Introduction
Esophageal carcinoma (ESCA) is the eighth most
frequently occurring cancer and the sixth most common cause of
cancer-associated mortality worldwide (1,2).
Definitive chemo-radiation therapy has become the standard
therapeutic scheme for advanced ESCA (3-5),
which has been demonstrated to result in a median survival time of
14 months and 5-year survival rate of 27% (6). However, radio-resistance threatens
treatment efficacy and leads to a poor prognosis. The most lethal
radiation-induced lesions are DNA double-strand breaks, which can
induce cellular DNA damage responses, some of which help cells
recover from radiation injury. These responses include cell cycle
arrest, DNA repair and the activation of DNA damage sensing and
early transduction pathways, and it is considered that these
protective DNA damage responses induce tumor-associated
radio-resistance (7). It is
urgently necessary to identify novel sensitizers that are able to
improve the radiosensitivity of the cells.
The appropriate subcellular location of proteins in
normal cells defines the physiology and homeostasis of the cells.
Chromosome maintenance protein-1 (CRM1) is a nuclear export
protein, which has >200 cargo proteins. Most nuclear export
molecules act solely as tumor suppressors (8-10).
CRM1 in the nucleus binds with RAs-related nuclear protein
(Ran)-GTP and cargo proteins to form a triplet-complex that
undergoes shuttling via the nuclear pore complex to the cytoplasm
(11,12). Ran-GTP is hydrolyzed into Ran-GDP
by Ran-GTPase in the cytoplasm, which releases CRM1 and cargo
proteins, after which CRM1 returns to the nucleus. It has been
shown that CRM1 is often upregulated in hematologic carcinomas and
numerous solid tumors (13-15).
The increased expression or activation of CRM1 can cause tumor
suppressor protein (TSP) dysfunction. The excessive transportation
of TSP protein to the cytoplasm triggers its degradation (14,16).
p53 is a multifunctional TSP, and its mutations constitute the most
common genetic alterations in human tumors. Studies have
demonstrated that the p53 protein mainly plays a role in the
nucleus and participates in a variety of antitumor processes,
including apoptosis, cell cycle arrest and DNA damage repair
(17,18). It has been identified that CRM1 is
dysregulated in esophageal squamous cell carcinoma, and the
inhibition of CRM1 can disturb the expression of TSPs and inhibit
NF-κB activity in esophageal squamous cell carcinoma cell lines
(19). Therefore, the proper
sub-localization of p53 protein in the cells is important for its
efficient antitumor functions. p53 is a nuclear transport cargo
protein of CRM1, and its ability to execute tumor suppressor
functions normally is closely associated with its nuclear transport
(20,21).
The use of a selective inhibitor of nuclear export
(SINE) is a novel strategy in the treatment of numerous tumors.
KPT-330, also known as selinexor, is a SINE that inhibits CRM1 with
high affinity and low toxicity. A previous study reported that
KPT-330 inhibits CRM1 and promotes the accumulation of p53 protein
in the nucleus of colorectal cancer cells (22). As a result, it has a synergistic
therapeutic effect with inhibitors of protein kinases such as BRAF
(23). Furthermore, studies have
demonstrated that the inhibition of CRM1 by KPT-330 increases the
radiosensitivity of rectal cancer and non-small cell lung cancer
cells (24,25). The potential mechanism may be
associated with the role of KPT-330 as an inhibitor of the
accumulation of DNA repair proteins (26). Phase I/II clinical trials of
KPT-330 have been carried out in non-Hodgkin's lymphoma (NHL)
(27) and other hematological
tumors (28). KPT-330 is approved
by the FDA and EMA in combination with dexamethasone for the
treatment of patients with relapsed and refractory multiple myeloma
(29).
Genomic studies on esophageal cancer have
demonstrated mutations and the abnormal upregulation of
CRM1(30). The postoperative
immunohistochemical staining of pathological sections of esophageal
cancer tissue has demonstrated that CRM1 expression is associated
with the poor prognosis of esophageal cancer (19,31).
However, data on the relationship between CRM1 and the prognosis of
patients with esophageal cancer undergoing radical radiotherapy
remains scarce. Therefore, the present study investigated the
association between CRM1 inhibition and radiosensitivity in
esophageal cancer. Furthermore, the study assessed the ability of
KPT-330 to increase the radiosensitivity of esophageal cancer cells
and explored its sensitization mechanisms.
Materials and methods
Clinical data and patient samples. The data
and tissues of 111 patients with esophageal squamous cell carcinoma
were collected from the First Affiliated Hospital of China Medical
University (Shenyang, China) from January 2009 to December 2012. As
the present research was a retrospective study, approval was
obtained from the ethics committee of the First Affiliated Hospital
of China Medical University (approval no. AF-SOP-07-1.1-01) and the
requirement for informed consent from all patients was waived. The
enrolled patients were diagnosed with primary esophageal squamous
cell carcinoma by pathological examination. The patients had not
received any other treatments prior to undergoing definitive
radiotherapy or concurrent radio-chemotherapy. Due to the
challenges of total surgical excision, samples were collected by
endoscopy. In addition, 10 pairs of tumor tissues and adjacent
normal tissues (located <5 cm from the cancer tissues and
without neoplasm invasiveness) were collected to evaluate the CRM1
expression cut-off point for high and low expression. The clinical
and pathological characteristics of these patients were analyzed
(Table I).
 | Table IAssociation of the relative
expression of CRM1 expression with clinical parameters. |
Table I
Association of the relative
expression of CRM1 expression with clinical parameters.
| Characteristic | Low CRM1 | High CRM1 | P-value |
|---|
| Number | 49 | 62 | |
| Sex, n (%) | | | 0.395 |
|
Female | 5 (4.5) | 11 (9.9) | |
|
Male | 44 (39.6) | 51 (45.9) | |
| Age, years, n
(%) | | | 1.000 |
|
≤65 | 25 (22.5) | 32 (28.8) | |
|
>65 | 24 (21.6) | 30 (27.0) | |
| Tumor site, n
(%) | | | 0.269 |
|
Cervical
esophagus and proximal third of the esophagus | 15 (13.5) | 15 (13.5) | |
|
Distal third
of esophagus and EGJ | 13 (11.7) | 11 (9.9) | |
|
Middle third
of the esophagus | 21 (18.9) | 36 (32.4) | |
| Tumor diameter, cm,
n (%) | | | 0.192 |
|
>5 to
≤7 | 8 (7.2) | 19 (17.1) | |
|
>7 | 15 (13.5) | 18 (16.2) | |
|
≤5 | 26 (23.4) | 25 (22.5) | |
| Radiation dose, Gy,
n (%) | | | 0.468 |
|
60 | 21 (18.9) | 32 (28.8) | |
|
66 | 28 (25.2) | 30 (27.0) | |
| Therapeutic method,
n (%) | | | 0.283 |
|
Concurrent
chemoradiation therapy | 19 (17.1) | 24 (21.6) | |
|
Radiation
therapy alone | 28 (25.2) | 30 (27.0) | |
|
Sequential
chemoradiation therapy | 2 (1.8) | 8 (7.2) | |
| T stage, n (%) | | | 0.891 |
|
T1 | 1 (0.9) | 2 (1.8) | |
|
T2 | 5 (4.5) | 8 (7.2) | |
|
T3 | 10 (9.0) | 9 (8.1) | |
|
T4 | 33 (29.7) | 43 (38.7) | |
| N stage, n (%) | | | 0.450 |
|
N0 | 19 (17.1) | 19 (17.1) | |
|
N1 | 29 (26.1) | 39 (35.1) | |
|
N2 | 1 (0.9) | 4 (3.6) | |
| Pathologic stage, n
(%) | | | 0.876 |
|
Stage I | 4 (3.6) | 4 (3.6) | |
|
Stage
II | 8 (7.2) | 12 (10.8) | |
|
Stage
III | 37 (33.3) | 46 (41.4) | |
| Survival status, n
(%) | | | 0.560 |
|
Dead | 39 (35.1) | 52 (46.8) | |
|
Alive | 10 (9.0) | 10 (9.0) | |
Bioinformatics analysis
The GEPIA database (http://gepia.cancer-pku.cn/index.html) was used to
profile CRM1 expression in 33 malignancies and compare it with that
in matched normal tissues in The Cancer Genome Atlas (TCGA)
datasets. The transcriptome data of different tumor tissues and
matched normal tissues were downloaded from TCGA database
(https://cancergenome.nih.gov). The Gene
Expression Omnibus (GEO) database (https://www.ncbi.nlm.nih.gov/gds/) was utilized to
find gene expression datasets for patients with esophageal squamous
cell carcinoma (GSE20347, https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE20347;
GSE23400, https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE23400)
(32,33). The CRM1 expression levels between
esophageal squamous cell carcinoma and adjacent normal tissues were
compared.
Immunohistochemical assays
The tissues collected from the patients were stained
using the streptavidin-peroxidase method using a commercially
available SP-kit (SP-9001, OriGene Technologies, Inc.).
Paraffin-embedded tissues were dewaxed at 65˚C for 4 h and hydrated
by passing the tissues through a series of solutions: Xylene I for
20 min, Xylene II for 10 min, 100% ethanol for 10 min, 95% ethanol
for 5 min, 80% ethanol for 5 min and 75% ethanol for 5 min at room
temperature. Tissues were incubated in 3% hydrogen peroxide at room
temperature for 10 min and then incubated with 5% sheep serum
(reagent A in the SP-kit) at room temperature for 10 min. The
slides were incubated with polyclonal rabbit anti-human CRM1
antibody (1:180; cat. no. ab24189; Abcam) overnight at 4˚C.
According to SP kit instructions, the tissues were incubated with
biotin-labeled goat anti-rabbit IgG secondary antibody (reagent B
in the SP-kit) at 37˚C for 15 min and streptavidin/peroxide complex
(reagent C in the SP-kit) at 37˚C for 15 min. Then, immunostaining
was developed using a DAB kit (PV-8000, OriGene Technologies,
Inc.). An Eclipse Ni microscope (Nikon Corporation) with a CCD
camera (Ds-Qi1Mc, Nikon Corporation) was used for imaging.
The percentage of positive cells was evaluated and
graded as the % stained cells among the total cells in cancer nests
as follows: 0, <1% stained cells; 1, 2-25% stained cells; 2,
26-50% stained cells; 3, 51-75% stained cells; and 4, >75%
stained cells. In addition, staining intensity was graded as
follows: 0+, no color; 1+, light yellow; 2+, light brown; and 3+,
brown. The expression of CRM1 was defined as the arithmetic product
of the positive percentage score and intensity grade. A receiver
operating characteristic (ROC) curve was built to obtain the
cut-off value for the high and low CRM1 expression groups.
Cell lines
The human ESCA cell line ECA109 was obtained from
the pathology laboratory of China Medical University. The ECA109
cells were cultured in RPMI-1640 (Hyclone; Cytiva) with 10% fetal
bovine serum (Clark Bioscience) in a humidified atmosphere with 5%
CO2 at 37˚C.
Reagents
KPT-330 was purchased from Selleck Chemicals,
dissolved in dimethylsulfoxide (DMSO) to a concentration of 1
mmol/l and stored at -80˚C. The working solution was diluted in
RPMI-1640.
Western blot analysis
The cells were treated with specific concentrations
(0.1 and 0.3 µmol/l) of KPT-330 for 12 h at 37˚C and then subjected
to a 0- or 4-Gy dose of radiation at a rate of 300 cGy/min using a
Siemens Accelerator (Siemens AG). After 24 h, the cells were
harvested and washed twice in phosphate-buffered saline. The cells
were then lysed in RIPA lysis buffer (Beyotime Institue of
Biotechnology) for 30 min on ice and centrifuged at 13,618 x g for
15 min at 4˚C to collect the total protein extract. Nuclear and
cytoplasmic proteins were separated using a Nuclear and Cytoplasm
Protein Extraction Kit (cat. no. P0028; Beyotime Institute of
Biotechnology) according to the manufacturer's instructions. The
proteins from each sample were quantified and normalized using a
bicinchoninic acid protein assay.
The proteins (10 µg/lane) were separated by 6-12%
SDS-PAGE gel and blotted onto a PVDF membrane. Each membrane was
then blocked with 5% non-fat milk at room temperature for 1 h and
incubated with primary antibodies targeting CRM1 (dilution 1:180;
ab24189; Abcam), lamin a/c (dilution 1:10,000; ab133256; Abcam),
β-actin (dilution 1:1,000; 3700; Cell Signaling Technology, Inc.),
p53 (dilution 1:1,000; sc-126; Santa Cruz Biotechnology, Inc.) and
GAPDH (dilution 1:1,000; sc-47724; Santa Cruz Biotechnology, Inc.)
overnight at 4˚C. Thereafter, the blots were washed three times
with Tris-buffered saline with 0.5% Tween-20, peroxidase conjugated
goat or rabbit IgG antibody (1:5,000, cat. nos. ab6721 and ab6728,
Abcam) were used as secondary antibodies and incubated at room
temperature for 1 h. Finally, chemiluminescent working solution
(cat. no. P0018A, Beyotime Institute of Biotechnology) was
introduced to the membrane, the membrane was exposed using
Tanon-5200 ECL film for 1-30 min.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
cell viability assay
Cells were seeded into 96-well plates at a density
of 4x103 cells/well and incubated overnight until
attachment occurred. The cells were then treated with increasing
concentrations of KPT-330 (0.01-50 µmol/l) or with the DMSO vehicle
as the control. After incubation for 72 h at 37˚C, 20 µl 1 µg/ml
MTT was added to each well and the plate was incubated for another
4 h at 37˚C. Thereafter, all the solution was aspirated and 150 µl
DMSO was added for 15 min to dissolve the purple formazan crystals.
The optical density (OD) at 570 nm was detected using a microplate
reader (Bio-Rad Laboratories, Inc.). Cell viability was calculated
using the following formula: Cell viability (%)=OD (experimental
group)/OD (control group) x100. The half-maximal inhibitory
concentration (IC50) values of KPT-330 were obtained
using GraphPad Prism 5.0 (GraphPad Software; Dotmatics).
Colony formation assay
The cell survival fraction (SF) was calculated and
cell survival curves were delineated for cells treated with KPT-330
and/or radiation. Cells (4x103 per well) were seeded in
6-well plates and incubated until cell attachment occurred. The
cells were pretreated with KPT-330 (0.1 µmol/l) for 12 h at 37˚C
followed by irradiation with different doses of radiation (0, 2, 4,
6 and 8 Gy) according to the aforementioned method for 24 h. Cells
without KPT-330 treatment and subjected to these radiation doses
served as controls. The incubation of the cells was continued for
10-14 days until colonies formed. Thereafter, the cell colonies
were stained with 0.1% crystal violet. The plating efficiency (PE)
was calculated as follows: PE (%)=(number of colonies/number of
plated cells) x100. The SF was calculated as follows:
SF=experimental group colonies ratio/PE. Survival curves were
plotted using the equation: SF=1-[1-e(-k*D)]N (34). For each experiment, K was the slope
of the straight part of cell survival curve, the lethal dose
D0 was the inverse of K (D0=1/K), N was the
section of the cell survival curve after the extension of the
straight line intersected the ordinate. Radiation sensitivity
enhancement ratio (SER) is the ratio of D0(radiation
group)/D0(KPT-330+radiation) and SF at 2 Gy (SF2) were
calculated.
Apoptosis assay
Cells were pretreated with 0.1 or 0.3 µmol/l KPT-330
for 12 h at 37˚C and then treated with 0 or 4 Gy radiotherapy.
After 48 h, the cells were harvested, 5 µl Annexin V and 5 µl
propidium iodide (PI) from a cell apoptosis detection kit (cat. no.
KGA107, Nanjing KeyGen Biotech Co., Ltd.) were added, and the cells
were incubated for 15 min at room temperature in darkness. The
proportion of apoptotic cells was then analyzed using a FACSAria
flow cytometer (BD Biosciences) and FlowJo (version, 7.6; FlowJo,
LLC.).
Cell cycle assay
To determine whether KPT-330 alone or in combination
with radiation therapy influenced the cell cycle distribution of
the cells, the ECA109 cells were treated with KPT-330 (0.1 or 0.3
µmol/l) for 12 h at 37˚C prior to irradiation with 4 Gy. Cells were
also treated with KPT-330 alone, without irradiation. After 24 h,
the cell cycle was assessed by flow cytometry (according to the
aforementioned method) using a cell cycle analysis kit (cat. no.
DKW41-CCK-010, Dakewe Biotech Co., Ltd.).
Statistical analysis
Continuous variables are presented as the mean ±
standard deviation (SD). Paired t-tests were used to identify
differences in matched tumor/normal sample expression. Survival
analysis was performed using Kaplan-Meier survival curves. The
significance of the survival differences between high- and low-CRM1
expression groups was assessed with the log-rank test. Univariate
and multivariate analyses of the risk factors for OS were performed
using the log-rank test and Cox proportional hazards model,
respectively. Multiple groups were analyzed using two-way ANOVA
followed by Bonferroni's post-hoc test using GraphPad Prism
software. P<0.05 was considered to indicate a statistically
significant result. The experiments were performed in
triplicates.
Results
CRM1 is universally upregulated in
human cancers
To characterize the expression of CRM1 in tumor
tissues, GEPIA was used to compare CRM1 expression in 33
malignancies with that in matched normal tissues in datasets from
TCGA. Results from the GEPIA analysis showed the upregulation of
CRM1 protein in six cancer types, including cholangiocarcinoma,
ESCA, pancreatic adenocarcinoma, sarcoma, stomach adenocarcinoma
and thymoma (P<0.05; Fig.
1).
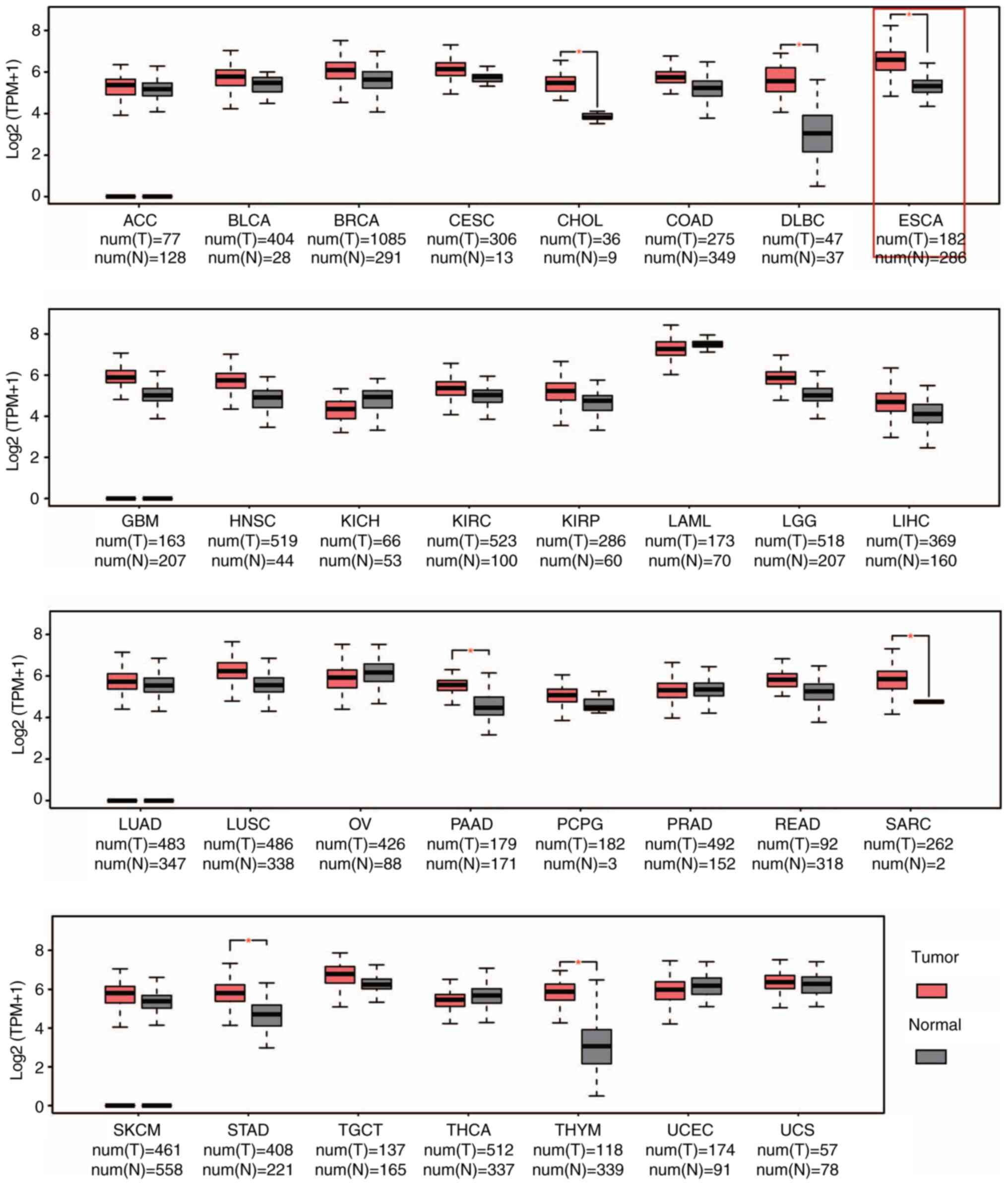 | Figure 1CRM1 is upregulated at the
transcriptional level in various cancers according to the GEPIA
database. GEPIA analysis showed that CRM1 was significantly
elevated in six types of cancer as compared with the respective
normal tissue. *P<0.05. CRM1, chromosome maintenance
protein-1; ACC, adrenocortical carcinoma; BLCA, bladder urothelial
carcinoma; BRCA, breast invasive carcinoma; CESC, cervical squamous
cell carcinoma; CHOL, cholangiocarcinoma; COAD, colon
adenocarcinoma; DLBC, lymphoid neoplasm diffuse large B-cell
lymphoma; ESCA, esophageal carcinoma; GBM, glioblastoma multiforme;
HNSC, head and neck squamous cell carcinoma; KICH, kidney
chromophobe; KIRC, kidney renal clear cell carcinoma; KIRP, kidney
renal papillary cell carcinoma; LAML, acute myeloid leukemia; LGG,
brain lower grade glioma; LIHC, liver hepatocellular carcinoma;
LUAD, lung adenocarcinoma; LUSC, lung squamous cell carcinoma; OV,
ovarian serous cystadenocarcinoma; PAAD, pancreatic adenocarcinoma;
PCPG, pheochromocytoma and paraganglioma; PRAD, prostate
adenocarcinoma; READ, rectum adenocarcinoma; SARC, sarcoma; SKCM,
skin cutaneous melanoma; STAD, stomach adenocarcinoma; TGCT,
testicular germ cell tumors; THCA, thyroid carcinoma; THYM,
thymoma; UCEC, uterine corpus endometrial carcinoma; UCS, uterine
carcinosarcoma; T, tumor; N, normal; TPM, transcripts per
million. |
Upregulation of CRM1 is associated
with poor patient survival in esophageal squamous cell
carcinoma
Differential CRM1 expression in esophageal squamous
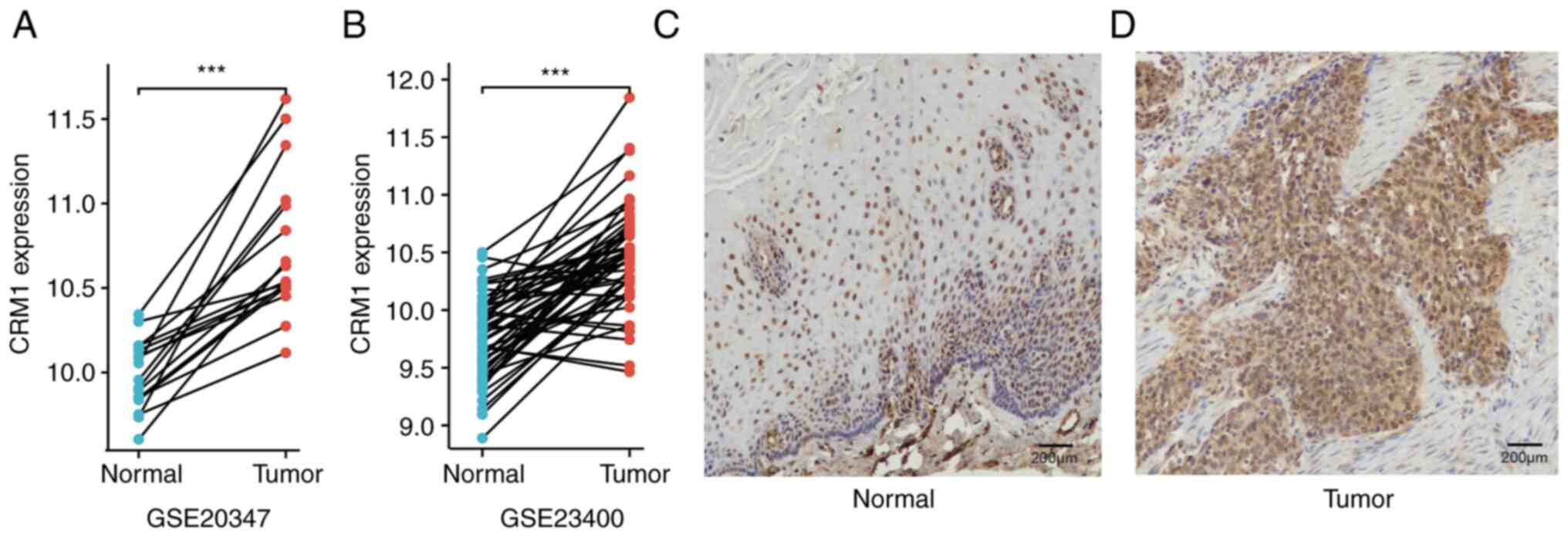
cell carcinoma was validated in two GEO datasets. The analysis
demonstrated significant upregulation of CRM1 in the tumor tissues
compared with the adjacent normal tissues in the GSE20347
(P=1.53x10-5) and GSE23400 (P=1.23x10-13)
datasets (Fig. 2A and B). To explore the expression of CRM1
protein, immunohistochemical analysis was performed in 10 pairs of
esophageal squamous cell carcinoma and matched adjacent tissues.
CRM1 expression was detected in the tumor and normal tissues.
However, in the normal tissues, CRM1 protein was localized in the
nucleus (Fig. 2C), while in the
tumor tissues, it was distributed in the nucleus and cytoplasm
(Fig. 2D).
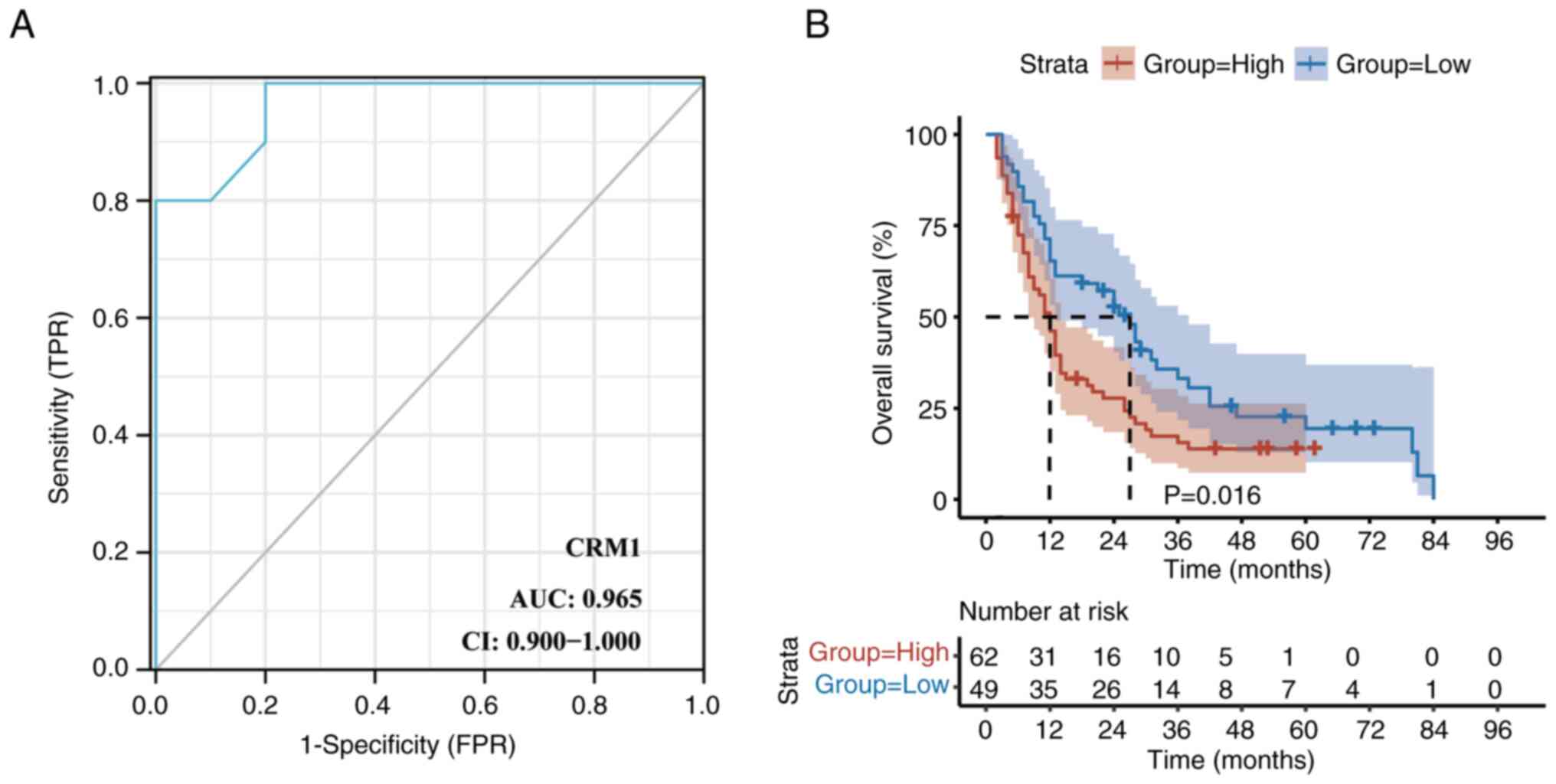
Based on CRM1 staining in the 10 pairs of esophageal
squamous cell carcinoma and adjacent tissues, a ROC curve was
constructed to determine the cut-off point for the
immunohistochemical staining score (Fig. 3A). A cut-off point of 4.5 provided
the maximum Youden index, with a specificity and sensitivity of 100
and 80%, respectively (area under the ROC curve, 0.965; P<0.05).
Therefore, a cut-off point of 4.5 was defined as the threshold for
distinguishing between the high- and low-expression states of CRM1.
High expression of CRM1 was detected in 62/111 (55.86%) of the
esophageal squamous cell carcinoma specimens. The basic
clinicopathological characteristics of the 111 patients are
presented in Table I. The analysis
demonstrated that the CRM1 expression was not associated with sex,
age, clinical stage, the diameter of the tumor or the tumor site
(P>0.05).
In addition, Kaplan-Meier analysis revealed that the
high expression of CRM1 was significantly associated with poor OS
(P=0.016; Fig. 3B). The results of
univariate analyses indicated that the OS of the patients was
significantly influenced by sex, therapeutic method, radiation dose
and CRM1 expression (P<0.05; Table
II). In addition, multivariate analyses showed that the OS was
significantly associated with gender, CRM1 expression, clinical
stage and therapeutic method (P<0.05; Table II). Patients with high CRM1
expression had a 2.38-fold increased risk of death compared with
those with low CRM1 expression.
 | Table IIUnivariate and multivariate analysis
of the risk factors for overall survival in patients with
esophageal squamous cell carcinoma. |
Table II
Univariate and multivariate analysis
of the risk factors for overall survival in patients with
esophageal squamous cell carcinoma.
| | Univariate
analysis | Multivariate
analysis |
|---|
| Variable | HR | P-value | HR | P-value |
|---|
| Age, years | | | | |
|
≤65 | Reference | | | |
|
>65 | 0.96 (0.63,
1.46) | 0.85 | | |
| Sex | | | | |
|
Female | Reference | | Reference | |
|
Male | 2.22 (1.14,
4.31) | 0.02 | 3.10 (1.39,
6.95) | 0.01 |
| Tumor site | | | | |
|
Upper | Reference | | | |
|
Middle | 1.30 (0.77,
2.17) | 0.33 | | |
|
Distal | 1.03 (0.55,
1.94) | 0.92 | | |
| Diameter of the
tumor, cm | | | | |
|
>5 to
≤7 | Reference | | | |
|
>7 | 0.87 (0.50,
1.54) | 0.64 | | |
|
≤5 | 0.66 (0.39,
1.09) | 0.11 | | |
| T stage | | | | |
|
T1 | Reference | | | |
|
T2 | 1.27 (0.27,
5.98) | 0.76 | | |
|
T3 | 1.44 (0.33,
6.36) | 0.63 | | |
|
T4 | 2.58 (0.63,
10.57) | 0.19 | | |
| N stage | | | | |
|
N0 | Reference | | | |
|
N1 | 1.58 (1.00,
2.49) | 0.05 | | |
|
N2 | 0.44 (0.10,
1.85) | 0.26 | | |
| Stage | | | | |
|
Stage I | Reference | | Reference | |
|
Stage
II | 2.10 (0.69,
6.40) | 0.19 | 1.62 (0.53,
4.99) | 0.40 |
|
Stage
III | 3.23 (1.18,
8.88) | 0.02 | 2.88 (1.04,
8.01) | 0.04 |
| Therapeutic
method | | | | |
|
Concurrent
chemoradiation therapy | Reference | | Reference | |
|
Radiation
therapy alone | 1.78 (1.14,
2.80) | 0.01 | 2.15 (1.34,
3.43) | <0.01 |
|
Sequential
chemoradiation therapy | 1.49 (0.68,
3.25) | 0.31 | 2.09 (0.83,
5.28) | 0.12 |
| Radiation dose,
Gy | | | | |
|
54-60 | Reference | | Reference | |
|
>60 | 1.66 (1.09,
2.54) | 0.02 | NA | NA |
| CRM1
expression | | | | |
|
High | Reference | | Reference | |
|
Low | 0.59 (0.39,
0.91) | 0.02 | 0.42 (0.26,
0.67) | <0.01 |
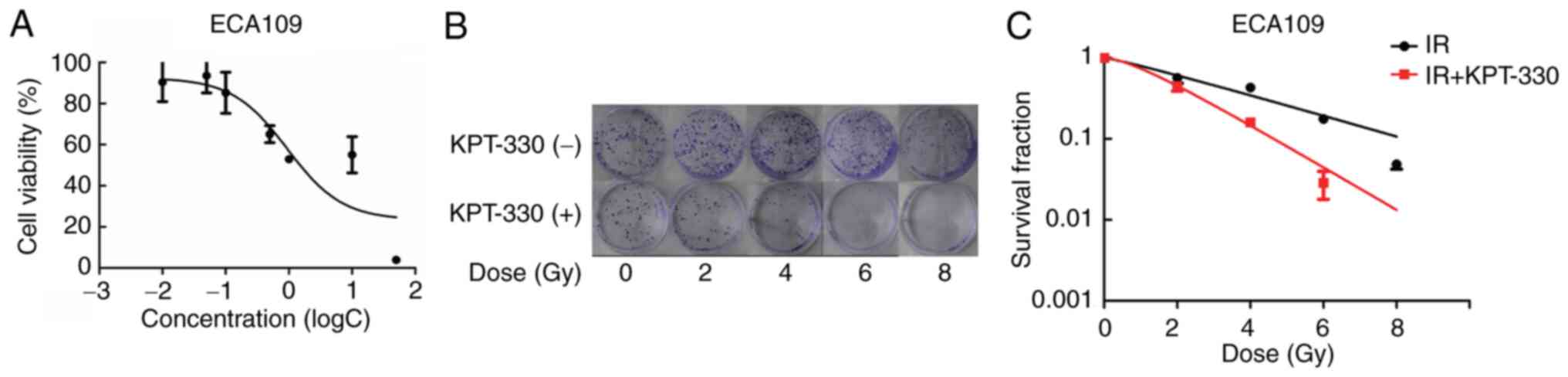
KPT-330 inhibits cell viability
The aforementioned results demonstrate that the CRM1
protein is upregulated in ESCA. To evaluate whether KPT-330
inhibits cell viability, ECA109 cancer cells were incubated with
various concentrations of KPT-330 in RPMI-1640 for 72 h. The
inhibitory effect of the treatment on cell viability was then
assessed using an MTT assay. The data showed a reduction in cell
viability as the concentration of KPT-330 increased. The
IC50 of KPT-330 in the ECA109 cell line was 0.9 µmol/l
(Fig. 4A).
Combination of KPT-330 and radiation
suppresses cell proliferation and decreases the colony formation
ability of ECA109 cells
To evaluate our hypothesis that the CRM1 inhibitor
is able to suppress cell proliferation and increase their radiation
sensitivity, 0.1 µmol/l KPT-330 was used in combination with
radiation to treat the ECA109 cells in a colony formation assay.
Cells treated with radiation alone served as controls. Images of
the plates were captured and cell SF curves were constructed
(Fig. 4B and C). Radiobiological parameters (Table III) were also obtained. The mean
D0 was 3.36 Gy for the irradiation group and 1.65 Gy for
the combined KPT-330 and irradiation group, while the SF2 values
were 56.71 and 44.89%, respectively. The SER was 2.04. Due to the
insensitivity of the ECA109 cell line to radiotherapy, relatively
small doses of radiotherapy including 2, 4 and 6 Gy were not able
reach a favorable treatment outcome. However, following the
application of KPT-330, the number of colonies formed by the ECA109
cells was markedly reduced, which may indicate an increase in the
sensitivity of the ECA109 cell line to radiotherapy. Thus, the data
demonstrated an improved inhibitory effect on proliferation and
lower SF in the cells treated with a combination of KPT-330 and
irradiation compared with those treated with radiation alone.
 | Table IIIRadiobiological parameters of ECA109
cells in the IR and IR + KPT-330 treatment groups. |
Table III
Radiobiological parameters of ECA109
cells in the IR and IR + KPT-330 treatment groups.
| | K value | N value | D0
(Gy) | SER | SF2
(%) |
|---|
| IR | 0.2977 | 1.169 | 3.36 | - | 56.71 |
| IR + KPT-330 | 0.6063 | 1.705 | 1.65 | 2.04 | 44.89 |
KPT-330 in combination with radiation
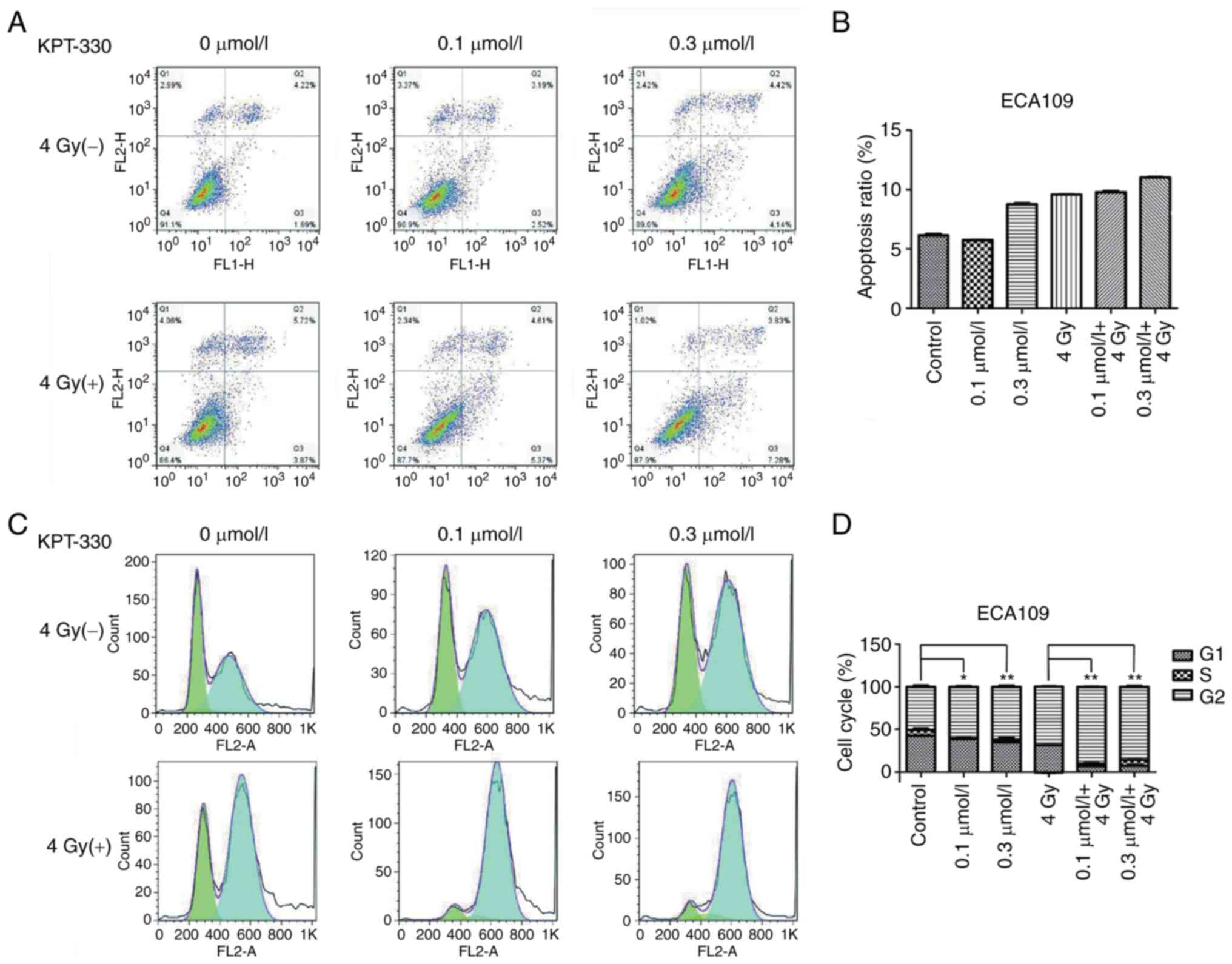
therapy induces apoptosis in ESCA cells
To assess the combined effect of KPT-330 and
radiation in the induction of apoptosis, ECA109 cells were
pretreated with KPT-330 for 12 h prior to being subjected to
irradiation. After 48 h the ECA109 cells were stained with
Annexin-Ⅴ and PI, and apoptosis was analyzed using flow cytometry.
The results illustrated that the ECA109 cells were sensitive to the
pro-apoptosis effect of KPT-330 to a certain extent, especially
when used in combination with radiation therapy (Fig. 5A and B); however, the effect of KPT-330 was not
statistically significant.
KPT-330 arrests the cell cycle in the
G2/M phase
To determine the mechanisms by which the inhibition
of cell growth occurred, changes in the cell cycle after treatment
with KPT-330 alone or in combination with irradiation were
evaluated in the ECA109 ESCA cells. The cell cycle was evaluated
using flow cytometry. KPT-330 induced cell cycle arrest and
increased the proportion of cells in the G2/M phase in
the ECA109 cell line (P<0.05; Fig.
5C and D), The cell cycle
distribution of cells treated with a combination of radiation and
KPT-330 was significantly different from that of cells treated with
radiation alone (P<0.01).
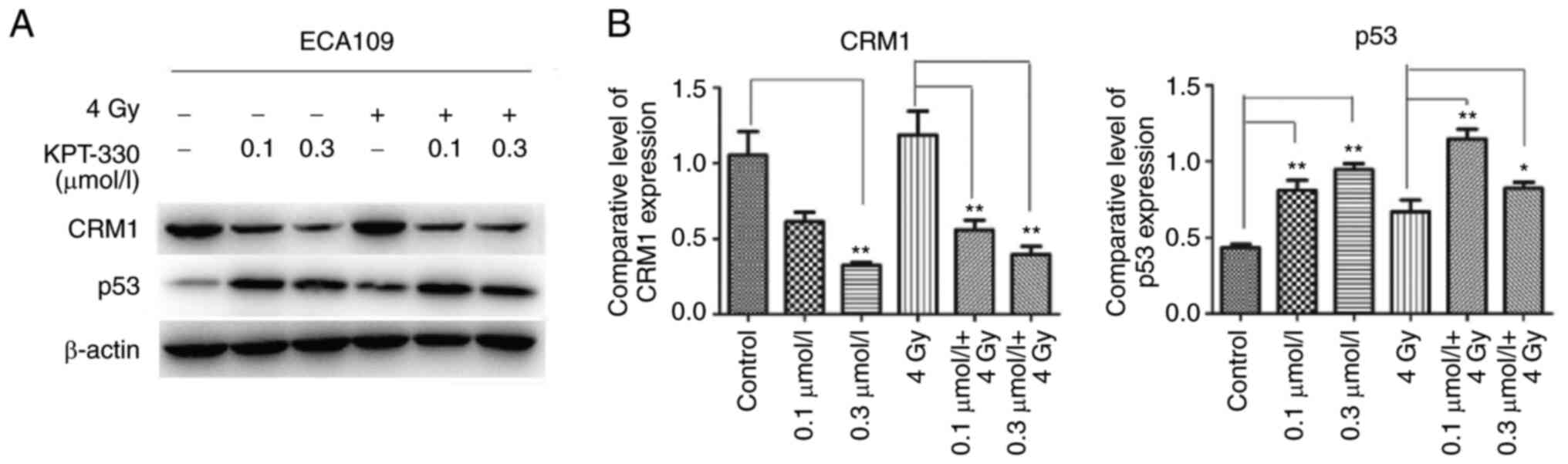
KPT-330 decreases CRM1 expression and
increases p53 expression
To elucidate the mechanism of apoptosis and cell
cycle arrest in the ECA109 cell line, western blot assays were used
to evaluate the CRM1-p53 signaling pathway in the whole cell
extracts. The results showed that CRM1 was highly expressed in the
ESCA cells and was downregulated following treatment with KPT-330,
while p53 was upregulated, particularly in the 0.3 µmol/l KPT-330
group (P<0.01). Upregulation of p53 expression was also observed
in the radiation plus KPT-330 combination group compared with
radiation alone (P<0.05), but the upregulation was not
significantly different from that achieved using KPT-330 alone
(Fig. 6).
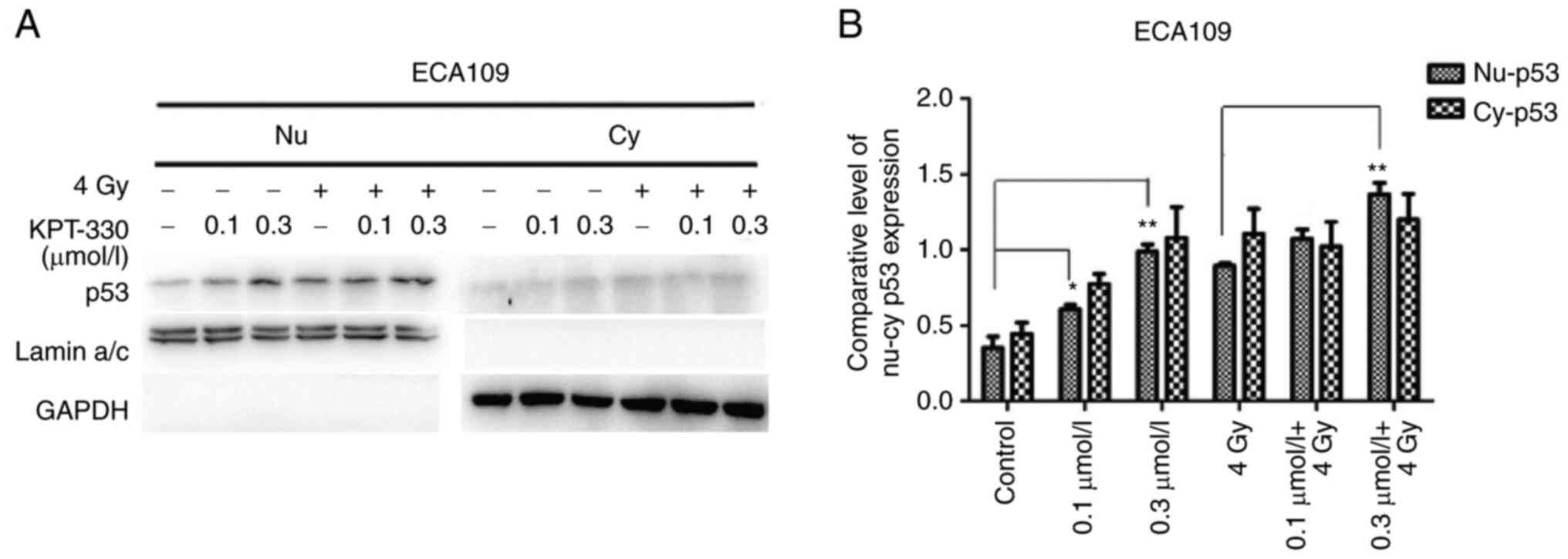
KPT-330 plus radiation induces p53
nuclear accumulation
Since CRM1 is a nuclear export protein, the nuclear
and cytoplasmic distribution of the known CRM1 cargo protein p53
was evaluated. KPT-330 significantly induced the nuclear
accumulation of p53 (P<0.05) as the concentration of KPT-330
increased, especially in the combination group. This effect was
more obvious when ECA109 cells were treated with radiation and
KPT-330 (Fig. 7). Although
cytoplasm p53 was also increased, p53 high-expression and its
nuclear location could perform its function. Nuclear p53 plays a
tumor suppressor role and participates in apoptosis and cell cycle
control (17,20,35).
Discussion
CRM1 is a member of the surface factor β family of
nuclear transport receptor karyopherins and the only nuclear export
protein of most TSPs (36-38).
CRM1 causes a variety of TSPs such as p53, p21 and FOXO1 to be
transported to the cytoplasm in excessive quantities, resulting in
the degradation of the TSPs. In the present study, the upregulation
of CRM1 in ESCA tissues was identified, and high expression levels
of CRM1 were found to be associated with decreased OS. In addition,
the study demonstrated that the application of the CRM1 inhibitor
KPT-330 increased the radiosensitivity of ECA109 cells, indicating
that this may be regarded as an alternative treatment approach for
patients with esophageal cancers.
The upregulation of CRM1 has been reported in
several types of solid tumor. For example, it was reported that the
high expression of CRM1 has a positive correlation with serum CEA
and CA19-9, and CRM1 protein expression is an independent
prognostic factor for OS and progression-free survival in patients
with pancreatic cancer (39). In
addition, Liu et al demonstrated that the expression of CRM1
was significantly increased in glioma tissues and associated with
the poor prognosis of glioma (40). Furthermore, a previous study
reported that patients with gastric cancer and high CRM1 expression
have poor postoperative prognoses (41). The results of the present study
demonstrated that the upregulation of CRM1 expression is associated
with poor survival in patients with esophageal squamous cell
carcinoma. In line with the present study, a study by Yang et
al (19) demonstrated that
increased expression of CRM1 in esophageal cancer is associated
with a poor postoperative prognosis. This previous study also found
that deletion of the CRM1 gene in ECA109 cells increased
5-fluorouracil-induced cell apoptosis, while also increasing the
expression levels of c-poly(ADP-ribose) polymerase and caspase-3.
Collectively, these findings indicate that CRM1 may play a key role
in the resistance of tumors, and result in a poor prognosis.
KPT-330 is the most studied and widely-applied CRM1
inhibitor. Compared with leptomycin B, the first CRM1 inhibitor,
KPT-330 has been shown to be less toxic and have improved
tolerability (42). However,
whether KPT-330 synergizes with radiation and its underlying
mechanisms are yet to be studied. Radiation resistance is a major
problem in advanced ESCA, which is mainly mediated by mechanisms
associated with apoptosis, the cell cycle and DNA damage repair. In
the present study, the in vitro results showed that KPT-330
decreased the viability of ECA109 cells and increased their
radiosensitivity, and suggested that the elevated radiosensitivity
may be associated with the elevated level of apoptosis and cell
cycle arrest at the G2/M phase detected in the cells.
Consistent with this, other studies have demonstrated that the
inhibition of CRM1 can increase the apoptosis of malignant melanoma
cells, induce the cell cycle arrest of liver cancer and renal cell
carcinoma cells, and reduce DNA damage repair (43-45).
In addition, Arango et al (46) showed that KPT-330 used alone or in
combination with chemotherapeutics such as paclitaxel or
carboplatin was able to increase the apoptosis of triple-negative
breast cancer cells and reduce cell colony formation. Furthermore,
Ranganathan et al (47)
showed that a combination of KPT-330 and topoisomerase II (topo II)
inhibitors increased the nuclear accumulation of topo IIα and
suppressed DNA damage repair, thereby reducing the chemotherapy
resistance of acute myeloid leukemia. Currently, KPT-330 is in
phase I/II clinical trials for the treatment of hematological and
some solid tumors, including NHL (27) and metastatic triple-negative breast
cancer (48). Recently, KPT-330
has been approved by the FDA as a novel therapy for treatment in
multiple myeloma and diffuse large B-cell lymphoma (29,49).
However, the large-scale application of KPT-330 in patients with
esophageal cancer has not yet been reported. Such application
appears to be worthy, based on the findings of the present study.
KPT-330 has the potential to be evaluated as a novel type of
radiosensitizer in the treatment of esophageal cancer.
The underlying mechanism of the enhanced
radiosensitivity of ECA109 cells induced by KPT-330 was explored in
the present study. The results demonstrated that KPT-330 induced
G2/M phase arrest in the ECA109 cells, particularly when
used in combination with radiation. Previous studies have indicated
that the G2/M phase is the most sensitive to
radiotherapy, followed by the G1 phase, while the S
phase is the most resistant (50,51).
The present findings suggest that KPT-330 increases the
G2/M phase arrest and radiosensitivity of esophageal
cancer cells. KPT-330 has been shown to change the cell cycle
distribution, reduce DNA damage repair protein and sensitize cells
to radiotherapy in non-small cell lung cancer cells (24). Inoue et al (52) demonstrated that KPT-185, another
CRM1 inhibitor, increased the G2/M phase arrest of von
Hippel-Landau (VHL)-wild-type renal cell carcinoma cell lines, but
increased G1 phase block in the VHL-negative cell line
786-O. A previous study reported that the treatment of
p53-deficient H1299 cells with KPT-330 and irradiation increased
G1 phase arrest, while p53 wild-type A549 cells
underwent G2 phase arrest following the same treatment
(24). Thus, both cell type and
p53 gene serve an important role in the cell cycle.
p53 is a typical TSP, which participates in a
variety of important biological processes, including apoptosis, the
cell cycle and DNA damage repair (21,53).
A previous study has shown that p53 inhibits CRM1 promoter
activity, and CRM1 inhibits the expression of p53(54). The present study showed that
following the treatment of ECA109 ESCA cells with KPT-330 alone,
CRM1 expression was suppressed and the expression of the p53
protein in the nucleus was upregulated. Furthermore, compared with
the cells that were only irradiated, the cells treated with a
combination of KPT-330 and radiation exhibited significantly
suppressed CRM1 protein expression, upregulated expression of p53
protein, and increased expression of p53 protein in the nucleus.
These results demonstrate that KPT-330 promoted nuclear
accumulation of the p53 protein.
The intracellular sub-localization of p53 plays an
important role in the regulation of tumorigenesis and development.
A previous study showed that KPT-330 combined with bortezomib, a
protein kinase inhibitor, altered the localization of the p53
protein in p53 wild-type colorectal cancer cells and increased the
expression of p53 in the nucleus (22). It has also been reported that in
p53 mutant colorectal cancer cell lines, transfection with a
plasmid expressing the wild-type p53 gene reduced the expression of
cyclin B mRNA, thereby increasing G2/M phase arrest;
this effect was not observed in cells transfected with a plasmid
expressing p53 mutations in the cyclin B DNA junction site
(55). Intriguingly, different
levels of p53 protein may display different functions (56-58).
For instance, low levels of p53 expression have been shown to cause
cell cycle arrest, while high levels of p53 expression lead to cell
apoptosis.
In the present study, the CRM1 inhibitor KPT-330 was
shown to increase the rate of radiotherapy-mediated apoptosis and
induce G2/M cell cycle arrest through the signal
transduction pathway protein p53. Since CRM1 is involved in the
nuclear export of a variety of proteins, it might also affect other
molecular pathways in CRM1 transportation. Kazim et al
(59) showed that KPT-330 plus
gemcitabine increased the apoptosis of pancreatic cancer cells by
inducing the expression of p27 and reducing the expression of the
anti-apoptotic protein survivin at the transcriptional level. In a
study of mantle cell lymphoma cells (60), the inhibition of CRM1 increased the
nuclear expression of cyclin D1 protein and reduced the level of
cyclin D1 in the cytoplasm. The study also showed that the
upregulation of cyclin D1 in the cytoplasm promoted the invasion
and metastasis of the cells. Therefore, the inhibition of CRM1
expression may slow the development of tumors and change the
radiosensitivity of tumor cells through multiple signal
transduction pathways.
In summary, in the current study it was identified
that KPT-330 inhibited CRM1 expression and increases p53 expression
in ESCA cells, which impacted the cell cycle distribution and
apoptosis, thereby improving radiosensitivity. However, due to the
large number of CRM1 cargo proteins, it is necessary to perform
additional experiments to explore the involvement of other proteins
in the mechanism of KPT-330 radio-sensitization. Furthermore, only
a single cell line and squamous cell carcinoma were explored in the
present study; further verification of the findings in other
esophageal squamous cell carcinoma cell lines such as TE1, KYSE-30,
EC-9706 and KYSE-70 and in other differentiated types of ESCA is
required.
Acknowledgements
The authors acknowledge the Laboratory Center of the
First Affiliated Hospital of China Medicine University (Shenyang,
China) for providing equipment used to perform experiments.
Funding
Funding: All reagents and consumables of the experiments were
provided through the postgraduate research budget of China Medical
University. No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
JX, SW and GL contributed to conception and design,
the collection and assembly of data, data analysis and
interpretation, and writing the manuscript. GL provided
administrative support, funded the project and provided study
materials and patients. All authors read and approved the final
manuscript. JX and SW confirm the authenticity of all the raw
data.
Ethics approval and consent to
participate
The study was conducted in accordance with the
Declaration of Helsinki as revised in 2013. The study was approved
by the ethics committee of the First Affiliated Hospital of China
Medical University (approval no. AF-SOP-07-1.1-01) and the
requirement for individual patient consent for this retrospective
analysis was waived.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Pennathur A, Gibson MK, Jobe BA and
Luketich JD: Oesophageal carcinoma. Lancet. 381:400–412.
2013.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Smyth EC, Lagergren J, Fitzgerald RC,
Lordick F, Shah MA, Lagergren P and Cunningham D: Oesophageal
cancer. Nat Rev Dis Primers. 3(17048)2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Arnold M, Soerjomataram I, Ferlay J and
Forman D: Global incidence of oesophageal cancer by histological
subtype in 2012. Gut. 64:381–387. 2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Rustgi A and El-Serag HB: Esophageal
carcinoma. N Engl J Med. 372:1472–1473. 2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Huang RX and Zhou PK: DNA damage response
signaling pathways and targets for radiotherapy sensitization in
cancer. Signal Transduct Target Ther. 5:1–27. 2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Conforti F, Zhang X, Rao G, Pas TD,
Yonemori Y, Rodriguez JA, McCutcheon JN, Rahhal R, Alberobello AT,
Wang Y, et al: Therapeutic effects of XPO1 inhibition in thymic
epithelial tumors. Cancer Res. 77:5614–5627. 2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Karki R, Sundaram B, Sharma BR, Lee S,
Malireddi RKS, Nguyen LN, Christgen S, Zheng M, Wang Y, Samir P, et
al: ADAR1 restricts ZBP1-mediated immune response and PANoptosis to
promote tumorigenesis. Cell Rep. 37(109858)2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Lv S, Song Q, Chen G, Cheng E, Chen W,
Cole R, Wu Z, Pascal LE, Wang K, Wipf P, et al: Regulation and
targeting of androgen receptor nuclear localization in
castration-resistant prostate cancer. J Clin Invest.
131(e141335)2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Hamed M, Caspar B, Port SA and Kehlenbach
RH: A nuclear export sequence promotes CRM1-dependent targeting of
the nucleoporin Nup214 to the nuclear pore complex. J Cell Sci.
134(jcs258095)2021.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Ferreira BI, Cautain B, Grenho I and Link
W: Small molecule inhibitors of CRM1. Front Pharmacol.
11(625)2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Landes JR, Moore SA, Bartley BR, Doan HQ,
Rady PL and Tyring SK: The efficacy of selinexor (KPT-330), an XPO1
inhibitor, on non-hematologic cancers: A comprehensive review. J
Cancer Res Clin Oncol. 149:2139–2155. 2022.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Gravina GL, Senapedis W, McCauley D,
Baloglu E, Shacham S and Festuccia C: Nucleo-cytoplasmic transport
as a therapeutic target of cancer. J Hematol Oncol.
7(85)2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Azizian NG and Li Y: XPO1-dependent
nuclear export as a target for cancer therapy. J Hematol Oncol.
13(61)2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Turner JG and Sullivan DM: CRM1-mediated
nuclear export of proteins and drug resistance in cancer. Curr Med
Chem. 15:2648–2655. 2008.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Bykov VJN, Eriksson SE, Bianchi J and
Wiman KG: Targeting mutant p53 for efficient cancer therapy. Nat
Rev Cancer. 18:89–102. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Borrero LJ and El-Deiry WS: Tumor
suppressor p53: Biology, signaling pathways, and therapeutic
targeting. Biochim Biophys Acta Rev Cancer.
1876(188556)2021.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Yang X, Cheng L, Yao L, Ren H, Zhang S,
Min X, Chen X, Zhang J and Li M: Involvement of chromosome region
maintenance 1 (CRM1) in the formation and progression of esophageal
squamous cell carcinoma. Med Oncol. 31(155)2014.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Fabbro M and Henderson BR: Regulation of
tumor suppressors by nuclear-cytoplasmic shuttling. Exp Cell Res.
282:59–69. 2003.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Kastan MB: Wild-type p53: Tumors can’t
stand it. Cell. 128:837–840. 2007.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Wu T, Chen W, Zhong Y, Hou X, Fang S, Liu
CY, Wang G, Yu T, Huang YY, Ouyang X, et al: Nuclear export of
ubiquitinated proteins determines the sensitivity of colorectal
cancer to proteasome inhibitor. Mol Cancer Ther. 16:717–728.
2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Fragomeni RA, Chung HW, Landesman Y,
Senapedis W, Saint-Martin JR, Tsao H, Flaherty KT, Shacham S,
Kauffman M and Cusack JC: CRM1 and BRAF inhibition synergize and
induce tumor regression in BRAF-mutant melanoma. Mol Cancer Ther.
12:1171–1179. 2013.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Rashal T, Elloul S, Crochiere M, Kashyap
T, Senapedis W, George R, Friedlander S, Ilouze M, Landesman Y,
Carlson R, et al: Selinexor (KPT-330) radio-sensitizes non-small
cell lung cancer cells in vitro and in vivo. Cancer Res. 75 (15
Suppl)(4490)2015.
|
|
25
|
Ferreiro-Neira I, Torres NE, Liesenfeld
LF, Chan CHF, Penson T, Landesman Y, Senapedis W, Shacham S, Hong
TS and Cusack JC: XPO1 inhibition enhances radiation response in
preclinical models of rectal cancer. Clin Cancer Res. 22:1663–1673.
2016.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Kazim S, Malafa MP, Coppola D, Husain K,
Zibadi S, Kashyap T, Crochiere M, Landesman Y, Rashal T, Sullivan
DM and Mahipal A: Selective nuclear export inhibitor KPT-330
enhances the antitumor activity of gemcitabine in human pancreatic
cancer. Mol Cancer Ther. 14:1570–1581. 2015.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Kuruvilla J, Savona M, Baz R, Mau-Sorensen
PM, Gabrail N, Garzon R, Stone R, Wang M, Savoie L, Martin P, et
al: Selective inhibition of nuclear export with selinexor in
patients with non-Hodgkin lymphoma. Blood. 129:3175–3183.
2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Conforti F, Wang Y, Rodriguez JA,
Alberobello AT, Zhang YW and Giaccone G: Molecular pathways:
Anticancer activity by inhibition of nucleocytoplasmic shuttling.
Clin Cancer Res. 21:4508–4513. 2015.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Dimopoulos MA, Richardson P and Lonial S:
Treatment options for patients with heavily pretreated relapsed and
refractory multiple myeloma. Clin Lymphoma Myeloma Leuk.
22:460–473. 2022.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Lin D, Hao J, Nagata Y, Xu L, Shang L,
Meng X, Sato Y, Okuno Y, Varela AM, Ding LW, et al: Genomic and
molecular characterization of esophageal squamous cell carcinoma.
Nat Genet. 46:467–473. 2014.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Van der Watt PJ, Zemanay W, Govender D,
Hendricks DT, Parker MI and Leaner VD: Elevated expression of the
nuclear export protein, Crm1 (exportin 1), associates with human
oesophageal squamous cell carcinoma. Oncol Rep. 32:730–738.
2014.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Hu N, Clifford RJ, Yang HH, Wang C,
Goldstein AM, Ding T, Taylor PR and Lee MP: Genome wide analysis of
DNA copy number neutral loss of heterozygosity (CNNLOH) and its
relation to gene expression in esophageal squamous cell carcinoma.
BMC Genomics. 11(576)2010.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Su H, Hu N, Yang HH, Wang C, Takikita M,
Wang QH, Giffen C, Clifford R, Hewitt SM, Shou JZ, et al: Global
gene expression profiling and validation in esophageal squamous
cell carcinoma and its association with clinical phenotypes. Clin
Cancer Res. 17:2955–2966. 2011.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Stephens TC, Peacock JH, Shipley WU and
Steel GG: Response to continuous irradiation (CI) in relation to
the initial slope of the cell survival curve for tumours and bone
marrow. Br J Cancer Suppl. 6:271–274. 1984.PubMed/NCBI
|
|
35
|
Ortega JF, de Conti A, Tryndyak V, Furtado
KS, Heidor R, Horst MA, Fernandes LHG, Tavares PELM, Pogribna M,
Shpyleva S, et al: Suppressing activity of tributyrin on
hepatocarcinogenesis is associated with inhibiting the p53-CRM1
interaction and changing the cellular compartmentalization of p53
protein. Oncotarget. 7:24339–24347. 2016.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Azmi AS, Muqbil I, Wu J, Aboukameel A,
Senapedis W, Baloglu E, Bollig-Fischer A, Dyson G, Kauffman M,
Landesman Y, et al: Targeting the nuclear export protein XPO1/CRM1
reverses epithelial to mesenchymal transition. Sci Rep. 5:1–13.
2015.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Sun H, Hattori N, Chien W, Sun Q, Sudo M,
E-Ling GL, Ding L, Lim SL, Shacham S, Kauffman M, et al: KPT-330
has antitumour activity against non-small cell lung cancer. Brit J
Cancer. 111:281–291. 2014.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Azmi AS, Aboukameel A, Bao B, Sarkar FH,
Philip PA, Kauffman M, Shacham S and Mohammad RM: Selective
inhibitors of nuclear export block pancreatic cancer cell
proliferation and reduce tumor growth in mice. Gastroenterology.
144:447–456. 2013.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Huang W, Yue L, Qiu WS, Wang LW, Zhou XH
and Sun YJ: Prognostic value of CRM1in pancreas cancer. Clin Invest
Med. 32(E315)2009.PubMed/NCBI
|
|
40
|
Liu X, Chong Y, Tu Y, Liu N, Yue C, Qi Z,
Liu H, Yao Y, Liu H, Gao S, et al: CRM1/XPO1 is associated with
clinical outcome in glioma and represents a therapeutic target by
perturbing multiple core pathways. J Hematol Oncol. 9:1–14.
2016.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Zhou F, Qiu W, Yao R, Xiang J, Sun X, Liu
S, Lv J and Yue L: CRM1 is a novel independent prognostic factor
for the poor prognosis of gastric carcinomas. Med Oncol.
30(726)2013.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Chen Y, Camacho SC, Silvers TR, Razak ARA,
Gabrail NY, Gerecitano JF, Kalir E, Pereira E, Evans BR, Ramus SJ,
et al: Inhibition of the nuclear export receptor XPO1 as a
therapeutic target for platinum-resistant ovarian cancer. Clin
Cancer Res. 23:1552–1563. 2017.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Marcus JM, Burke RT, DeSisto JA, Landesman
Y and Orth JD: Longitudinal tracking of single live cancer cells to
understand cell cycle effects of the nuclear export inhibitor,
selinexor. Sci Rep. 5(14391)2015.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Wettersten HI, Landesman Y, Friedlander S,
Shacham S, Kauffman M and Weiss RH: Specific inhibition of the
nuclear exporter exportin-1 attenuates kidney cancer growth. PLoS
One. 9(e113867)2014.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Yang J, Bill MA, Young GS, La Perle K,
Landesman Y, Shacham S, Kauffman M, Senapedis W, Kashyap T,
Saint-Martin JR, et al: Novel small molecule XPO1/CRM1 inhibitors
induce nuclear accumulation of TP53, phosphorylated MAPK and
apoptosis in human melanoma cells. PLoS One.
9(e102983)2014.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Arango NP, Yuca E, Zhao M, Evans KW, Scott
S, Kim C, Gonzalez-Angulo AM, Janku F, Ueno NT, Tripathy D, et al:
Selinexor (KPT-330) demonstrates anti-tumor efficacy in preclinical
models of triple-negative breast cancer. Breast Cancer Res.
19:1–10. 2017.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Ranganathan P, Kashyap T, Yu X, Meng X,
Lai TH, McNeil B, Bhatnagar B, Shacham S, Kauffman M, Dorrance AM,
et al: XPO1 inhibition using selinexor synergizes with chemotherapy
in acute myeloid leukemia by targeting DNA repair and restoring
topoisomerase IIα to the nucleus. Clin Cancer Res. 22:6142–6152.
2016.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Shafique M, Ismail-Khan R, Extermann M,
Sullivan D, Goodridge D, Boulware D, Hogue D, Soliman H, Khong H
and Han HS: A phase II trial of selinexor (KPT-330) for metastatic
triple-negative breast cancer. Oncologist. 24:887–e416.
2019.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Cheson BD, Nowakowski G and Salles G:
Diffuse large B-cell lymphoma: New targets and novel therapies.
Blood Cancer J. 11(68)2021.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Wang D, Veo B, Pierce A, Fosmire S,
Madhavan K, Balakrishnan I, Donson A, Alimova I, Sullivan KD, Joshi
M, et al: A novel PLK1 inhibitor onvansertib effectively sensitizes
MYC-driven medulloblastoma to radiotherapy. Neuro Oncol.
24:414–426. 2022.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Pawlik TM and Keyomarsi K: Role of cell
cycle in mediating sensitivity to radiotherapy. Int J Radiat Oncol
Biol Phys. 59:928–942. 2004.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Inoue H, Kauffman M, Shacham S, Landesman
Y, Yang J, Evans CP and Weiss RH: CRM1 blockade by selective
inhibitors of nuclear export attenuates kidney cancer growth. J
Urol. 189:2317–2326. 2013.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Stein Y, Rotter V and Aloni-Grinstein R:
Gain-of-function mutant p53: All the roads lead to tumorigenesis.
Int J Mol Sci. 20(6197)2019.PubMed/NCBI View Article : Google Scholar
|
|
54
|
van der Watt PJ and Leaner VD: The nuclear
exporter, Crm1, is regulated by NFY and Sp1 in cancer cells and
repressed by p53 in response to DNA damage. Biochim Biophys Acta.
1809:316–326. 2011.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Krause K, Wasner M, Reinhard W, Haugwitz
U, Dohna CL, Mössner J and Engeland K: The tumour suppressor
protein p53 can repress transcription of cyclin B. Nucleic Acids
Res. 28:4410–4418. 2000.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Tang X, Milyavsky M, Shats I, Erez N,
Goldfinger N and Rotter V: Activated p53 suppresses the histone
methyltransferase EZH2 gene. Oncogene. 23:5759–5769.
2004.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Schmidt AK, Pudelko K, Boekenkamp JE,
Berger K, Kschischo M and Bastians H: The p53/p73-p21(CIP1) tumor
suppressor axis guards against chromosomal instability by
restraining CDK1 in human cancer cells. Oncogene. 40:436–451.
2021.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Li C, Qin T, Liu Y, Wen H, Zhao J, Luo Z,
Peng W, Lu H, Duan C, Cao Y and Hu J: Microglia-derived exosomal
microRNA-151-3p enhances functional healing after spinal cord
injury by attenuating neuronal apoptosis via regulating the
p53/p21/CDK1 signaling pathway. Front Cell Dev Biol.
9(783017)2021.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Kazim S, Malafa MP, Coppola D, Husain K,
Zibadi S, Kashyap T, Crochiere M, Landesman Y, Rashal T, Sullivan
DM and Mahipal A: Selective nuclear export inhibitor KPT-330
enhances the antitumor activity of gemcitabine in human pancreatic
cancer. Mol Cancer Ther. 14:1570–1581. 2015.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Body S, Esteve-Arenys A, Miloudi H,
Recasens-Zorzo C, Tchakarska G, Moros A, Bustany S, Vidal-Crespo A,
Rodriguez V, Lavigne R, et al: Cytoplasmic cyclin D1 controls the
migration and invasiveness of mantle lymphoma cells. Sci Rep.
7:1–12. 2017.PubMed/NCBI View Article : Google Scholar
|