulation of TRPV1 receptors
on perivascular CGRP containing nerves. J Cardiovasc Pharmacol.
47:303–309. 2006.PubMed/NCBI View Article : Google Scholar
|