Introduction
Mesenchymal stem cells (MSCs), exhibit a high degree
of self-renewal and multipotential differentiation, and are widely
used in regenerative medicine (1,2).
Further investigations into the molecular mechanisms underlying the
therapeutic effects of MSCs may lead to increased clinical
application of cell-based therapy, and may aid in uncovering the
MSC directional differentiation at the molecular level (3). These investigations require precise
modulation of the signaling pathways involved in MSC
differentiation, in which the Wnt signaling pathway is one of the
key players (4).
The canonical Wnt signaling pathway depends on the
accumulation of β-catenin, and is one of the fundamental pathways
in cell proliferation and migration, cell fate determination,
polarization during embryonic development and tissue homeostasis
(5). The canonical Wnt signaling
pathway also exerts critical effects on the self-renewal and
differentiation of MSCs (6).
Results of a previous study demonstrated that lithium chloride
(LiCl), an antagonist of glycogen synthase kinase 3β (GSK 3β),
reduces the degradation of β-catenin through phosphorylation of GSK
3β at Ser9, subsequently activating the canonical Wnt-β-catenin
pathway (7). In addition,
Dickkopf-related protein 1 (DKK-1), a member of the Dickkopf
family, secretes proteins with two cysteine-rich domains separated
by a linker region. Thus, DKK-1 antagonizes canonical Wnt signaling
through inhibiting the interaction of low-density lipoprotein
receptor-related proteins 5/6 with Wnt (8).
Results of a previous study demonstrated that bone
marrow-derived MSCs develop the immunophenotypic and
ultrastructural characteristics of type II alveolar epithelial (AT
II) cells when cocultured with lung tissue (9). Moreover, MSCs protect against
hyperoxic lung injury through increasing the number of distal lung
epithelial cells and alveolar differentiation, and decreasing
self-renewal (10).
Results of our previous study demonstrated that
exogenous addition of Wnt3a or LiCl activated the canonical
Wnt/β-catenin pathway, and differentiation of MSCs into AT II cells
was promoted using a modified coculture system with murine lung
epithelial-12 (MLE-12) cells incubated in small airway growth
medium (SAGM) in vitro. However, DKK-1-mediated inhibition
of the canonical Wnt pathway reduced the expression of AT
II-associated markers (11).
Moreover, the downstream molecular mechanism by which the canonical
Wnt signaling pathway regulates the differentiation of MSCs into AT
II cells during mitosis remains unclear.
In the majority of cell lines, checkpoints that
regulate cell cycle progression and cell differentiation occur in
the G1/G0 phase. p130, as a member of the
retinoblastoma gene product (pRb) family, forms a repressor complex
with the transcription repressor E2F4, initiating the G1
to S phase transformation in the cell cycle, or maintaining cell
cycle arrest in G0/G1 phase (12). Activation of the Wnt/β-catenin
pathway in mouse MSCs (mMSCs) was associated with the accumulation
of β-catenin and pRb family members during MSC cycle progression
(13). In addition, results of our
previous study revealed that p130 or E2F4 overexpression in mMSCs
improved osteogenesis, and inhibited adipogenesis and
chondrogenesis through regulating the G1 phase (14). However, whether the directional
differentiation of MSCs and activation of Wnt/β-catenin resulted
from the p130/E2F4 pathway remains to be fully elucidated.
Thus, we hypothesized that activation of the
canonical Wnt/β-catenin signaling pathway in MSCs may affect the
p130/E2F4 pathway through regulating the cell cycle and
participating in the differentiation into AT II cells.
Materials and methods
Mesenchymal stem cell transduction and
culture
Bone marrow-derived mMSCs of C57BL/6 mice were
purchased from Cyagen Biosciences, Inc. These MSCs were isolated
from the bone marrow of the femurs of C57BL/6 mice. They were
uniformly positive for CD29, CD44, and Sca-1 antigens and negative
for CD117 and CD31 antigens, and possessed the potential to
differentiate into osteocytes, adipocytes, and chondrocytes, as
demonstrated in the instructions provided by the supplier. The
transduction of mMSCs using lentiviral vectors has been described
in our previous study (14).
Following transduction, mMSCs tagged with green fluorescent protein
(GFP), p130 or E2F4 with GFP; namely, normal control mMSCs
(MSC-NC), MSCs overexpressing p130 (MSC-p130) or MSCs
overexpressing E2F3 (MSC-E2F4), were cultured in Dulbecco's
modified Eagle's medium/nutrient mixture F-12 (DMEM/F12; Wisent,
Inc.) containing 10% foetal bovine serum (FBS; Wisent, Inc.) and 1%
antibiotic-antimycotic solution (streptomycin, penicillin and
amphotericin B; Wisent, Inc.). Following incubation at 37˚C in a
humidified atmosphere with 5% CO2, cells at passages
6-10 were used for in vitro experiments. MLE-12 cells
purchased from Xiangfbio Ltd. were cultured in DMEM/F12 with 2% FBS
and 1% antibiotic-antimycotic solution (Wisent, Inc.) in a
humidified 5% CO2 incubator at 37˚C.
Differentiation of mMSCs into AT II
cells
The coculture system for induced differentiation of
mMSCs into AT II cells was described in our previous study
(11). Briefly, mMSCs were
indirectly cocultured with MLE-12 cells in SAGM (Lonza Group,
Ltd.). SAGM was supplemented with 0.5 mg/ml bovine serum albumin
(BSA), 30 mg/ml bovine pituitary extract, 0.5 mg/ml hydrocortisone,
0.5 ng/ml epithelial growth factor, 0.5 mg/ml epinephrine, 5 mg/ml
insulin, 6.5 ng/ml triiodothyronine, 10 mg/ml transferrin and 0.1
ng/ml retinoic acid. A total of 1x104 mMSCs in 1.5 ml
DMEM/F12 supplemented with 10% FBS were seeded in each well of
six-well plates. Subsequently, Transwell inserts (0.4-mm pore size,
4.5 cm2; Corning, Inc.) loaded with 1x104
MLE-12 cells in 1 ml DMEM/F12 supplemented with 10% FBS were added
to establish the coculture system. When mMSCs reached 80%
confluence after 3 days incubation, the culture medium was replaced
with SAGM. Following a further 7 or 14 days, the inserts were
removed, and the mMSCs were harvested for subsequent analysis. All
of the incubation steps above were at 37˚C in a humidified
atmosphere with 5% CO2. To investigate the role of
canonical Wnt signaling in differentiation, 4 mmol/l LiCl
(Sigma-Aldrich; Merck KGaA) or 200 ng/ml DKK-1 (PeproTech, Inc.)
was used to activate or inhibit the canonical Wnt pathway,
respectively. LiCl or DKK-1 were added in the cocultured conditions
prior to the addition of SAGM, according to a previous study
(11).
Immunofluorescent staining
To analyse the differentiation of mMSCs into AT II
cells in vitro, immunofluorescent staining was performed.
Briefly, following induced differentiation, mMSCs were fixed in 4%
paraformaldehyde at 37˚C for 30 min. Following lysis with 0.5%
Triton X-100 and blocking with 3% BSA (Roche Diagnostics) for 30
min at room temperature, cells were stained with surfactant protein
C (SP-C) primary antibodies (1:100, Santa Cruz Biotechnology, Inc.
cat. no. sc-13979) at 4˚C overnight. Cells were subsequently
incubated with goat anti-rabbit Alexa Fluor® 647
secondary antibodies (1:250; Abcam; cat. no. ab150079) in 2% BSA
for 30 min at room temperature in the dark. Nuclei were stained
with 4,6-diamidino-2-phenylindole (DAPI; Beyotime Institute of
Biotechnology) at room temperature for 5-10 min. Images were
captured using fluorescence microscopy (Olympus Corporation).
Transmission electron microscopy
assay
Trypsin-digested cells were rinsed in
phosphate-buffered saline (PBS) and pelleted using centrifugation.
Cells were subsequently fixed in 1 ml PBS containing 2.5%
glutaraldehyde and 1% osmium tetroxide at 4˚C overnight. Samples
were dehydrated in gradient ethanol and embedded in an Araldite
resin block at 37, 42, and 60˚C for 24 h each. Ultrathin sections
of 80 nm excised from the block were stained with uranyl acetate at
room temperature for 3 min prior to imaging with a transmission
electron microscope (Hitachi H-7650).
Protein expression of SP-C and
p130/E2F4 in MSCs
To analyse the expression of SP-C, phosphorylated
(p)-p130, p130 and E2F4 in cells following differentiation, RIPA
lysis buffer (Beyotime Institute of Biotechnology) was used to
extract total protein lysates, followed by protein determination
using a BCA protein assay kit (Beyotime Institute of
Biotechnology). Proteins were separated using 10% sodium dodecyl
sulfate-polyacrylamide gel electrophoresis (Beyotime Institute of
Biotechnology) with the mass of protein loaded per lane of 100 µg
and transferred onto PVDF membranes (MilliporeSigma). Following
blocking in 5% BSA for 1 h at room temperature, membranes were
incubated at 4˚C overnight with primary antibodies against SP-C
(1:1,500; Abcam; cat. no. ab90716), p-p130 (1:1,000; Abcam; cat.
no. ab76255), p130 (1:2,000; Abcam; cat. no. ab76234), E2F4
(1:1,500; Proteintech; cat. no. 10923-1-AP) or β-actin (1:4,000;
Hangzhou HuaAn Biotechnology Co., Ltd. cat. no. M1210-2). Following
primary incubation, membranes were incubated with goat anti-rabbit
or goat anti-mouse IgG conjugated with horseradish peroxidase
(1:5,000; Zoonbio Biotechnology) for 1 h at room temperature.
Protein bands were visualized using Pierce ECL western blotting
substrate (Thermo Fisher Scientific, Inc.) and a chemiluminescence
imaging system (Bioshine ChemiQ 4800 mini; Ouxiang). Protein
expression was quantified using ImageJ software (National
Institutes of Health, version 1.52a).
Cell cycle analysis
Cells were centrifuged at 1,500 rpm and washed twice
with ice-cold PBS. Cells were fixed with 70% ethanol and incubated
at 4˚C for 30 min. Following fixation, cells were incubated with
RNase A (20 µg/ml) for 30 min at 37˚C, and stained with propidium
iodide (50 µg/ml; BD Biosciences) for 30 min at room temperature in
the dark. Cell cycle analysis was conducted using a BD FACS flow
cytometer (BD Biosciences) (14).
Statistical analysis
Data are presented as the mean ± standard deviation.
Statistical analyses were performed using GraphPad Prism 8
(GraphPad Software, Inc.). If the data were normally distributed,
comparisons among multiple groups were performed using one-way
ANOVA followed by Bonferroni's post hoc test. P<0.05 was
considered to indicate a statistically significant difference.
Results
Overexpression of p130 or E2F4
promotes the differentiation of mMSCs into AT II cells
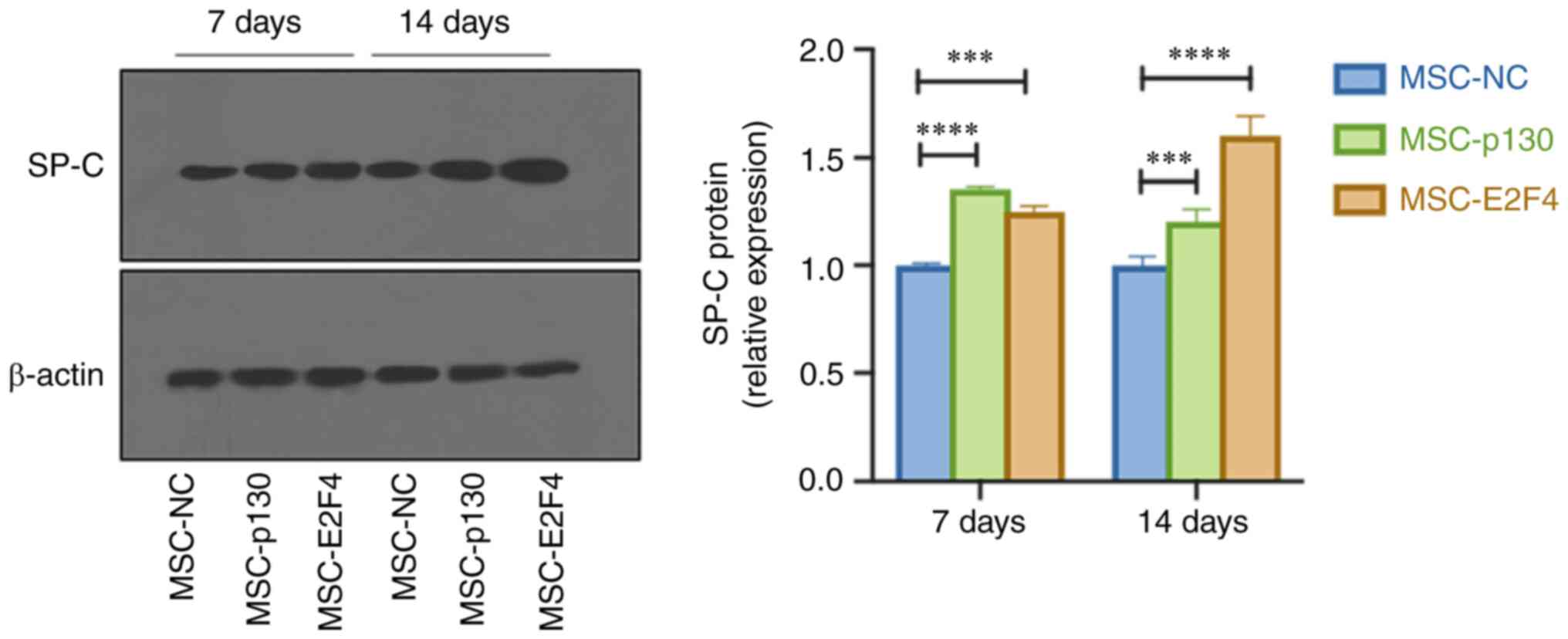
SP-C protein was analysed to detect the
differentiation of mMSCs into AT II cells after 7 and 14 days.
Results of the present study demonstrated that SP-C protein
expression in the MSC-p130 and MSC-E2F4 groups was significantly
higher than in the MSC-NC group (Fig.
1).
Immunofluorescence staining was also performed at 7
and 14 days following the differentiation of mMSCs. Red
fluorescence was indicative of SP-C-positive cells, and these were
present in both MSC-p130 and MSC-E2F4 groups. The numbers of
SP-C-positive cells and DAPI-SP-C double-positive cells (blue and
red fusion) in the MSC-p130 and MSC-E2F4 groups were higher than
that in the MSC-NC group (Fig.
2A).
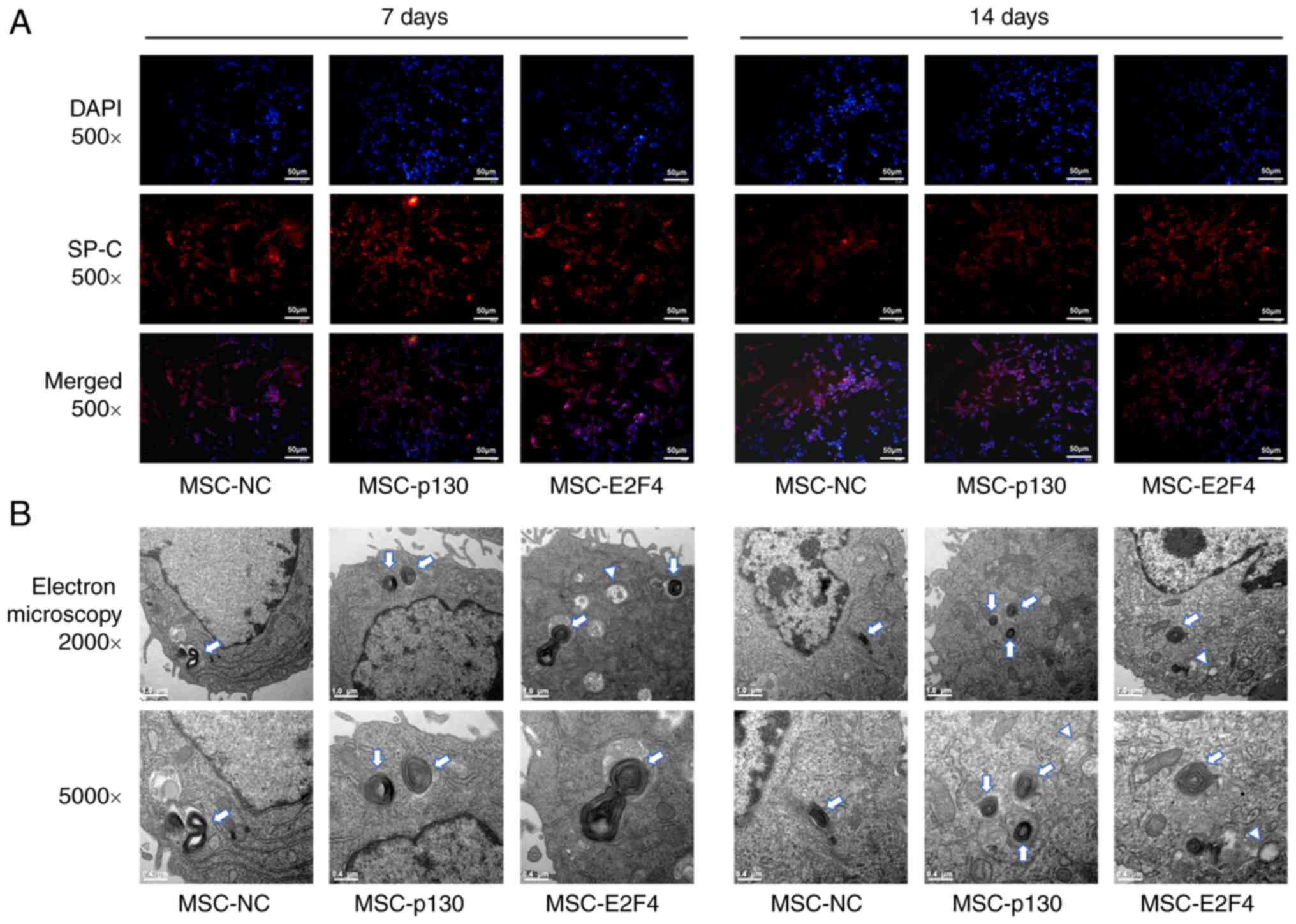 | Figure 2Effects of p130 or E2F4
overexpression on the differentiation of mMSCs into AT II cells
in vitro. (A) Immunofluorescence staining was used to detect
the differentiation of mMSCs. SP-C-positive cells are red. The
nuclei stained with DAPI are blue. Representative images at 7 and
14 days after the induced differentiation of mMSCs (magnification,
x500; scale bar, 50 µm). (B) Morphological changes in mMSCs
following differentiation into AT II cells under a transmission
electron microscope. Typical ultrastructure images of mMSCs after 7
and 14 days of differentiation driven by coculture with murine lung
epithelial-12 cells and small airway growth medium were observed as
lamellar body-like structures and vacuoles [magnification, x2,000
(top row) or x5,000 (bottom row); scale bar, 1.0 µm (top row) or
0.4 µm (bottom row); arrows, lamellar body-like structures;
triangles, vacuoles]. AT II cells, type II alveolar epithelial
cells; DAPI, 4,6-diamidino-2-phenylindole; E2F4, E2F transcription
factor 4; mMSCs, mouse mesenchymal stem cells; MSC-p130, MSCs
overexpressing p130; MSC-E2F4, MSCs overexpressing E2F4; NC, normal
control; SP-C, surfactant protein C. |
In addition, at 7 and 14 days following
differentiation, the morphology of numerous mMSCs diversified from
fusiform fibroblasts to paving-like epithelial structures. The
ultrastructure of the cells was further observed using electron
microscopy. Increased levels of vacuolization were observed in the
cytoplasm and the vicinity of the membrane in the MSC-p130 and
MSC-E2F4 groups. A small number of characteristic organelles of AT
II-lamellar bodies were observed, while there were fewer vacuoles
or lamellar structures present in the control group (Fig. 2B).
Activation of the canonical Wnt
signaling pathway promotes the differentiation of mMSCs into AT II
cells
In our previous study (11), dose-dependent assays using LiCl
activation (0.4, 1 or 4 mmol/l) and DKK-1 inhibition were used to
modulate the canonical Wnt pathway in mMSCs. Based on the
aforementioned findings, 4 mmol/l LiCl, which activated β-catenin
to the maximum extent, and 200 ng/ml DKK-1, which effectively
inhibited the Wnt signaling pathway, were used in the
differentiation experiments of the present study. Compared with the
control group, LiCl-mediated activation of the canonical Wnt
pathway significantly promoted the expression of SP-C in mMSCs,
while the expression of SP-C was significantly decreased following
treatment with DKK-1 (Fig. 3A and
B) at 7 and 14 days following
differentiation.
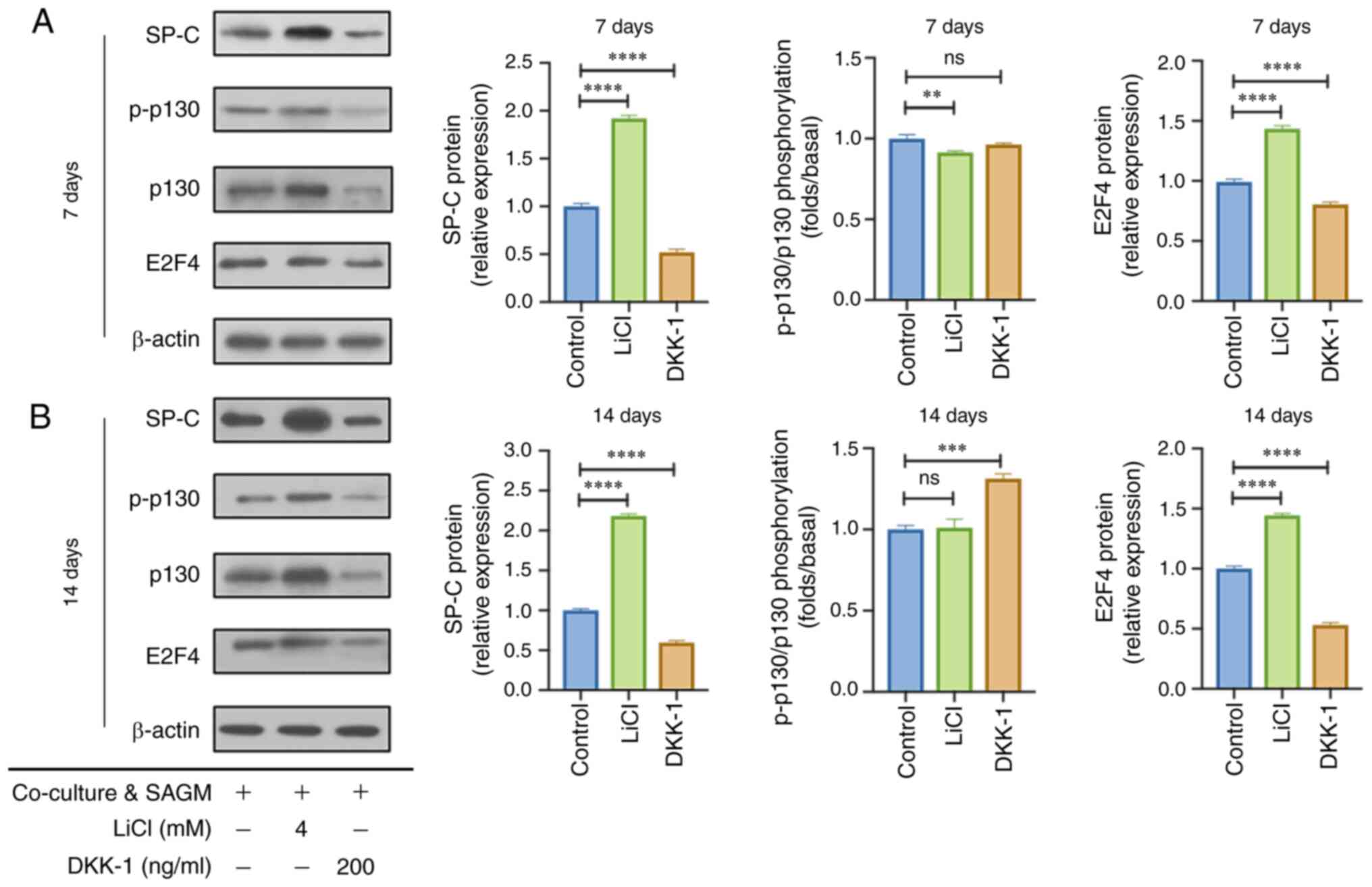 | Figure 3Effects of the canonical Wnt
signaling pathway on the expression levels of p130/E2F4 in mMSCs
during differentiation into type II alveolar epithelial cells in
vitro. (A) SP-C, p130 and E2F4 expression was evaluated in
mMSCs at 7 days after coculturing with MLE-12 cells and SAGM
supplemented with 4 mM LiCl or 200 ng/ml DKK-1 (n=3). (B) SP-C,
p130 and E2F4 expression was evaluated in mMSCs at 14 days after
coculturing with MLE-12 cells and SAGM supplemented with 4 mM LiCl
or 200 ng/ml DKK-1 (n=3). **P<0.01 vs. Control group
(mMSCs co-cultured with MLE-12 cells and SAGM),
***P<0.001 vs. Control group,
****P<0.0001 vs. Control group. DKK-1,
Dickkopf-related protein 1; E2F4, E2F transcription factor 4; LiCl,
lithium chloride; mMSCs, mouse mesenchymal stem cells; p-,
phosphorylated; SAGM, small airway growth medium; SP-C, surfactant
protein C; ns, not significant. |
Activation of the canonical Wnt
signaling pathway increases p130/E2F4 expression during mMSC
differentiation into AT II cells
To verify whether the differentiation of MSC-p130
and MSC-E2F4 groups into AT II cells was associated with activation
of the canonical Wnt signaling pathway, the expression levels of
p130 and E2F4 were detected. Results of the present study
demonstrated that the phosphorylation level of p130, which was
shown by p-p130/p130, was slightly decreased following LiCl
activation at 7 days (Fig. 3A)
while significantly increased following treatment with DKK-1 at 14
days after differentiation of mMSCs (Fig. 3B). Moreover, the expression level
of E2F4 was increased following LiCl activation, compared with the
control group but was inhibited following treatment with DKK-1 at 7
days after differentiation (Fig.
3A). Comparable results of E2F4 were also found at 14 days
after differentiation of mMSCs in each group (Fig. 3B).
To further elucidate the effects of the canonical
Wnt signaling pathway on the differentiation of MSC-p130 and
MSC-E2F4 groups into AT II cells, SP-C expression was examined
during differentiation using western blotting. The expression
levels of SP-C in the DKK-1 + MSC-p130 group were significantly
higher than that in the DKK-1 + MSC-NC group at 7 days following
differentiation. Moreover, there was no significant change in SP-C
expression in the DKK-1 + MSC-E2F4 group (Fig. S1). Comparable results were
observed among the three groups at 14 days after differentiation
(Fig. S1). These results
suggested that overexpression of p130, but not E2F4, may improve
the differentiation of mMSCs into AT II cells, while the canonical
Wnt signaling pathway was inhibited.
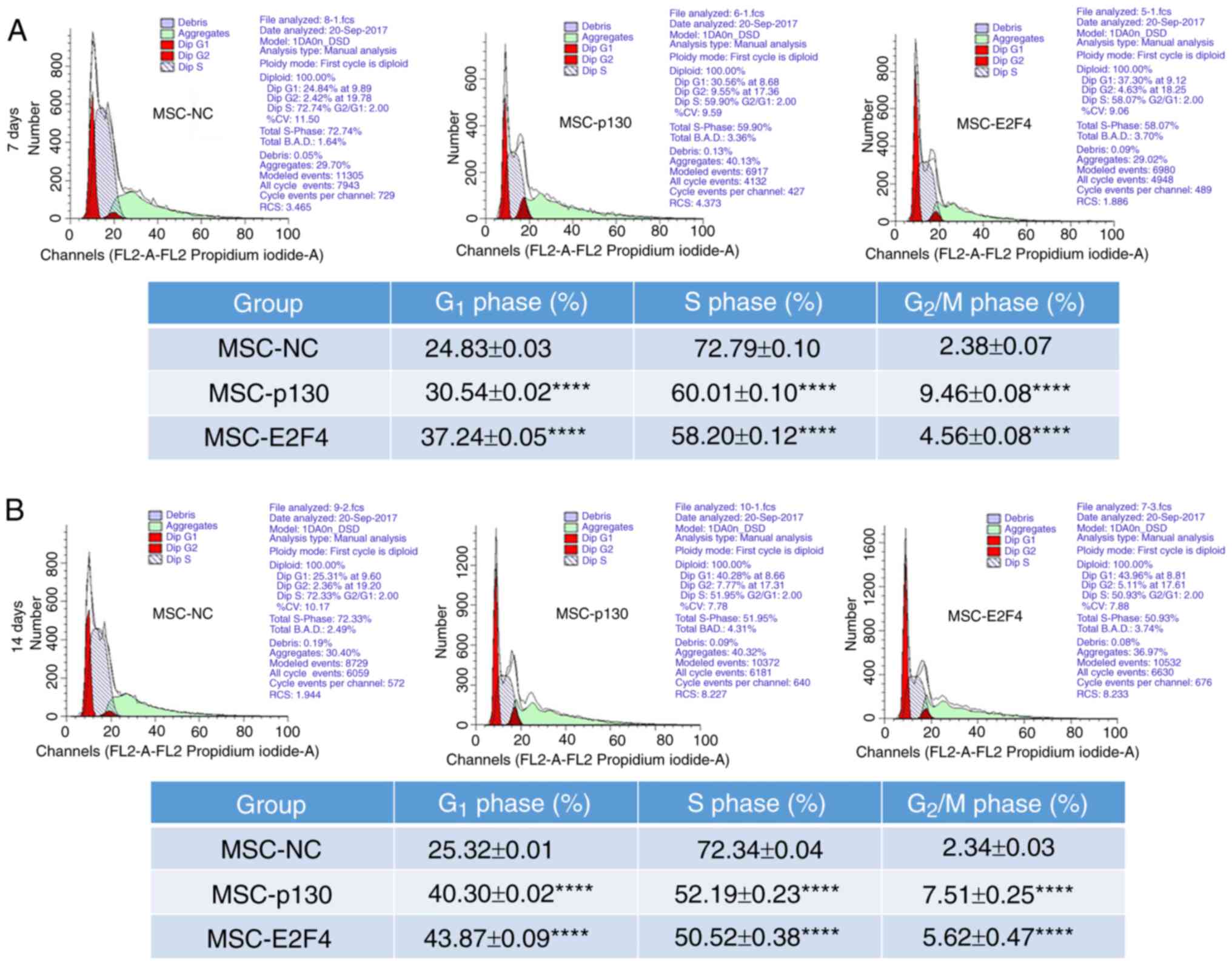
Overexpression of p130 or E2F4
prolongs the G1 phase of mMSCs during differentiation
into AT II cells
To determine whether the differentiation of MSC-p130
and MSC-E2F4 groups was associated with regulation of the cell
cycle, cell cycle stages were detected using flow cytometry. The
proportion of G1-phase cells in the MSC-p130 group was
30.54±0.02% at 7 days following differentiation, which was
significantly higher than that in the MSC-NC group. Moreover, the
proportion of S-phase cells significantly decreased, and the
proportion of G2/M-phase cells significantly increased
in the MSC-p130 group, compared with the MSC-NC group (Fig. 4A). In addition, compared with those
in the MSC-NC group, the G1 and G2/M phases
in the MSC-E2F4 group were also significantly delayed, and the
proportion of cells in S phase was significantly reduced (Fig. 4A). Comparable trends were also
observed at 14 days following differentiation of mMSCs in each
group. Compared with the control group, the proportion of cells in
the G1 and G2/M phases in the MSC-p130 group
was significantly increased, and the proportion of cells in S phase
was significantly decreased (Fig.
4B). Comparable trends were observed in the MSC-E2F4 group
(Fig. 4B).
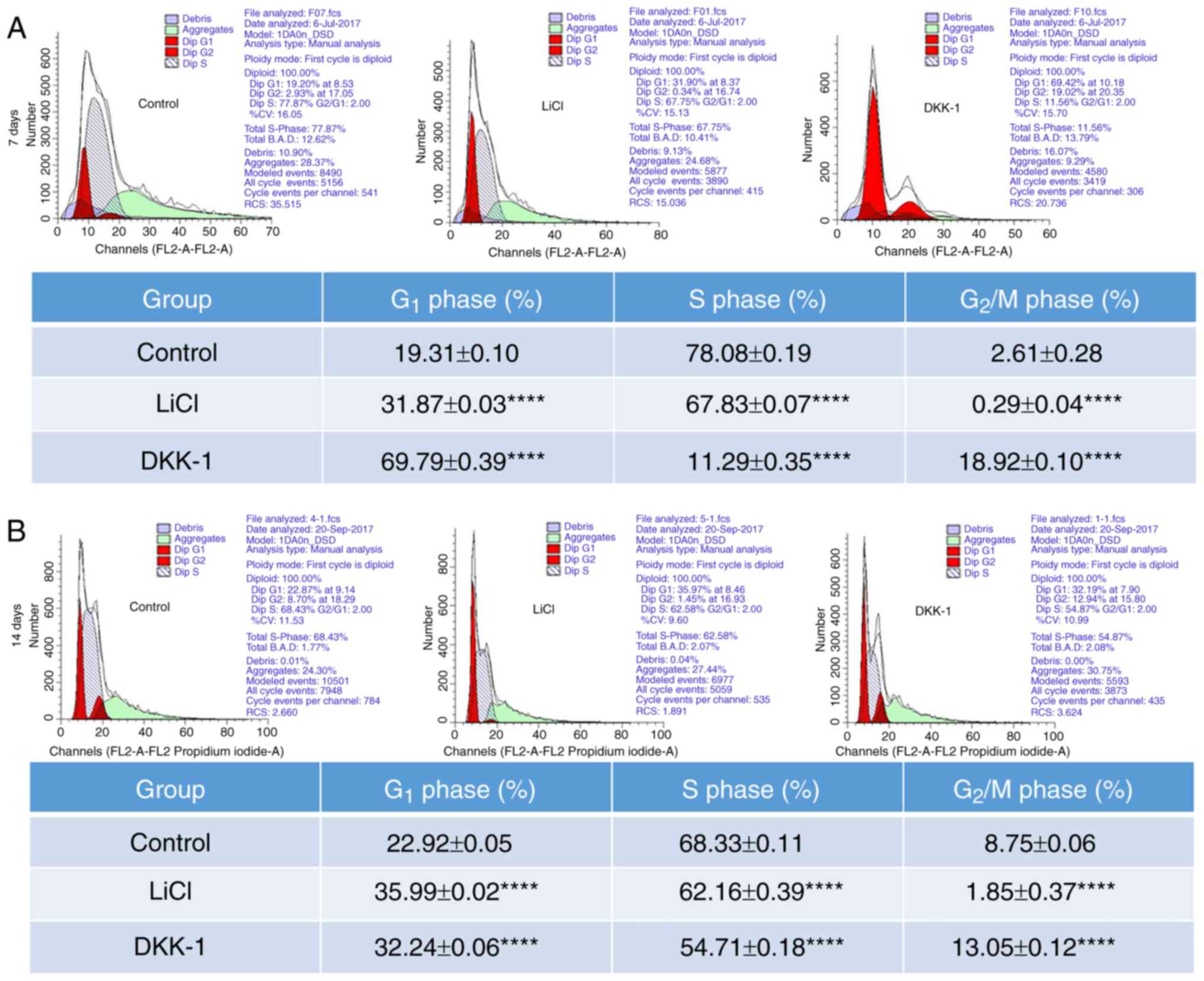
Activation of the canonical Wnt
signaling pathway prolongs the G1 + S phase of mMSCs
during differentiation into AT II cells
To further evaluate whether the cell cycle was
associated with the canonical Wnt signaling pathway-mediated
differentiation of mMSCs into AT II cells, changes in the cell
cycle in mMSCs were detected using flow cytometry. The proportion
of cells in the G2/M phase in the LiCl group was
0.29±0.04% at 7 days following differentiation of mMSCs, which was
significantly lower than that in the control group (2.61±0.28%). In
addition, DKK-1 significantly increased the proportion of cells in
the G2/M phase in mMSCs at 7 days following
differentiation (Fig. 5A).
Comparable results were observed at 14 days following the
differentiation of mMSCs. Compared with the control group, the
proportion of cells in the G2/M phase was significantly
decreased in the LiCl group, and significantly increased in the
DKK-1 group (Fig. 5B).
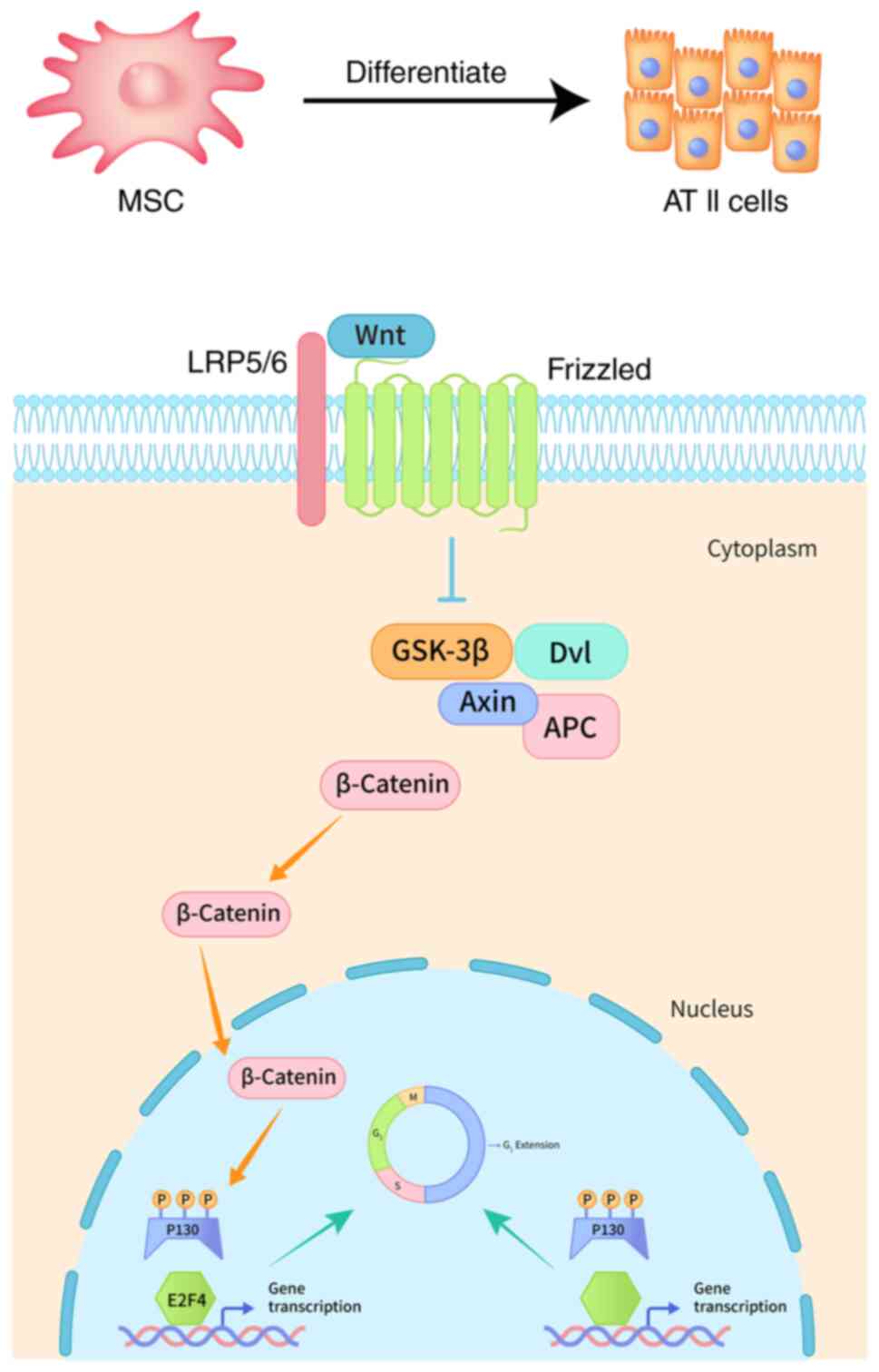
To sum up, a graphical representation to illustrate
the molecular mechanism of MSCs differentiation into AT II cells
was shown in Fig. 6.
Discussion
MSCs exhibit potential in tissue repair, and in the
treatment of cardiovascular, pulmonary and renal diseases. However,
the clinical application of MSCs remains limited to phase 2a
clinical trials for acute respiratory distress syndrome (ARDS)
(15). Large-scale clinical
research demonstrating the therapeutic value of MSCs is lacking,
due to complexities in the directional differentiation of MSCs
(16-18).
In the present study, a coculture system of mMSCs
and MLE-12 cells combined with SAGM was established in
vitro, and the Wnt signaling pathway in mMSCs was specifically
activated or inhibited. Subsequently, the directed differentiation
of mMSCs was observed, and the role of the canonical Wnt signaling
pathway in the cell cycle of mMSCs during differentiation into AT
II cells was detected. Moreover, the association between the
canonical Wnt signaling pathway and p130/E2F4 during the
differentiation of mMSCs was also identified, to establish a
downstream molecular mechanism during the directed differentiation
into AT II cells. To the best of our knowledge, the present study
was the first to assess the directional differentiation of mMSCs
into AT II cells via regulation of p130/E2F4 and the canonical Wnt
pathway.
Results of the present study demonstrated that
overexpression of p130 or E2F4 promoted the differentiation of
mMSCs into AT II cells, and Wnt signaling pathway-mediated
differentiation may be associated with changes in the G1
+ S phase. Moreover, results of the present study demonstrated that
regulation of the canonical Wnt signaling pathway specifically
regulated the expression of p130 and E2F4 in mMSCs during
differentiation, and the canonical Wnt signaling pathway promoted
the differentiation of mMSCs into AT II cells via regulating
p130/E2F4. The specific underlying mechanisms may be associated
with a prolonged G1 phase in mMSCs (Fig. 6).
The Wnt signaling pathway is a fundamental
regulatory pathway in the development, differentiation and other
physiological functions of cells and organisms (5). According to different transduction
signals, the Wnt pathway is classified into canonical and
noncanonical pathways (19). The
canonical Wnt pathway, which is also referred to as the
Wnt/β-catenin pathway, plays important regulatory roles in numerous
biological processes, such as cell proliferation, differentiation
and embryonic development. It is highly conserved in the whole
evolutionary process of cells (19). Previous studies demonstrated that
both canonical and noncanonical Wnt signaling pathways are involved
in the regulation of mMSC differentiation (11,20,21).
Results of our previous study demonstrated that activation of the
canonical or noncanonical Wnt signaling pathway improved the
differentiation of MSCs into AT II cells in
lipopolysaccharide-induced ARDS mice (22). Results of the present study also
confirmed that regulation of the canonical Wnt signaling pathway
specifically regulated the differentiation of MSCs into AT II
cells. Cell cycle changes detected using flow cytometry also
revealed that the differentiation of mMSCs into AT II cells
regulated by the canonical Wnt signaling pathway may be associated
with the delay of the G1 + S phase. Notably, associated
literature is lacking at present, and further investigations are
required.
p130 plays a role in regulating mitosis, and is an
important nuclear transcription factor that initiates the cell
cycle and promotes cell differentiation. E2F4, which interacts with
p130, is involved in regulation of the cell cycle, from
G1 to S phase transformation (12). Capasso et al (23) revealed that the inactivation of
retinoblastoma proteins (Rb1 or Rb2/p130) delayed the onset of the
last cell division and impaired the terminal adipocyte
differentiation of MSCs. Petrov et al (13) confirmed that the Wnt/β-catenin and
pRb signaling pathways interact and form a common
p130/Gsk3β/β-catenin complex during MSC cycle progression.
Moreover, results of a previous study demonstrated that levels of
p130 and E2F4 in MSCs treated with LiCl were also increased. The
p130/E2F4 complex in MSCs was associated with β-catenin, which was
co-precipitated from the extracts of asynchronously growing or
synchronized at G0/G1- and S-phase cells.
These results are consistent with those of the present study.
However, the effects of the canonical Wnt and p130/E2F4 pathways on
the differentiation of MSCs into AT II cells remain unclear.
Results of the present study demonstrated that the canonical Wnt
signaling pathway impacted the differentiation of mMSCs into AT II
cells through regulating p130/E2F4. The specific underlying
mechanism may be associated with a prolonged G1 phase in
mMSCs. In addition, results of a previous study demonstrated that
mMSCs overexpressing p130 or E2F4 promoted osteogenesis, and
inhibited adipogenesis and chondrogenesis. The specific underlying
mechanism was associated with regulation of the G1 phase
(14). These results are
comparable with those of the present study.
AT II cells synthesize and secrete alveolar
surfactant, and differentiate into type I alveolar epithelial
cells. They act as progenitor cells for re-epithelialization of
injured alveoli (24). The repair
and regeneration of injured AT II cells are therefore critical for
patients with acute and chronic pulmonary diseases (25,26).
MSCs exhibit potential as therapeutic sources for tissue repair.
Results of previous studies demonstrated that MSCs differentiate
into AT II cells in vivo and in vitro (27,28).
Moreover, murine bone marrow-derived MSCs and human bone
marrow-derived MSCs, with common cell surface markers
CD105+ and CD45- (29), possess similar characteristics,
including differentiation into alveolar epithelial cells,
alleviation of inflammation, and improvement of pathological
impairment both in vivo and in vitro (30-33).
Results of a previous study demonstrated that regulation of the
canonical and noncanonical Wnt signaling pathways promoted
MSC-mediated protection against epithelial impairment in ARDS mice
(22). Moreover, results of the
present study revealed that changes in the cell cycle may play a
role in the therapeutic effects of MSC differentiation,
specifically in repairing lung injuries. Regulating cell cycle
changes in MSCs may contribute to further development of their
clinical application for the treatment of pulmonary diseases.
MSCs have potential to differentiate into alveolar
epithelial cells in vitro and in vivo. Different
media containing epithelial differentiation inducers or matrix were
tested in vitro to prove MSCs differentiation into specific
epithelial tissue-related properties (34,35).
Recently, Zeng et al (36)
proved that overexpression of Fork head box protein M1 facilitates
MSCs differentiation into AT II cells in vitro using the
co-culture system combined with SAGM. Besides, Perng et al
(37) demonstrated that the
addition of transforming growth factor-β1 and extracellular matrix
collagen are required to facilitate MSCs differentiation into lung
epithelial-like cells in vitro. And Chanda et al
(38) established an organoid
model in Matrigel mixed with MTEC/plus media to study stem cell
behavior and intercellular communication between lung resident MSCs
and AT II with age-related phenotypes, revealing that young MSCs
formed normal 3D structures with both young and aged AT II cells,
but aged L-MSCs developed abnormal, loose structures with AT II
cells. In addition, different models were also used to prove the
therapeutic effects of MSCs by differentiation into AT II cells
in vivo. Recently, Shao et al (39) found that overexpression of CXCR7
promoted differentiation of MSCs into AT II cells and enhanced the
ability of MSCs to modulate the inflammatory response in
phosgene-induced ALI. And Huang et al (40) demonstrated that MSCs possess the
differentiation potential into AT II cells, as a primary mechanism
through which MSCs exert a therapeutic effect, specifically within
a microenvironment of injury of pulmonary fibrosis.
Surfactant protein is a specific protein in the
lung, which is divided into four subtypes; namely, SP-A, -B, -C and
-D. SP-A and SP-D directly interact with pathogens and allergens,
stimulate immune cells, and influence cytokine and chemokine
profiles during the host defense response. In addition, SP-B and
SP-C play critical roles in reducing the surface tension of the
lung, and in the regulation of intracellular processes required for
the production of surfactant and for the maintenance of AT II cell
function (41). SP-C mRNA is
limited to lung tissue and is detectable in isolated AT II cells.
Compared with SP-C mRNA, SP-A, SP-B and SP-D are mainly present in
type II cells and Clara cells (42).
Notably, the present study exhibits numerous
limitations. Results of the present study demonstrated that changes
in the cell cycle were not synchronized between canonical Wnt
pathway activation and p130/E2F4 overexpression during the
differentiation of mMSCs into AT II cells. This may be due to the
complex crosstalk that occurs during cell signaling, making it
difficult to target specific factors. Moreover, further
investigations are required to determine the interaction between
β-catenin and p130 during the differentiation of MSCs into AT II
cells, including specific cell cycle changes and interaction
domains. In addition, p130 and E2F4 on Day 7 and Day 14 in the
MSC-NC, MSC-p130 and MSC-E2F4 groups after induced differentiation
is required to further determine the effects of overexpression of
p130 or E2F4. However, results of our previous study demonstrated
that lentivirus-mediated p130 or E2F4 transduction is stable and
efficient, even after 20 passages of mMSCs (14). A further limitation of the present
study included the low magnification used during fluorescence
microscopy, which may lead to bias during fluorescent counting.
However, the protein expression levels of SP-C verified the
increased levels of differentiation.
In conclusion, results of the present study
demonstrated that the canonical Wnt signaling pathway may impact
the differentiation of MSCs into AT II cells through regulating
downstream p130/E2F4. The specific underlying mechanism may be
associated with regulation of the cell cycle. Results of the
present study may provide a novel theoretical basis for promoting
the differentiation of MSCs into AT II cells for the treatment of
pulmonary diseases.
Supplementary Material
SP-C expression following induced
differentiation of MSC-p130 and MSC-E2F4 into type II alveolar
epithelial cells in vitro upon canonical Wnt signaling
pathway inhibition (n=3). ****P<0.0001 vs. DKK-1 +
MSC-NC group. Co-culture with MSC-E2F4, MSC-E2F4 co-cultured with
MLE-12 cells with SAGM and DKK-1 added; Co-culture with MSC-NC,
MSC-NC co-cultured with MLE-12 cells with SAGM and DKK-1 added;
Co-culture with MSC-p130, MSC-p130 co-cultured with MLE-12 cells
with SAGM and DKK-1 added; DKK-1, Dickkopf-related protein 1; MSCs,
mouse mesenchymal stem cells; MSC-p130, MSCs overexpressing p130;
MSC-E2F4, MSCs overexpressing E2F transcription factor 4; NC,
normal control; SAGM, small airway growth medium; SP-C, surfactant
protein C.
Acknowledgements
The authors would like to thank Dr Shixia Cai
(Department of Critical Care Medicine, The Affiliated Hospital of
Qingdao University, Qingdao, China) and Dr Jianxiao Chen
(Department of Critical Care Medicine, Renji Hospital, School of
Medicine, Shanghai Jiaotong University, Shanghai, China) for their
advice and guidance on the design of the experiments.
Funding
Funding: The study was supported by the National Natural Science
Foundation of China (grant nos. 81871602 and 81930058), the Jiangsu
Provincial Special Program of Medical Science (grant no.
BE2019749), National Science and Technology Major Project for
Control and Prevention of Major Infectious Diseases of China (grant
no. 2017ZX10103004), Natural Science Foundation of Jiangsu Province
(grant no. BK20200356), and the Fundamental Research Funds for the
Central Universities of China (grant no. ZDYYZXKT2020027).
Availability of data and materials
All data generated and/or analyzed during this study
are included in this published article.
Authors' contributions
XZ participated in the study design, performed
laboratory work and statistical analysis, prepared the drafts of
the manuscript, and revised the manuscript according to advice from
the other authors. MX participated in the laboratory work,
performed statistical analysis and drafted the manuscript. AL
participated in the study design and helped revise the manuscript.
HQ and FG confirm the authenticity of all the raw data and were
responsible for the study design and revised the manuscript for
important intellectual content. All authors have read and approved
the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Galipeau J and Sensébé L: Mesenchymal
stromal cells: Clinical challenges and therapeutic opportunities.
Cell Stem Cell. 22:824–833. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Huppert LA, Liu KD and Matthay MA:
Therapeutic potential of mesenchymal stromal cells in the treatment
of ARDS. Transfusion. 59:869–875. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Schwede M, Wilfong EM, Zemans RL, Lee PJ,
Dos Santos C, Fang X and Matthay MA: Effects of bone marrow-derived
mesenchymal stromal cells on gene expression in human alveolar type
II cells exposed to TNF-α, IL-1β, and IFN-γ. Physiol Rep.
6(e13831)2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Visweswaran M, Pohl S, Arfuso F, Newsholme
P, Dilley R, Pervaiz S and Dharmarajan A: Multi-lineage
differentiation of mesenchymal stem cells-to Wnt, or not Wnt. Int J
Biochem Cell Biol. 68:139–147. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Logan CY and Nusse R: The Wnt signaling
pathway in development and disease. Annu Rev Cell Dev Biol.
20:781–810. 2004.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Etheridge SL, Spencer GJ, Heath DJ and
Genever PG: Expression profiling and functional analysis of wnt
signaling mechanisms in mesenchymal stem cells. Stem Cells.
22:849–860. 2004.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Sasaki T, Han F, Shioda N, Moriguchi S,
Kasahara J, Ishiguro K and Fukunaga K: Lithium-induced activation
of Akt and CaM kinase II contributes to its neuroprotective action
in a rat microsphere embolism model. Brain Res. 1108:98–106.
2006.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Mao B, Wu W, Davidson G, Marhold J, Li M,
Mechler BM, Delius H, Hoppe D, Stannek P, Walter C, et al: Kremen
proteins are Dickkopf receptors that regulate Wnt/beta-catenin
signaling. Nature. 417:664–667. 2002.PubMed/NCBI View Article : Google Scholar
|
|
9
|
van Haaften T, Byrne R, Bonnet S,
Rochefort GY, Akabutu J, Bouchentouf M, Rey-Parra GJ, Galipeau J,
Haromy A, Eaton F, et al: Airway delivery of mesenchymal stem cells
prevents arrested alveolar growth in neonatal lung injury in rats.
Am J Respir Crit Care Med. 180:1131–1142. 2009.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Leeman KT, Pessina P, Lee JH and Kim CF:
Mesenchymal stem cells increase alveolar differentiation in lung
progenitor organoid cultures. Sci Rep. 9(6479)2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Liu AR, Liu L, Chen S, Yang Y, Zhao HJ,
Liu L, Guo FM, Lu XM and Qiu HB: Activation of canonical wnt
pathway promotes differentiation of mouse bone marrow-derived MSCs
into type II alveolar epithelial cells, confers resistance to
oxidative stress, and promotes their migration to injured lung
tissue in vitro. J Cell Physiol. 228:1270–1283. 2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Beshiri ML, Holmes KB, Richter WF, Hess S,
Islam AB, Yan Q, Plante L, Litovchick L, Gévry N, Lopez-Bigas N, et
al: Coordinated repression of cell cycle genes by KDM5A and E2F4
during differentiation. Proc Natl Acad Sci USA. 109:18499–18504.
2012.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Petrov N, Zhidkova O, Serikov V, Zenin V
and Popov B: Induction of Wnt/β-catenin signaling in mouse
mesenchymal stem cells is associated with activation of the p130
and E2f4 and formation of the p130/Gsk3β/β-catenin complex. Stem
Cells Dev. 21:589–597. 2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Zhang X, Chen J, Liu A, Xu X, Xue M, Xu J,
Yang Y, Qiu H and Guo F: Stable overexpression of p130/E2F4 affects
the multipotential abilities of bone-marrow-derived mesenchymal
stem cells. J Cell Physiol. 233:9739–9749. 2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Matthay MA, Calfee CS, Zhuo H, Thompson
BT, Wilson JG, Levitt JE, Rogers AJ, Gotts JE, Wiener-Kronish JP,
Bajwa EK, et al: Treatment with allogeneic mesenchymal stromal
cells for moderate to severe acute respiratory distress syndrome
(START study): A randomised phase 2a safety trial. Lancet Respir
Med. 7:154–162. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Laffey JG and Matthay MA: Fifty years of
research in ARDS. Cell-based therapy for acute respiratory distress
syndrome. biology and potential therapeutic value. Am J Respir Crit
Care Med. 196:266–273. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Wilson JG, Liu KD, Zhuo H, Caballero L,
McMillan M, Fang X, Cosgrove K, Vojnik R, Calfee CS, Lee JW, et al:
Mesenchymal stem (stromal) cells for treatment of ARDS: A phase 1
clinical trial. Lancet Respir Med. 3:24–32. 2015.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Walter J, Ware LB and Matthay MA:
Mesenchymal stem cells: Mechanisms of potential therapeutic benefit
in ARDS and sepsis. Lancet Respir Med. 2:1016–1026. 2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ling L, Nurcombe V and Cool SM: Wnt
signaling controls the fate of mesenchymal stem cells. Gene.
433:1–7. 2009.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Cai SX, Liu AR, He HL, Chen QH, Yang Y,
Guo FM, Huang YZ, Liu L and Qiu HB: Stable genetic alterations of
β-catenin and ROR2 regulate the Wnt pathway, affect the fate of
MSCs. J Cell Physiol. 229:791–800. 2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Liu A, Chen S, Cai S, Dong L, Liu L, Yang
Y, Guo F, Lu X, He H, Chen Q, et al: Wnt5a through noncanonical
Wnt/JNK or Wnt/PKC signaling contributes to the differentiation of
mesenchymal stem cells into type II alveolar epithelial cells in
vitro. PLoS One. 9(e90229)2014.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Cai SX, Liu AR, Chen S, He HL, Chen QH, Xu
JY, Pan C, Yang Y, Guo FM, Huang YZ, et al: The orphan receptor
tyrosine kinase ROR2 facilitates MSCs to repair lung injury in ARDS
animal model. Cell Transplant. 25:1561–1574. 2016.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Capasso S, Alessio N, Di Bernardo G,
Cipollaro M, Melone MA, Peluso G, Giordano A and Galderisi U:
Silencing of RB1 and RB2/P130 during adipogenesis of bone marrow
stromal cells results in dysregulated differentiation. Cell Cycle.
13:482–490. 2014.PubMed/NCBI View
Article : Google Scholar
|
|
24
|
Matthay MA, Robriquet L and Fang X:
Alveolar epithelium: Role in lung fluid balance and acute lung
injury. Proc Am Thorac Soc. 2:206–213. 2005.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Mason RJ: Thoughts on the alveolar phase
of COVID-19. Am J Physiol Lung Cell Mol Physiol. 319:L115–L120.
2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Ruaro B, Salton F, Braga L, Wade B,
Confalonieri P, Volpe MC, Baratella E, Maiocchi S and Confalonieri
M: The history and mystery of alveolar epithelial type II cells:
Focus on their physiologic and pathologic role in lung. Int J Mol
Sci. 22(2566)2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Rojas M, Xu J, Woods CR, Mora AL, Spears
W, Roman J and Brigham KL: Bone marrow-derived mesenchymal stem
cells in repair of the injured lung. Am J Respir Cell Mol Biol.
33:145–152. 2005.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Ma N, Gai H, Mei J, Ding FB, Bao CR,
Nguyen DM and Zhong H: Bone marrow mesenchymal stem cells can
differentiate into type II alveolar epithelial cells in vitro. Cell
Biol Int. 35:1261–1266. 2011.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Li H, Ghazanfari R, Zacharaki D, Lim HC
and Scheding S: Isolation and characterization of primary bone
marrow mesenchymal stromal cells. Ann N Y Acad Sci. 1370:109–118.
2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Ortiz LA, Gambelli F, McBride C, Gaupp D,
Baddoo M, Kaminski N and Phinney DG: Mesenchymal stem cell
engraftment in lung is enhanced in response to bleomycin exposure
and ameliorates its fibrotic effects. Proc Natl Acad Sci USA.
100:8407–8411. 2003.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Yamada M, Kubo H, Kobayashi S, Ishizawa K,
Numasaki M, Ueda S, Suzuki T and Sasaki H: Bone marrow-derived
progenitor cells are important for lung repair after
lipopolysaccharide-induced lung injury. J Immunol. 172:1266–1272.
2004.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kotton DN, Ma BY, Cardoso WV, Sanderson
EA, Summer RS, Williams MC and Fine A: Bone marrow-derived cells as
progenitors of lung alveolar epithelium. Development.
128:5181–5188. 2001.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Chen QH, Liu AR, Qiu HB and Yang Y:
Interaction between mesenchymal stem cells and endothelial cells
restores endothelial permeability via paracrine hepatocyte growth
factor in vitro. Stem Cell Res Ther. 6(44)2015.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Shannon JM, Mason RJ and Jennings SD:
Functional differentiation of alveolar type II epithelial cells in
vitro: Effects of cell shape, cell-matrix interactions and
cell-cell interactions. Biochim Biophys Acta. 931:143–156.
1987.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Olsen CO, Isakson BE, Seedorf GJ, Lubman
RL and Boitano S: Extracellular matrix-driven alveolar epithelial
cell differentiation in vitro. Exp Lung Res. 31:461–482.
2005.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Zeng M, Chen Q, Ge S, He W, Zhang L, Yi H
and Lin S: Overexpression of FoxM1 promotes differentiation of bone
marrow mesenchymal stem cells into alveolar type II cells through
activating Wnt/β-catenin signaling. Biochem Biophys Res Commun.
528:311–317. 2020.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Perng DW, Yang DM, Hsiao YH, Lo T, Lee OK,
Wu MT, Wu YC and Lee YC: miRNA-146a expression positively regulates
tumor necrosis factor-α-induced interleukin-8 production in
mesenchymal stem cells and differentiated lung epithelial-like
cells. Tissue Eng Part A. 18:2259–2267. 2012.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Chanda D, Rehan M, Smith SR, Dsouza KG,
Wang Y, Bernard K, Kurundkar D, Memula V, Kojima K, Mobley JA, et
al: Mesenchymal stromal cell aging impairs the self-organizing
capacity of lung alveolar epithelial stem cells. Elife.
10(e68049)2021.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Shao Y, Zhou F, He D, Zhang L and Shen J:
Overexpression of CXCR7 promotes mesenchymal stem cells to repair
phosgene-induced acute lung injury in rats. Biomed Pharmacother.
109:1233–1239. 2019.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Huang K, Kang X, Wang X, Wu S, Xiao J, Li
Z, Wu X and Zhang W: Conversion of bone marrow mesenchymal stem
cells into type II alveolar epithelial cells reduces pulmonary
fibrosis by decreasing oxidative stress in rats. Mol Med Rep.
11:1685–1692. 2015.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Mulugeta S and Beers MF: Surfactant
protein C: Its unique properties and emerging immunomodulatory role
in the lung. Microbes Infect. 8:2317–2323. 2006.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Beers MF and Fisher AB: Surfactant protein
C: A review of its unique properties and metabolism. Am J Physiol.
263:L151–L160. 1992.PubMed/NCBI View Article : Google Scholar
|